Proparacaine Hydrochloride
b699f838-8b69-4bac-953b-ea8822ec19fc
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
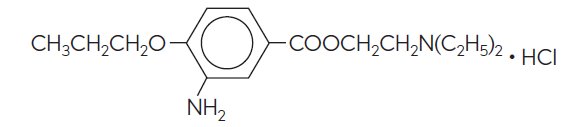
Proparacaine hydrochloride ophthalmic solution USP, 0.5% is a topical local anesthetic for ophthalmic use. The active ingredient is represented by the structural formula: Established name: Proparacaine Hydrochloride Chemical name: Benzoic acid, 3-amino-4-propoxy-,2-(diethylamino) ethyl ester, monohydrochloride Molecular weight: 330.86 Each mL contains: Active: proparacaine hydrochloride 5 mg (0.5%). Inactives: glycerin and purified water. The pH may be adjusted with hydrochloric acid and/or sodium hydroxide. Preservative: benzalkonium chloride (0.01%).
Indications and Usage
Proparacaine hydrochloride ophthalmic solution is indicated for procedures in which a topical ophthalmic anesthetic is indicated; corneal anesthesia of short duration, e.g. tonometry, gonioscopy, removal of corneal foreign bodies and for short corneal and conjunctival procedures.
Dosage and Administration
Usual Dosage: Removal of foreign bodies and sutures, and for tonometry: 1 to 2 drops (in single instillations) in each eye before operating. Short corneal and conjunctival procedures: 1 drop in each eye every 5 to 10 minutes for 5 to 7 doses. NOTE: Proparacaine hydrochloride ophthalmic solution USP, 0.5% should be clear, colorless to faint yellow color. If the solution becomes darker, discard the solution. FOR TOPICAL OPHTHALMIC USE ONLY
Contraindications
Proparacaine hydrochloride ophthalmic solution is contraindicated in patients with known hypersensitivity to any of the ingredients of this preparation.
Adverse Reactions
Occasional temporary stinging, burning and conjunctival redness may occur with the use of proparacaine. A rare, severe, immediate-type, apparently hyperallergic corneal reaction characterized by acute, intense and diffuse epithelial keratitis, a gray, ground glass appearance, sloughing of large areas of necrotic epithelium, corneal filaments and sometimes iritis with descemetitis has been reported. Allergic contact dermatitis from proparacaine with drying and fissuring of the fingertips has been reported. To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
How Supplied
Proparacaine hydrochloride ophthalmic solution USP, 0.5% is supplied in a plastic bottle with a controlled drop tip and a white polypropylene cap in the following size: NDC 24208-730-06 15 mL bottle
Medication Information
Indications and Usage
Proparacaine hydrochloride ophthalmic solution is indicated for procedures in which a topical ophthalmic anesthetic is indicated; corneal anesthesia of short duration, e.g. tonometry, gonioscopy, removal of corneal foreign bodies and for short corneal and conjunctival procedures.
Dosage and Administration
Usual Dosage: Removal of foreign bodies and sutures, and for tonometry: 1 to 2 drops (in single instillations) in each eye before operating.
Short corneal and conjunctival procedures: 1 drop in each eye every 5 to 10 minutes for 5 to 7 doses.
NOTE: Proparacaine hydrochloride ophthalmic solution USP, 0.5% should be clear, colorless to faint yellow color. If the solution becomes darker, discard the solution.
FOR TOPICAL OPHTHALMIC USE ONLY
Contraindications
Proparacaine hydrochloride ophthalmic solution is contraindicated in patients with known hypersensitivity to any of the ingredients of this preparation.
Adverse Reactions
Occasional temporary stinging, burning and conjunctival redness may occur with the use of proparacaine. A rare, severe, immediate-type, apparently hyperallergic corneal reaction characterized by acute, intense and diffuse epithelial keratitis, a gray, ground glass appearance, sloughing of large areas of necrotic epithelium, corneal filaments and sometimes iritis with descemetitis has been reported.
Allergic contact dermatitis from proparacaine with drying and fissuring of the fingertips has been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Proparacaine hydrochloride ophthalmic solution USP, 0.5% is supplied in a plastic bottle with a controlled drop tip and a white polypropylene cap in the following size:
-
NDC 24208-730-06 15 mL bottle
Description
Proparacaine hydrochloride ophthalmic solution USP, 0.5% is a topical local anesthetic for ophthalmic use. The active ingredient is represented by the structural formula:
Established name:
Proparacaine Hydrochloride
Chemical name:
Benzoic acid, 3-amino-4-propoxy-,2-(diethylamino) ethyl ester, monohydrochloride
Molecular weight: 330.86
Each mL contains:
Active: proparacaine hydrochloride 5 mg (0.5%). Inactives: glycerin and purified water. The pH may be adjusted with hydrochloric acid and/or sodium hydroxide. Preservative: benzalkonium chloride (0.01%).
Storage:
Refrigerate at 2°C to 8°C (36°F to 46°F). Protect from light. Keep tightly closed.
|
DO NOT USE IF IMPRINTED NECKBAND IS NOT INTACT. |
Keep out of reach of children.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
© 2023 Bausch & Lomb Incorporated or its affiliates
Revised: September 2023
-
9114405 (Folded)
9114505 (Flat)
Warnings
NOT FOR INJECTION INTO THE EYE - FOR TOPICAL OPHTHALMIC USE ONLY
Prolonged use of a topical ocular anesthetic is not recommended. It may produce permanent corneal opacification with accompanying visual loss.
Pregnancy
Animal reproduction studies have not been conducted with proparacaine hydrochloride ophthalmic solution USP, 0.5%. It is also not known whether proparacaine hydrochloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Proparacaine hydrochloride should be administered to a pregnant woman only if clearly needed.
Geriatric Use
No overall clinical differences in safety of effectiveness have been observed between the elderly and other adult patients.
Pediatric Use
Safety and effectiveness of proparacaine hydrochloride ophthalmic solution in pediatric patients have been established. Use of proparacaine hydrochloride is supported by evidence from adequate and well-controlled studies in adults and children over the age of twelve, and safety information in neonates and other pediatric patients.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when proparacaine hydrochloride is administered to a nursing woman.
Clinical Pharmacology
Proparacaine hydrochloride ophthalmic solution is a rapidly-acting topical anesthetic, with induced anesthesia lasting approximately 10-20 minutes.
Principal Display Panel Carton
NDC 24208-730-06
Proparacaine
Hydrochloride
Ophthalmic
Solution, USP
0.5%
(Sterile)
FOR TOPICAL OPHTHALMIC
USE ONLY
Rx only
15 mL
BAUSCH + LOMB
9536602
AB04711
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential, mutagenicity or possible impairment of fertility in males or females.
Structured Label Content
Storage:
Refrigerate at 2°C to 8°C (36°F to 46°F). Protect from light. Keep tightly closed.
|
DO NOT USE IF IMPRINTED NECKBAND IS NOT INTACT. |
Keep out of reach of children.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
© 2023 Bausch & Lomb Incorporated or its affiliates
Revised: September 2023
-
9114405 (Folded)
9114505 (Flat)
Warnings (WARNINGS)
NOT FOR INJECTION INTO THE EYE - FOR TOPICAL OPHTHALMIC USE ONLY
Prolonged use of a topical ocular anesthetic is not recommended. It may produce permanent corneal opacification with accompanying visual loss.
Pregnancy
Animal reproduction studies have not been conducted with proparacaine hydrochloride ophthalmic solution USP, 0.5%. It is also not known whether proparacaine hydrochloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Proparacaine hydrochloride should be administered to a pregnant woman only if clearly needed.
Description (DESCRIPTION)
Proparacaine hydrochloride ophthalmic solution USP, 0.5% is a topical local anesthetic for ophthalmic use. The active ingredient is represented by the structural formula:
Established name:
Proparacaine Hydrochloride
Chemical name:
Benzoic acid, 3-amino-4-propoxy-,2-(diethylamino) ethyl ester, monohydrochloride
Molecular weight: 330.86
Each mL contains:
Active: proparacaine hydrochloride 5 mg (0.5%). Inactives: glycerin and purified water. The pH may be adjusted with hydrochloric acid and/or sodium hydroxide. Preservative: benzalkonium chloride (0.01%).
How Supplied (HOW SUPPLIED)
Proparacaine hydrochloride ophthalmic solution USP, 0.5% is supplied in a plastic bottle with a controlled drop tip and a white polypropylene cap in the following size:
-
NDC 24208-730-06 15 mL bottle
Geriatric Use
No overall clinical differences in safety of effectiveness have been observed between the elderly and other adult patients.
Pediatric Use
Safety and effectiveness of proparacaine hydrochloride ophthalmic solution in pediatric patients have been established. Use of proparacaine hydrochloride is supported by evidence from adequate and well-controlled studies in adults and children over the age of twelve, and safety information in neonates and other pediatric patients.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when proparacaine hydrochloride is administered to a nursing woman.
Adverse Reactions (ADVERSE REACTIONS)
Occasional temporary stinging, burning and conjunctival redness may occur with the use of proparacaine. A rare, severe, immediate-type, apparently hyperallergic corneal reaction characterized by acute, intense and diffuse epithelial keratitis, a gray, ground glass appearance, sloughing of large areas of necrotic epithelium, corneal filaments and sometimes iritis with descemetitis has been reported.
Allergic contact dermatitis from proparacaine with drying and fissuring of the fingertips has been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Contraindications (CONTRAINDICATIONS)
Proparacaine hydrochloride ophthalmic solution is contraindicated in patients with known hypersensitivity to any of the ingredients of this preparation.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Proparacaine hydrochloride ophthalmic solution is a rapidly-acting topical anesthetic, with induced anesthesia lasting approximately 10-20 minutes.
Indications and Usage (INDICATIONS AND USAGE)
Proparacaine hydrochloride ophthalmic solution is indicated for procedures in which a topical ophthalmic anesthetic is indicated; corneal anesthesia of short duration, e.g. tonometry, gonioscopy, removal of corneal foreign bodies and for short corneal and conjunctival procedures.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Usual Dosage: Removal of foreign bodies and sutures, and for tonometry: 1 to 2 drops (in single instillations) in each eye before operating.
Short corneal and conjunctival procedures: 1 drop in each eye every 5 to 10 minutes for 5 to 7 doses.
NOTE: Proparacaine hydrochloride ophthalmic solution USP, 0.5% should be clear, colorless to faint yellow color. If the solution becomes darker, discard the solution.
FOR TOPICAL OPHTHALMIC USE ONLY
Principal Display Panel Carton (PRINCIPAL DISPLAY PANEL-Carton)
NDC 24208-730-06
Proparacaine
Hydrochloride
Ophthalmic
Solution, USP
0.5%
(Sterile)
FOR TOPICAL OPHTHALMIC
USE ONLY
Rx only
15 mL
BAUSCH + LOMB
9536602
AB04711
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential, mutagenicity or possible impairment of fertility in males or females.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:10.580306 · Updated: 2026-03-14T21:56:49.665802