These Highlights Do Not Include All The Information Needed To Use Fosphenytoin Sodium Injection Safely And Effectively. See Full Prescribing Information For Fosphenytoin Sodium Injection.
b60c9c82-e5c7-4e05-98c7-5bbba4af04b2
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.9 ) 4/2022
Indications and Usage
Fosphenytoin sodium injection is indicated for the treatment of generalized tonic-clonic status epilepticus and prevention and treatment of seizures occurring during neurosurgery. Fosphenytoin sodium injection can also be substituted, short-term, for oral phenytoin. Fosphenytoin sodium injection should be used only when oral phenytoin administration is not possible [see Dosage and Administration ( 2.4 ) and Warnings and Precautions ( 5.2 )] .
Dosage and Administration
The dose, concentration, and infusion rate of fosphenytoin sodium injection should always be expressed as phenytoin sodium equivalents (PE) ( 2.1 ) For Status Epilepticus: Adult loading dose is 15 to 20 mg PE/kg at a rate of 100 to 150 mg PE/min ( 2.3 ) Pediatric loading dose is 15 to 20 mg PE/kg at a rate of 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) ( 2.3 ) For Non-emergent Loading and Maintenance Dosing: Adult loading dose is 10 to 20 mg PE/kg given IV or IM; initial maintenance dose is 4 to 6 mg PE/kg/day in divided doses ( 2.4 ) Pediatric loading dose is 10 to 15 mg PE/kg at a rate of 1 to 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower); initial maintenance dose is 2 to 4 mg PE/kg every 12 hours at a rate of 1 to 2 mg PE/kg/min (or 100 mg PE/min, whichever is slower) ( 2.4 ) Intramuscular Administration: Fosphenytoin sodium injection should ordinarily not be given intramuscularly ( 2.3 , 2.4 )
Warnings and Precautions
Dosing Errors: Do not confuse the amount of drug to be given in PE with the concentration of the drug in the vial. Ensure the appropriate volume is withdrawn from the vial when preparing for administration. ( 5.1 ) Withdrawal Precipitated Seizure: May precipitate status epilepticus. Dose reductions or discontinuation should be done gradually. ( 5.3 ) Serious Dermatologic Reactions: Discontinue at the first sign of a rash, unless clearly not drug-related. If signs or symptoms suggest SJS/TEN, fosphenytoin sodium injection should not be resumed; consider alternative therapy. ( 5.4 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: If signs or symptoms of hypersensitivity are present, evaluate the patient immediately. Discontinue if an alternative etiology cannot be established. ( 5.5 ) Angioedema: Discontinue immediately if symptoms of angioedema such as facial, perioral, or upper airway swelling occur. ( 5.7 ) Hematopoietic Complications: If occurs, follow-up observation is indicated and an alternative antiepileptic treatment should be used. ( 5.9 )
Contraindications
Fosphenytoin sodium injection is contraindicated in patients with: A history of hypersensitivity to fosphenytoin sodium injection or its inactive ingredients, or to phenytoin or other hydantoins [see Warnings and Precautions ( 5.6 )] . Reactions have included angioedema. Sinus bradycardia, sino-atrial block, second and third degree A-V block, or Adams-Stokes syndrome because of the effect of parenteral phenytoin or fosphenytoin sodium injection on ventricular automaticity. A history of prior acute hepatotoxicity attributable to fosphenytoin sodium injection or phenytoin [see Warnings and Precautions ( 5.8 )] . Coadministration with delavirdine because of the potential for loss of virologic response and possible resistance to delavirdine or to the class of non-nucleoside reverse transcriptase inhibitors.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Cardiovascular Risk Associated with Rapid Infusion [see Warnings and Precautions ( 5.2 )] Withdrawal Precipitated Seizure, Status Epilepticus [see Warnings and Precautions ( 5.3 )] Serious Dermatologic Reactions [see Warnings and Precautions ( 5.4 )] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions ( 5.5 )] Hypersensitivity [see Warnings and Precautions ( 5.6 )] Angioedema [see Warnings and Precautions ( 5.7 )] Hepatic Injury [see Warnings and Precautions ( 5.8 )] Hematopoietic Complications [see Warnings and Precautions ( 5.9 )] Sensory Disturbances [see Warnings and Precautions ( 5.10 )] Local Toxicity (Including Purple Glove Syndrome) [see Warnings and Precautions ( 5.11 )] Exacerbation of Porphyria [see Warnings and Precautions ( 5.14 )] Teratogenicity and Other Harm to the Newborn [see Warnings and Precautions ( 5.15 )] Hyperglycemia [see Warnings and Precautions ( 5.16 )]
Drug Interactions
Fosphenytoin is extensively bound to human plasma proteins. Drugs highly bound to albumin could increase the unbound fraction of fosphenytoin. Although, it is unknown whether this could result in clinically significant effects, caution is advised when administering fosphenytoin sodium injection with other drugs that significantly bind to serum albumin. The most significant drug interactions following administration of fosphenytoin sodium injection are expected to occur with drugs that interact with phenytoin. Phenytoin is extensively bound to serum plasma proteins and is prone to competitive displacement. Phenytoin is primarily metabolized by the hepatic cytochrome P450 enzyme CYP2C9 and to a lesser extent by CYP2C19 and is particularly susceptible to inhibitory drug interactions because it is subject to saturable metabolism. Inhibition of metabolism may produce significant increases in circulating phenytoin concentrations and enhance the risk of drug toxicity. Monitoring of phenytoin serum levels is recommended when a drug interaction is suspected. Phenytoin or fosphenytoin sodium injection is a potent inducer of hepatic drug-metabolizing enzymes.
Storage and Handling
Store under refrigeration at 2°C to 8°C (36°F to 46°F). The product should not be stored at room temperature for more than 48 hours. Vials that develop particulate matter should not be used. Injection vials are single-dose only. After opening, any unused product should be discarded.
How Supplied
Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow solution supplied as follows: Product Code Unit of Sale Strength Each 400302 NDC 63323-403-02 Unit of 25 100 mg PE per 2 mL (50 mg PE per mL) NDC 63323-403-01 2 mL fill in a 5 mL Single Dose Vial 400310 NDC 63323-403-10 Unit of 10 500 mg PE per 10 mL (50 mg PE per mL) NDC 63323-403-04 10 mL Single Dose Vial The container closure is not made with natural rubber latex. Both sizes of vials contain Tromethamine, USP (TRIS), Hydrochloric Acid, NF, or Sodium Hydroxide, NF, and Water for Injection, USP. Fosphenytoin sodium injection, USP should always be prescribed in phenytoin sodium equivalents (PE) [see Dosage and Administration ( 2.1 ) and Warnings and Precautions ( 5.1 )] . 1.5 mg of fosphenytoin sodium is equivalent to 1 mg phenytoin sodium, and is referred to as 1 mg PE. The amount and concentration of fosphenytoin is always expressed in terms of mg of phenytoin sodium equivalents (PE). Fosphenytoin's weight is expressed as phenytoin sodium equivalents to avoid the need to perform molecular weight-based adjustments when substituting fosphenytoin for phenytoin or vice versa.
Medication Information
Warnings and Precautions
Dosing Errors: Do not confuse the amount of drug to be given in PE with the concentration of the drug in the vial. Ensure the appropriate volume is withdrawn from the vial when preparing for administration. ( 5.1 ) Withdrawal Precipitated Seizure: May precipitate status epilepticus. Dose reductions or discontinuation should be done gradually. ( 5.3 ) Serious Dermatologic Reactions: Discontinue at the first sign of a rash, unless clearly not drug-related. If signs or symptoms suggest SJS/TEN, fosphenytoin sodium injection should not be resumed; consider alternative therapy. ( 5.4 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: If signs or symptoms of hypersensitivity are present, evaluate the patient immediately. Discontinue if an alternative etiology cannot be established. ( 5.5 ) Angioedema: Discontinue immediately if symptoms of angioedema such as facial, perioral, or upper airway swelling occur. ( 5.7 ) Hematopoietic Complications: If occurs, follow-up observation is indicated and an alternative antiepileptic treatment should be used. ( 5.9 )
Indications and Usage
Fosphenytoin sodium injection is indicated for the treatment of generalized tonic-clonic status epilepticus and prevention and treatment of seizures occurring during neurosurgery. Fosphenytoin sodium injection can also be substituted, short-term, for oral phenytoin. Fosphenytoin sodium injection should be used only when oral phenytoin administration is not possible [see Dosage and Administration ( 2.4 ) and Warnings and Precautions ( 5.2 )] .
Dosage and Administration
The dose, concentration, and infusion rate of fosphenytoin sodium injection should always be expressed as phenytoin sodium equivalents (PE) ( 2.1 ) For Status Epilepticus: Adult loading dose is 15 to 20 mg PE/kg at a rate of 100 to 150 mg PE/min ( 2.3 ) Pediatric loading dose is 15 to 20 mg PE/kg at a rate of 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) ( 2.3 ) For Non-emergent Loading and Maintenance Dosing: Adult loading dose is 10 to 20 mg PE/kg given IV or IM; initial maintenance dose is 4 to 6 mg PE/kg/day in divided doses ( 2.4 ) Pediatric loading dose is 10 to 15 mg PE/kg at a rate of 1 to 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower); initial maintenance dose is 2 to 4 mg PE/kg every 12 hours at a rate of 1 to 2 mg PE/kg/min (or 100 mg PE/min, whichever is slower) ( 2.4 ) Intramuscular Administration: Fosphenytoin sodium injection should ordinarily not be given intramuscularly ( 2.3 , 2.4 )
Contraindications
Fosphenytoin sodium injection is contraindicated in patients with: A history of hypersensitivity to fosphenytoin sodium injection or its inactive ingredients, or to phenytoin or other hydantoins [see Warnings and Precautions ( 5.6 )] . Reactions have included angioedema. Sinus bradycardia, sino-atrial block, second and third degree A-V block, or Adams-Stokes syndrome because of the effect of parenteral phenytoin or fosphenytoin sodium injection on ventricular automaticity. A history of prior acute hepatotoxicity attributable to fosphenytoin sodium injection or phenytoin [see Warnings and Precautions ( 5.8 )] . Coadministration with delavirdine because of the potential for loss of virologic response and possible resistance to delavirdine or to the class of non-nucleoside reverse transcriptase inhibitors.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Cardiovascular Risk Associated with Rapid Infusion [see Warnings and Precautions ( 5.2 )] Withdrawal Precipitated Seizure, Status Epilepticus [see Warnings and Precautions ( 5.3 )] Serious Dermatologic Reactions [see Warnings and Precautions ( 5.4 )] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions ( 5.5 )] Hypersensitivity [see Warnings and Precautions ( 5.6 )] Angioedema [see Warnings and Precautions ( 5.7 )] Hepatic Injury [see Warnings and Precautions ( 5.8 )] Hematopoietic Complications [see Warnings and Precautions ( 5.9 )] Sensory Disturbances [see Warnings and Precautions ( 5.10 )] Local Toxicity (Including Purple Glove Syndrome) [see Warnings and Precautions ( 5.11 )] Exacerbation of Porphyria [see Warnings and Precautions ( 5.14 )] Teratogenicity and Other Harm to the Newborn [see Warnings and Precautions ( 5.15 )] Hyperglycemia [see Warnings and Precautions ( 5.16 )]
Drug Interactions
Fosphenytoin is extensively bound to human plasma proteins. Drugs highly bound to albumin could increase the unbound fraction of fosphenytoin. Although, it is unknown whether this could result in clinically significant effects, caution is advised when administering fosphenytoin sodium injection with other drugs that significantly bind to serum albumin. The most significant drug interactions following administration of fosphenytoin sodium injection are expected to occur with drugs that interact with phenytoin. Phenytoin is extensively bound to serum plasma proteins and is prone to competitive displacement. Phenytoin is primarily metabolized by the hepatic cytochrome P450 enzyme CYP2C9 and to a lesser extent by CYP2C19 and is particularly susceptible to inhibitory drug interactions because it is subject to saturable metabolism. Inhibition of metabolism may produce significant increases in circulating phenytoin concentrations and enhance the risk of drug toxicity. Monitoring of phenytoin serum levels is recommended when a drug interaction is suspected. Phenytoin or fosphenytoin sodium injection is a potent inducer of hepatic drug-metabolizing enzymes.
Storage and Handling
Store under refrigeration at 2°C to 8°C (36°F to 46°F). The product should not be stored at room temperature for more than 48 hours. Vials that develop particulate matter should not be used. Injection vials are single-dose only. After opening, any unused product should be discarded.
How Supplied
Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow solution supplied as follows: Product Code Unit of Sale Strength Each 400302 NDC 63323-403-02 Unit of 25 100 mg PE per 2 mL (50 mg PE per mL) NDC 63323-403-01 2 mL fill in a 5 mL Single Dose Vial 400310 NDC 63323-403-10 Unit of 10 500 mg PE per 10 mL (50 mg PE per mL) NDC 63323-403-04 10 mL Single Dose Vial The container closure is not made with natural rubber latex. Both sizes of vials contain Tromethamine, USP (TRIS), Hydrochloric Acid, NF, or Sodium Hydroxide, NF, and Water for Injection, USP. Fosphenytoin sodium injection, USP should always be prescribed in phenytoin sodium equivalents (PE) [see Dosage and Administration ( 2.1 ) and Warnings and Precautions ( 5.1 )] . 1.5 mg of fosphenytoin sodium is equivalent to 1 mg phenytoin sodium, and is referred to as 1 mg PE. The amount and concentration of fosphenytoin is always expressed in terms of mg of phenytoin sodium equivalents (PE). Fosphenytoin's weight is expressed as phenytoin sodium equivalents to avoid the need to perform molecular weight-based adjustments when substituting fosphenytoin for phenytoin or vice versa.
Description
Warnings and Precautions ( 5.9 ) 4/2022
Section 42229-5
Phenytoin Sodium Equivalents (PE)
The dose, concentration, and infusion rate of fosphenytoin sodium injection should always be expressed as phenytoin sodium equivalents (PE). There is no need to perform molecular weight-based adjustments when converting between fosphenytoin and phenytoin sodium doses. Fosphenytoin sodium injection should always be prescribed and dispensed in phenytoin sodium equivalent units (PE). The amount and concentration of fosphenytoin is always expressed in terms of mg of phenytoin sodium equivalents (mg PE).
Section 43683-2
| Warnings and Precautions (5.9) | 4/2022 |
Section 51945-4
PACKAGE LABEL - PRINCIPAL DISPLAY - Fosphenytoin 2 mL Single Dose Vial Label
NDC 63323-403-01 400302
Fosphenytoin Sodium Injection, USP
100 mg PE per 2 mL (50 mg PE per mL)
(PE = phenytoin sodium equivalents)
2 mL Single Dose Vial Rx only
10 Overdosage
Nausea, vomiting, lethargy, tachycardia, bradycardia, asystole, cardiac arrest, hypotension, syncope, hypocalcemia, metabolic acidosis, and death have been reported in cases of overdosage with fosphenytoin sodium injection.
Because fosphenytoin sodium injection is a prodrug of phenytoin, the following information about phenytoin overdosage may be helpful. Initial symptoms of acute phenytoin toxicity are nystagmus, ataxia, and dysarthria. Other signs include tremor, hyperreflexia, lethargy, slurred speech, nausea, vomiting, coma, and hypotension. Death is caused by respiratory and circulatory depression. The lethal dose of phenytoin in adults is estimated to be 2 to 5 grams. The lethal dose in pediatrics is not known.
There are marked variations among individuals with respect to serum phenytoin concentrations where toxicity occurs. Lateral gaze nystagmus usually appears at 20 mcg/mL, ataxia at 30 mcg/mL, and dysarthria and lethargy appear when the serum concentration is over 40 mcg/mL. However, phenytoin concentrations as high as 50 mcg/mL have been reported without evidence of toxicity. As much as 25 times the therapeutic phenytoin dose has been taken, resulting in serum phenytoin concentrations over 100 mcg/mL, with complete recovery. Irreversible cerebellar dysfunction and atrophy have been reported after overdosage.
Formate and phosphate are metabolites of fosphenytoin sodium injection and therefore may contribute to signs of toxicity following overdosage. Signs of formate toxicity are similar to those of methanol toxicity and are associated with severe anion-gap metabolic acidosis. Large amounts of phosphate, delivered rapidly, could potentially cause hypocalcemia with paresthesia, muscle spasms, and seizures. Ionized free calcium levels can be measured and, if low, used to guide treatment.
Treatment: Treatment is nonspecific since there is no known antidote to fosphenytoin sodium injection or phenytoin overdosage.
The adequacy of the respiratory and circulatory systems should be carefully observed, and appropriate supportive measures employed. Hemodialysis can be considered since phenytoin (the active metabolite of fosphenytoin sodium injection) is not completely bound to plasma proteins. Total exchange transfusion has been used in the treatment of severe intoxication in children.
In acute overdosage the possibility of other CNS depressants, including alcohol, should be borne in mind.
11 Description
Fosphenytoin Sodium Injection, USP is a prodrug intended for parenteral administration; its active metabolite is phenytoin. 1.5 mg of fosphenytoin sodium is equivalent to 1 mg phenytoin sodium, and is referred to as 1 mg phenytoin sodium equivalents (PE). The amount and concentration of fosphenytoin is always expressed in terms of mg PE.
The pharmacological class of the fosphenytoin sodium is hydantoin derivative, and the therapeutic class is anticonvulsant.
Fosphenytoin sodium injection, USP is marketed as a 2 mL fill in 5 mL vials containing a total of 100 mg PE/2 mL (50 mg PE/mL) and 10 mL vials containing a total of 500 mg PE/10 mL (50 mg PE/mL), for intravenous or intramuscular administration. The concentration of each vial is 50 mg PE/mL. Fosphenytoin Sodium Injection, USP is supplied in vials as a sterile solution in Water for Injection, USP, and Tromethamine, USP (TRIS) (12.11 mg/mL), buffer adjusted to pH 8.6 to 9.0 with either Hydrochloric Acid, NF, or Sodium Hydroxide, NF. Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow, sterile solution.
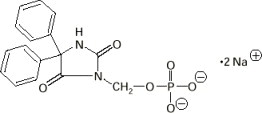
The chemical name of fosphenytoin is 5,5-diphenyl-3-[(phosphonooxy)methyl]-2,4-imidazolidinedione disodium salt. The molecular structure of fosphenytoin is:
The molecular weight of fosphenytoin is 406.24.
5.7 Angioedema
Angioedema has been reported in patients treated with phenytoin and fosphenytoin sodium injection in the postmarketing setting. Fosphenytoin sodium injection should be discontinued immediately if symptoms of angioedema, such as facial, perioral, or upper airway swelling occur. Fosphenytoin sodium injection should be discontinued permanently if a clear alternative etiology for the reaction cannot be established.
2.2 Preparation
Prior to intravenous (IV) infusion, dilute fosphenytoin sodium injection in 5% Dextrose Injection or 0.9% Sodium Chloride Injection to a concentration ranging from 1.5 to 25 mg PE/mL. The maximum concentration of fosphenytoin sodium in any solution should be 25 mg PE/mL. When fosphenytoin sodium injection is given as an IV infusion, fosphenytoin sodium injection needs to be diluted and should only be administered at a rate not exceeding 150 mg PE/min.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
For single-dose only. After opening, any unused product should be discarded.
16.1 How Supplied
Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow solution supplied as follows:
|
Product
Code |
Unit of Sale |
Strength |
Each |
| 400302 | NDC 63323-403-02 Unit of 25 |
100 mg PE per 2 mL (50 mg PE per mL) |
NDC 63323-403-01 2 mL fill in a 5 mL Single Dose Vial |
| 400310 | NDC 63323-403-10 Unit of 10 |
500 mg PE per 10 mL (50 mg PE per mL) |
NDC 63323-403-04 10 mL Single Dose Vial |
The container closure is not made with natural rubber latex.
Both sizes of vials contain Tromethamine, USP (TRIS), Hydrochloric Acid, NF, or Sodium Hydroxide, NF, and Water for Injection, USP.
Fosphenytoin sodium injection, USP should always be prescribed in phenytoin sodium equivalents (PE) [see Dosage and Administration (2.1) and Warnings and Precautions (5.1)].
1.5 mg of fosphenytoin sodium is equivalent to 1 mg phenytoin sodium, and is referred to as 1 mg PE. The amount and concentration of fosphenytoin is always expressed in terms of mg of phenytoin sodium equivalents (PE). Fosphenytoin's weight is expressed as phenytoin sodium equivalents to avoid the need to perform molecular weight-based adjustments when substituting fosphenytoin for phenytoin or vice versa.
8.4 Pediatric Use
Fosphenytoin sodium injection is indicated for the treatment of generalized tonic-clonic status epilepticus and prevention and treatment of seizures occurring during neurosurgery in all pediatric age groups [see Indications and Usage (1) and Dosage and Administration (2.3, 2.4)]. Because rapid intravenous administration of fosphenytoin sodium injection increases the risk of adverse cardiovascular reactions, the rate of administration should not exceed 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients [see Dosage and Administration (2.3, 2.4) and Warnings and Precautions (5.2)].
8.5 Geriatric Use
5.16 Hyperglycemia
Hyperglycemia, resulting from the inhibitory effect of phenytoin (the active metabolite of fosphenytoin sodium injection) on insulin release, has been reported. Phenytoin may also raise the serum glucose concentrations in diabetic patients.
5.8 Hepatic Injury
Cases of acute hepatotoxicity, including infrequent cases of acute hepatic failure, have been reported with phenytoin (the active metabolite of fosphenytoin sodium injection). These events may be part of the spectrum of DRESS or may occur in isolation [see Warnings and Precautions (5.5)]. Other common manifestations include jaundice, hepatomegaly, elevated serum transaminase levels, leukocytosis, and eosinophilia. The clinical course of acute phenytoin hepatotoxicity ranges from prompt recovery to fatal outcomes. In these patients with acute hepatotoxicity, fosphenytoin sodium injection should be immediately discontinued and not re-administered.
14 Clinical Studies
Infusion tolerance was evaluated in clinical studies. One double-blind study assessed infusion-site tolerance of equivalent loading doses (15 to 20 mg PE/kg) of fosphenytoin sodium infused at 150 mg PE/min or phenytoin infused at 50 mg/min. The study demonstrated better local tolerance (pain and burning at the infusion site), fewer disruptions of the infusion, and a shorter infusion period for fosphenytoin sodium-treated patients (Table 8).
|
a Percent of patients |
||
| IV Fosphenytoin Sodium N=90 |
IV Phenytoin N=22 |
|
| Local Intolerance | 9%a | 90% |
| Infusion Disrupted | 21% | 67% |
| Average Infusion Time | 13 min | 44 min |
Fosphenytoin sodium-treated patients, however, experienced more systemic sensory disturbances [see Warnings and Precautions (5.10)]. Infusion disruptions in fosphenytoin sodium-treated patients were primarily due to systemic burning, pruritus, and/or paresthesia while those in phenytoin-treated patients were primarily due to pain and burning at the infusion site (see Table 8). In a double-blind study investigating temporary substitution of fosphenytoin sodium injection for oral phenytoin, IM fosphenytoin sodium was as well-tolerated as IM placebo. IM fosphenytoin sodium injection resulted in a slight increase in transient, mild to moderate local itching (23% of fosphenytoin sodium-treated patients vs 11% of IM placebo-treated patients at any time during the study). This study also demonstrated that equimolar doses of IM fosphenytoin sodium injection may be substituted for oral phenytoin sodium with no dosage adjustments needed when initiating IM or returning to oral therapy. In contrast, switching between IM and oral phenytoin requires dosage adjustments because of slow and erratic phenytoin absorption from muscle.
4 Contraindications
Fosphenytoin sodium injection is contraindicated in patients with:
- A history of hypersensitivity to fosphenytoin sodium injection or its inactive ingredients, or to phenytoin or other hydantoins [see Warnings and Precautions (5.6)]. Reactions have included angioedema.
- Sinus bradycardia, sino-atrial block, second and third degree A-V block, or Adams-Stokes syndrome because of the effect of parenteral phenytoin or fosphenytoin sodium injection on ventricular automaticity.
- A history of prior acute hepatotoxicity attributable to fosphenytoin sodium injection or phenytoin [see Warnings and Precautions (5.8)].
- Coadministration with delavirdine because of the potential for loss of virologic response and possible resistance to delavirdine or to the class of non-nucleoside reverse transcriptase inhibitors.
5.12 Phosphate Load
The phosphate load provided by fosphenytoin sodium injection (0.0037 mmol phosphate/mg PE fosphenytoin sodium injection) should be considered when treating patients who require phosphate restriction, such as those with severe renal impairment.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
- Cardiovascular Risk Associated with Rapid Infusion [see Warnings and Precautions (5.2)]
- Withdrawal Precipitated Seizure, Status Epilepticus [see Warnings and Precautions (5.3)]
- Serious Dermatologic Reactions [see Warnings and Precautions (5.4)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5)]
- Hypersensitivity [see Warnings and Precautions (5.6)]
- Angioedema [see Warnings and Precautions (5.7)]
- Hepatic Injury [see Warnings and Precautions (5.8)]
- Hematopoietic Complications [see Warnings and Precautions (5.9)]
- Sensory Disturbances [see Warnings and Precautions (5.10)]
- Local Toxicity (Including Purple Glove Syndrome) [see Warnings and Precautions (5.11)]
- Exacerbation of Porphyria [see Warnings and Precautions (5.14)]
- Teratogenicity and Other Harm to the Newborn [see Warnings and Precautions (5.15)]
- Hyperglycemia [see Warnings and Precautions (5.16)]
7 Drug Interactions
Fosphenytoin is extensively bound to human plasma proteins. Drugs highly bound to albumin could increase the unbound fraction of fosphenytoin. Although, it is unknown whether this could result in clinically significant effects, caution is advised when administering fosphenytoin sodium injection with other drugs that significantly bind to serum albumin. The most significant drug interactions following administration of fosphenytoin sodium injection are expected to occur with drugs that interact with phenytoin. Phenytoin is extensively bound to serum plasma proteins and is prone to competitive displacement. Phenytoin is primarily metabolized by the hepatic cytochrome P450 enzyme CYP2C9 and to a lesser extent by CYP2C19 and is particularly susceptible to inhibitory drug interactions because it is subject to saturable metabolism. Inhibition of metabolism may produce significant increases in circulating phenytoin concentrations and enhance the risk of drug toxicity. Monitoring of phenytoin serum levels is recommended when a drug interaction is suspected.
Phenytoin or fosphenytoin sodium injection is a potent inducer of hepatic drug-metabolizing enzymes.
5.6 Hypersensitivity
Fosphenytoin sodium and other hydantoins are contraindicated in patients who have experienced phenytoin hypersensitivity [see Contraindications (4) and Warnings and Precautions (5.7)]. Additionally, consider alternatives to structurally similar drugs such as carboxamides (e.g., carbamazepine), barbiturates, succinimides, and oxazolidinediones (e.g., trimethadione) in these same patients. Similarly, if there is a history of hypersensitivity reactions to these structurally similar drugs in the patient or immediate family members, consider alternatives to fosphenytoin sodium injection.
12.5 Pharmacogenomics
CYP2C9 activity is decreased in individuals with genetic variants such as the CYP2C9*2 and CYP2C9*3 alleles. Carriers of variant alleles, resulting in intermediate (e.g., *1/*3, *2/*2) or poor metabolism (e.g., *2/*3, *3/*3) have decreased clearance of phenytoin. Other decreased or nonfunctional CYP2C9 alleles may also result in decreased clearance of phenytoin (e.g., *5, *6, *8, *11).
The prevalence of the CYP2C9 poor metabolizer phenotype is approximately 2-3% in the White population, 0.5-4% in the Asian population, and <1% in the African American population. The CYP2C9 intermediate phenotype prevalence is approximately 35% in the White population, 24% in the African American population, and 15-36% in the Asian population [see Warnings and Precautions (5.4) and Use in Specific Populations (8.7)].
2.3 Status Epilepticus
- Because of the risk of hypotension and cardiac arrhythmias, the rate of administration for IV fosphenytoin sodium injection should be no greater than 150 mg PE/min in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients [see Warnings and Precautions (5.2)]. Continuous monitoring of the electrocardiogram, blood pressure, and respiratory function is essential, and the patient should be observed throughout the period where maximal serum phenytoin concentrations occur, approximately 10 to 20 minutes after the end of fosphenytoin sodium infusions.
- Because the full antiepileptic effect of phenytoin, whether given as fosphenytoin sodium injection or parenteral phenytoin, is not immediate, other measures, including concomitant administration of an IV benzodiazepine, will usually be necessary for the control of status epilepticus.
- The loading dose should be followed by maintenance doses of either fosphenytoin sodium injection or phenytoin [see Dosage and Administration (2.4)].
- If administration of fosphenytoin sodium injection does not terminate seizures, the use of other anticonvulsants and other appropriate measures should be considered.
1 Indications and Usage
Fosphenytoin sodium injection is indicated for the treatment of generalized tonic-clonic status epilepticus and prevention and treatment of seizures occurring during neurosurgery. Fosphenytoin sodium injection can also be substituted, short-term, for oral phenytoin. Fosphenytoin sodium injection should be used only when oral phenytoin administration is not possible [see Dosage and Administration (2.4) and Warnings and Precautions (5.2)].
12.1 Mechanism of Action
Fosphenytoin is a prodrug of phenytoin and accordingly, its anticonvulsant effects are attributable to phenytoin. The precise mechanism by which phenytoin exerts its therapeutic effect has not been established but is thought to involve the voltage-dependent blockade of membrane sodium channels resulting in a reduction in sustained high-frequency neuronal discharges.
2.8 Dosing in Geriatrics
The clearance of phenytoin (the active metabolite of fosphenytoin sodium injection) is decreased slightly in elderly patients and lower or less frequent dosing may be required [see Clinical Pharmacology (12.3)].
16.2 Storage and Handling
Store under refrigeration at 2°C to 8°C (36°F to 46°F). The product should not be stored at room temperature for more than 48 hours. Vials that develop particulate matter should not be used.
Injection vials are single-dose only. After opening, any unused product should be discarded.
5.10 Sensory Disturbances
Severe burning, itching, and/or paresthesia were reported by 7 of 16 normal volunteers administered IV fosphenytoin sodium injection at a dose of 1,200 mg PE at the maximum rate of administration (150 mg PE/min). The severe sensory disturbance lasted from 3 to 50 minutes in 6 of these subjects and for 14 hours in the seventh subject. In some cases, milder sensory disturbances persisted for as long as 24 hours. The location of the discomfort varied among subjects with the groin mentioned most frequently as an area of discomfort. In a separate cohort of 16 normal volunteers (taken from 2 other studies) who were administered IV fosphenytoin sodium injection at a dose of 1,200 mg PE at the maximum rate of administration (150 mg PE/min), none experienced severe disturbances, but most experienced mild to moderate itching or tingling. Patients administered fosphenytoin sodium injection at doses of 20 mg PE/kg at 150 mg PE/min are expected to experience discomfort of some degree. The occurrence and intensity of the discomfort can be lessened by slowing or temporarily stopping the infusion. The effect of continuing infusion unaltered in the presence of these sensations is unknown. No permanent sequelae have been reported thus far. The pharmacologic basis for these positive sensory phenomena is unknown, but other phosphate ester drugs, which deliver smaller phosphate loads, have been associated with burning, itching, and/or tingling predominantly in the groin area.
5 Warnings and Precautions
- Dosing Errors: Do not confuse the amount of drug to be given in PE with the concentration of the drug in the vial. Ensure the appropriate volume is withdrawn from the vial when preparing for administration. (5.1)
- Withdrawal Precipitated Seizure: May precipitate status epilepticus. Dose reductions or discontinuation should be done gradually. (5.3)
- Serious Dermatologic Reactions: Discontinue at the first sign of a rash, unless clearly not drug-related. If signs or symptoms suggest SJS/TEN, fosphenytoin sodium injection should not be resumed; consider alternative therapy. (5.4)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: If signs or symptoms of hypersensitivity are present, evaluate the patient immediately. Discontinue if an alternative etiology cannot be established. (5.5)
- Angioedema: Discontinue immediately if symptoms of angioedema such as facial, perioral, or upper airway swelling occur. (5.7)
- Hematopoietic Complications: If occurs, follow-up observation is indicated and an alternative antiepileptic treatment should be used. (5.9)
2 Dosage and Administration
- The dose, concentration, and infusion rate of fosphenytoin sodium injection should always be expressed as phenytoin sodium equivalents (PE) (2.1)
- For Status Epilepticus:
-
For Non-emergent Loading and Maintenance Dosing:
- Adult loading dose is 10 to 20 mg PE/kg given IV or IM; initial maintenance dose is 4 to 6 mg PE/kg/day in divided doses (2.4)
- Pediatric loading dose is 10 to 15 mg PE/kg at a rate of 1 to 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower); initial maintenance dose is 2 to 4 mg PE/kg every 12 hours at a rate of 1 to 2 mg PE/kg/min (or 100 mg PE/min, whichever is slower) (2.4)
- Intramuscular Administration:
2.9 Dosing During Pregnancy
Decreased serum concentrations of phenytoin (the active metabolite of fosphenytoin sodium injection) may occur during pregnancy because of altered phenytoin pharmacokinetics [see Clinical Pharmacology (12.3)]. Periodic measurement of serum phenytoin concentrations should be performed during pregnancy, and the fosphenytoin sodium injection dosage should be adjusted as necessary. Postpartum restoration of the original dosage will probably be indicated [see Use in Specific Populations (8.1)]. Because of potential changes in protein binding during pregnancy, the monitoring of phenytoin serum levels should be based on the unbound fraction.
3 Dosage Forms and Strengths
Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow solution available as 50 mg phenytoin sodium equivalents (PE) per mL in:
- 10 mL single-dose injection vials, each containing 500 mg PE/10 mL (50 mg PE/mL)
- 2 mL single-dose injection vials, each containing 100 mg PE/2 mL (50 mg PE/mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of phenytoin or fosphenytoin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole: Anaphylaxis, angioedema [see Warnings and Precautions (5.7)]
Hematologic: Pure red cell aplasia [see Warnings and Precautions (5.9)]
Laboratory Test Abnormality: Phenytoin or fosphenytoin sodium injection may decrease serum concentrations of T4. It may also produce lower than normal values for dexamethasone or metyrapone tests. Phenytoin may also cause increased serum levels of gamma glutamyl transpeptidase (GGT).
Nervous System Disorders: Dyskinesia
8 Use in Specific Populations
5.14 Exacerbation of Porphyria
In view of isolated reports associating phenytoin (the active metabolite of fosphenytoin sodium injection) with exacerbation of porphyria, caution should be exercised in using fosphenytoin sodium injection in patients suffering from this disease.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The more important adverse clinical reactions caused by the IV use of fosphenytoin sodium injection or phenytoin are cardiovascular collapse and/or CNS depression. Hypotension can occur when either drug is administered rapidly by the IV route. The rate of administration is very important; for fosphenytoin sodium injection, it should not exceed 150 mg PE/min [see Warnings and Precautions (5.2)]. The adverse reactions most commonly observed with the use of fosphenytoin sodium injection in clinical trials were nystagmus, dizziness, pruritus, somnolence, and ataxia. With one exception, these reactions are commonly associated with the administration of IV phenytoin. Pruritus, however, was seen much more often following fosphenytoin sodium injection administration and occurred more often with IV fosphenytoin sodium administration than with IM fosphenytoin sodium administration. These reactions were dose and rate related; most alert patients (41 of 64; 64%) administered doses of ≥15 mg PE/kg at 150 mg PE/min experienced discomfort of some degree. These sensations, generally described as itching, burning, or tingling, were usually not at the infusion site. The location of the discomfort varied with the groin mentioned most frequently as a site of involvement. The paresthesia and pruritus were transient events that occurred within several minutes of the start of infusion and generally resolved within 10 minutes after completion of fosphenytoin sodium infusion. Some patients experienced symptoms for hours. These reactions did not increase in severity with repeated administration. Concurrent adverse events or clinical laboratory change suggesting an allergic process were not seen [see Warnings and Precautions (5.10)]. Approximately 2% of the 859 patients who received fosphenytoin sodium injection in premarketing clinical trials discontinued treatment because of an adverse event. The adverse events most commonly associated with withdrawal were pruritus (0.5%), hypotension (0.3%), and bradycardia (0.2%).
Dose and Rate Dependency of Adverse Reactions Following IV Fosphenytoin Sodium Injection: The incidence of adverse reactions tended to increase as both dose and infusion rate increased. In particular, at doses of ≥15mg PE/kg and rates ≥150 mg PE/min, transient pruritus, tinnitus, nystagmus, somnolence, and ataxia occurred 2 to 3 times more often than at lower doses or rates.
5.9 Hematopoietic Complications
Hematopoietic complications, some fatal, have occasionally been reported in association with administration of phenytoin (the active metabolite of fosphenytoin sodium injection). These have included thrombocytopenia, leukopenia, granulocytopenia, agranulocytosis, and pancytopenia with or without bone marrow suppression.
There have been a number of reports that have suggested a relationship between phenytoin and the development of lymphadenopathy (local or generalized), including benign lymph node hyperplasia, pseudolymphoma, lymphoma, and Hodgkin's disease. Although a cause and effect relationship has not been established, the occurrence of lymphadenopathy indicates the need to differentiate such a condition from other types of lymph node pathology. Lymph node involvement may occur with or without symptoms and signs resembling DRESS [see Warnings and Precautions (5.5)].
In all cases of lymphadenopathy, follow-up observation for an extended period is indicated and every effort should be made to achieve seizure control using alternative antiepileptic drugs. Macrocytosis and megaloblastic anemia have occurred, these conditions usually respond to folic acid therapy. Pure red cell aplasia has also been reported with phenytoin.
5.4 Serious Dermatologic Reactions
Fosphenytoin sodium can cause severe cutaneous adverse reactions (SCARs), which may be fatal. Reported reactions in phenytoin (the active metabolite of fosphenytoin sodium)-treated patients have included toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), acute generalized exanthematous pustulosis (AGEP), and Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.5)]. The onset of symptoms is usually within 28 days, but can occur later. Fosphenytoin sodium injection should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest a severe cutaneous adverse reaction, use of this drug should not be resumed and alternative therapy should be considered. If a rash occurs, the patient should be evaluated for signs and symptoms of SCARs.
Studies in patients of Chinese ancestry have found a strong association between the risk of developing SJS/TEN and the presence of HLA-B*1502, an inherited allelic variant of the HLA B gene, in patients using carbamazepine. Limited evidence suggests that HLA-B*1502 may be a risk factor for the development of SJS/TEN in patients of Asian ancestry taking other antiepileptic drugs associated with SJS/TEN, including phenytoin. In addition, retrospective, case-control, genome-wide association studies in patients of southeast Asian ancestry have also identified an increased risk of SCARs in carriers of the decreased function CYP2C9*3 variant, which has also been associated with decreased clearance of phenytoin. Consider avoiding fosphenytoin sodium injection as an alternative to carbamazepine in patients who are positive for HLA-B*1502 or in CYP2C9*3 carriers.
Should fosphenytoin sodium injection be utilized for CYP2C9*3 carriers, consider starting at the lower end of the dosage range [see Use in Specific Populations (8.7)].
The use of HLA-B*1502 or CYP2C9 genotyping has important limitations and must never substitute for appropriate clinical vigilance and patient management. The role of other possible factors in the development of, and morbidity from, SJS/TEN, such as antiepileptic drug (AED) dose, compliance, concomitant medications, comorbidities, and the level of dermatologic monitoring have not been studied.
7.4 Drug/laboratory Test Interactions
Care should be taken when using immunoanalytical methods to measure serum phenytoin concentrations following fosphenytoin sodium injection administration.
2.4 Non Emergent Loading and Maintenance Dosing
- Because of the risk of hypotension and cardiac arrhythmias, the rate of administration for IV fosphenytoin sodium injection should be no greater than 150 mg PE/min in adults. For loading doses in pediatric patients, the rate should not exceed 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower). For maintenance doses in pediatric patients, the rate should not exceed 1 to 2 mg PE/kg/min (or 100 mg PE/min, whichever is slower). Continuous monitoring of the electrocardiogram, blood pressure, and respiratory function is essential, and the patient should be observed throughout the period where maximal serum phenytoin concentrations occur (approximately 10 to 20 minutes after the end of fosphenytoin sodium infusions).
- After the initial maintenance dose, subsequent maintenance doses should be individualized by monitoring serum phenytoin concentrations to achieve a target therapeutic concentration of phenytoin [see Dosage and Administration (2.5) and Warnings and Precautions (5.17)].
5.13 Renal Or Hepatic Disease Or Hypoalbuminemia
Because the fraction of unbound phenytoin (the active metabolite of fosphenytoin sodium injection) is increased in patients with renal or hepatic disease, or in those with hypoalbuminemia, the monitoring of phenytoin serum levels should be based on the unbound fraction in those patients. After IV administration to patients with renal and/or hepatic disease, or in those with hypoalbuminemia, fosphenytoin clearance to phenytoin may be increased without a similar increase in phenytoin clearance. This has the potential to increase the frequency and severity of adverse events.
5.15 Teratogenicity and Other Harm to the Newborn
Fosphenytoin sodium injection may cause fetal harm when administered to a pregnant woman. Prenatal exposure to phenytoin (the active metabolite of fosphenytoin sodium injection) may increase the risks for congenital malformations and other adverse developmental outcomes [see Use in Specific Populations (8.1)].
Increased frequencies of major malformations (such as orofacial clefts and cardiac defects), and abnormalities characteristic of fetal hydantoin syndrome, including dysmorphic skull and facial features, nail and digit hypoplasia, growth abnormalities (including microcephaly), and cognitive deficits, have been reported among children born to epileptic women who took phenytoin alone or in combination with other antiepileptic drugs during pregnancy. There have been several reported cases of malignancies, including neuroblastoma.
A potentially life-threatening bleeding disorder related to decreased levels of vitamin K-dependent clotting factors may occur in newborns exposed to phenytoin in utero. This drug-induced condition can be prevented with vitamin K administration to the mother before delivery and to the neonate after birth.
8.7 Use in Patients With Decreased Cyp2c9 Function
Patients who are intermediate or poor metabolizers of CYP2C9 substrates (e.g., *1/*3, *2/*2, *3/*3) may exhibit increased phenytoin serum concentrations compared to patients who are normal metabolizers (e.g., *1/*1). Thus, patients who are known to be intermediate or poor metabolizers may ultimately require lower doses to maintain similar steady-state concentrations compared to normal metabolizers. In patients who are known to be carriers of the decreased function CYP2C9*2 or *3 alleles (intermediate and poor metabolizers), consider starting at the low end of the dosage range and monitor serum concentrations to maintain total phenytoin concentrations of 10 to 20 mcg/mL. If early signs of dose-related central nervous system (CNS) toxicity develop, serum concentrations should be checked immediately [see Clinical Pharmacology (12.5)].
5.17 Serum Phenytoin Levels Above Therapeutic Range
Serum levels of phenytoin (the active metabolite of fosphenytoin sodium injection) sustained above the therapeutic range may produce confusional states referred to as “delirium,” “psychosis,” or “encephalopathy,” or rarely, irreversible cerebellar dysfunction and/or cerebellar atrophy. Accordingly, at the first sign of acute toxicity, serum levels should be immediately checked. Fosphenytoin sodium injection dose reduction is indicated if serum levels are excessive; if symptoms persist, administration of fosphenytoin sodium injection should be discontinued.
7.3 Hyperammonemia With Concomitant Use of Valproate
Concomitant administration of phenytoin and valproate has been associated with an increased risk of valproate-associated hyperammonemia. Patients treated concomitantly with these two drugs should be monitored for signs and symptoms of hyperammonemia.
5.11 Local Toxicity (including Purple Glove Syndrome)
Edema, discoloration, and pain distal to the site of injection (described as “purple glove syndrome”) have also been reported following peripheral intravenous fosphenytoin sodium injection. This may or may not be associated with extravasation. The syndrome may not develop for several days after injection.
2.6 Parenteral Substitution for Oral Phenytoin Therapy
When treatment with oral phenytoin is not possible, fosphenytoin sodium injection can be substituted for oral phenytoin at the same total daily phenytoin sodium equivalents (PE) dose. Phenytoin sodium capsules are approximately 90% bioavailable by the oral route. Phenytoin, derived from administration of fosphenytoin sodium injection, is 100% bioavailable by both the IM and IV routes. For this reason, serum phenytoin concentrations may increase modestly when IM or IV fosphenytoin sodium injection is substituted for oral phenytoin sodium therapy. The rate of administration for IV fosphenytoin sodium injection should be no greater than 150 mg PE/min in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients. In controlled trials, IM fosphenytoin sodium injection was administered as a single daily dose utilizing either 1 or 2 injection sites. Some patients may require more frequent dosing. Intramuscular administration of fosphenytoin sodium injection should ordinarily not be used in pediatric patients.
5.2 Cardiovascular Risk Associated With Rapid Infusion
Rapid intravenous administration of fosphenytoin sodium injection increases the risk of adverse cardiovascular reactions, including severe hypotension and cardiac arrhythmias. Cardiac arrhythmias have included bradycardia, heart block, QT interval prolongation, ventricular tachycardia, and ventricular fibrillation which have resulted in asystole, cardiac arrest, and death. Severe complications are most commonly encountered in critically ill patients, elderly patients, and patients with hypotension and severe myocardial insufficiency. However, cardiac events have also been reported in adults and children without underlying cardiac disease or comorbidities and at recommended doses and infusion rates.
The rate of intravenous fosphenytoin sodium injection administration should not exceed 150 mg phenytoin sodium equivalents (PE) per minute in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients [see Dosage and Administration (2.3, 2.4)].
Although the risk of cardiovascular toxicity increases with infusion rates above the recommended infusion rate, these events have also been reported at or below the recommended infusion rate.
As non-emergency therapy, intravenous fosphenytoin sodium injection should be administered more slowly. Because of the risks of cardiac and local toxicity associated with IV fosphenytoin sodium injection, oral phenytoin should be used whenever possible.
Because adverse cardiovascular reactions have occurred during and after infusions, careful cardiac and respiratory monitoring is needed during and after the administration of intravenous fosphenytoin sodium injection. Reduction in rate of administration or discontinuation of dosing may be needed.
5.3 Withdrawal Precipitated Seizure, Status Epilepticus
Antiepileptic drugs should not be abruptly discontinued because of the possibility of increased seizure frequency, including status epilepticus. When, in the judgment of the clinician, the need for dosage reduction, discontinuation, or substitution of alternative antiepileptic medication arises, this should be done gradually. However, in the event of an allergic or hypersensitivity reaction, rapid substitution of alternative therapy may be necessary. In this case, alternative therapy should be an antiepileptic drug not belonging to the hydantoin chemical class.
8.6 Renal And/or Hepatic Impairment, Or Hypoalbuminemia
The liver is the site of biotransformation. Patients with impaired liver function, elderly patients, or those who are gravely ill may show early toxicity.
Because the fraction of unbound phenytoin (the active metabolite of fosphenytoin sodium injection) is increased in patients with renal or hepatic disease, or in those with hypoalbuminemia, the monitoring of phenytoin serum levels should be based on the unbound fraction in those patients.
After IV administration to patients with renal and/or hepatic disease, or in those with hypoalbuminemia, fosphenytoin clearance to phenytoin may be increased without a similar increase in phenytoin clearance. This has the potential to increase the frequency and severity of adverse events.
2.1 Important Administration Instructions to Avoid Dosing Errors
Use caution when administering fosphenytoin sodium injection because of the risk of dosing errors [see Warnings and Precautions (5.1)].
7.1 Drugs That Affect Phenytoin Or Fosphenytoin Sodium Injection
Table 6 includes commonly occurring drug interactions that affect phenytoin (the active metabolite of fosphenytoin sodium injection) concentrations. However, this list is not intended to be inclusive or comprehensive. Individual prescribing information from relevant drugs should be consulted.
The addition or withdrawal of these agents in patients on phenytoin therapy may require an adjustment of the phenytoin dose to achieve optimal clinical outcome.
|
a The induction potency of St. John's wort may vary widely based on preparation. |
|
| Interacting Agent | Examples |
| Drugs that may increase phenytoin serum levels | |
| Antiepileptic drugs | Ethosuximide, felbamate, oxcarbazepine, methsuximide, topiramate |
| Azoles | Fluconazole, ketoconazole, itraconazole, miconazole, voriconazole |
| Antineoplastic agents | Capecitabine, fluorouracil |
| Antidepressants | Fluoxetine, fluvoxamine, sertraline |
| Gastric acid reducing agents |
H2 antagonists (cimetidine), omeprazole |
|
Sulfonamides |
Sulfamethizole, sulfaphenazole, sulfadiazine, sulfamethoxazole- trimethoprim |
|
Other |
Acute alcohol intake, amiodarone, chloramphenicol, chlordiazepoxide, disulfiram, estrogen, fluvastatin, isoniazid, methylphenidate, phenothiazines, salicylates, ticlopidine, tolbutamide, trazodone, warfarin |
| Drugs that may decrease phenytoin serum levels | |
| Antineoplastic agents usually in combination |
Bleomycin, carboplatin, cisplatin, doxorubicin, methotrexate |
| Antiviral agents | Fosamprenavir, nelfinavir, ritonavir |
| Antiepileptic drugs | Carbamazepine, vigabatrin |
|
Other |
Chronic alcohol abuse, diazepam, diazoxide, folic acid, reserpine, rifampin, St. John's wort,a theophylline |
| Drugs that may either increase or decrease phenytoin serum levels | |
| Antiepileptic drugs | Phenobarbital, valproate sodium, valproic acid |
7.2 Drugs Affected By Phenytoin Or Fosphenytoin Sodium Injection
Table 7 includes commonly occurring drug interactions affected by phenytoin (the active metabolite of fosphenytoin sodium injection). However, this list is not intended to be inclusive or comprehensive. Individual drug package inserts should be consulted. The addition or withdrawal of phenytoin during concomitant therapy with these agents may require adjustment of the dose of these agents to achieve optimal clinical outcome.
|
a The effect of phenytoin on phenobarbital, valproic acid and sodium valproate serum levels is unpredictable. |
|
| Interacting Agent | Examples |
| Drugs whose efficacy is impaired by phenytoin | |
| Azoles | Fluconazole, ketoconazole, itraconazole, posaconazole, voriconazole |
| Antineoplastic agents | Irinotecan, paclitaxel, teniposide |
|
Delavirdine |
Phenytoin can substantially reduce the concentrations of delavirdine. This can lead to loss of virologic response and possible resistance [see Contraindications (4)]. |
|
Neuromuscular blocking agents |
Cisatracurium, pancuronium, rocuronium and vecuronium: resistance to the neuromuscular blocking action of the nondepolarizing neuromuscular blocking agents has occurred in patients chronically administered phenytoin. Whether or not phenytoin has the same effect on other non-depolarizing agents is unknown. Prevention or Management: Patients should be monitored closely for more rapid recovery from neuromuscular blockade than expected, and infusion rate requirements may be higher. |
|
Warfarin |
Increased and decreased PT/INR responses have been reported when phenytoin is coadministered with warfarin. |
|
Other |
Corticosteroids, doxycycline, estrogens, furosemide, oral contraceptives, paroxetine, quinidine, rifampin, sertraline, theophylline, and vitamin D |
| Drugs whose level is decreased by phenytoin | |
| Anticoagulants | Apixaban, dabigatran, edoxaban, rivaroxaban |
| Antiepileptic drugsa | Carbamazepine, felbamate, lamotrigine, topiramate, oxcarbazepine, lacosamide |
| Antilipidemic agents | Atorvastatin, fluvastatin, simvastatin |
| Antiplatelets | Ticagrelor |
|
Antiviral agents |
Efavirenz, lopinavir/ritonavir, indinavir, nelfinavir, ritonavir, saquinavir Fosamprenavir: phenytoin when given with fosamprenavir alone may decrease the concentration of amprenavir, the active metabolite. Phenytoin when given with the combination of fosamprenavir and ritonavir may increase the concentration of amprenavir |
| Calcium channel blockers | Nifedipine, nimodipine, nisoldipine, verapamil |
| Other | Albendazole (decreases active metabolite), chlorpropamide, clozapine, cyclosporine, digoxin, disopyramide, folic acid, methadone, mexiletine, praziquantel, quetiapine |
Warning: Cardiovascular Risk Associated With Rapid Infusion Rates
The rate of intravenous fosphenytoin sodium injection administration should not exceed 150 mg phenytoin sodium equivalents (PE) per minute in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients because of the risk of severe hypotension and cardiac arrhythmias. Careful cardiac monitoring is needed during and after administering intravenous fosphenytoin sodium. Although the risk of cardiovascular toxicity increases with infusion rates above the recommended infusion rate, these events have also been reported at or below the recommended infusion rate. Reduction in rate of administration or discontinuation of dosing may be needed [see Dosage and Administration (2.3, 2.4) and Warnings and Precautions (5.2)].
2.7 Dosing in Patients With Renal Or Hepatic Impairment Or Hypoalbuminemia
Because the fraction of unbound phenytoin (the active metabolite of fosphenytoin sodium injection) is increased in patients with renal or hepatic disease, or in those with hypoalbuminemia, the monitoring of phenytoin serum levels should be based on the unbound fraction in those patients. After IV fosphenytoin sodium administration to patients with renal and/or hepatic disease, or in those with hypoalbuminemia, fosphenytoin clearance to phenytoin may be increased without a similar increase in phenytoin clearance. This has the potential to increase the frequency and severity of adverse events [see Warnings and Precautions (5.13)].
5.5 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as Multiorgan hypersensitivity, has been reported in patients taking antiepileptic drugs, including phenytoin and fosphenytoin sodium. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Fosphenytoin sodium injection should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
Structured Label Content
Section 42229-5 (42229-5)
Phenytoin Sodium Equivalents (PE)
The dose, concentration, and infusion rate of fosphenytoin sodium injection should always be expressed as phenytoin sodium equivalents (PE). There is no need to perform molecular weight-based adjustments when converting between fosphenytoin and phenytoin sodium doses. Fosphenytoin sodium injection should always be prescribed and dispensed in phenytoin sodium equivalent units (PE). The amount and concentration of fosphenytoin is always expressed in terms of mg of phenytoin sodium equivalents (mg PE).
Section 43683-2 (43683-2)
| Warnings and Precautions (5.9) | 4/2022 |
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY - Fosphenytoin 2 mL Single Dose Vial Label
NDC 63323-403-01 400302
Fosphenytoin Sodium Injection, USP
100 mg PE per 2 mL (50 mg PE per mL)
(PE = phenytoin sodium equivalents)
2 mL Single Dose Vial Rx only
10 Overdosage (10 OVERDOSAGE)
Nausea, vomiting, lethargy, tachycardia, bradycardia, asystole, cardiac arrest, hypotension, syncope, hypocalcemia, metabolic acidosis, and death have been reported in cases of overdosage with fosphenytoin sodium injection.
Because fosphenytoin sodium injection is a prodrug of phenytoin, the following information about phenytoin overdosage may be helpful. Initial symptoms of acute phenytoin toxicity are nystagmus, ataxia, and dysarthria. Other signs include tremor, hyperreflexia, lethargy, slurred speech, nausea, vomiting, coma, and hypotension. Death is caused by respiratory and circulatory depression. The lethal dose of phenytoin in adults is estimated to be 2 to 5 grams. The lethal dose in pediatrics is not known.
There are marked variations among individuals with respect to serum phenytoin concentrations where toxicity occurs. Lateral gaze nystagmus usually appears at 20 mcg/mL, ataxia at 30 mcg/mL, and dysarthria and lethargy appear when the serum concentration is over 40 mcg/mL. However, phenytoin concentrations as high as 50 mcg/mL have been reported without evidence of toxicity. As much as 25 times the therapeutic phenytoin dose has been taken, resulting in serum phenytoin concentrations over 100 mcg/mL, with complete recovery. Irreversible cerebellar dysfunction and atrophy have been reported after overdosage.
Formate and phosphate are metabolites of fosphenytoin sodium injection and therefore may contribute to signs of toxicity following overdosage. Signs of formate toxicity are similar to those of methanol toxicity and are associated with severe anion-gap metabolic acidosis. Large amounts of phosphate, delivered rapidly, could potentially cause hypocalcemia with paresthesia, muscle spasms, and seizures. Ionized free calcium levels can be measured and, if low, used to guide treatment.
Treatment: Treatment is nonspecific since there is no known antidote to fosphenytoin sodium injection or phenytoin overdosage.
The adequacy of the respiratory and circulatory systems should be carefully observed, and appropriate supportive measures employed. Hemodialysis can be considered since phenytoin (the active metabolite of fosphenytoin sodium injection) is not completely bound to plasma proteins. Total exchange transfusion has been used in the treatment of severe intoxication in children.
In acute overdosage the possibility of other CNS depressants, including alcohol, should be borne in mind.
11 Description (11 DESCRIPTION)
Fosphenytoin Sodium Injection, USP is a prodrug intended for parenteral administration; its active metabolite is phenytoin. 1.5 mg of fosphenytoin sodium is equivalent to 1 mg phenytoin sodium, and is referred to as 1 mg phenytoin sodium equivalents (PE). The amount and concentration of fosphenytoin is always expressed in terms of mg PE.
The pharmacological class of the fosphenytoin sodium is hydantoin derivative, and the therapeutic class is anticonvulsant.
Fosphenytoin sodium injection, USP is marketed as a 2 mL fill in 5 mL vials containing a total of 100 mg PE/2 mL (50 mg PE/mL) and 10 mL vials containing a total of 500 mg PE/10 mL (50 mg PE/mL), for intravenous or intramuscular administration. The concentration of each vial is 50 mg PE/mL. Fosphenytoin Sodium Injection, USP is supplied in vials as a sterile solution in Water for Injection, USP, and Tromethamine, USP (TRIS) (12.11 mg/mL), buffer adjusted to pH 8.6 to 9.0 with either Hydrochloric Acid, NF, or Sodium Hydroxide, NF. Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow, sterile solution.
The chemical name of fosphenytoin is 5,5-diphenyl-3-[(phosphonooxy)methyl]-2,4-imidazolidinedione disodium salt. The molecular structure of fosphenytoin is:
The molecular weight of fosphenytoin is 406.24.
5.7 Angioedema
Angioedema has been reported in patients treated with phenytoin and fosphenytoin sodium injection in the postmarketing setting. Fosphenytoin sodium injection should be discontinued immediately if symptoms of angioedema, such as facial, perioral, or upper airway swelling occur. Fosphenytoin sodium injection should be discontinued permanently if a clear alternative etiology for the reaction cannot be established.
2.2 Preparation
Prior to intravenous (IV) infusion, dilute fosphenytoin sodium injection in 5% Dextrose Injection or 0.9% Sodium Chloride Injection to a concentration ranging from 1.5 to 25 mg PE/mL. The maximum concentration of fosphenytoin sodium in any solution should be 25 mg PE/mL. When fosphenytoin sodium injection is given as an IV infusion, fosphenytoin sodium injection needs to be diluted and should only be administered at a rate not exceeding 150 mg PE/min.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
For single-dose only. After opening, any unused product should be discarded.
16.1 How Supplied
Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow solution supplied as follows:
|
Product
Code |
Unit of Sale |
Strength |
Each |
| 400302 | NDC 63323-403-02 Unit of 25 |
100 mg PE per 2 mL (50 mg PE per mL) |
NDC 63323-403-01 2 mL fill in a 5 mL Single Dose Vial |
| 400310 | NDC 63323-403-10 Unit of 10 |
500 mg PE per 10 mL (50 mg PE per mL) |
NDC 63323-403-04 10 mL Single Dose Vial |
The container closure is not made with natural rubber latex.
Both sizes of vials contain Tromethamine, USP (TRIS), Hydrochloric Acid, NF, or Sodium Hydroxide, NF, and Water for Injection, USP.
Fosphenytoin sodium injection, USP should always be prescribed in phenytoin sodium equivalents (PE) [see Dosage and Administration (2.1) and Warnings and Precautions (5.1)].
1.5 mg of fosphenytoin sodium is equivalent to 1 mg phenytoin sodium, and is referred to as 1 mg PE. The amount and concentration of fosphenytoin is always expressed in terms of mg of phenytoin sodium equivalents (PE). Fosphenytoin's weight is expressed as phenytoin sodium equivalents to avoid the need to perform molecular weight-based adjustments when substituting fosphenytoin for phenytoin or vice versa.
8.4 Pediatric Use
Fosphenytoin sodium injection is indicated for the treatment of generalized tonic-clonic status epilepticus and prevention and treatment of seizures occurring during neurosurgery in all pediatric age groups [see Indications and Usage (1) and Dosage and Administration (2.3, 2.4)]. Because rapid intravenous administration of fosphenytoin sodium injection increases the risk of adverse cardiovascular reactions, the rate of administration should not exceed 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients [see Dosage and Administration (2.3, 2.4) and Warnings and Precautions (5.2)].
8.5 Geriatric Use
5.16 Hyperglycemia
Hyperglycemia, resulting from the inhibitory effect of phenytoin (the active metabolite of fosphenytoin sodium injection) on insulin release, has been reported. Phenytoin may also raise the serum glucose concentrations in diabetic patients.
5.8 Hepatic Injury
Cases of acute hepatotoxicity, including infrequent cases of acute hepatic failure, have been reported with phenytoin (the active metabolite of fosphenytoin sodium injection). These events may be part of the spectrum of DRESS or may occur in isolation [see Warnings and Precautions (5.5)]. Other common manifestations include jaundice, hepatomegaly, elevated serum transaminase levels, leukocytosis, and eosinophilia. The clinical course of acute phenytoin hepatotoxicity ranges from prompt recovery to fatal outcomes. In these patients with acute hepatotoxicity, fosphenytoin sodium injection should be immediately discontinued and not re-administered.
14 Clinical Studies (14 CLINICAL STUDIES)
Infusion tolerance was evaluated in clinical studies. One double-blind study assessed infusion-site tolerance of equivalent loading doses (15 to 20 mg PE/kg) of fosphenytoin sodium infused at 150 mg PE/min or phenytoin infused at 50 mg/min. The study demonstrated better local tolerance (pain and burning at the infusion site), fewer disruptions of the infusion, and a shorter infusion period for fosphenytoin sodium-treated patients (Table 8).
|
a Percent of patients |
||
| IV Fosphenytoin Sodium N=90 |
IV Phenytoin N=22 |
|
| Local Intolerance | 9%a | 90% |
| Infusion Disrupted | 21% | 67% |
| Average Infusion Time | 13 min | 44 min |
Fosphenytoin sodium-treated patients, however, experienced more systemic sensory disturbances [see Warnings and Precautions (5.10)]. Infusion disruptions in fosphenytoin sodium-treated patients were primarily due to systemic burning, pruritus, and/or paresthesia while those in phenytoin-treated patients were primarily due to pain and burning at the infusion site (see Table 8). In a double-blind study investigating temporary substitution of fosphenytoin sodium injection for oral phenytoin, IM fosphenytoin sodium was as well-tolerated as IM placebo. IM fosphenytoin sodium injection resulted in a slight increase in transient, mild to moderate local itching (23% of fosphenytoin sodium-treated patients vs 11% of IM placebo-treated patients at any time during the study). This study also demonstrated that equimolar doses of IM fosphenytoin sodium injection may be substituted for oral phenytoin sodium with no dosage adjustments needed when initiating IM or returning to oral therapy. In contrast, switching between IM and oral phenytoin requires dosage adjustments because of slow and erratic phenytoin absorption from muscle.
4 Contraindications (4 CONTRAINDICATIONS)
Fosphenytoin sodium injection is contraindicated in patients with:
- A history of hypersensitivity to fosphenytoin sodium injection or its inactive ingredients, or to phenytoin or other hydantoins [see Warnings and Precautions (5.6)]. Reactions have included angioedema.
- Sinus bradycardia, sino-atrial block, second and third degree A-V block, or Adams-Stokes syndrome because of the effect of parenteral phenytoin or fosphenytoin sodium injection on ventricular automaticity.
- A history of prior acute hepatotoxicity attributable to fosphenytoin sodium injection or phenytoin [see Warnings and Precautions (5.8)].
- Coadministration with delavirdine because of the potential for loss of virologic response and possible resistance to delavirdine or to the class of non-nucleoside reverse transcriptase inhibitors.
5.12 Phosphate Load
The phosphate load provided by fosphenytoin sodium injection (0.0037 mmol phosphate/mg PE fosphenytoin sodium injection) should be considered when treating patients who require phosphate restriction, such as those with severe renal impairment.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
- Cardiovascular Risk Associated with Rapid Infusion [see Warnings and Precautions (5.2)]
- Withdrawal Precipitated Seizure, Status Epilepticus [see Warnings and Precautions (5.3)]
- Serious Dermatologic Reactions [see Warnings and Precautions (5.4)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5)]
- Hypersensitivity [see Warnings and Precautions (5.6)]
- Angioedema [see Warnings and Precautions (5.7)]
- Hepatic Injury [see Warnings and Precautions (5.8)]
- Hematopoietic Complications [see Warnings and Precautions (5.9)]
- Sensory Disturbances [see Warnings and Precautions (5.10)]
- Local Toxicity (Including Purple Glove Syndrome) [see Warnings and Precautions (5.11)]
- Exacerbation of Porphyria [see Warnings and Precautions (5.14)]
- Teratogenicity and Other Harm to the Newborn [see Warnings and Precautions (5.15)]
- Hyperglycemia [see Warnings and Precautions (5.16)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Fosphenytoin is extensively bound to human plasma proteins. Drugs highly bound to albumin could increase the unbound fraction of fosphenytoin. Although, it is unknown whether this could result in clinically significant effects, caution is advised when administering fosphenytoin sodium injection with other drugs that significantly bind to serum albumin. The most significant drug interactions following administration of fosphenytoin sodium injection are expected to occur with drugs that interact with phenytoin. Phenytoin is extensively bound to serum plasma proteins and is prone to competitive displacement. Phenytoin is primarily metabolized by the hepatic cytochrome P450 enzyme CYP2C9 and to a lesser extent by CYP2C19 and is particularly susceptible to inhibitory drug interactions because it is subject to saturable metabolism. Inhibition of metabolism may produce significant increases in circulating phenytoin concentrations and enhance the risk of drug toxicity. Monitoring of phenytoin serum levels is recommended when a drug interaction is suspected.
Phenytoin or fosphenytoin sodium injection is a potent inducer of hepatic drug-metabolizing enzymes.
5.6 Hypersensitivity
Fosphenytoin sodium and other hydantoins are contraindicated in patients who have experienced phenytoin hypersensitivity [see Contraindications (4) and Warnings and Precautions (5.7)]. Additionally, consider alternatives to structurally similar drugs such as carboxamides (e.g., carbamazepine), barbiturates, succinimides, and oxazolidinediones (e.g., trimethadione) in these same patients. Similarly, if there is a history of hypersensitivity reactions to these structurally similar drugs in the patient or immediate family members, consider alternatives to fosphenytoin sodium injection.
12.5 Pharmacogenomics
CYP2C9 activity is decreased in individuals with genetic variants such as the CYP2C9*2 and CYP2C9*3 alleles. Carriers of variant alleles, resulting in intermediate (e.g., *1/*3, *2/*2) or poor metabolism (e.g., *2/*3, *3/*3) have decreased clearance of phenytoin. Other decreased or nonfunctional CYP2C9 alleles may also result in decreased clearance of phenytoin (e.g., *5, *6, *8, *11).
The prevalence of the CYP2C9 poor metabolizer phenotype is approximately 2-3% in the White population, 0.5-4% in the Asian population, and <1% in the African American population. The CYP2C9 intermediate phenotype prevalence is approximately 35% in the White population, 24% in the African American population, and 15-36% in the Asian population [see Warnings and Precautions (5.4) and Use in Specific Populations (8.7)].
2.3 Status Epilepticus
- Because of the risk of hypotension and cardiac arrhythmias, the rate of administration for IV fosphenytoin sodium injection should be no greater than 150 mg PE/min in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients [see Warnings and Precautions (5.2)]. Continuous monitoring of the electrocardiogram, blood pressure, and respiratory function is essential, and the patient should be observed throughout the period where maximal serum phenytoin concentrations occur, approximately 10 to 20 minutes after the end of fosphenytoin sodium infusions.
- Because the full antiepileptic effect of phenytoin, whether given as fosphenytoin sodium injection or parenteral phenytoin, is not immediate, other measures, including concomitant administration of an IV benzodiazepine, will usually be necessary for the control of status epilepticus.
- The loading dose should be followed by maintenance doses of either fosphenytoin sodium injection or phenytoin [see Dosage and Administration (2.4)].
- If administration of fosphenytoin sodium injection does not terminate seizures, the use of other anticonvulsants and other appropriate measures should be considered.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Fosphenytoin sodium injection is indicated for the treatment of generalized tonic-clonic status epilepticus and prevention and treatment of seizures occurring during neurosurgery. Fosphenytoin sodium injection can also be substituted, short-term, for oral phenytoin. Fosphenytoin sodium injection should be used only when oral phenytoin administration is not possible [see Dosage and Administration (2.4) and Warnings and Precautions (5.2)].
12.1 Mechanism of Action
Fosphenytoin is a prodrug of phenytoin and accordingly, its anticonvulsant effects are attributable to phenytoin. The precise mechanism by which phenytoin exerts its therapeutic effect has not been established but is thought to involve the voltage-dependent blockade of membrane sodium channels resulting in a reduction in sustained high-frequency neuronal discharges.
2.8 Dosing in Geriatrics
The clearance of phenytoin (the active metabolite of fosphenytoin sodium injection) is decreased slightly in elderly patients and lower or less frequent dosing may be required [see Clinical Pharmacology (12.3)].
16.2 Storage and Handling
Store under refrigeration at 2°C to 8°C (36°F to 46°F). The product should not be stored at room temperature for more than 48 hours. Vials that develop particulate matter should not be used.
Injection vials are single-dose only. After opening, any unused product should be discarded.
5.10 Sensory Disturbances
Severe burning, itching, and/or paresthesia were reported by 7 of 16 normal volunteers administered IV fosphenytoin sodium injection at a dose of 1,200 mg PE at the maximum rate of administration (150 mg PE/min). The severe sensory disturbance lasted from 3 to 50 minutes in 6 of these subjects and for 14 hours in the seventh subject. In some cases, milder sensory disturbances persisted for as long as 24 hours. The location of the discomfort varied among subjects with the groin mentioned most frequently as an area of discomfort. In a separate cohort of 16 normal volunteers (taken from 2 other studies) who were administered IV fosphenytoin sodium injection at a dose of 1,200 mg PE at the maximum rate of administration (150 mg PE/min), none experienced severe disturbances, but most experienced mild to moderate itching or tingling. Patients administered fosphenytoin sodium injection at doses of 20 mg PE/kg at 150 mg PE/min are expected to experience discomfort of some degree. The occurrence and intensity of the discomfort can be lessened by slowing or temporarily stopping the infusion. The effect of continuing infusion unaltered in the presence of these sensations is unknown. No permanent sequelae have been reported thus far. The pharmacologic basis for these positive sensory phenomena is unknown, but other phosphate ester drugs, which deliver smaller phosphate loads, have been associated with burning, itching, and/or tingling predominantly in the groin area.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Dosing Errors: Do not confuse the amount of drug to be given in PE with the concentration of the drug in the vial. Ensure the appropriate volume is withdrawn from the vial when preparing for administration. (5.1)
- Withdrawal Precipitated Seizure: May precipitate status epilepticus. Dose reductions or discontinuation should be done gradually. (5.3)
- Serious Dermatologic Reactions: Discontinue at the first sign of a rash, unless clearly not drug-related. If signs or symptoms suggest SJS/TEN, fosphenytoin sodium injection should not be resumed; consider alternative therapy. (5.4)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: If signs or symptoms of hypersensitivity are present, evaluate the patient immediately. Discontinue if an alternative etiology cannot be established. (5.5)
- Angioedema: Discontinue immediately if symptoms of angioedema such as facial, perioral, or upper airway swelling occur. (5.7)
- Hematopoietic Complications: If occurs, follow-up observation is indicated and an alternative antiepileptic treatment should be used. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The dose, concentration, and infusion rate of fosphenytoin sodium injection should always be expressed as phenytoin sodium equivalents (PE) (2.1)
- For Status Epilepticus:
-
For Non-emergent Loading and Maintenance Dosing:
- Adult loading dose is 10 to 20 mg PE/kg given IV or IM; initial maintenance dose is 4 to 6 mg PE/kg/day in divided doses (2.4)
- Pediatric loading dose is 10 to 15 mg PE/kg at a rate of 1 to 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower); initial maintenance dose is 2 to 4 mg PE/kg every 12 hours at a rate of 1 to 2 mg PE/kg/min (or 100 mg PE/min, whichever is slower) (2.4)
- Intramuscular Administration:
2.9 Dosing During Pregnancy (2.9 Dosing during Pregnancy)
Decreased serum concentrations of phenytoin (the active metabolite of fosphenytoin sodium injection) may occur during pregnancy because of altered phenytoin pharmacokinetics [see Clinical Pharmacology (12.3)]. Periodic measurement of serum phenytoin concentrations should be performed during pregnancy, and the fosphenytoin sodium injection dosage should be adjusted as necessary. Postpartum restoration of the original dosage will probably be indicated [see Use in Specific Populations (8.1)]. Because of potential changes in protein binding during pregnancy, the monitoring of phenytoin serum levels should be based on the unbound fraction.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Fosphenytoin Sodium Injection, USP is a clear, colorless to pale yellow solution available as 50 mg phenytoin sodium equivalents (PE) per mL in:
- 10 mL single-dose injection vials, each containing 500 mg PE/10 mL (50 mg PE/mL)
- 2 mL single-dose injection vials, each containing 100 mg PE/2 mL (50 mg PE/mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of phenytoin or fosphenytoin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole: Anaphylaxis, angioedema [see Warnings and Precautions (5.7)]
Hematologic: Pure red cell aplasia [see Warnings and Precautions (5.9)]
Laboratory Test Abnormality: Phenytoin or fosphenytoin sodium injection may decrease serum concentrations of T4. It may also produce lower than normal values for dexamethasone or metyrapone tests. Phenytoin may also cause increased serum levels of gamma glutamyl transpeptidase (GGT).
Nervous System Disorders: Dyskinesia
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.14 Exacerbation of Porphyria
In view of isolated reports associating phenytoin (the active metabolite of fosphenytoin sodium injection) with exacerbation of porphyria, caution should be exercised in using fosphenytoin sodium injection in patients suffering from this disease.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The more important adverse clinical reactions caused by the IV use of fosphenytoin sodium injection or phenytoin are cardiovascular collapse and/or CNS depression. Hypotension can occur when either drug is administered rapidly by the IV route. The rate of administration is very important; for fosphenytoin sodium injection, it should not exceed 150 mg PE/min [see Warnings and Precautions (5.2)]. The adverse reactions most commonly observed with the use of fosphenytoin sodium injection in clinical trials were nystagmus, dizziness, pruritus, somnolence, and ataxia. With one exception, these reactions are commonly associated with the administration of IV phenytoin. Pruritus, however, was seen much more often following fosphenytoin sodium injection administration and occurred more often with IV fosphenytoin sodium administration than with IM fosphenytoin sodium administration. These reactions were dose and rate related; most alert patients (41 of 64; 64%) administered doses of ≥15 mg PE/kg at 150 mg PE/min experienced discomfort of some degree. These sensations, generally described as itching, burning, or tingling, were usually not at the infusion site. The location of the discomfort varied with the groin mentioned most frequently as a site of involvement. The paresthesia and pruritus were transient events that occurred within several minutes of the start of infusion and generally resolved within 10 minutes after completion of fosphenytoin sodium infusion. Some patients experienced symptoms for hours. These reactions did not increase in severity with repeated administration. Concurrent adverse events or clinical laboratory change suggesting an allergic process were not seen [see Warnings and Precautions (5.10)]. Approximately 2% of the 859 patients who received fosphenytoin sodium injection in premarketing clinical trials discontinued treatment because of an adverse event. The adverse events most commonly associated with withdrawal were pruritus (0.5%), hypotension (0.3%), and bradycardia (0.2%).
Dose and Rate Dependency of Adverse Reactions Following IV Fosphenytoin Sodium Injection: The incidence of adverse reactions tended to increase as both dose and infusion rate increased. In particular, at doses of ≥15mg PE/kg and rates ≥150 mg PE/min, transient pruritus, tinnitus, nystagmus, somnolence, and ataxia occurred 2 to 3 times more often than at lower doses or rates.
5.9 Hematopoietic Complications
Hematopoietic complications, some fatal, have occasionally been reported in association with administration of phenytoin (the active metabolite of fosphenytoin sodium injection). These have included thrombocytopenia, leukopenia, granulocytopenia, agranulocytosis, and pancytopenia with or without bone marrow suppression.
There have been a number of reports that have suggested a relationship between phenytoin and the development of lymphadenopathy (local or generalized), including benign lymph node hyperplasia, pseudolymphoma, lymphoma, and Hodgkin's disease. Although a cause and effect relationship has not been established, the occurrence of lymphadenopathy indicates the need to differentiate such a condition from other types of lymph node pathology. Lymph node involvement may occur with or without symptoms and signs resembling DRESS [see Warnings and Precautions (5.5)].
In all cases of lymphadenopathy, follow-up observation for an extended period is indicated and every effort should be made to achieve seizure control using alternative antiepileptic drugs. Macrocytosis and megaloblastic anemia have occurred, these conditions usually respond to folic acid therapy. Pure red cell aplasia has also been reported with phenytoin.
5.4 Serious Dermatologic Reactions
Fosphenytoin sodium can cause severe cutaneous adverse reactions (SCARs), which may be fatal. Reported reactions in phenytoin (the active metabolite of fosphenytoin sodium)-treated patients have included toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), acute generalized exanthematous pustulosis (AGEP), and Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.5)]. The onset of symptoms is usually within 28 days, but can occur later. Fosphenytoin sodium injection should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest a severe cutaneous adverse reaction, use of this drug should not be resumed and alternative therapy should be considered. If a rash occurs, the patient should be evaluated for signs and symptoms of SCARs.
Studies in patients of Chinese ancestry have found a strong association between the risk of developing SJS/TEN and the presence of HLA-B*1502, an inherited allelic variant of the HLA B gene, in patients using carbamazepine. Limited evidence suggests that HLA-B*1502 may be a risk factor for the development of SJS/TEN in patients of Asian ancestry taking other antiepileptic drugs associated with SJS/TEN, including phenytoin. In addition, retrospective, case-control, genome-wide association studies in patients of southeast Asian ancestry have also identified an increased risk of SCARs in carriers of the decreased function CYP2C9*3 variant, which has also been associated with decreased clearance of phenytoin. Consider avoiding fosphenytoin sodium injection as an alternative to carbamazepine in patients who are positive for HLA-B*1502 or in CYP2C9*3 carriers.
Should fosphenytoin sodium injection be utilized for CYP2C9*3 carriers, consider starting at the lower end of the dosage range [see Use in Specific Populations (8.7)].
The use of HLA-B*1502 or CYP2C9 genotyping has important limitations and must never substitute for appropriate clinical vigilance and patient management. The role of other possible factors in the development of, and morbidity from, SJS/TEN, such as antiepileptic drug (AED) dose, compliance, concomitant medications, comorbidities, and the level of dermatologic monitoring have not been studied.
7.4 Drug/laboratory Test Interactions (7.4 Drug/Laboratory Test Interactions)
Care should be taken when using immunoanalytical methods to measure serum phenytoin concentrations following fosphenytoin sodium injection administration.
2.4 Non Emergent Loading and Maintenance Dosing (2.4 Non-emergent Loading and Maintenance Dosing)
- Because of the risk of hypotension and cardiac arrhythmias, the rate of administration for IV fosphenytoin sodium injection should be no greater than 150 mg PE/min in adults. For loading doses in pediatric patients, the rate should not exceed 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower). For maintenance doses in pediatric patients, the rate should not exceed 1 to 2 mg PE/kg/min (or 100 mg PE/min, whichever is slower). Continuous monitoring of the electrocardiogram, blood pressure, and respiratory function is essential, and the patient should be observed throughout the period where maximal serum phenytoin concentrations occur (approximately 10 to 20 minutes after the end of fosphenytoin sodium infusions).
- After the initial maintenance dose, subsequent maintenance doses should be individualized by monitoring serum phenytoin concentrations to achieve a target therapeutic concentration of phenytoin [see Dosage and Administration (2.5) and Warnings and Precautions (5.17)].
5.13 Renal Or Hepatic Disease Or Hypoalbuminemia (5.13 Renal or Hepatic Disease or Hypoalbuminemia)
Because the fraction of unbound phenytoin (the active metabolite of fosphenytoin sodium injection) is increased in patients with renal or hepatic disease, or in those with hypoalbuminemia, the monitoring of phenytoin serum levels should be based on the unbound fraction in those patients. After IV administration to patients with renal and/or hepatic disease, or in those with hypoalbuminemia, fosphenytoin clearance to phenytoin may be increased without a similar increase in phenytoin clearance. This has the potential to increase the frequency and severity of adverse events.
5.15 Teratogenicity and Other Harm to the Newborn
Fosphenytoin sodium injection may cause fetal harm when administered to a pregnant woman. Prenatal exposure to phenytoin (the active metabolite of fosphenytoin sodium injection) may increase the risks for congenital malformations and other adverse developmental outcomes [see Use in Specific Populations (8.1)].
Increased frequencies of major malformations (such as orofacial clefts and cardiac defects), and abnormalities characteristic of fetal hydantoin syndrome, including dysmorphic skull and facial features, nail and digit hypoplasia, growth abnormalities (including microcephaly), and cognitive deficits, have been reported among children born to epileptic women who took phenytoin alone or in combination with other antiepileptic drugs during pregnancy. There have been several reported cases of malignancies, including neuroblastoma.
A potentially life-threatening bleeding disorder related to decreased levels of vitamin K-dependent clotting factors may occur in newborns exposed to phenytoin in utero. This drug-induced condition can be prevented with vitamin K administration to the mother before delivery and to the neonate after birth.
8.7 Use in Patients With Decreased Cyp2c9 Function (8.7 Use in Patients with Decreased CYP2C9 Function)
Patients who are intermediate or poor metabolizers of CYP2C9 substrates (e.g., *1/*3, *2/*2, *3/*3) may exhibit increased phenytoin serum concentrations compared to patients who are normal metabolizers (e.g., *1/*1). Thus, patients who are known to be intermediate or poor metabolizers may ultimately require lower doses to maintain similar steady-state concentrations compared to normal metabolizers. In patients who are known to be carriers of the decreased function CYP2C9*2 or *3 alleles (intermediate and poor metabolizers), consider starting at the low end of the dosage range and monitor serum concentrations to maintain total phenytoin concentrations of 10 to 20 mcg/mL. If early signs of dose-related central nervous system (CNS) toxicity develop, serum concentrations should be checked immediately [see Clinical Pharmacology (12.5)].
5.17 Serum Phenytoin Levels Above Therapeutic Range (5.17 Serum Phenytoin Levels above Therapeutic Range)
Serum levels of phenytoin (the active metabolite of fosphenytoin sodium injection) sustained above the therapeutic range may produce confusional states referred to as “delirium,” “psychosis,” or “encephalopathy,” or rarely, irreversible cerebellar dysfunction and/or cerebellar atrophy. Accordingly, at the first sign of acute toxicity, serum levels should be immediately checked. Fosphenytoin sodium injection dose reduction is indicated if serum levels are excessive; if symptoms persist, administration of fosphenytoin sodium injection should be discontinued.
7.3 Hyperammonemia With Concomitant Use of Valproate (7.3 Hyperammonemia with Concomitant Use of Valproate)
Concomitant administration of phenytoin and valproate has been associated with an increased risk of valproate-associated hyperammonemia. Patients treated concomitantly with these two drugs should be monitored for signs and symptoms of hyperammonemia.
5.11 Local Toxicity (including Purple Glove Syndrome) (5.11 Local Toxicity (Including Purple Glove Syndrome))
Edema, discoloration, and pain distal to the site of injection (described as “purple glove syndrome”) have also been reported following peripheral intravenous fosphenytoin sodium injection. This may or may not be associated with extravasation. The syndrome may not develop for several days after injection.
2.6 Parenteral Substitution for Oral Phenytoin Therapy
When treatment with oral phenytoin is not possible, fosphenytoin sodium injection can be substituted for oral phenytoin at the same total daily phenytoin sodium equivalents (PE) dose. Phenytoin sodium capsules are approximately 90% bioavailable by the oral route. Phenytoin, derived from administration of fosphenytoin sodium injection, is 100% bioavailable by both the IM and IV routes. For this reason, serum phenytoin concentrations may increase modestly when IM or IV fosphenytoin sodium injection is substituted for oral phenytoin sodium therapy. The rate of administration for IV fosphenytoin sodium injection should be no greater than 150 mg PE/min in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients. In controlled trials, IM fosphenytoin sodium injection was administered as a single daily dose utilizing either 1 or 2 injection sites. Some patients may require more frequent dosing. Intramuscular administration of fosphenytoin sodium injection should ordinarily not be used in pediatric patients.
5.2 Cardiovascular Risk Associated With Rapid Infusion (5.2 Cardiovascular Risk Associated with Rapid Infusion)
Rapid intravenous administration of fosphenytoin sodium injection increases the risk of adverse cardiovascular reactions, including severe hypotension and cardiac arrhythmias. Cardiac arrhythmias have included bradycardia, heart block, QT interval prolongation, ventricular tachycardia, and ventricular fibrillation which have resulted in asystole, cardiac arrest, and death. Severe complications are most commonly encountered in critically ill patients, elderly patients, and patients with hypotension and severe myocardial insufficiency. However, cardiac events have also been reported in adults and children without underlying cardiac disease or comorbidities and at recommended doses and infusion rates.
The rate of intravenous fosphenytoin sodium injection administration should not exceed 150 mg phenytoin sodium equivalents (PE) per minute in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients [see Dosage and Administration (2.3, 2.4)].
Although the risk of cardiovascular toxicity increases with infusion rates above the recommended infusion rate, these events have also been reported at or below the recommended infusion rate.
As non-emergency therapy, intravenous fosphenytoin sodium injection should be administered more slowly. Because of the risks of cardiac and local toxicity associated with IV fosphenytoin sodium injection, oral phenytoin should be used whenever possible.
Because adverse cardiovascular reactions have occurred during and after infusions, careful cardiac and respiratory monitoring is needed during and after the administration of intravenous fosphenytoin sodium injection. Reduction in rate of administration or discontinuation of dosing may be needed.
5.3 Withdrawal Precipitated Seizure, Status Epilepticus
Antiepileptic drugs should not be abruptly discontinued because of the possibility of increased seizure frequency, including status epilepticus. When, in the judgment of the clinician, the need for dosage reduction, discontinuation, or substitution of alternative antiepileptic medication arises, this should be done gradually. However, in the event of an allergic or hypersensitivity reaction, rapid substitution of alternative therapy may be necessary. In this case, alternative therapy should be an antiepileptic drug not belonging to the hydantoin chemical class.
8.6 Renal And/or Hepatic Impairment, Or Hypoalbuminemia (8.6 Renal and/or Hepatic Impairment, or Hypoalbuminemia)
The liver is the site of biotransformation. Patients with impaired liver function, elderly patients, or those who are gravely ill may show early toxicity.
Because the fraction of unbound phenytoin (the active metabolite of fosphenytoin sodium injection) is increased in patients with renal or hepatic disease, or in those with hypoalbuminemia, the monitoring of phenytoin serum levels should be based on the unbound fraction in those patients.
After IV administration to patients with renal and/or hepatic disease, or in those with hypoalbuminemia, fosphenytoin clearance to phenytoin may be increased without a similar increase in phenytoin clearance. This has the potential to increase the frequency and severity of adverse events.
2.1 Important Administration Instructions to Avoid Dosing Errors
Use caution when administering fosphenytoin sodium injection because of the risk of dosing errors [see Warnings and Precautions (5.1)].
7.1 Drugs That Affect Phenytoin Or Fosphenytoin Sodium Injection (7.1 Drugs that Affect Phenytoin or Fosphenytoin Sodium Injection)
Table 6 includes commonly occurring drug interactions that affect phenytoin (the active metabolite of fosphenytoin sodium injection) concentrations. However, this list is not intended to be inclusive or comprehensive. Individual prescribing information from relevant drugs should be consulted.
The addition or withdrawal of these agents in patients on phenytoin therapy may require an adjustment of the phenytoin dose to achieve optimal clinical outcome.
|
a The induction potency of St. John's wort may vary widely based on preparation. |
|
| Interacting Agent | Examples |
| Drugs that may increase phenytoin serum levels | |
| Antiepileptic drugs | Ethosuximide, felbamate, oxcarbazepine, methsuximide, topiramate |
| Azoles | Fluconazole, ketoconazole, itraconazole, miconazole, voriconazole |
| Antineoplastic agents | Capecitabine, fluorouracil |
| Antidepressants | Fluoxetine, fluvoxamine, sertraline |
| Gastric acid reducing agents |
H2 antagonists (cimetidine), omeprazole |
|
Sulfonamides |
Sulfamethizole, sulfaphenazole, sulfadiazine, sulfamethoxazole- trimethoprim |
|
Other |
Acute alcohol intake, amiodarone, chloramphenicol, chlordiazepoxide, disulfiram, estrogen, fluvastatin, isoniazid, methylphenidate, phenothiazines, salicylates, ticlopidine, tolbutamide, trazodone, warfarin |
| Drugs that may decrease phenytoin serum levels | |
| Antineoplastic agents usually in combination |
Bleomycin, carboplatin, cisplatin, doxorubicin, methotrexate |
| Antiviral agents | Fosamprenavir, nelfinavir, ritonavir |
| Antiepileptic drugs | Carbamazepine, vigabatrin |
|
Other |
Chronic alcohol abuse, diazepam, diazoxide, folic acid, reserpine, rifampin, St. John's wort,a theophylline |
| Drugs that may either increase or decrease phenytoin serum levels | |
| Antiepileptic drugs | Phenobarbital, valproate sodium, valproic acid |
7.2 Drugs Affected By Phenytoin Or Fosphenytoin Sodium Injection (7.2 Drugs Affected by Phenytoin or Fosphenytoin Sodium Injection)
Table 7 includes commonly occurring drug interactions affected by phenytoin (the active metabolite of fosphenytoin sodium injection). However, this list is not intended to be inclusive or comprehensive. Individual drug package inserts should be consulted. The addition or withdrawal of phenytoin during concomitant therapy with these agents may require adjustment of the dose of these agents to achieve optimal clinical outcome.
|
a The effect of phenytoin on phenobarbital, valproic acid and sodium valproate serum levels is unpredictable. |
|
| Interacting Agent | Examples |
| Drugs whose efficacy is impaired by phenytoin | |
| Azoles | Fluconazole, ketoconazole, itraconazole, posaconazole, voriconazole |
| Antineoplastic agents | Irinotecan, paclitaxel, teniposide |
|
Delavirdine |
Phenytoin can substantially reduce the concentrations of delavirdine. This can lead to loss of virologic response and possible resistance [see Contraindications (4)]. |
|
Neuromuscular blocking agents |
Cisatracurium, pancuronium, rocuronium and vecuronium: resistance to the neuromuscular blocking action of the nondepolarizing neuromuscular blocking agents has occurred in patients chronically administered phenytoin. Whether or not phenytoin has the same effect on other non-depolarizing agents is unknown. Prevention or Management: Patients should be monitored closely for more rapid recovery from neuromuscular blockade than expected, and infusion rate requirements may be higher. |
|
Warfarin |
Increased and decreased PT/INR responses have been reported when phenytoin is coadministered with warfarin. |
|
Other |
Corticosteroids, doxycycline, estrogens, furosemide, oral contraceptives, paroxetine, quinidine, rifampin, sertraline, theophylline, and vitamin D |
| Drugs whose level is decreased by phenytoin | |
| Anticoagulants | Apixaban, dabigatran, edoxaban, rivaroxaban |
| Antiepileptic drugsa | Carbamazepine, felbamate, lamotrigine, topiramate, oxcarbazepine, lacosamide |
| Antilipidemic agents | Atorvastatin, fluvastatin, simvastatin |
| Antiplatelets | Ticagrelor |
|
Antiviral agents |
Efavirenz, lopinavir/ritonavir, indinavir, nelfinavir, ritonavir, saquinavir Fosamprenavir: phenytoin when given with fosamprenavir alone may decrease the concentration of amprenavir, the active metabolite. Phenytoin when given with the combination of fosamprenavir and ritonavir may increase the concentration of amprenavir |
| Calcium channel blockers | Nifedipine, nimodipine, nisoldipine, verapamil |
| Other | Albendazole (decreases active metabolite), chlorpropamide, clozapine, cyclosporine, digoxin, disopyramide, folic acid, methadone, mexiletine, praziquantel, quetiapine |
Warning: Cardiovascular Risk Associated With Rapid Infusion Rates (WARNING: CARDIOVASCULAR RISK ASSOCIATED WITH RAPID INFUSION RATES)
The rate of intravenous fosphenytoin sodium injection administration should not exceed 150 mg phenytoin sodium equivalents (PE) per minute in adults and 2 mg PE/kg/min (or 150 mg PE/min, whichever is slower) in pediatric patients because of the risk of severe hypotension and cardiac arrhythmias. Careful cardiac monitoring is needed during and after administering intravenous fosphenytoin sodium. Although the risk of cardiovascular toxicity increases with infusion rates above the recommended infusion rate, these events have also been reported at or below the recommended infusion rate. Reduction in rate of administration or discontinuation of dosing may be needed [see Dosage and Administration (2.3, 2.4) and Warnings and Precautions (5.2)].
2.7 Dosing in Patients With Renal Or Hepatic Impairment Or Hypoalbuminemia (2.7 Dosing in Patients with Renal or Hepatic Impairment or Hypoalbuminemia)
Because the fraction of unbound phenytoin (the active metabolite of fosphenytoin sodium injection) is increased in patients with renal or hepatic disease, or in those with hypoalbuminemia, the monitoring of phenytoin serum levels should be based on the unbound fraction in those patients. After IV fosphenytoin sodium administration to patients with renal and/or hepatic disease, or in those with hypoalbuminemia, fosphenytoin clearance to phenytoin may be increased without a similar increase in phenytoin clearance. This has the potential to increase the frequency and severity of adverse events [see Warnings and Precautions (5.13)].
5.5 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity (5.5 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as Multiorgan hypersensitivity, has been reported in patients taking antiepileptic drugs, including phenytoin and fosphenytoin sodium. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Fosphenytoin sodium injection should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:38:16.931050 · Updated: 2026-03-14T21:53:59.849930