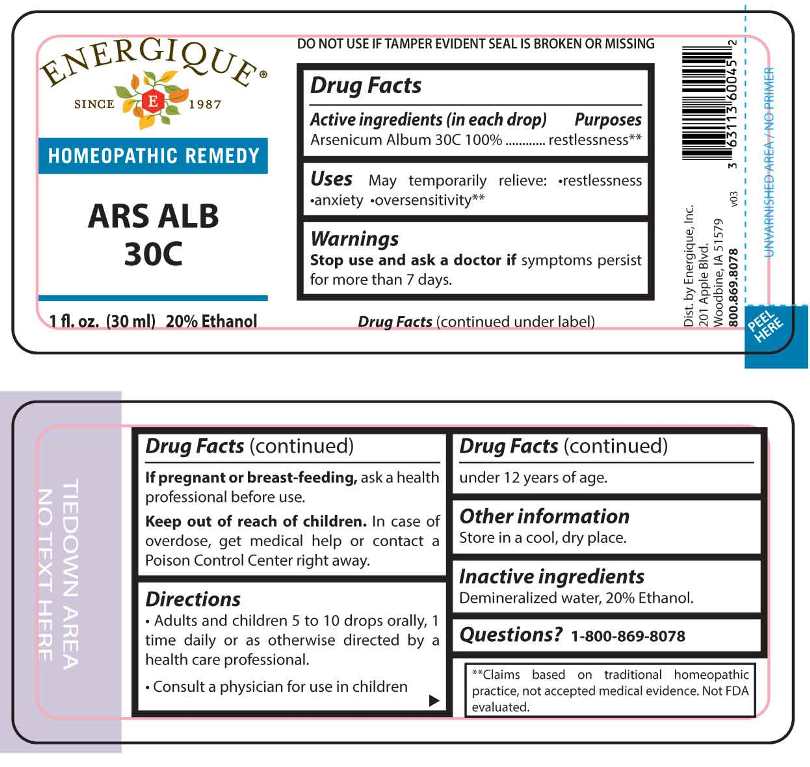
Drug Facts:
b874a9d8-7ce3-4a7e-8c47-e56961ec19ff
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
(in each drop) Arsenicum Album 30C 100%.
Purpose
Arsenicum Album - restlessness** **Claims based on traditional homeopathic practices, not accepted medical evidence. Not FDA evaluated.
Medication Information
Purpose
Arsenicum Album - restlessness** **Claims based on traditional homeopathic practices, not accepted medical evidence. Not FDA evaluated.
Description
(in each drop) Arsenicum Album 30C 100%.
Uses:
May temporarily relieve:
•restlessness •anxiety oversensitivity.**
**Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
Purposes:
Arsenicum Album - restlessness**
**Claims based on traditional homeopathic practices, not accepted medical evidence. Not FDA evaluated.
Warnings:
Stop use and ask a doctor if symptoms persist for more than 7 days.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
DO NOT USE IF TAMPER EVIDENT SEAL IS BROKEN OR MISSING
Store in a cool, dry place.
Questions:
Dist. by Energique, Inc.
201 Apple Blvd
Woodbine, IA 51579
800-869-8078
Directions:
• Adults and children 5 to 10 drops orally, 1 time daily or as otherwise directed by a health care professional.
• Consult a physician for use in children under 12 years of age.
Active Ingredient:
(in each drop) Arsenicum Album 30C 100%.
Inactive Ingredients:
Demineralized water, 20% Ethanol.
Package Label Display:
ENERGIQUE
SINCE 1987
HOMEOPATHIC REMEDY
ARS ALB
30C
1 fl. oz. (30 ml)
Keep Out of Reach of Children:
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses: (USES:)
May temporarily relieve:
•restlessness •anxiety oversensitivity.**
**Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
Purposes: (PURPOSES:)
Arsenicum Album - restlessness**
**Claims based on traditional homeopathic practices, not accepted medical evidence. Not FDA evaluated.
Warnings: (WARNINGS:)
Stop use and ask a doctor if symptoms persist for more than 7 days.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
DO NOT USE IF TAMPER EVIDENT SEAL IS BROKEN OR MISSING
Store in a cool, dry place.
Questions: (QUESTIONS:)
Dist. by Energique, Inc.
201 Apple Blvd
Woodbine, IA 51579
800-869-8078
Directions: (DIRECTIONS:)
• Adults and children 5 to 10 drops orally, 1 time daily or as otherwise directed by a health care professional.
• Consult a physician for use in children under 12 years of age.
Active Ingredient: (ACTIVE INGREDIENT:)
(in each drop) Arsenicum Album 30C 100%.
Inactive Ingredients: (INACTIVE INGREDIENTS:)
Demineralized water, 20% Ethanol.
Package Label Display: (PACKAGE LABEL DISPLAY:)
ENERGIQUE
SINCE 1987
HOMEOPATHIC REMEDY
ARS ALB
30C
1 fl. oz. (30 ml)
Keep Out of Reach of Children: (KEEP OUT OF REACH OF CHILDREN:)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:02.800312 · Updated: 2026-03-14T23:17:21.910853