enoxaparin sodium injection, for subcutaneous and intravenous use
b4d9e4d4-4fdb-48d6-a169-7bc5eebc51e9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
NDC 11797-757-06 Enoxaparin Sodium Injection USP 30 mg/0.3 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.3 mL Syringes NDC 11797-758-06 Enoxaparin Sodium Injection USP 40 mg/0.4 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.4 mL Syringes NDC 11797-759-06 Enoxaparin Sodium Injection USP 60 mg/0.6 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.6 mL Syringes NDC 11797-760-06 Enoxaparin Sodium Injection USP 80 mg/0.8 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.8 mL Syringes NDC 11797-761-06 Enoxaparin Sodium Injection USP 100 mg/1 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 1 mL Syringes NDC 11797-762-06 Enoxaparin Sodium Injection USP 120 mg/0.8 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.8 mL Syringes NDC 11797-763-06 Enoxaparin Sodium Injection USP 150 mg/1 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 1 mL Syringes
Medication Information
Description
NDC 11797-757-06 Enoxaparin Sodium Injection USP 30 mg/0.3 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.3 mL Syringes NDC 11797-758-06 Enoxaparin Sodium Injection USP 40 mg/0.4 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.4 mL Syringes NDC 11797-759-06 Enoxaparin Sodium Injection USP 60 mg/0.6 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.6 mL Syringes NDC 11797-760-06 Enoxaparin Sodium Injection USP 80 mg/0.8 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.8 mL Syringes NDC 11797-761-06 Enoxaparin Sodium Injection USP 100 mg/1 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 1 mL Syringes NDC 11797-762-06 Enoxaparin Sodium Injection USP 120 mg/0.8 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 0.8 mL Syringes NDC 11797-763-06 Enoxaparin Sodium Injection USP 150 mg/1 mL SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE FOR SUBCUTANEOUS INJECTION Ten 1 mL Syringes
Package Label.principal Display Panel
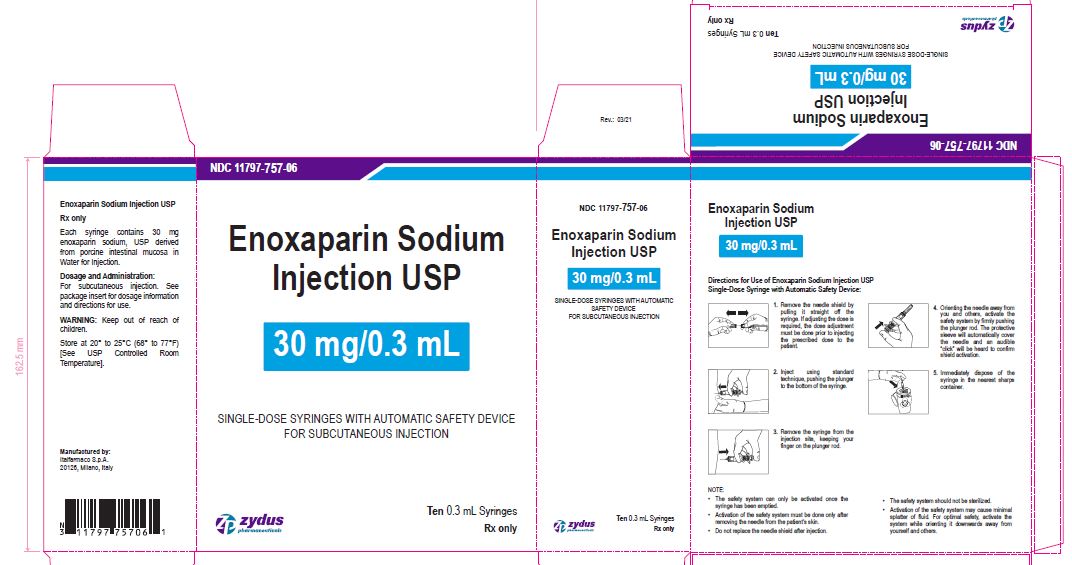
NDC 11797-757-06
Enoxaparin Sodium Injection USP
30 mg/0.3 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.3 mL Syringes
NDC 11797-758-06
Enoxaparin Sodium Injection USP
40 mg/0.4 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.4 mL Syringes
NDC 11797-759-06
Enoxaparin Sodium Injection USP
60 mg/0.6 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.6 mL Syringes
NDC 11797-760-06
Enoxaparin Sodium Injection USP
80 mg/0.8 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.8 mL Syringes
NDC 11797-761-06
Enoxaparin Sodium Injection USP
100 mg/1 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 1 mL Syringes
NDC 11797-762-06
Enoxaparin Sodium Injection USP
120 mg/0.8 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.8 mL Syringes
NDC 11797-763-06
Enoxaparin Sodium Injection USP
150 mg/1 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 1 mL Syringes
Structured Label Content
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 11797-757-06
Enoxaparin Sodium Injection USP
30 mg/0.3 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.3 mL Syringes
NDC 11797-758-06
Enoxaparin Sodium Injection USP
40 mg/0.4 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.4 mL Syringes
NDC 11797-759-06
Enoxaparin Sodium Injection USP
60 mg/0.6 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.6 mL Syringes
NDC 11797-760-06
Enoxaparin Sodium Injection USP
80 mg/0.8 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.8 mL Syringes
NDC 11797-761-06
Enoxaparin Sodium Injection USP
100 mg/1 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 1 mL Syringes
NDC 11797-762-06
Enoxaparin Sodium Injection USP
120 mg/0.8 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 0.8 mL Syringes
NDC 11797-763-06
Enoxaparin Sodium Injection USP
150 mg/1 mL
SINGLE-DOSE SYRINGES WITH AUTOMATIC SAFETY DEVICE
FOR SUBCUTANEOUS INJECTION
Ten 1 mL Syringes
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:28.980958 · Updated: 2026-03-14T21:50:25.596456