hempvana endtag
b5e2d18c-cce8-6619-e053-2a95a90a8643
34390-5
HUMAN OTC DRUG LABEL
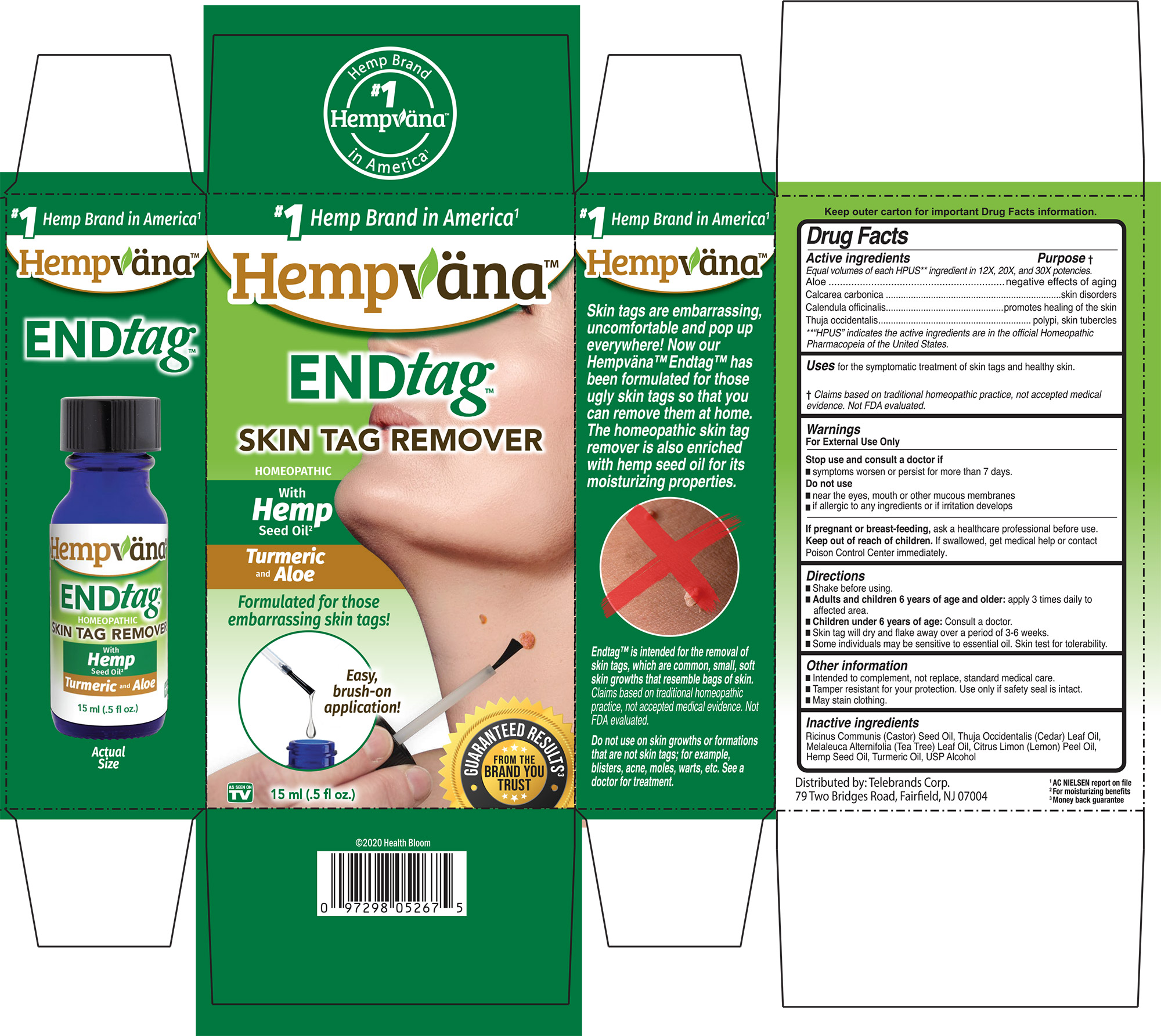
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Equal Volumes of each HPUS ** ingredient in 12x, 20x, and 30x potencies. Aloe Calcarea carbonica Calendula officinalis Thuja occidentalis **HPUS indicates the active ingredie ts are in the official Homeopathic Pharmacopeia of the United States
Medication Information
Warnings
For External Use Only
Stop use and consult a doctor if
- symptoms worsen or persist for more than 7 days.
Do not use
- near the eyes, mouth or other mucous membranes
- if allergic to any ingredients or if irritation develops
Uses
for the symptomatic treatment of skin tags and healthy skin.
Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
Directions
- Shake before using
- Adults and children 6 years of age and older: apply 3 times daily to affected area.
- Children under 6 years of age: Consult a doctor.
- Skin tag will dry and flake away over a period of 3-6 weeks.
- Some individuals may be senstitive to essential oil. Skin test for tolerability.
Other Information
- Intended to compliment, not replace, standard medcial care
- Tamper resistant for your protection. Use only if safety seal is intact.
- May stain clothing.
Inactive Ingredients
Ricinus Communis (Castor) Seed Oil, Thuja Occidentalis (Cedar) Leaf Oil, Melaleuca Alternifolia (Tea Tree) Leaf Oil, Citrus Limon (Lemon) Peel Oil, Hemp Seed Oil, Turmeric Oil, USP Alcohol.
Description
Equal Volumes of each HPUS ** ingredient in 12x, 20x, and 30x potencies. Aloe Calcarea carbonica Calendula officinalis Thuja occidentalis **HPUS indicates the active ingredie ts are in the official Homeopathic Pharmacopeia of the United States
Section 42229-5
Distributed by Telebrands Corp.
79 Two Bridges Road, Fairfield, NJ 07004
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact Poison Control Center immediately.
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a healthcare professional before use.
Section 55105-1
Active ingredients ................. Purpose
Equal volumes of each HPUS** ingredient in 12X, 20X, and 30X potencies.
Aloe...........................................negative effects of againg
Calcarea carbonica...............skin disorders
Calendula officinalis.............promotes healing of the skin
Thuja occidentalis.................polypi, skin, tubercles
**“HPUS” indicates the active ingredients are in the official Homeopathic Pharmacopeia of the United States.
Hpus Active Ingredients
Equal Volumes of each HPUS ** ingredient in 12x, 20x, and 30x potencies.
Aloe
Calcarea carbonica
Calendula officinalis
Thuja occidentalis
**HPUS indicates the active ingredie ts are in the official Homeopathic Pharmacopeia of the United States
Structured Label Content
Warnings
For External Use Only
Stop use and consult a doctor if
- symptoms worsen or persist for more than 7 days.
Do not use
- near the eyes, mouth or other mucous membranes
- if allergic to any ingredients or if irritation develops
Uses
for the symptomatic treatment of skin tags and healthy skin.
Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
Directions
- Shake before using
- Adults and children 6 years of age and older: apply 3 times daily to affected area.
- Children under 6 years of age: Consult a doctor.
- Skin tag will dry and flake away over a period of 3-6 weeks.
- Some individuals may be senstitive to essential oil. Skin test for tolerability.
Other Information (Other information)
- Intended to compliment, not replace, standard medcial care
- Tamper resistant for your protection. Use only if safety seal is intact.
- May stain clothing.
Inactive Ingredients (Inactive ingredients)
Ricinus Communis (Castor) Seed Oil, Thuja Occidentalis (Cedar) Leaf Oil, Melaleuca Alternifolia (Tea Tree) Leaf Oil, Citrus Limon (Lemon) Peel Oil, Hemp Seed Oil, Turmeric Oil, USP Alcohol.
Section 42229-5 (42229-5)
Distributed by Telebrands Corp.
79 Two Bridges Road, Fairfield, NJ 07004
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact Poison Control Center immediately.
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a healthcare professional before use.
Section 55105-1 (55105-1)
Active ingredients ................. Purpose
Equal volumes of each HPUS** ingredient in 12X, 20X, and 30X potencies.
Aloe...........................................negative effects of againg
Calcarea carbonica...............skin disorders
Calendula officinalis.............promotes healing of the skin
Thuja occidentalis.................polypi, skin, tubercles
**“HPUS” indicates the active ingredients are in the official Homeopathic Pharmacopeia of the United States.
Hpus Active Ingredients (HPUS Active Ingredients)
Equal Volumes of each HPUS ** ingredient in 12x, 20x, and 30x potencies.
Aloe
Calcarea carbonica
Calendula officinalis
Thuja occidentalis
**HPUS indicates the active ingredie ts are in the official Homeopathic Pharmacopeia of the United States
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:57.280834 · Updated: 2026-03-14T23:15:01.065113