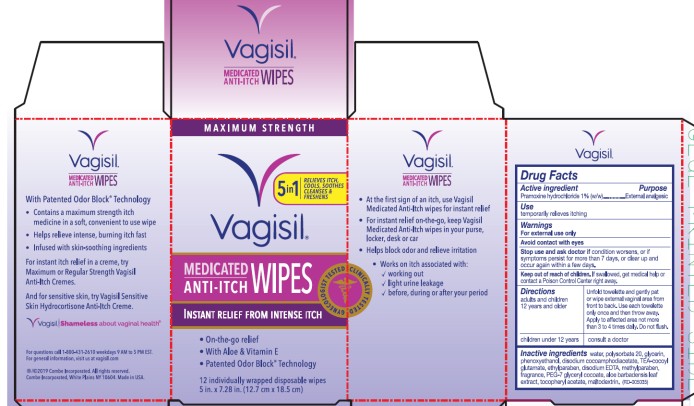
Vagisil Medicated Anti-itch Wipes Maximum Strength
b848f53d-8aea-4efe-aa2c-009d5121fc33
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
VAGISIL Anti-Itch Medicated Wipes Drug Facts
Purpose
External analgesic
Medication Information
Purpose
External analgesic
Description
VAGISIL Anti-Itch Medicated Wipes Drug Facts
Use
temporarily relieves itching
Section 42229-5
VAGISIL Anti-Itch Medicated Wipes
Drug Facts
Warnings
For external use only
Avoid contact with eyes
Directions
| adults and children 12 years and older |
Unfold towelette and gently pat or wipe external vaginal area from front to back. Use each towelette only once and then throw away. Apply to affected area not more than 3 to 4 times daily. Do not flush. |
| children under 12 years | consult a doctor |
Active Ingredient
Pramoxine hydrochloride 1% (w/w)
Other Information
store at room temperature
discard within 2 months of opening
Inactive Ingredients
water, polysorbate 20, glycerin, phenoxyethanol, disodium cocoamphodiacetate, TEA-cocoyl glutamate, ethylparaben, disodium EDTA, methylparaben, fragrance, PEG-7 glyceryl cocoate, aloe barbadensis leaf extract, tocopheryl acetate, maltodextrin
Principal Display Panel
Maximum Strength
Vagisil
®
Medicated Anti-Itch Wipes
Instant Relief From Intense Itch
Gynecologist Tested
Clinically Tested
-
On-the-go relief
-
With Aloe & Vitamin E
- Patented Odor Block Technology
12 individually wrapped disposable wipes
5 in. x 7.28 in. (12.7 cm x 18.5 cm)
Stop Use and Ask Doctor If
condition worsens, or if symptoms persist for more than 7 days, or clear up and occur again within a few days
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use
temporarily relieves itching
Section 42229-5 (42229-5)
VAGISIL Anti-Itch Medicated Wipes
Drug Facts
Purpose
External analgesic
Warnings
For external use only
Avoid contact with eyes
Directions
| adults and children 12 years and older |
Unfold towelette and gently pat or wipe external vaginal area from front to back. Use each towelette only once and then throw away. Apply to affected area not more than 3 to 4 times daily. Do not flush. |
| children under 12 years | consult a doctor |
Active Ingredient (Active ingredient)
Pramoxine hydrochloride 1% (w/w)
Other Information (Other information)
store at room temperature
discard within 2 months of opening
Inactive Ingredients (Inactive ingredients)
water, polysorbate 20, glycerin, phenoxyethanol, disodium cocoamphodiacetate, TEA-cocoyl glutamate, ethylparaben, disodium EDTA, methylparaben, fragrance, PEG-7 glyceryl cocoate, aloe barbadensis leaf extract, tocopheryl acetate, maltodextrin
Principal Display Panel
Maximum Strength
Vagisil
®
Medicated Anti-Itch Wipes
Instant Relief From Intense Itch
Gynecologist Tested
Clinically Tested
-
On-the-go relief
-
With Aloe & Vitamin E
- Patented Odor Block Technology
12 individually wrapped disposable wipes
5 in. x 7.28 in. (12.7 cm x 18.5 cm)
Stop Use and Ask Doctor If (Stop use and ask doctor if)
condition worsens, or if symptoms persist for more than 7 days, or clear up and occur again within a few days
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:41.280817 · Updated: 2026-03-14T23:05:35.877028