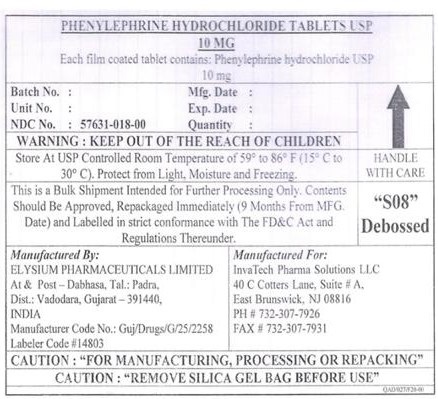
Phenylephrine Hydrochloride
b8d360fa-0d80-f6a8-e053-2a95a90a8bd4
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Medication Information
Warnings and Precautions
Indications and Usage
Dosage and Administration
Section 51727-6
Section 51945-4
Section 55105-1
Structured Label Content
Indications and Usage (34067-9)
Dosage and Administration (34068-7)
Warnings and Precautions (34071-1)
Section 51727-6 (51727-6)
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:08.622866 · Updated: 2026-03-14T23:10:23.192014