Alogliptin Tablets
b25f155a-1259-47c2-aa3b-7c1356e4c7f6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Alogliptin tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Dosage and Administration
The recommended dosage in patients with normal renal function or mild renal impairment is 25 mg orally once daily. ( 2.1 ) Can be taken with or without food. ( 2.1 ) Adjust dosage if moderate or severe renal impairment or end-stage renal disease (ESRD). ( 2.2 ) Degree of Renal Impairment Creatinine Clearance (mL/min) Recommended Dosage Moderate ≥30 to <60 12.5 mg once daily Severe/ESRD <30 6.25 mg once daily
Contraindications
Alogliptin tablets is contraindicated in patients with a history of serious hypersensitivity to alogliptin or any of the excipients in Alogliptin tablets. Reactions such as anaphylaxis, angioedema and severe cutaneous adverse reactions have been reported [see Warnings and Precautions (5.3) , Adverse Reactions (6.2) ] .
Warnings and Precautions
Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue alogliptin tablets. ( 5.1 ) Heart failure: Consider the risks and benefits of alogliptin tablets prior to initiating treatment in patients at risk for heart failure. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of alogliptin tablets. ( 5.2 ) Hypersensitivity: There have been postmarketing reports of serious hypersensitivity reactions in patients treated with alogliptin tablets such as anaphylaxis, angioedema and severe cutaneous adverse reactions, including Stevens-Johnson syndrome. If hypersensitivity reactions occur, discontinue alogliptin tablets, treat promptly and monitor until signs and symptoms resolve. ( 5.3 ) Hepatic effects: Postmarketing reports of hepatic failure, sometimes fatal. Causality cannot be excluded. If liver injury is detected, promptly interrupt alogliptin tablets and assess patient for probable cause, then treat cause if possible, to resolution or stabilization. Do not restart alogliptin tablets if liver injury is confirmed and no alternative etiology can be found. ( 5.4 ) Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating Alogliptin tablets. ( 5.5 ) Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.6 ) Bullous pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue alogliptin tablets. ( 5.7 )
Adverse Reactions
The following serious adverse reactions are described below or elsewhere in the prescribing information: Pancreatitis [see Warnings and Precautions (5.1) ] Heart Failure [see Warnings and Precautions (5.2) ] Hypersensitivity Reactions [see Warnings and Precautions (5.3) ] Hepatic Effects [see Warnings and Precautions (5.4) ] Severe and Disabling Arthralgia [see Warnings and Precautions (5.6) ] Bullous Pemphigoid [see Warnings and Precautions (5.7) ]
How Supplied
Alogliptin tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows: 25 mg tablet: light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side, available in: NDC 45802-150-65 Bottles of 30 tablets 12.5 mg tablet: yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side, available in: NDC 45802-103-65 Bottles of 30 tablets 6.25 mg tablet: light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side, available in: NDC 45802-087-65 Bottles of 30 tablets
Storage and Handling
Alogliptin tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows: 25 mg tablet: light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side, available in: NDC 45802-150-65 Bottles of 30 tablets 12.5 mg tablet: yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side, available in: NDC 45802-103-65 Bottles of 30 tablets 6.25 mg tablet: light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side, available in: NDC 45802-087-65 Bottles of 30 tablets
Description
Alogliptin tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Medication Information
Warnings and Precautions
Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue alogliptin tablets. ( 5.1 ) Heart failure: Consider the risks and benefits of alogliptin tablets prior to initiating treatment in patients at risk for heart failure. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of alogliptin tablets. ( 5.2 ) Hypersensitivity: There have been postmarketing reports of serious hypersensitivity reactions in patients treated with alogliptin tablets such as anaphylaxis, angioedema and severe cutaneous adverse reactions, including Stevens-Johnson syndrome. If hypersensitivity reactions occur, discontinue alogliptin tablets, treat promptly and monitor until signs and symptoms resolve. ( 5.3 ) Hepatic effects: Postmarketing reports of hepatic failure, sometimes fatal. Causality cannot be excluded. If liver injury is detected, promptly interrupt alogliptin tablets and assess patient for probable cause, then treat cause if possible, to resolution or stabilization. Do not restart alogliptin tablets if liver injury is confirmed and no alternative etiology can be found. ( 5.4 ) Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating Alogliptin tablets. ( 5.5 ) Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.6 ) Bullous pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue alogliptin tablets. ( 5.7 )
Indications and Usage
Alogliptin tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Dosage and Administration
The recommended dosage in patients with normal renal function or mild renal impairment is 25 mg orally once daily. ( 2.1 ) Can be taken with or without food. ( 2.1 ) Adjust dosage if moderate or severe renal impairment or end-stage renal disease (ESRD). ( 2.2 ) Degree of Renal Impairment Creatinine Clearance (mL/min) Recommended Dosage Moderate ≥30 to <60 12.5 mg once daily Severe/ESRD <30 6.25 mg once daily
Contraindications
Alogliptin tablets is contraindicated in patients with a history of serious hypersensitivity to alogliptin or any of the excipients in Alogliptin tablets. Reactions such as anaphylaxis, angioedema and severe cutaneous adverse reactions have been reported [see Warnings and Precautions (5.3) , Adverse Reactions (6.2) ] .
Adverse Reactions
The following serious adverse reactions are described below or elsewhere in the prescribing information: Pancreatitis [see Warnings and Precautions (5.1) ] Heart Failure [see Warnings and Precautions (5.2) ] Hypersensitivity Reactions [see Warnings and Precautions (5.3) ] Hepatic Effects [see Warnings and Precautions (5.4) ] Severe and Disabling Arthralgia [see Warnings and Precautions (5.6) ] Bullous Pemphigoid [see Warnings and Precautions (5.7) ]
Storage and Handling
Alogliptin tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows: 25 mg tablet: light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side, available in: NDC 45802-150-65 Bottles of 30 tablets 12.5 mg tablet: yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side, available in: NDC 45802-103-65 Bottles of 30 tablets 6.25 mg tablet: light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side, available in: NDC 45802-087-65 Bottles of 30 tablets
How Supplied
Alogliptin tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows: 25 mg tablet: light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side, available in: NDC 45802-150-65 Bottles of 30 tablets 12.5 mg tablet: yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side, available in: NDC 45802-103-65 Bottles of 30 tablets 6.25 mg tablet: light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side, available in: NDC 45802-087-65 Bottles of 30 tablets
Description
Alogliptin tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Section 34077-8
Animal Data
Alogliptin administered to pregnant rabbits and rats during the period of organogenesis did not cause adverse developmental effects at doses of up to 200 mg/kg and 500 mg/kg, or 149 times and 180 times, the 25 mg clinical dose, respectively, based on plasma drug exposure (AUC). Placental transfer of alogliptin into the fetus was observed following oral dosing to pregnant rats.
No adverse developmental outcomes were observed in offspring when alogliptin was administered to pregnant rats during gestation and lactation at doses up to 250 mg/kg (~ 95 times the 25 mg clinical dose, based on AUC).
Section 42229-5
Limitations of Use
Alogliptin tablet is not recommended for use in patients with type 1 diabetes mellitus.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ALO332 R9 | April 2025 | |||
|
MEDICATION GUIDE Alogliptin Tablets |
|||||
| Read this Medication Guide carefully before you start taking alogliptin tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment. If you have any questions about alogliptin tablets, ask your doctor or pharmacist. | |||||
|
What is the most important information I should know about alogliptin tablets? Serious side effects can happen to people taking alogliptin tablets, including: |
|||||
|
|||||
|
|
|
|||
| Stop taking alogliptin tablets and call your doctor right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen through to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis. | |||||
|
|||||
|
|
|
|||
| These may be symptoms of heart failure. | |||||
| What are alogliptin tablets? | |||||
|
|||||
| It is not known if alogliptin tablets are safe and effective in children under the age of 18. | |||||
|
Who should not take alogliptin tablets? Do not take alogliptin tablets if you: |
|||||
|
|||||
|
|
||||
| If you have any of these symptoms, stop taking alogliptin tablets and contact your doctor or go to the nearest hospital emergency room right away. | |||||
| What should I tell my doctor before and during treatment with alogliptin tablets? | |||||
| Before you take alogliptin tablets, tell your doctor if you: | |||||
|
|||||
| Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. | |||||
| Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist before you start any new medicine. | |||||
| Alogliptin tablets may affect the way other medicines work, and other medicines may affect how alogliptin tablets works. Contact your doctor before you start or stop other types of medicines. | |||||
| How should I take alogliptin tablets? | |||||
|
|||||
| What are the possible side effects of alogliptin tablets? | |||||
| Alogliptin tablets can cause serious side effects, including: | |||||
| See " What is the most important information I should know about alogliptin tablets? " | |||||
|
|||||
|
|
||||
| If you have these symptoms, stop taking alogliptin tablets and contact your doctor right away or go the nearest hospital emergency room. | |||||
|
|||||
|
|
|
|||
|
|||||
|
|
|
|
|
|
|
|||||
| The most common side effects of alogliptin tablets include stuffy or runny nose and sore throat, headache, or cold-like symptoms (upper respiratory tract infection). | |||||
| Tell your doctor if you have any side effect that bothers you or that does not go away. | |||||
| These are not all the possible side effects of alogliptin tablets. For more information, ask your doctor or pharmacist. | |||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||||
| How should I store alogliptin tablets? | |||||
| Store alogliptin tablets at room temperature between 68°F to 77°F (20°C to 25°C). | |||||
| Keep alogliptin tablets and all medicines out of the reach of children. | |||||
| General information about the safe and effective use of alogliptin tablets. | |||||
| Medicines are sometimes prescribed for purposes other than those listed in the Medication Guide. Do not take alogliptin tablets for a condition for which it was not prescribed. Do not give alogliptin tablets to other people, even if they have the same symptoms you have. It may harm them. | |||||
| This Medication Guide summarizes the most important information about alogliptin tablets. If you would like to know more information, talk with your doctor. You can ask your doctor or pharmacist for information about alogliptin tablets that is written for health professionals. | |||||
| For more information go to www.padagis.com or call 1-877-TAKEDA-7 (1-877-825-3327). | |||||
|
What are the ingredients in alogliptin tablets?
Active ingredient: alogliptin Inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, mannitol, and microcrystalline cellulose. In addition, the film-coating contains the following inactive ingredients: ferric oxide (red or yellow), hypromellose, polyethylene glycol and titanium dioxide is marked with printing ink (Gray F1). |
|||||
| Manufactured for Padagis®, Minneapolis, MN 55427 • www.padagis.com | |||||
|
and PADAGIS are registered trademarks of Padagis US LLC. |
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
10 Overdosage
In the event of an overdose, it is reasonable to institute the necessary clinical monitoring and supportive therapy as dictated by the patient's clinical status. Per clinical judgment, it may be reasonable to initiate removal of unabsorbed material from the gastrointestinal tract.
Alogliptin is minimally dialyzable; over a three-hour hemodialysis session, approximately 7% of the drug was removed. Therefore, hemodialysis is unlikely to be beneficial in an overdose situation. It is not known if alogliptin tablets are dialyzable by peritoneal dialysis.
In the event of an overdose, contact the Poison Help Line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
11 Description
Alogliptin tablets contain the active ingredient alogliptin, which is a selective, orally bioavailable inhibitor of the enzymatic activity of DPP-4.
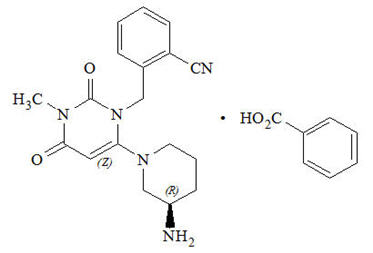
Chemically, alogliptin is prepared as a benzoate salt, which is identified as 2-({6-[(3R)-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl}methyl)benzonitrile monobenzoate. It has a molecular formula of C18H21N5O2∙C7H6O2 and a molecular weight of 461.51 daltons. The structural formula is:
Alogliptin benzoate is a white to off-white crystalline powder containing one asymmetric carbon in the aminopiperidine moiety. It is soluble in dimethylsulfoxide, sparingly soluble in water and methanol, slightly soluble in ethanol and very slightly soluble in octanol and isopropyl acetate.
Each alogliptin tablet contains 34 mg, 17 mg or 8.5 mg alogliptin benzoate, which is equivalent to 25 mg, 12.5 mg or 6.25 mg, respectively, of alogliptin and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, mannitol, and microcrystalline cellulose. In addition, the film coating contains the following inactive ingredients: ferric oxide (red or yellow), hypromellose, polyethylene glycol, and titanium dioxide and is marked with printing ink (Gray F1).
5.1 Pancreatitis
Acute pancreatitis has been reported in the postmarketing setting and in randomized clinical trials. In glycemic control trials in patients with type 2 diabetes mellitus, acute pancreatitis was reported in 6 (0.2%) patients treated with alogliptin tablets 25 mg and 2 (<0.1%) patients treated with active comparators or placebo. In the EXAMINE trial (a cardiovascular outcomes trial of patients with type 2 diabetes mellitus and high cardiovascular (CV) risk), acute pancreatitis was reported in 10 (0.4%) of patients treated with alogliptin tablets and in 7 (0.3%) of patients treated with placebo.
It is unknown whether patients with a history of pancreatitis are at increased risk for pancreatitis while using alogliptin tablets.
After initiation of alogliptin tablets, patients should be observed for signs and symptoms of pancreatitis. If pancreatitis is suspected, alogliptin tablets should promptly be discontinued and appropriate management should be initiated.
5.2 Heart Failure
In the EXAMINE trial which enrolled patients with type 2 diabetes mellitus and recent acute coronary syndrome, 106 (3.9%) of patients treated with alogliptin tablets and 89 (3.3%) of patients treated with placebo were hospitalized for congestive heart failure.
Consider the risks and benefits of alogliptin tablets prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment, and observe these patients for signs and symptoms of heart failure during therapy. Patients should be advised of the characteristic symptoms of heart failure and should be instructed to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of alogliptin tablets.
8.4 Pediatric Use
The safety and effectiveness of alogliptin tablets have not been established in pediatric patients.
Effectiveness of alogliptin tablets was not demonstrated in a 52 week, randomized, double-blind, placebo-controlled trial (NCT02856113) in 151 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes mellitus.
8.5 Geriatric Use
Of the total number of patients (N=9052) in clinical safety and efficacy trials treated with alogliptin tablets, 2,257 (24.9%) patients were 65 years and older and 386 (4.3%) patients were 75 years and older. No overall differences in safety or effectiveness were observed between patients 65 years and over and younger patients.
4 Contraindications
Alogliptin tablets is contraindicated in patients with a history of serious hypersensitivity to alogliptin or any of the excipients in Alogliptin tablets. Reactions such as anaphylaxis, angioedema and severe cutaneous adverse reactions have been reported [see Warnings and Precautions (5.3), Adverse Reactions (6.2)].
5.4 Hepatic Effects
There have been postmarketing reports of fatal and nonfatal hepatic failure in patients taking alogliptin tablets, although some of the reports contain insufficient information necessary to establish the probable cause [see Adverse Reactions (6.2)].
In glycemic control trials in patients with type 2 diabetes mellitus, serum alanine aminotransferase (ALT) elevations greater than three times the upper limit of normal (ULN) were reported in 1.3% of patients treated with alogliptin tablets 25 mg and 1.7% of patients treated with active comparators or placebo. In the EXAMINE trial (a cardiovascular outcomes trial of patients with type 2 diabetes mellitus and high cardiovascular (CV) risk), increases in serum alanine aminotransferase three times the upper limit of the reference range occurred in 2.4% of patients treated with alogliptin tablets and in 1.8% of patients treated with placebo.
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient is found to have clinically significant liver enzyme elevations and if abnormal liver tests persist or worsen, alogliptin tablets should be interrupted and investigation done to establish the probable cause. Alogliptin tablets should not be restarted in these patients without another explanation for the liver test abnormalities.
6 Adverse Reactions
The following serious adverse reactions are described below or elsewhere in the prescribing information:
- Pancreatitis [see Warnings and Precautions (5.1)]
- Heart Failure [see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Hepatic Effects [see Warnings and Precautions (5.4)]
- Severe and Disabling Arthralgia [see Warnings and Precautions (5.6)]
- Bullous Pemphigoid [see Warnings and Precautions (5.7)]
8.6 Renal Impairment
A total of 602 adult patients with moderate renal impairment (eGFR ≥30 and <60 mL/min/1.73 m2) and 4 patients with severe renal impairment/end-stage renal disease (eGFR <30 mL/min/1.73 m2 or <15 mL/min/1.73 m2, respectively) at baseline were treated with alogliptin tablets in clinical trials in patients with type 2 diabetes mellitus.
In the EXAMINE trial of high CV risk type 2 diabetes mellitus patients, 694 patients had moderate renal impairment and 78 patients had severe renal impairment or end-stage renal disease at baseline.
The recommended dose is 12.5 mg once daily in patients with moderate renal impairment and 6.25 mg once daily in patients with severe renal impairment, as well as in patients with ESRD requiring dialysis. Alogliptin tablets may be administered without regard to the timing of the dialysis.
12.2 Pharmacodynamics
Dose-Response Relationships
Single-dose administration of alogliptin tablets to healthy subjects resulted in a peak inhibition of DPP-4 within two to three hours after dosing. The peak inhibition of DPP-4 exceeded 93% across doses of 12.5 mg to 800 mg. Inhibition of DPP-4 remained above 80% at 24 hours for doses greater than or equal to 25 mg. Peak and total exposure over 24 hours to active GLP-1 were three- to four-fold greater with alogliptin tablets (at doses of 25 to 200 mg) than placebo. In a 16 week, double-blind, placebo-controlled study, alogliptin tablets 25 mg demonstrated decreases in postprandial glucagon while increasing postprandial active GLP-1 levels compared to placebo over an eight hour period following a standardized meal. It is unclear how these findings relate to changes in overall glycemic control in patients with type 2 diabetes mellitus. In this study, alogliptin tablets 25 mg demonstrated decreases in two hour postprandial glucose compared to placebo (-30 mg/dL versus 17 mg/dL, respectively).
Multiple-dose administration of alogliptin to patients with type 2 diabetes mellitus also resulted in a peak inhibition of DPP-4 within one to two hours and exceeded 93% across all doses (25 mg, 100 mg and 400 mg) after a single dose and after 14 days of once-daily dosing. At these doses of alogliptin tablets, inhibition of DPP-4 remained above 81% at 24 hours after 14 days of dosing.
12.3 Pharmacokinetics
The pharmacokinetics of alogliptin tablets has been studied in healthy subjects and in patients with type 2 diabetes mellitus. The pharmacokinetics of alogliptin tablets were similar in healthy subjects and in patients with type 2 diabetes mellitus.
After multiple-dose administration up to 400 mg for 14 days in patients with type 2 diabetes mellitus, accumulation of alogliptin was minimal with an increase in total [e.g., area under the plasma concentration curve (AUC)] and peak (i.e., Cmax) alogliptin exposures of 34% and 9%, respectively. Total and peak exposure to alogliptin increased proportionally across single doses and multiple doses of alogliptin ranging from 25 mg to 400 mg. The intersubject coefficient of variation for alogliptin AUC was 17%.
2.1 Recommended Dosage
- The recommended dosage of alogliptin tablets is 25 mg taken orally once daily. Do not spilt tablet.
- Alogliptin tablets may be taken with or without food. [see Clinical Pharmacology (12.3)]
- Instruct patients if a dose is missed, not to double their next dose.
5.7 Bullous Pemphigoid
Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving alogliptin tablets. If bullous pemphigoid is suspected, alogliptin tablets should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
No dose adjustments are required in patients with mild to moderate hepatic impairment (Child-Pugh Grade A and B) [see Clinical Pharmacology (12.3)]. Alogliptin tablets have not been studied in patients with severe hepatic impairment (Child-Pugh Grade C). Use caution when administering alogliptin tablets to patients with liver disease [see Warnings and Precautions (5.4)].
1 Indications and Usage
Alogliptin tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
12.1 Mechanism of Action
Increased concentrations of the incretin hormones such as glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are released into the bloodstream from the small intestine in response to meals. These hormones cause insulin release from the pancreatic beta cells in a glucose-dependent manner but are inactivated by the DPP-4 enzyme within minutes. GLP-1 also lowers glucagon secretion from pancreatic alpha cells, reducing hepatic glucose production. In patients with type 2 diabetes mellitus, concentrations of GLP-1 are reduced but the insulin response to GLP-1 is preserved. Alogliptin is a DPP-4 inhibitor that slows the inactivation of the incretin hormones, thereby increasing their bloodstream concentrations and reducing fasting and postprandial glucose concentrations in a glucose-dependent manner in patients with type 2 diabetes mellitus. Alogliptin selectively binds to and inhibits DPP-4 but not DPP-8 or DPP-9 activity in vitro at concentrations approximating therapeutic exposures.
5 Warnings and Precautions
- Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue alogliptin tablets. (5.1)
- Heart failure: Consider the risks and benefits of alogliptin tablets prior to initiating treatment in patients at risk for heart failure. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of alogliptin tablets. (5.2)
- Hypersensitivity: There have been postmarketing reports of serious hypersensitivity reactions in patients treated with alogliptin tablets such as anaphylaxis, angioedema and severe cutaneous adverse reactions, including Stevens-Johnson syndrome. If hypersensitivity reactions occur, discontinue alogliptin tablets, treat promptly and monitor until signs and symptoms resolve. (5.3)
- Hepatic effects: Postmarketing reports of hepatic failure, sometimes fatal. Causality cannot be excluded. If liver injury is detected, promptly interrupt alogliptin tablets and assess patient for probable cause, then treat cause if possible, to resolution or stabilization. Do not restart alogliptin tablets if liver injury is confirmed and no alternative etiology can be found. (5.4)
- Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating Alogliptin tablets. (5.5)
- Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.6)
- Bullous pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue alogliptin tablets. (5.7)
2 Dosage and Administration
- The recommended dosage in patients with normal renal function or mild renal impairment is 25 mg orally once daily. (2.1)
- Can be taken with or without food. (2.1)
- Adjust dosage if moderate or severe renal impairment or end-stage renal disease (ESRD). (2.2)
| Degree of Renal Impairment | Creatinine Clearance (mL/min) |
Recommended Dosage |
|---|---|---|
| Moderate | ≥30 to <60 | 12.5 mg once daily |
| Severe/ESRD | <30 | 6.25 mg once daily |
3 Dosage Forms and Strengths
- 25 mg tablets are light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side.
- 12.5 mg tablets are yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side.
- 6.25 mg tablets are light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during the postmarketing use of alogliptin tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: acute pancreatitis, diarrhea, constipation, nausea, ileus
Hepatobiliary Disorders: fulminant hepatic failure
Immune System Disorders: hypersensitivity reactions including anaphylaxis
Investigations: hepatic enzyme elevations
Musculoskeletal and Connective Tissue Disorders: severe and disabling arthralgia, rhabdomyolysis
Renal and Urinary Disorders: tubulointerstitial nephritis
Skin and Subcutaneous Tissue Disorders: angioedema, rash, urticaria and severe cutaneous adverse reactions including Stevens-Johnson syndrome, bullous pemphigoid
5.3 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions in patients treated with alogliptin tablets [see Adverse Reactions (6.2)]. These reactions include anaphylaxis, angioedema, and severe cutaneous adverse reactions, including Stevens-Johnson syndrome. If a serious hypersensitivity reaction is suspected, discontinue alogliptin tablets, assess for other potential causes for the event and institute alternative treatment for diabetes mellitus. Use caution in patients with a history of angioedema with another dipeptidyl peptidase-4 (DPP-4) inhibitor because it is unknown whether such patients will be predisposed to angioedema with alogliptin tablets.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 14,778 patients with type 2 diabetes mellitus participated in 14 randomized, double-blind, controlled clinical trials of whom 9,052 subjects were treated with alogliptin tablets, 3,469 subjects were treated with placebo and 2,257 were treated with an active comparator. The racial distribution of patients exposed to trial medication was 71% White, 17% Asian, 6% Black or African American, 2% American Indian or Alaska Native, 0% Native Hawaiian/Other Pacific Islander and 5% Multiracial or other racial groups. The ethnic distribution was 30% Hispanic or Latino and 70% was not Hispanic or Latino. The mean duration of diabetes mellitus was seven years, the mean body mass index (BMI) was 31 kg/m2 (49% of patients had a BMI ≥30 kg/m2), and the mean age was 58 years (26% of patients ≥65 years of age). The mean exposure to alogliptin tablets was 49 weeks with 3,348 subjects treated for more than one year.
In a pooled analysis of these 14 controlled clinical trials, the overall incidence of adverse reactions was 73% in patients treated with alogliptin tablets 25 mg compared to 75% with placebo and 70% with active comparator. Overall discontinuation of therapy due to adverse reactions was 6.8% with alogliptin tablets 25 mg compared to 8.4% with placebo or 6.2% with active comparator.
Adverse reactions reported in ≥4% of adult patients treated with alogliptin tablets 25 mg and more frequently than in patients who received placebo are summarized in Table 1.
| Number of Patients (%) | |||
|---|---|---|---|
| Alogliptin Tablets 25 mg |
Placebo | Active Comparator | |
| N=6447 | N=3469 | N=2257 | |
| Nasopharyngitis | 309 (5) | 152 (4) | 113 (5) |
| Upper Respiratory Tract Infection | 287 (4) | 121 (4) | 113 (5) |
| Headache | 278 (4) | 101 (3) | 121 (5) |
14.4 Cardiovascular Safety Trial
A randomized, double-blind, placebo-controlled cardiovascular outcomes trial (EXAMINE) was conducted to evaluate the cardiovascular risk of alogliptin tablets. The trial compared the risk of major adverse cardiovascular events (MACE) between alogliptin tablets (N=2701) and placebo (N=2679) when added to standard of care therapies for diabetes mellitus and atherosclerotic vascular disease (ASCVD). The trial was event driven and patients were followed until a sufficient number of primary outcome events accrued.
Eligible patients were adults with type 2 diabetes mellitus who had inadequate glycemic control at baseline (e.g., HbA1c >6.5%) and had been hospitalized for an acute coronary syndrome event (e.g., acute myocardial infarction or unstable angina requiring hospitalization) 15 to 90 days prior to randomization. The dose of alogliptin tablets was based on estimated renal function at baseline per dosage and administration recommendations [see Dosage and Administration (2.2)]. The average time between an acute coronary syndrome event and randomization was approximately 48 days.
The mean age of the population was 61 years. Most patients were male (68%), White (73%), and were recruited from outside of the United States (86%). Asian and Black or African American patients contributed 20% and 4% of the total population, respectively. At the time of randomization patients had a diagnosis of type 2 diabetes mellitus for approximately 9 years, 87% had a prior myocardial infarction and 14% were current smokers. Hypertension (83%) and renal impairment (27% with an eGFR ≤60 mL/min/1.73 m2) were prevalent co-morbid conditions. Use of medications to treat diabetes mellitus (e.g., metformin 73%, sulfonylurea 54%, insulin 41%), and ASCVD (e.g., statin 94%, aspirin 93%, renin-angiotensin system blocker 88%, beta-blocker 87%) was similar between patients randomized to alogliptin tablets and placebo at baseline. During the trial, medications to treat diabetes mellitus and ASCVD could be adjusted to ensure care for these conditions adhered to standard of care recommendations set by local practice guidelines.
The primary endpoint in EXAMINE was the time to first occurrence of a MACE defined as the composite of cardiovascular death, nonfatal myocardial infarction (MI), or nonfatal stroke. The trial was designed to exclude a pre-specified risk margin of 1.3 for the hazard ratio of MACE. The median exposure to trial drug was 526 days and 95% of the patients were followed to trial completion or death.
Table 12 shows the trial results for the primary MACE composite endpoint and the contribution of each component to the primary MACE endpoint. The upper bound of the confidence interval was 1.16 and excluded a risk margin larger than 1.3.
| Composite of first event of CV death, nonfatal MI or nonfatal stroke (MACE) | Alogliptin | Placebo | Hazard Ratio | ||
| Number of Patients (%) |
Rate per 100 PY
Patient Years (PY)
|
Number of Patients (%) | Rate per 100 PY | (98% CI) | |
| N=2701 | N=2679 | ||||
| 305 (11.3) | 7.6 | 316 (11.8) | 7.9 | 0.96 (0.80, 1.16) | |
| CV Death | 89 (3.3) | 2.2 | 111 (4.1) | 2.8 | |
| Non-fatal MI | 187 (6.9) | 4.6 | 173 (6.5) | 4.3 | |
| Non-fatal stroke | 29 (1.1) | 0.7 | 32 (1.2) | 0.8 |
The Kaplan-Meier based cumulative event probability is presented in Figure 4 for the time to first occurrence of the primary MACE composite endpoint by treatment arm. The curves for placebo and alogliptin tablets overlap throughout the duration of the trial. The observed incidence of MACE was highest within the first 60 days after randomization in both treatment arms (14.8 MACE per 100 PY), decreased from day 60 to the end of the first year (8.4 per 100 PY) and was lowest after one year of follow-up (5.2 per 100 PY).
| Figure 4. Observed Cumulative Rate of MACE in EXAMINE |
The rate of all cause death was similar between treatment arms with 153 (3.6 per 100 PY) recorded among patients randomized to alogliptin tablets and 173 (4.1 per 100 PY) among patients randomized to placebo. A total of 112 deaths (2.9 per 100 PY) among patients on alogliptin tablets and 130 among patients on placebo (3.5 per 100 PY) were adjudicated as cardiovascular deaths.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.2 Patients With Renal Impairment
- Assess renal function prior to initiation of alogliptin tablets and periodically thereafter [see Use in Specific Populations (8.6)].
- No dose adjustment of alogliptin tablets is necessary for patients with mild renal impairment (creatinine clearance [CrCl] ≥60 mL/min).
- The dose of alogliptin tablets is 12.5 mg once daily for patients with moderate renal impairment (CrCl ≥30 to <60 mL/min).
- The dose of alogliptin tablets is 6.25 mg once daily for patients with severe renal impairment (CrCl ≥15 to <30 mL/min) or with end-stage renal disease (ESRD) (CrCl <15 mL/min or requiring hemodialysis). Alogliptin tablets may be administered without regard to the timing of dialysis. Alogliptin tablets have not been studied in patients undergoing peritoneal dialysis [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
5.6 Severe and Disabling Arthralgia
There have been postmarketing reports of severe and disabling arthralgia in patients taking DPP-4 inhibitors. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider DPP-4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling
Alogliptin tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows:
25 mg tablet: light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side, available in:
| NDC 45802-150-65 | Bottles of 30 tablets |
12.5 mg tablet: yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side, available in:
| NDC 45802-103-65 | Bottles of 30 tablets |
6.25 mg tablet: light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side, available in:
| NDC 45802-087-65 | Bottles of 30 tablets |
7.1 Insulin Secretagogues and Insulin
Insulin and insulin secretagogues are known to cause hypoglycemia. Coadministration of alogliptin tablets with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue and insulin to reduce the risk of hypoglycemia [see Warnings and Precautions (5.5)].
Principal Display Panel 25 Mg Tablet Bottle Label
NDC 45802-150-65
Rx Only
Alogliptin
Tablets
25 mg
DISPENSE WITH
MEDICATION GUIDE
30 Tablets
Padagis®
Principal Display Panel 12.5 Mg Tablet Bottle Label
NDC 45802-103-65
Rx Only
Alogliptin
Tablets
12.5 mg
DISPENSE WITH
MEDICATION GUIDE
30 Tablets
Padagis®
Principal Display Panel 6.25 Mg Tablet Bottle Label
NDC 45802-087-65
Rx Only
Alogliptin
Tablets
6.25 mg
DISPENSE WITH
MEDICATION GUIDE
30 Tablets
Padagis®
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Rats were administered oral doses of 75, 400 and 800 mg/kg alogliptin for two years. No drug-related tumors were observed up to 75 mg/kg or approximately 32 times the maximum recommended clinical dose of 25 mg, based on area under the plasma concentration curve (AUC) exposure. At higher doses (approximately 308 times the maximum recommended clinical dose of 25 mg), a combination of thyroid C-cell adenomas and carcinomas increased in male but not female rats. No drug-related tumors were observed in mice after administration of 50, 150 or 300 mg/kg alogliptin for two years, or up to approximately 51 times the maximum recommended clinical dose of 25 mg, based on AUC exposure.
Mutagenesis
Alogliptin was not mutagenic or clastogenic, with and without metabolic activation, in the Ames test with S. typhimurium and E. coli or the cytogenetic assay in mouse lymphoma cells. Alogliptin was negative in the in vivo mouse micronucleus study.
Impairment of Fertility
In a fertility study in rats, alogliptin had no adverse effects on early embryonic development, mating or fertility at doses up to 500 mg/kg, or approximately 172 times the clinical dose based on plasma drug exposure (AUC).
14.2 Patients With Inadequate Glycemic Control On Diet and Exercise
A total of 1,768 patients with type 2 diabetes mellitus participated in three double-blind trials to evaluate the efficacy and safety of alogliptin tablets in patients with inadequate glycemic control on diet and exercise. All three trials had a four week, single-blind, placebo run-in period followed by a 26 week randomized treatment period. Patients who failed to meet prespecified hyperglycemic goals during the 26 week treatment periods received glycemic rescue therapy.
In a 26 week, double-blind, placebo-controlled trial, a total of 329 patients (mean baseline A1C = 8%) were randomized to receive alogliptin tablets 12.5 mg, alogliptin tablets 25 mg or placebo once daily. Treatment with alogliptin tablets 25 mg resulted in statistically significant improvements from baseline in A1C and fasting plasma glucose (FPG) compared to placebo at Week 26 (Table 3). A total of 8% of patients receiving alogliptin tablets 25 mg and 30% of those receiving placebo required glycemic rescue therapy.
Improvements in A1C were not affected by gender, age or baseline body mass index (BMI).
The mean change in body weight with alogliptin tablets was similar to placebo.
| Alogliptin Tablets 25 mg |
Placebo | |
|---|---|---|
| A1C (%) | N=128 | N=63 |
| Baseline (mean) | 7.9 | 8.0 |
| Change from baseline (adjusted mean Least squares means adjusted for treatment, baseline value, geographic region and duration of diabetes mellitus ) |
-0.6 | 0 |
| Difference from placebo (adjusted mean with 95% confidence interval) | -0.6 p<0.01 compared to placebo (-0.8, -0.3) |
˗ |
| % of patients (n/N) achieving A1C ≤7% | 44% (58/131) | 23% (15/64) |
| FPG (mg/dL) | N=129 | N=64 |
| Baseline (mean) | 172 | 173 |
| Change from baseline (adjusted mean) | -16 | 11 |
| Difference from placebo (adjusted mean with 95% confidence interval) | -28 (-40, -15) | ˗ |
In a 26 week, double-blind, active-controlled trial, a total of 655 patients (mean baseline A1C = 8.8%) were randomized to receive alogliptin tablets 25 mg alone, pioglitazone 30 mg alone, alogliptin tablets 12.5 mg with pioglitazone 30 mg or alogliptin tablets 25 mg with pioglitazone 30 mg once daily. Coadministration of alogliptin tablets 25 mg with pioglitazone 30 mg resulted in statistically significant improvements from baseline in A1C and FPG compared to alogliptin tablets 25 mg alone and to pioglitazone 30 mg alone (Table 4). A total of 3% of patients receiving alogliptin tablets 25 mg coadministered with pioglitazone 30 mg, 11% of those receiving alogliptin tablets 25 mg alone and 6% of those receiving pioglitazone 30 mg alone required glycemic rescue.
Improvements in A1C were not affected by gender, age or baseline BMI.
The mean increase in body weight was similar between pioglitazone alone and alogliptin tablets when coadministered with pioglitazone.
| Alogliptin Tablets 25 mg |
Pioglitazone 30 mg |
Alogliptin Tablets 25 mg + Pioglitazone 30 mg |
|
|---|---|---|---|
| A1C (%) | N=160 | N=153 | N=158 |
| Baseline (mean) | 8.8 | 8.8 | 8.8 |
| Change from baseline (adjusted mean Least squares means adjusted for treatment, geographic region and baseline value ) |
-1.0 | -1.2 | -1.7 |
| Difference from alogliptin tablets 25 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -0.8 p<0.01 compared to alogliptin tablets 25 mg or pioglitazone 30 mg (-1.0, -0.5) |
| Difference from pioglitazone 30 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -0.6 (-0.8, -0.3) |
| % of Patients (n/N) achieving A1C ≤7% | 24% (40/164) | 34% (55/163) | 63% (103/164) |
| FPG (mg/dL) | N=162 | N=157 | N=162 |
| Baseline (mean) | 189 | 189 | 185 |
| Change from baseline (adjusted mean) | -26 | -37 | -50 |
| Difference from alogliptin tablets 25 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -24 (-34, -15) |
| Difference from pioglitazone 30 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -13 (-22, -4) |
In a 26 week, double-blind, placebo-controlled trial, a total of 784 patients inadequately controlled on diet and exercise alone (mean baseline A1C = 8.4%) were randomized to one of seven treatment groups: placebo; metformin HCl 500 mg or metformin HCl 1000 mg twice daily; alogliptin tablets 12.5 mg twice daily; alogliptin tablets 25 mg daily; or alogliptin tablets 12.5 mg in combination with metformin HCl 500 mg or metformin HCl 1000 mg twice daily. Both coadministration treatment arms (alogliptin tablets 12.5 mg + metformin HCl 500 mg and alogliptin tablets 12.5 mg + metformin HCl 1000 mg) resulted in statistically significant improvements in A1C and FPG when compared with their respective individual alogliptin and metformin component regimens (Table 5). Coadministration treatment arms demonstrated improvements in two hour postprandial glucose (PPG) compared to alogliptin tablets alone or metformin alone (Table 5). A total of 12.3% of patients receiving alogliptin tablets 12.5 mg + metformin HCl 500 mg, 2.6% of patients receiving alogliptin tablets 12.5 mg + metformin HCl 1000 mg, 17.3% of patients receiving alogliptin tablets 12.5 mg, 22.9% of patients receiving metformin HCl 500 mg, 10.8% of patients receiving metformin HCl 1000 mg and 38.7% of patients receiving placebo required glycemic rescue.
Improvements in A1C were not affected by gender, age, race or baseline BMI. The mean decrease in body weight was similar between metformin alone and alogliptin tablets when coadministered with metformin.
| Placebo | Alogliptin Tablets 12.5 mg Twice Daily |
Metformin HCl 500 mg Twice Daily |
Metformin HCl 1000 mg Twice Daily |
Alogliptin Tablets 12.5 mg + Metformin HCl 500 mg Twice Daily |
Alogliptin Tablets 12.5 mg + Metformin HCl 1000 mg Twice Daily |
|
|---|---|---|---|---|---|---|
|
A1C (%) Intent-to-treat population using last observation on trial prior to discontinuation of double-blind trial medication or sulfonylurea rescue therapy for patients needing rescue
|
N=102 | N=104 | N=103 | N=108 | N=102 | N=111 |
| Baseline (mean) | 8.5 | 8.4 | 8.5 | 8.4 | 8.5 | 8.4 |
| Change from baseline (adjusted mean Least squares means adjusted for treatment, geographic region and baseline value ) |
0.1 | -0.6 | -0.7 | -1.1 | -1.2 | -1.6 |
| Difference from metformin (adjusted mean with 95% confidence interval) | - | - | - | - | -0.6 p<0.05 when compared to metformin and alogliptin tablets alone
(-0.9, -0.3) |
-0.4
(-0.7, -0.2) |
| Difference from Alogliptin Tablets (adjusted mean with 95% confidence interval) | - | - | - | - | -0.7
(-1.0, -0.4) |
-1.0
(-1.3, -0.7) |
| % of Patients (n/N) achieving A1C <7% Compared using logistic regression
|
4% (4/102) |
20% (21/104) |
27% (28/103) |
34% (37/108) |
47%
(48/102) |
59%
(66/111) |
| FPG (mg/dL) | N=105 | N=106 | N=106 | N=110 | N=106 | N=112 |
| Baseline (mean) | 187 | 177 | 180 | 181 | 176 | 185 |
| Change from baseline (adjusted mean) | 12 | -10 | -12 | -32 | -32 | -46 |
| Difference from metformin (adjusted mean with 95% confidence interval) | - | - | - | - | -20
(-33, -8) |
-14
(-26, -2) |
| Difference from Alogliptin Tablets (adjusted mean with 95% confidence interval) | - | - | - | - | -22
(-35, -10) |
-36
(-49, -24) |
|
2-Hour PPG (mg/dL)
Intent-to-treat population using data available at Week 26
|
N=26 | N=34 | N=28 | N=37 | N=31 | N=37 |
| Baseline (mean) | 263 | 272 | 247 | 266 | 261 | 268 |
| Change from baseline (adjusted mean) | -21 | -43 | -49 | -54 | -68 | -86 |
| Difference from metformin (adjusted mean with 95% confidence interval) | - | - | - | - | -19 (-49, 11) |
-32
(-58, -5) |
| Difference from Alogliptin Tablets (adjusted mean with 95% confidence interval) | - | - | - | - | -25 (-53, -3) |
-43
(-70, -16) |
14.1 Overview of Clinical Trials in Adults With Type 2 Diabetes Mellitus
Alogliptin tablets have been studied as monotherapy and in combination with metformin, a sulfonylurea, a thiazolidinedione (either alone or in combination with metformin or a sulfonylurea) and insulin (either alone or in combination with metformin).
A total of 14,053 patients with type 2 diabetes mellitus were randomized in 11 double-blind, placebo- or active-controlled clinical safety and efficacy trials conducted to evaluate the effects of alogliptin tablets on glycemic control. The racial distribution of patients exposed to trials medication was 70% White, 17% Asian, 6% Black or African American, 2% American Indian or Alaska Native, 0% Native Hawaiian/Other Pacific Islander and 5% Multiracial or other racial groups. The ethnic distribution was 30% Hispanic or Latino. Patients had an overall mean age of 57 years (range 21 to 91 years).
In patients with type 2 diabetes mellitus, treatment with alogliptin tablets produced clinically meaningful and statistically significant improvements in hemoglobin A1c (A1C) compared to placebo. As is typical for trials of agents to treat type 2 diabetes mellitus, the mean reduction in A1C with alogliptin tablets appears to be related to the degree of A1C elevation at baseline.
Alogliptin tablets had similar changes from baseline in serum lipids compared to placebo.
5.5 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues
Insulin and insulin secretagogues, such as sulfonylureas, are known to cause hypoglycemia. Therefore, a lower dosage of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with alogliptin tablets.
Structured Label Content
Section 34077-8 (34077-8)
Animal Data
Alogliptin administered to pregnant rabbits and rats during the period of organogenesis did not cause adverse developmental effects at doses of up to 200 mg/kg and 500 mg/kg, or 149 times and 180 times, the 25 mg clinical dose, respectively, based on plasma drug exposure (AUC). Placental transfer of alogliptin into the fetus was observed following oral dosing to pregnant rats.
No adverse developmental outcomes were observed in offspring when alogliptin was administered to pregnant rats during gestation and lactation at doses up to 250 mg/kg (~ 95 times the 25 mg clinical dose, based on AUC).
Section 42229-5 (42229-5)
Limitations of Use
Alogliptin tablet is not recommended for use in patients with type 1 diabetes mellitus.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ALO332 R9 | April 2025 | |||
|
MEDICATION GUIDE Alogliptin Tablets |
|||||
| Read this Medication Guide carefully before you start taking alogliptin tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment. If you have any questions about alogliptin tablets, ask your doctor or pharmacist. | |||||
|
What is the most important information I should know about alogliptin tablets? Serious side effects can happen to people taking alogliptin tablets, including: |
|||||
|
|||||
|
|
|
|||
| Stop taking alogliptin tablets and call your doctor right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen through to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis. | |||||
|
|||||
|
|
|
|||
| These may be symptoms of heart failure. | |||||
| What are alogliptin tablets? | |||||
|
|||||
| It is not known if alogliptin tablets are safe and effective in children under the age of 18. | |||||
|
Who should not take alogliptin tablets? Do not take alogliptin tablets if you: |
|||||
|
|||||
|
|
||||
| If you have any of these symptoms, stop taking alogliptin tablets and contact your doctor or go to the nearest hospital emergency room right away. | |||||
| What should I tell my doctor before and during treatment with alogliptin tablets? | |||||
| Before you take alogliptin tablets, tell your doctor if you: | |||||
|
|||||
| Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. | |||||
| Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist before you start any new medicine. | |||||
| Alogliptin tablets may affect the way other medicines work, and other medicines may affect how alogliptin tablets works. Contact your doctor before you start or stop other types of medicines. | |||||
| How should I take alogliptin tablets? | |||||
|
|||||
| What are the possible side effects of alogliptin tablets? | |||||
| Alogliptin tablets can cause serious side effects, including: | |||||
| See " What is the most important information I should know about alogliptin tablets? " | |||||
|
|||||
|
|
||||
| If you have these symptoms, stop taking alogliptin tablets and contact your doctor right away or go the nearest hospital emergency room. | |||||
|
|||||
|
|
|
|||
|
|||||
|
|
|
|
|
|
|
|||||
| The most common side effects of alogliptin tablets include stuffy or runny nose and sore throat, headache, or cold-like symptoms (upper respiratory tract infection). | |||||
| Tell your doctor if you have any side effect that bothers you or that does not go away. | |||||
| These are not all the possible side effects of alogliptin tablets. For more information, ask your doctor or pharmacist. | |||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||||
| How should I store alogliptin tablets? | |||||
| Store alogliptin tablets at room temperature between 68°F to 77°F (20°C to 25°C). | |||||
| Keep alogliptin tablets and all medicines out of the reach of children. | |||||
| General information about the safe and effective use of alogliptin tablets. | |||||
| Medicines are sometimes prescribed for purposes other than those listed in the Medication Guide. Do not take alogliptin tablets for a condition for which it was not prescribed. Do not give alogliptin tablets to other people, even if they have the same symptoms you have. It may harm them. | |||||
| This Medication Guide summarizes the most important information about alogliptin tablets. If you would like to know more information, talk with your doctor. You can ask your doctor or pharmacist for information about alogliptin tablets that is written for health professionals. | |||||
| For more information go to www.padagis.com or call 1-877-TAKEDA-7 (1-877-825-3327). | |||||
|
What are the ingredients in alogliptin tablets?
Active ingredient: alogliptin Inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, mannitol, and microcrystalline cellulose. In addition, the film-coating contains the following inactive ingredients: ferric oxide (red or yellow), hypromellose, polyethylene glycol and titanium dioxide is marked with printing ink (Gray F1). |
|||||
| Manufactured for Padagis®, Minneapolis, MN 55427 • www.padagis.com | |||||
|
and PADAGIS are registered trademarks of Padagis US LLC. |
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose, it is reasonable to institute the necessary clinical monitoring and supportive therapy as dictated by the patient's clinical status. Per clinical judgment, it may be reasonable to initiate removal of unabsorbed material from the gastrointestinal tract.
Alogliptin is minimally dialyzable; over a three-hour hemodialysis session, approximately 7% of the drug was removed. Therefore, hemodialysis is unlikely to be beneficial in an overdose situation. It is not known if alogliptin tablets are dialyzable by peritoneal dialysis.
In the event of an overdose, contact the Poison Help Line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
11 Description (11 DESCRIPTION)
Alogliptin tablets contain the active ingredient alogliptin, which is a selective, orally bioavailable inhibitor of the enzymatic activity of DPP-4.
Chemically, alogliptin is prepared as a benzoate salt, which is identified as 2-({6-[(3R)-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl}methyl)benzonitrile monobenzoate. It has a molecular formula of C18H21N5O2∙C7H6O2 and a molecular weight of 461.51 daltons. The structural formula is:
Alogliptin benzoate is a white to off-white crystalline powder containing one asymmetric carbon in the aminopiperidine moiety. It is soluble in dimethylsulfoxide, sparingly soluble in water and methanol, slightly soluble in ethanol and very slightly soluble in octanol and isopropyl acetate.
Each alogliptin tablet contains 34 mg, 17 mg or 8.5 mg alogliptin benzoate, which is equivalent to 25 mg, 12.5 mg or 6.25 mg, respectively, of alogliptin and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, mannitol, and microcrystalline cellulose. In addition, the film coating contains the following inactive ingredients: ferric oxide (red or yellow), hypromellose, polyethylene glycol, and titanium dioxide and is marked with printing ink (Gray F1).
5.1 Pancreatitis
Acute pancreatitis has been reported in the postmarketing setting and in randomized clinical trials. In glycemic control trials in patients with type 2 diabetes mellitus, acute pancreatitis was reported in 6 (0.2%) patients treated with alogliptin tablets 25 mg and 2 (<0.1%) patients treated with active comparators or placebo. In the EXAMINE trial (a cardiovascular outcomes trial of patients with type 2 diabetes mellitus and high cardiovascular (CV) risk), acute pancreatitis was reported in 10 (0.4%) of patients treated with alogliptin tablets and in 7 (0.3%) of patients treated with placebo.
It is unknown whether patients with a history of pancreatitis are at increased risk for pancreatitis while using alogliptin tablets.
After initiation of alogliptin tablets, patients should be observed for signs and symptoms of pancreatitis. If pancreatitis is suspected, alogliptin tablets should promptly be discontinued and appropriate management should be initiated.
5.2 Heart Failure
In the EXAMINE trial which enrolled patients with type 2 diabetes mellitus and recent acute coronary syndrome, 106 (3.9%) of patients treated with alogliptin tablets and 89 (3.3%) of patients treated with placebo were hospitalized for congestive heart failure.
Consider the risks and benefits of alogliptin tablets prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment, and observe these patients for signs and symptoms of heart failure during therapy. Patients should be advised of the characteristic symptoms of heart failure and should be instructed to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of alogliptin tablets.
8.4 Pediatric Use
The safety and effectiveness of alogliptin tablets have not been established in pediatric patients.
Effectiveness of alogliptin tablets was not demonstrated in a 52 week, randomized, double-blind, placebo-controlled trial (NCT02856113) in 151 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes mellitus.
8.5 Geriatric Use
Of the total number of patients (N=9052) in clinical safety and efficacy trials treated with alogliptin tablets, 2,257 (24.9%) patients were 65 years and older and 386 (4.3%) patients were 75 years and older. No overall differences in safety or effectiveness were observed between patients 65 years and over and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
Alogliptin tablets is contraindicated in patients with a history of serious hypersensitivity to alogliptin or any of the excipients in Alogliptin tablets. Reactions such as anaphylaxis, angioedema and severe cutaneous adverse reactions have been reported [see Warnings and Precautions (5.3), Adverse Reactions (6.2)].
5.4 Hepatic Effects
There have been postmarketing reports of fatal and nonfatal hepatic failure in patients taking alogliptin tablets, although some of the reports contain insufficient information necessary to establish the probable cause [see Adverse Reactions (6.2)].
In glycemic control trials in patients with type 2 diabetes mellitus, serum alanine aminotransferase (ALT) elevations greater than three times the upper limit of normal (ULN) were reported in 1.3% of patients treated with alogliptin tablets 25 mg and 1.7% of patients treated with active comparators or placebo. In the EXAMINE trial (a cardiovascular outcomes trial of patients with type 2 diabetes mellitus and high cardiovascular (CV) risk), increases in serum alanine aminotransferase three times the upper limit of the reference range occurred in 2.4% of patients treated with alogliptin tablets and in 1.8% of patients treated with placebo.
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient is found to have clinically significant liver enzyme elevations and if abnormal liver tests persist or worsen, alogliptin tablets should be interrupted and investigation done to establish the probable cause. Alogliptin tablets should not be restarted in these patients without another explanation for the liver test abnormalities.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below or elsewhere in the prescribing information:
- Pancreatitis [see Warnings and Precautions (5.1)]
- Heart Failure [see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Hepatic Effects [see Warnings and Precautions (5.4)]
- Severe and Disabling Arthralgia [see Warnings and Precautions (5.6)]
- Bullous Pemphigoid [see Warnings and Precautions (5.7)]
8.6 Renal Impairment
A total of 602 adult patients with moderate renal impairment (eGFR ≥30 and <60 mL/min/1.73 m2) and 4 patients with severe renal impairment/end-stage renal disease (eGFR <30 mL/min/1.73 m2 or <15 mL/min/1.73 m2, respectively) at baseline were treated with alogliptin tablets in clinical trials in patients with type 2 diabetes mellitus.
In the EXAMINE trial of high CV risk type 2 diabetes mellitus patients, 694 patients had moderate renal impairment and 78 patients had severe renal impairment or end-stage renal disease at baseline.
The recommended dose is 12.5 mg once daily in patients with moderate renal impairment and 6.25 mg once daily in patients with severe renal impairment, as well as in patients with ESRD requiring dialysis. Alogliptin tablets may be administered without regard to the timing of the dialysis.
12.2 Pharmacodynamics
Dose-Response Relationships
Single-dose administration of alogliptin tablets to healthy subjects resulted in a peak inhibition of DPP-4 within two to three hours after dosing. The peak inhibition of DPP-4 exceeded 93% across doses of 12.5 mg to 800 mg. Inhibition of DPP-4 remained above 80% at 24 hours for doses greater than or equal to 25 mg. Peak and total exposure over 24 hours to active GLP-1 were three- to four-fold greater with alogliptin tablets (at doses of 25 to 200 mg) than placebo. In a 16 week, double-blind, placebo-controlled study, alogliptin tablets 25 mg demonstrated decreases in postprandial glucagon while increasing postprandial active GLP-1 levels compared to placebo over an eight hour period following a standardized meal. It is unclear how these findings relate to changes in overall glycemic control in patients with type 2 diabetes mellitus. In this study, alogliptin tablets 25 mg demonstrated decreases in two hour postprandial glucose compared to placebo (-30 mg/dL versus 17 mg/dL, respectively).
Multiple-dose administration of alogliptin to patients with type 2 diabetes mellitus also resulted in a peak inhibition of DPP-4 within one to two hours and exceeded 93% across all doses (25 mg, 100 mg and 400 mg) after a single dose and after 14 days of once-daily dosing. At these doses of alogliptin tablets, inhibition of DPP-4 remained above 81% at 24 hours after 14 days of dosing.
12.3 Pharmacokinetics
The pharmacokinetics of alogliptin tablets has been studied in healthy subjects and in patients with type 2 diabetes mellitus. The pharmacokinetics of alogliptin tablets were similar in healthy subjects and in patients with type 2 diabetes mellitus.
After multiple-dose administration up to 400 mg for 14 days in patients with type 2 diabetes mellitus, accumulation of alogliptin was minimal with an increase in total [e.g., area under the plasma concentration curve (AUC)] and peak (i.e., Cmax) alogliptin exposures of 34% and 9%, respectively. Total and peak exposure to alogliptin increased proportionally across single doses and multiple doses of alogliptin ranging from 25 mg to 400 mg. The intersubject coefficient of variation for alogliptin AUC was 17%.
2.1 Recommended Dosage
- The recommended dosage of alogliptin tablets is 25 mg taken orally once daily. Do not spilt tablet.
- Alogliptin tablets may be taken with or without food. [see Clinical Pharmacology (12.3)]
- Instruct patients if a dose is missed, not to double their next dose.
5.7 Bullous Pemphigoid
Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving alogliptin tablets. If bullous pemphigoid is suspected, alogliptin tablets should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
No dose adjustments are required in patients with mild to moderate hepatic impairment (Child-Pugh Grade A and B) [see Clinical Pharmacology (12.3)]. Alogliptin tablets have not been studied in patients with severe hepatic impairment (Child-Pugh Grade C). Use caution when administering alogliptin tablets to patients with liver disease [see Warnings and Precautions (5.4)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Alogliptin tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
12.1 Mechanism of Action
Increased concentrations of the incretin hormones such as glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are released into the bloodstream from the small intestine in response to meals. These hormones cause insulin release from the pancreatic beta cells in a glucose-dependent manner but are inactivated by the DPP-4 enzyme within minutes. GLP-1 also lowers glucagon secretion from pancreatic alpha cells, reducing hepatic glucose production. In patients with type 2 diabetes mellitus, concentrations of GLP-1 are reduced but the insulin response to GLP-1 is preserved. Alogliptin is a DPP-4 inhibitor that slows the inactivation of the incretin hormones, thereby increasing their bloodstream concentrations and reducing fasting and postprandial glucose concentrations in a glucose-dependent manner in patients with type 2 diabetes mellitus. Alogliptin selectively binds to and inhibits DPP-4 but not DPP-8 or DPP-9 activity in vitro at concentrations approximating therapeutic exposures.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue alogliptin tablets. (5.1)
- Heart failure: Consider the risks and benefits of alogliptin tablets prior to initiating treatment in patients at risk for heart failure. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of alogliptin tablets. (5.2)
- Hypersensitivity: There have been postmarketing reports of serious hypersensitivity reactions in patients treated with alogliptin tablets such as anaphylaxis, angioedema and severe cutaneous adverse reactions, including Stevens-Johnson syndrome. If hypersensitivity reactions occur, discontinue alogliptin tablets, treat promptly and monitor until signs and symptoms resolve. (5.3)
- Hepatic effects: Postmarketing reports of hepatic failure, sometimes fatal. Causality cannot be excluded. If liver injury is detected, promptly interrupt alogliptin tablets and assess patient for probable cause, then treat cause if possible, to resolution or stabilization. Do not restart alogliptin tablets if liver injury is confirmed and no alternative etiology can be found. (5.4)
- Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating Alogliptin tablets. (5.5)
- Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.6)
- Bullous pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue alogliptin tablets. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The recommended dosage in patients with normal renal function or mild renal impairment is 25 mg orally once daily. (2.1)
- Can be taken with or without food. (2.1)
- Adjust dosage if moderate or severe renal impairment or end-stage renal disease (ESRD). (2.2)
| Degree of Renal Impairment | Creatinine Clearance (mL/min) |
Recommended Dosage |
|---|---|---|
| Moderate | ≥30 to <60 | 12.5 mg once daily |
| Severe/ESRD | <30 | 6.25 mg once daily |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 25 mg tablets are light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side.
- 12.5 mg tablets are yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side.
- 6.25 mg tablets are light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during the postmarketing use of alogliptin tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: acute pancreatitis, diarrhea, constipation, nausea, ileus
Hepatobiliary Disorders: fulminant hepatic failure
Immune System Disorders: hypersensitivity reactions including anaphylaxis
Investigations: hepatic enzyme elevations
Musculoskeletal and Connective Tissue Disorders: severe and disabling arthralgia, rhabdomyolysis
Renal and Urinary Disorders: tubulointerstitial nephritis
Skin and Subcutaneous Tissue Disorders: angioedema, rash, urticaria and severe cutaneous adverse reactions including Stevens-Johnson syndrome, bullous pemphigoid
5.3 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions in patients treated with alogliptin tablets [see Adverse Reactions (6.2)]. These reactions include anaphylaxis, angioedema, and severe cutaneous adverse reactions, including Stevens-Johnson syndrome. If a serious hypersensitivity reaction is suspected, discontinue alogliptin tablets, assess for other potential causes for the event and institute alternative treatment for diabetes mellitus. Use caution in patients with a history of angioedema with another dipeptidyl peptidase-4 (DPP-4) inhibitor because it is unknown whether such patients will be predisposed to angioedema with alogliptin tablets.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 14,778 patients with type 2 diabetes mellitus participated in 14 randomized, double-blind, controlled clinical trials of whom 9,052 subjects were treated with alogliptin tablets, 3,469 subjects were treated with placebo and 2,257 were treated with an active comparator. The racial distribution of patients exposed to trial medication was 71% White, 17% Asian, 6% Black or African American, 2% American Indian or Alaska Native, 0% Native Hawaiian/Other Pacific Islander and 5% Multiracial or other racial groups. The ethnic distribution was 30% Hispanic or Latino and 70% was not Hispanic or Latino. The mean duration of diabetes mellitus was seven years, the mean body mass index (BMI) was 31 kg/m2 (49% of patients had a BMI ≥30 kg/m2), and the mean age was 58 years (26% of patients ≥65 years of age). The mean exposure to alogliptin tablets was 49 weeks with 3,348 subjects treated for more than one year.
In a pooled analysis of these 14 controlled clinical trials, the overall incidence of adverse reactions was 73% in patients treated with alogliptin tablets 25 mg compared to 75% with placebo and 70% with active comparator. Overall discontinuation of therapy due to adverse reactions was 6.8% with alogliptin tablets 25 mg compared to 8.4% with placebo or 6.2% with active comparator.
Adverse reactions reported in ≥4% of adult patients treated with alogliptin tablets 25 mg and more frequently than in patients who received placebo are summarized in Table 1.
| Number of Patients (%) | |||
|---|---|---|---|
| Alogliptin Tablets 25 mg |
Placebo | Active Comparator | |
| N=6447 | N=3469 | N=2257 | |
| Nasopharyngitis | 309 (5) | 152 (4) | 113 (5) |
| Upper Respiratory Tract Infection | 287 (4) | 121 (4) | 113 (5) |
| Headache | 278 (4) | 101 (3) | 121 (5) |
14.4 Cardiovascular Safety Trial
A randomized, double-blind, placebo-controlled cardiovascular outcomes trial (EXAMINE) was conducted to evaluate the cardiovascular risk of alogliptin tablets. The trial compared the risk of major adverse cardiovascular events (MACE) between alogliptin tablets (N=2701) and placebo (N=2679) when added to standard of care therapies for diabetes mellitus and atherosclerotic vascular disease (ASCVD). The trial was event driven and patients were followed until a sufficient number of primary outcome events accrued.
Eligible patients were adults with type 2 diabetes mellitus who had inadequate glycemic control at baseline (e.g., HbA1c >6.5%) and had been hospitalized for an acute coronary syndrome event (e.g., acute myocardial infarction or unstable angina requiring hospitalization) 15 to 90 days prior to randomization. The dose of alogliptin tablets was based on estimated renal function at baseline per dosage and administration recommendations [see Dosage and Administration (2.2)]. The average time between an acute coronary syndrome event and randomization was approximately 48 days.
The mean age of the population was 61 years. Most patients were male (68%), White (73%), and were recruited from outside of the United States (86%). Asian and Black or African American patients contributed 20% and 4% of the total population, respectively. At the time of randomization patients had a diagnosis of type 2 diabetes mellitus for approximately 9 years, 87% had a prior myocardial infarction and 14% were current smokers. Hypertension (83%) and renal impairment (27% with an eGFR ≤60 mL/min/1.73 m2) were prevalent co-morbid conditions. Use of medications to treat diabetes mellitus (e.g., metformin 73%, sulfonylurea 54%, insulin 41%), and ASCVD (e.g., statin 94%, aspirin 93%, renin-angiotensin system blocker 88%, beta-blocker 87%) was similar between patients randomized to alogliptin tablets and placebo at baseline. During the trial, medications to treat diabetes mellitus and ASCVD could be adjusted to ensure care for these conditions adhered to standard of care recommendations set by local practice guidelines.
The primary endpoint in EXAMINE was the time to first occurrence of a MACE defined as the composite of cardiovascular death, nonfatal myocardial infarction (MI), or nonfatal stroke. The trial was designed to exclude a pre-specified risk margin of 1.3 for the hazard ratio of MACE. The median exposure to trial drug was 526 days and 95% of the patients were followed to trial completion or death.
Table 12 shows the trial results for the primary MACE composite endpoint and the contribution of each component to the primary MACE endpoint. The upper bound of the confidence interval was 1.16 and excluded a risk margin larger than 1.3.
| Composite of first event of CV death, nonfatal MI or nonfatal stroke (MACE) | Alogliptin | Placebo | Hazard Ratio | ||
| Number of Patients (%) |
Rate per 100 PY
Patient Years (PY)
|
Number of Patients (%) | Rate per 100 PY | (98% CI) | |
| N=2701 | N=2679 | ||||
| 305 (11.3) | 7.6 | 316 (11.8) | 7.9 | 0.96 (0.80, 1.16) | |
| CV Death | 89 (3.3) | 2.2 | 111 (4.1) | 2.8 | |
| Non-fatal MI | 187 (6.9) | 4.6 | 173 (6.5) | 4.3 | |
| Non-fatal stroke | 29 (1.1) | 0.7 | 32 (1.2) | 0.8 |
The Kaplan-Meier based cumulative event probability is presented in Figure 4 for the time to first occurrence of the primary MACE composite endpoint by treatment arm. The curves for placebo and alogliptin tablets overlap throughout the duration of the trial. The observed incidence of MACE was highest within the first 60 days after randomization in both treatment arms (14.8 MACE per 100 PY), decreased from day 60 to the end of the first year (8.4 per 100 PY) and was lowest after one year of follow-up (5.2 per 100 PY).
| Figure 4. Observed Cumulative Rate of MACE in EXAMINE |
The rate of all cause death was similar between treatment arms with 153 (3.6 per 100 PY) recorded among patients randomized to alogliptin tablets and 173 (4.1 per 100 PY) among patients randomized to placebo. A total of 112 deaths (2.9 per 100 PY) among patients on alogliptin tablets and 130 among patients on placebo (3.5 per 100 PY) were adjudicated as cardiovascular deaths.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.2 Patients With Renal Impairment (2.2 Patients with Renal Impairment)
- Assess renal function prior to initiation of alogliptin tablets and periodically thereafter [see Use in Specific Populations (8.6)].
- No dose adjustment of alogliptin tablets is necessary for patients with mild renal impairment (creatinine clearance [CrCl] ≥60 mL/min).
- The dose of alogliptin tablets is 12.5 mg once daily for patients with moderate renal impairment (CrCl ≥30 to <60 mL/min).
- The dose of alogliptin tablets is 6.25 mg once daily for patients with severe renal impairment (CrCl ≥15 to <30 mL/min) or with end-stage renal disease (ESRD) (CrCl <15 mL/min or requiring hemodialysis). Alogliptin tablets may be administered without regard to the timing of dialysis. Alogliptin tablets have not been studied in patients undergoing peritoneal dialysis [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
5.6 Severe and Disabling Arthralgia
There have been postmarketing reports of severe and disabling arthralgia in patients taking DPP-4 inhibitors. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider DPP-4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Alogliptin tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows:
25 mg tablet: light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side, available in:
| NDC 45802-150-65 | Bottles of 30 tablets |
12.5 mg tablet: yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side, available in:
| NDC 45802-103-65 | Bottles of 30 tablets |
6.25 mg tablet: light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side, available in:
| NDC 45802-087-65 | Bottles of 30 tablets |
7.1 Insulin Secretagogues and Insulin
Insulin and insulin secretagogues are known to cause hypoglycemia. Coadministration of alogliptin tablets with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue and insulin to reduce the risk of hypoglycemia [see Warnings and Precautions (5.5)].
Principal Display Panel 25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label)
NDC 45802-150-65
Rx Only
Alogliptin
Tablets
25 mg
DISPENSE WITH
MEDICATION GUIDE
30 Tablets
Padagis®
Principal Display Panel 12.5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 12.5 mg Tablet Bottle Label)
NDC 45802-103-65
Rx Only
Alogliptin
Tablets
12.5 mg
DISPENSE WITH
MEDICATION GUIDE
30 Tablets
Padagis®
Principal Display Panel 6.25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 6.25 mg Tablet Bottle Label)
NDC 45802-087-65
Rx Only
Alogliptin
Tablets
6.25 mg
DISPENSE WITH
MEDICATION GUIDE
30 Tablets
Padagis®
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Rats were administered oral doses of 75, 400 and 800 mg/kg alogliptin for two years. No drug-related tumors were observed up to 75 mg/kg or approximately 32 times the maximum recommended clinical dose of 25 mg, based on area under the plasma concentration curve (AUC) exposure. At higher doses (approximately 308 times the maximum recommended clinical dose of 25 mg), a combination of thyroid C-cell adenomas and carcinomas increased in male but not female rats. No drug-related tumors were observed in mice after administration of 50, 150 or 300 mg/kg alogliptin for two years, or up to approximately 51 times the maximum recommended clinical dose of 25 mg, based on AUC exposure.
Mutagenesis
Alogliptin was not mutagenic or clastogenic, with and without metabolic activation, in the Ames test with S. typhimurium and E. coli or the cytogenetic assay in mouse lymphoma cells. Alogliptin was negative in the in vivo mouse micronucleus study.
Impairment of Fertility
In a fertility study in rats, alogliptin had no adverse effects on early embryonic development, mating or fertility at doses up to 500 mg/kg, or approximately 172 times the clinical dose based on plasma drug exposure (AUC).
14.2 Patients With Inadequate Glycemic Control On Diet and Exercise (14.2 Patients with Inadequate Glycemic Control on Diet and Exercise)
A total of 1,768 patients with type 2 diabetes mellitus participated in three double-blind trials to evaluate the efficacy and safety of alogliptin tablets in patients with inadequate glycemic control on diet and exercise. All three trials had a four week, single-blind, placebo run-in period followed by a 26 week randomized treatment period. Patients who failed to meet prespecified hyperglycemic goals during the 26 week treatment periods received glycemic rescue therapy.
In a 26 week, double-blind, placebo-controlled trial, a total of 329 patients (mean baseline A1C = 8%) were randomized to receive alogliptin tablets 12.5 mg, alogliptin tablets 25 mg or placebo once daily. Treatment with alogliptin tablets 25 mg resulted in statistically significant improvements from baseline in A1C and fasting plasma glucose (FPG) compared to placebo at Week 26 (Table 3). A total of 8% of patients receiving alogliptin tablets 25 mg and 30% of those receiving placebo required glycemic rescue therapy.
Improvements in A1C were not affected by gender, age or baseline body mass index (BMI).
The mean change in body weight with alogliptin tablets was similar to placebo.
| Alogliptin Tablets 25 mg |
Placebo | |
|---|---|---|
| A1C (%) | N=128 | N=63 |
| Baseline (mean) | 7.9 | 8.0 |
| Change from baseline (adjusted mean Least squares means adjusted for treatment, baseline value, geographic region and duration of diabetes mellitus ) |
-0.6 | 0 |
| Difference from placebo (adjusted mean with 95% confidence interval) | -0.6 p<0.01 compared to placebo (-0.8, -0.3) |
˗ |
| % of patients (n/N) achieving A1C ≤7% | 44% (58/131) | 23% (15/64) |
| FPG (mg/dL) | N=129 | N=64 |
| Baseline (mean) | 172 | 173 |
| Change from baseline (adjusted mean) | -16 | 11 |
| Difference from placebo (adjusted mean with 95% confidence interval) | -28 (-40, -15) | ˗ |
In a 26 week, double-blind, active-controlled trial, a total of 655 patients (mean baseline A1C = 8.8%) were randomized to receive alogliptin tablets 25 mg alone, pioglitazone 30 mg alone, alogliptin tablets 12.5 mg with pioglitazone 30 mg or alogliptin tablets 25 mg with pioglitazone 30 mg once daily. Coadministration of alogliptin tablets 25 mg with pioglitazone 30 mg resulted in statistically significant improvements from baseline in A1C and FPG compared to alogliptin tablets 25 mg alone and to pioglitazone 30 mg alone (Table 4). A total of 3% of patients receiving alogliptin tablets 25 mg coadministered with pioglitazone 30 mg, 11% of those receiving alogliptin tablets 25 mg alone and 6% of those receiving pioglitazone 30 mg alone required glycemic rescue.
Improvements in A1C were not affected by gender, age or baseline BMI.
The mean increase in body weight was similar between pioglitazone alone and alogliptin tablets when coadministered with pioglitazone.
| Alogliptin Tablets 25 mg |
Pioglitazone 30 mg |
Alogliptin Tablets 25 mg + Pioglitazone 30 mg |
|
|---|---|---|---|
| A1C (%) | N=160 | N=153 | N=158 |
| Baseline (mean) | 8.8 | 8.8 | 8.8 |
| Change from baseline (adjusted mean Least squares means adjusted for treatment, geographic region and baseline value ) |
-1.0 | -1.2 | -1.7 |
| Difference from alogliptin tablets 25 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -0.8 p<0.01 compared to alogliptin tablets 25 mg or pioglitazone 30 mg (-1.0, -0.5) |
| Difference from pioglitazone 30 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -0.6 (-0.8, -0.3) |
| % of Patients (n/N) achieving A1C ≤7% | 24% (40/164) | 34% (55/163) | 63% (103/164) |
| FPG (mg/dL) | N=162 | N=157 | N=162 |
| Baseline (mean) | 189 | 189 | 185 |
| Change from baseline (adjusted mean) | -26 | -37 | -50 |
| Difference from alogliptin tablets 25 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -24 (-34, -15) |
| Difference from pioglitazone 30 mg (adjusted mean with 95% confidence interval) | ˗ | ˗ | -13 (-22, -4) |
In a 26 week, double-blind, placebo-controlled trial, a total of 784 patients inadequately controlled on diet and exercise alone (mean baseline A1C = 8.4%) were randomized to one of seven treatment groups: placebo; metformin HCl 500 mg or metformin HCl 1000 mg twice daily; alogliptin tablets 12.5 mg twice daily; alogliptin tablets 25 mg daily; or alogliptin tablets 12.5 mg in combination with metformin HCl 500 mg or metformin HCl 1000 mg twice daily. Both coadministration treatment arms (alogliptin tablets 12.5 mg + metformin HCl 500 mg and alogliptin tablets 12.5 mg + metformin HCl 1000 mg) resulted in statistically significant improvements in A1C and FPG when compared with their respective individual alogliptin and metformin component regimens (Table 5). Coadministration treatment arms demonstrated improvements in two hour postprandial glucose (PPG) compared to alogliptin tablets alone or metformin alone (Table 5). A total of 12.3% of patients receiving alogliptin tablets 12.5 mg + metformin HCl 500 mg, 2.6% of patients receiving alogliptin tablets 12.5 mg + metformin HCl 1000 mg, 17.3% of patients receiving alogliptin tablets 12.5 mg, 22.9% of patients receiving metformin HCl 500 mg, 10.8% of patients receiving metformin HCl 1000 mg and 38.7% of patients receiving placebo required glycemic rescue.
Improvements in A1C were not affected by gender, age, race or baseline BMI. The mean decrease in body weight was similar between metformin alone and alogliptin tablets when coadministered with metformin.
| Placebo | Alogliptin Tablets 12.5 mg Twice Daily |
Metformin HCl 500 mg Twice Daily |
Metformin HCl 1000 mg Twice Daily |
Alogliptin Tablets 12.5 mg + Metformin HCl 500 mg Twice Daily |
Alogliptin Tablets 12.5 mg + Metformin HCl 1000 mg Twice Daily |
|
|---|---|---|---|---|---|---|
|
A1C (%) Intent-to-treat population using last observation on trial prior to discontinuation of double-blind trial medication or sulfonylurea rescue therapy for patients needing rescue
|
N=102 | N=104 | N=103 | N=108 | N=102 | N=111 |
| Baseline (mean) | 8.5 | 8.4 | 8.5 | 8.4 | 8.5 | 8.4 |
| Change from baseline (adjusted mean Least squares means adjusted for treatment, geographic region and baseline value ) |
0.1 | -0.6 | -0.7 | -1.1 | -1.2 | -1.6 |
| Difference from metformin (adjusted mean with 95% confidence interval) | - | - | - | - | -0.6 p<0.05 when compared to metformin and alogliptin tablets alone
(-0.9, -0.3) |
-0.4
(-0.7, -0.2) |
| Difference from Alogliptin Tablets (adjusted mean with 95% confidence interval) | - | - | - | - | -0.7
(-1.0, -0.4) |
-1.0
(-1.3, -0.7) |
| % of Patients (n/N) achieving A1C <7% Compared using logistic regression
|
4% (4/102) |
20% (21/104) |
27% (28/103) |
34% (37/108) |
47%
(48/102) |
59%
(66/111) |
| FPG (mg/dL) | N=105 | N=106 | N=106 | N=110 | N=106 | N=112 |
| Baseline (mean) | 187 | 177 | 180 | 181 | 176 | 185 |
| Change from baseline (adjusted mean) | 12 | -10 | -12 | -32 | -32 | -46 |
| Difference from metformin (adjusted mean with 95% confidence interval) | - | - | - | - | -20
(-33, -8) |
-14
(-26, -2) |
| Difference from Alogliptin Tablets (adjusted mean with 95% confidence interval) | - | - | - | - | -22
(-35, -10) |
-36
(-49, -24) |
|
2-Hour PPG (mg/dL)
Intent-to-treat population using data available at Week 26
|
N=26 | N=34 | N=28 | N=37 | N=31 | N=37 |
| Baseline (mean) | 263 | 272 | 247 | 266 | 261 | 268 |
| Change from baseline (adjusted mean) | -21 | -43 | -49 | -54 | -68 | -86 |
| Difference from metformin (adjusted mean with 95% confidence interval) | - | - | - | - | -19 (-49, 11) |
-32
(-58, -5) |
| Difference from Alogliptin Tablets (adjusted mean with 95% confidence interval) | - | - | - | - | -25 (-53, -3) |
-43
(-70, -16) |
14.1 Overview of Clinical Trials in Adults With Type 2 Diabetes Mellitus (14.1 Overview of Clinical Trials in Adults with Type 2 Diabetes Mellitus)
Alogliptin tablets have been studied as monotherapy and in combination with metformin, a sulfonylurea, a thiazolidinedione (either alone or in combination with metformin or a sulfonylurea) and insulin (either alone or in combination with metformin).
A total of 14,053 patients with type 2 diabetes mellitus were randomized in 11 double-blind, placebo- or active-controlled clinical safety and efficacy trials conducted to evaluate the effects of alogliptin tablets on glycemic control. The racial distribution of patients exposed to trials medication was 70% White, 17% Asian, 6% Black or African American, 2% American Indian or Alaska Native, 0% Native Hawaiian/Other Pacific Islander and 5% Multiracial or other racial groups. The ethnic distribution was 30% Hispanic or Latino. Patients had an overall mean age of 57 years (range 21 to 91 years).
In patients with type 2 diabetes mellitus, treatment with alogliptin tablets produced clinically meaningful and statistically significant improvements in hemoglobin A1c (A1C) compared to placebo. As is typical for trials of agents to treat type 2 diabetes mellitus, the mean reduction in A1C with alogliptin tablets appears to be related to the degree of A1C elevation at baseline.
Alogliptin tablets had similar changes from baseline in serum lipids compared to placebo.
5.5 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues (5.5 Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues)
Insulin and insulin secretagogues, such as sulfonylureas, are known to cause hypoglycemia. Therefore, a lower dosage of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with alogliptin tablets.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:55.426510 · Updated: 2026-03-14T22:25:56.100461