Drug Facts
b406f41a-5a81-762d-e053-2995a90a064c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
salicylic acid, tocopheryl acetate, niacinamide
Medication Information
Warnings and Precautions
■ For external use only.
■ Do not use in eyes.
■ lf swallowed, get medical help promptly.
■ Stop use, ask doctor lf irritation occurs.
■ Keep out of reach of children.
Indications and Usage
apply proper amount to the subject area
Dosage and Administration
for external use only
Description
salicylic acid, tocopheryl acetate, niacinamide
Section 50565-1
KEEP OUT OF REACH OF THE CHILDREN
Section 51727-6
Water
Alcohol
Polysorbate 80
Benzyl Glycol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Avena Sativa (Oat) Leaf Extract
Trideceth-10
Salicylic Acid
Butylene Glycol
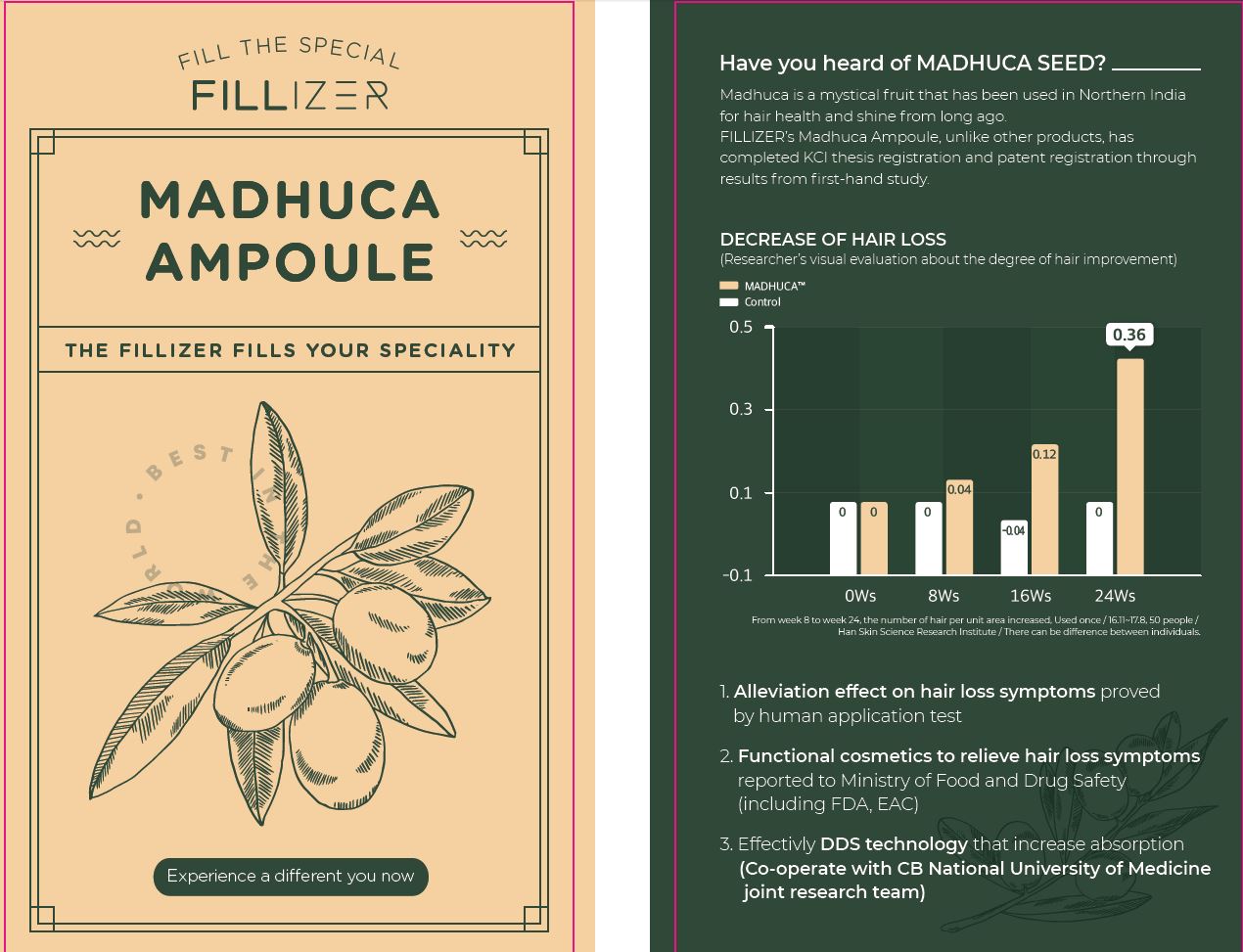
Madhuca Longifolia Sophorolipids(0.8%)
Fragrance
Hydrolyzed Collagen
Citrus Unshiu Peel Extract
Dipotassium Glycyrrhizate
Sodium Citrate
Ethylhexylglycerin
Pyridoxine HCl
1,2-Hexanediol
Raspberry Ketone
Menthol
Althaea Rosea Flower Extract
Coffea Arabica (Coffee) Seed Extract
Camellia Sinensis Leaf Extract
Morus Alba Bark Extract
Perilla Frutescens Leaf Extract
Houttuynia Cordata Extract
Polygonum Multiflorum Root Extract
Limonene
Geraniol
Citronellol
Butylphenyl Methylpropional
Linalool
Alpha-Isomethyl Ionone
Hydroxycitronellal
Amyl Cinnamal
Benzyl Benzoate
Citral
Section 51945-4
Section 55105-1
antiseptic
Section 55106-9
salicylic acid, tocopheryl acetate, niacinamide
Structured Label Content
Indications and Usage (34067-9)
apply proper amount to the subject area
Dosage and Administration (34068-7)
for external use only
Warnings and Precautions (34071-1)
■ For external use only.
■ Do not use in eyes.
■ lf swallowed, get medical help promptly.
■ Stop use, ask doctor lf irritation occurs.
■ Keep out of reach of children.
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF THE CHILDREN
Section 51727-6 (51727-6)
Water
Alcohol
Polysorbate 80
Benzyl Glycol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Avena Sativa (Oat) Leaf Extract
Trideceth-10
Salicylic Acid
Butylene Glycol
Madhuca Longifolia Sophorolipids(0.8%)
Fragrance
Hydrolyzed Collagen
Citrus Unshiu Peel Extract
Dipotassium Glycyrrhizate
Sodium Citrate
Ethylhexylglycerin
Pyridoxine HCl
1,2-Hexanediol
Raspberry Ketone
Menthol
Althaea Rosea Flower Extract
Coffea Arabica (Coffee) Seed Extract
Camellia Sinensis Leaf Extract
Morus Alba Bark Extract
Perilla Frutescens Leaf Extract
Houttuynia Cordata Extract
Polygonum Multiflorum Root Extract
Limonene
Geraniol
Citronellol
Butylphenyl Methylpropional
Linalool
Alpha-Isomethyl Ionone
Hydroxycitronellal
Amyl Cinnamal
Benzyl Benzoate
Citral
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
antiseptic
Section 55106-9 (55106-9)
salicylic acid, tocopheryl acetate, niacinamide
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:03.254752 · Updated: 2026-03-14T22:58:52.562418