Dendracin Neurodendraxcin®, Topical Pain Relief Lotion
b36c7977-557d-4ab4-bc4a-35875f1848e6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
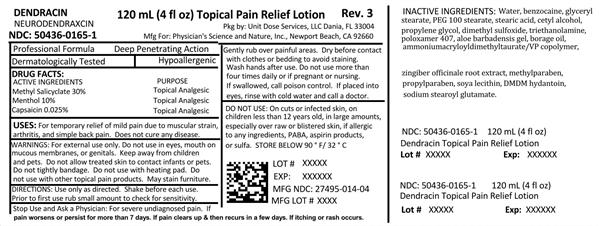
Methyl Salicylate 30% Menthol 10% Capsaicin 0.025%
Purpose
Topical Analgesic
How Supplied
Product: 50436-0165 NDC: 50436-0165-1 120 mL in a BOTTLE
Medication Information
Purpose
Topical Analgesic
How Supplied
Product: 50436-0165
NDC: 50436-0165-1 120 mL in a BOTTLE
Description
Methyl Salicylate 30% Menthol 10% Capsaicin 0.025%
Uses:
For temporary relief of mild pain due to muscular strain, arthritis, and simple back pain. Does not cure any disease.
Section 42229-5
Manufactured for Physicians' Science and Nature, Inc.
220 Newport Center Drive 11-634, Newport Beach, CA 92660
Made in the USA
Patent Pending
Warnings:
For external use only. Do not use in eyes, mouth, on mucous membranes, or genitals. Do not tightly bandage. Do not use with heating pad. Do not use with other topical pain products.
Directions:
Use only as directed. Shake before each use. Prior to first use, rub small amount to check for sensitivity. Gently rub over painful areas. Dry before contact with clothes or bedding to avoid staining. Wash hands after use. Do not use more than 4 times daily or if pregnant or nursing. If swallowed, call poison control. If placed into eyes, rinse with cold water and call a doctor.
Do Not Use:
On cuts or infected skin, on children less than 12 years old, in large amounts, especially over raw or blistered skin, if allergic to any ingredients, PABA, aspirin products, or sulfa.
Store below 90°F/32°C.
Active Ingredients
Methyl Salicylate 30%
Menthol 10%
Capsaicin 0.025%
Inactive Ingredients:
Water, benzocaine, glyceryl stearate, PEG 100 stearate, stearic acid, cetyl alcohol, propylene glycol, dimethyl sulfoxide, triethanolamine, poloxamer 407, aloe barbadensis gel, borage oil, ammonium acryloyldimethyltaurate, zingiber officinale root extract, methylparaben, propylparaben, soya lecithin, DMDM hydantoin, sodium stearoyl glutamate.
Dendracin Topical Pain Relief
Stop Use and Ask A Physician:
For severe undiagnosed pain. If pain worsens or persist for more than 7 days. If pain clears up and then recurs in a few days. If itching or rash occurs.
Structured Label Content
Uses:
For temporary relief of mild pain due to muscular strain, arthritis, and simple back pain. Does not cure any disease.
Section 42229-5 (42229-5)
Manufactured for Physicians' Science and Nature, Inc.
220 Newport Center Drive 11-634, Newport Beach, CA 92660
Made in the USA
Patent Pending
Purpose
Topical Analgesic
Warnings:
For external use only. Do not use in eyes, mouth, on mucous membranes, or genitals. Do not tightly bandage. Do not use with heating pad. Do not use with other topical pain products.
Directions:
Use only as directed. Shake before each use. Prior to first use, rub small amount to check for sensitivity. Gently rub over painful areas. Dry before contact with clothes or bedding to avoid staining. Wash hands after use. Do not use more than 4 times daily or if pregnant or nursing. If swallowed, call poison control. If placed into eyes, rinse with cold water and call a doctor.
Do Not Use:
On cuts or infected skin, on children less than 12 years old, in large amounts, especially over raw or blistered skin, if allergic to any ingredients, PABA, aspirin products, or sulfa.
Store below 90°F/32°C.
How Supplied (HOW SUPPLIED)
Product: 50436-0165
NDC: 50436-0165-1 120 mL in a BOTTLE
Active Ingredients (Active ingredients)
Methyl Salicylate 30%
Menthol 10%
Capsaicin 0.025%
Inactive Ingredients: (Inactive ingredients:)
Water, benzocaine, glyceryl stearate, PEG 100 stearate, stearic acid, cetyl alcohol, propylene glycol, dimethyl sulfoxide, triethanolamine, poloxamer 407, aloe barbadensis gel, borage oil, ammonium acryloyldimethyltaurate, zingiber officinale root extract, methylparaben, propylparaben, soya lecithin, DMDM hydantoin, sodium stearoyl glutamate.
Dendracin Topical Pain Relief (DENDRACIN TOPICAL PAIN RELIEF)
Stop Use and Ask A Physician: (Stop Use and Ask a Physician:)
For severe undiagnosed pain. If pain worsens or persist for more than 7 days. If pain clears up and then recurs in a few days. If itching or rash occurs.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:36.958361 · Updated: 2026-03-14T22:55:12.436150