Itk Body Blemish Treatment Mist
b67b910b-2475-4c39-85a9-7a46ed39eeab
34390-5
HUMAN OTC DRUG LABEL
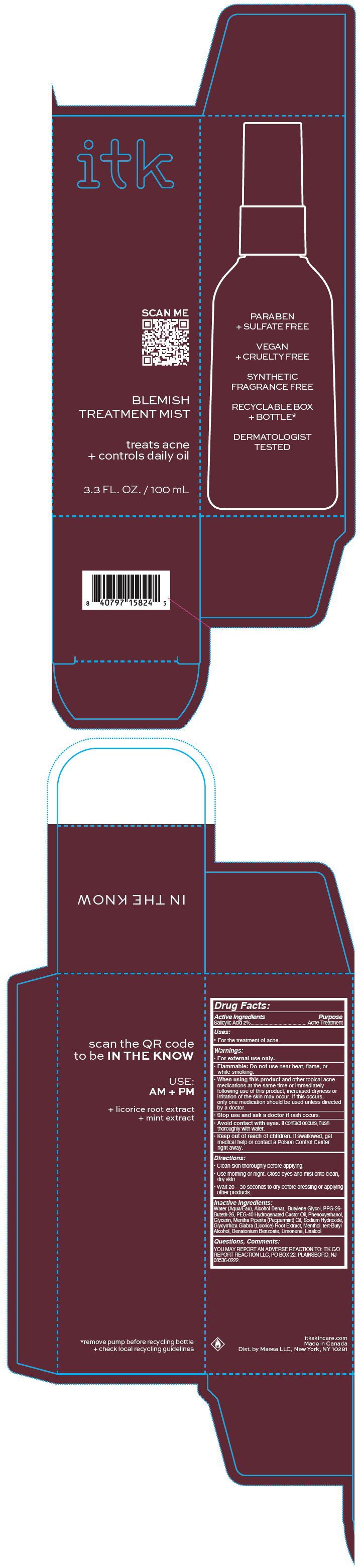
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts:
Purpose
Acne Treatment
Medication Information
Purpose
Acne Treatment
Description
Drug Facts:
Uses
- For the treatment of acne.
Section 42229-5
Drug Facts:
Section 50565-1
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
- Stop use and ask a doctor if rash occurs.
- Avoid contact with eyes. If contact occurs, flush thoroughly with water.
Section 50567-7
- When using this product and other topical acne medications at the same time or immediately following use of this product, increased dryness or irritation of the skin may occur. If this occurs, only one medication should be used unless directed by a doctor.
Warnings
- For external use only.
- Flammable: Do not use near heat, flame, or while smoking.
Directions
- Clean skin thoroughly before applying.
- Use morning or night. Close eyes and mist onto clean, dry skin.
- Wait 20 – 30 seconds to dry before dressing or applying other products.
Active Ingredients
Salicylic Acid 2%
Questions, Comments
YOU MAY REPORT AN ADVERSE REACTION TO: ITK C/O REPORT REACTION LLC, PO BOX 22, PLAINSBORO, NJ 08536-0222.
Inactive Ingredients
Water (Aqua/Eau), Alcohol Denat., Butylene Glycol, PPG-26- Buteth-26, PEG-40 Hydrogenated Castor Oil, Phenoxyethanol, Glycerin, Mentha Piperita (Peppermint) Oil, Sodium Hydroxide, Glycyrrhiza Glabra (Licorice) Root Extract, Menthol, tert-Butyl Alcohol, Denatonium Benzoate, Limonene, Linalool.
Principal Display Panel 100 Ml Bottle Carton
itk
SCAN ME
BLEMISH
TREATMENT MIST
treats acne
+ controls daily oil
3.3 FL. OZ. / 100 mL
Structured Label Content
Uses
- For the treatment of acne.
Section 42229-5 (42229-5)
Drug Facts:
Section 50565-1 (50565-1)
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
- Stop use and ask a doctor if rash occurs.
- Avoid contact with eyes. If contact occurs, flush thoroughly with water.
Section 50567-7 (50567-7)
- When using this product and other topical acne medications at the same time or immediately following use of this product, increased dryness or irritation of the skin may occur. If this occurs, only one medication should be used unless directed by a doctor.
Purpose
Acne Treatment
Warnings
- For external use only.
- Flammable: Do not use near heat, flame, or while smoking.
Directions
- Clean skin thoroughly before applying.
- Use morning or night. Close eyes and mist onto clean, dry skin.
- Wait 20 – 30 seconds to dry before dressing or applying other products.
Active Ingredients
Salicylic Acid 2%
Questions, Comments
YOU MAY REPORT AN ADVERSE REACTION TO: ITK C/O REPORT REACTION LLC, PO BOX 22, PLAINSBORO, NJ 08536-0222.
Inactive Ingredients
Water (Aqua/Eau), Alcohol Denat., Butylene Glycol, PPG-26- Buteth-26, PEG-40 Hydrogenated Castor Oil, Phenoxyethanol, Glycerin, Mentha Piperita (Peppermint) Oil, Sodium Hydroxide, Glycyrrhiza Glabra (Licorice) Root Extract, Menthol, tert-Butyl Alcohol, Denatonium Benzoate, Limonene, Linalool.
Principal Display Panel 100 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 100 mL Bottle Carton)
itk
SCAN ME
BLEMISH
TREATMENT MIST
treats acne
+ controls daily oil
3.3 FL. OZ. / 100 mL
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:43.530314 · Updated: 2026-03-14T23:02:25.522673