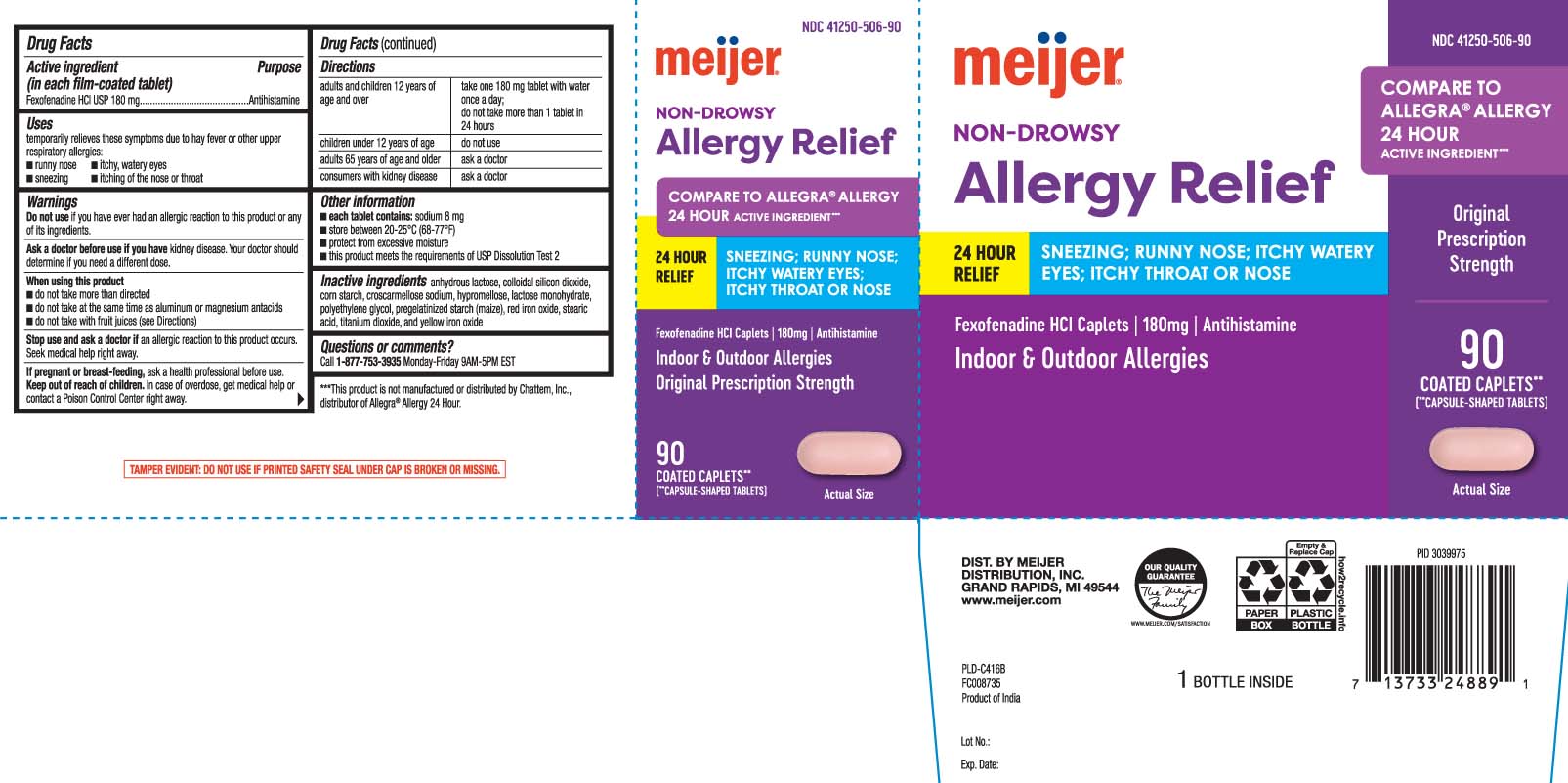
Drug Facts
b3395454-65db-4d8b-abce-2475b049a114
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Fexofenadine HCl USP 180 mg
Purpose
Antihistamine
Medication Information
Purpose
Antihistamine
Description
Fexofenadine HCl USP 180 mg
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Directions
| adults and children 12 years of age and over | take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Do Not Use
if you have ever had an allergic reaction to this product or any of its ingredients.
Product Label
Other Information
- each tablet contains: sodium 8 mg
- store between 20-25ºC (68-77ºF)
- protect from excessive moisture
- this product meets the requirements of USP Dissolution Test 2
Inactive Ingredients
anhydrous lactose,colloidal silicon dioxide, corn starch, croscarmellose sodium, hypromellose, lactose monohydrate, polyethylene glycol, pregelatinized starch (maize), red iron oxide, stearic acid, titanium dioxide, and yellow iron oxide
Questions Or Comments?
call 1-877-753-3935 Monday- Friday 9AM- 5PM EST
Principal Display Panel
COMPARE TO ALLEGRA® ALLERGY 24 HOUR ACTIVE INGREDIENT***
NON-DROWSY
24 HOUR RELIEF
SNEEZING; RUNNY NOSE; ITCHY WATERY EYES; ITCHY THROAT OR NOSE
Fexofenadine HCI Caplets | 180 mg | Antihistamine
Indoor & Outdoor Allergies
Original Prescription Strength
PRESCRIPTION STRENGTH
COATED CAPLETS**
(**CAPSULE-SHAPED TABLETS)
***This product is not manufactured or distributed by Chattem Inc., distributor of Allegra® Allergy 24 hour.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
DIST. BY MEIJER
DISTRIBUTION, INC.
GRAND RAPIDS, MI 49544
When Using This Product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Stop Use and Ask A Doctor If
an allergic reaction to this product occurs. Seek medical help right away.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have
kidney disease. Your doctor should determine if you need a different dose.
Active Ingredient (in Each Film Coated tablet)
Fexofenadine HCl USP 180 mg
Structured Label Content
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Purpose
Antihistamine
Directions
| adults and children 12 years of age and over | take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Do Not Use (Do not use)
if you have ever had an allergic reaction to this product or any of its ingredients.
Product Label
Other Information (Other information)
- each tablet contains: sodium 8 mg
- store between 20-25ºC (68-77ºF)
- protect from excessive moisture
- this product meets the requirements of USP Dissolution Test 2
Inactive Ingredients (Inactive ingredients)
anhydrous lactose,colloidal silicon dioxide, corn starch, croscarmellose sodium, hypromellose, lactose monohydrate, polyethylene glycol, pregelatinized starch (maize), red iron oxide, stearic acid, titanium dioxide, and yellow iron oxide
Questions Or Comments? (Questions or comments?)
call 1-877-753-3935 Monday- Friday 9AM- 5PM EST
Principal Display Panel
COMPARE TO ALLEGRA® ALLERGY 24 HOUR ACTIVE INGREDIENT***
NON-DROWSY
24 HOUR RELIEF
SNEEZING; RUNNY NOSE; ITCHY WATERY EYES; ITCHY THROAT OR NOSE
Fexofenadine HCI Caplets | 180 mg | Antihistamine
Indoor & Outdoor Allergies
Original Prescription Strength
PRESCRIPTION STRENGTH
COATED CAPLETS**
(**CAPSULE-SHAPED TABLETS)
***This product is not manufactured or distributed by Chattem Inc., distributor of Allegra® Allergy 24 hour.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
DIST. BY MEIJER
DISTRIBUTION, INC.
GRAND RAPIDS, MI 49544
When Using This Product (When using this product)
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
an allergic reaction to this product occurs. Seek medical help right away.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
kidney disease. Your doctor should determine if you need a different dose.
Active Ingredient (in Each Film Coated tablet) (Active ingredient (in each film-coated tablet))
Fexofenadine HCl USP 180 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:27.597899 · Updated: 2026-03-14T23:09:13.832086