Doxorubicin Hydrochloride Liposome Injection
b24afa8c-636b-4ace-99d5-51f445ad3123
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
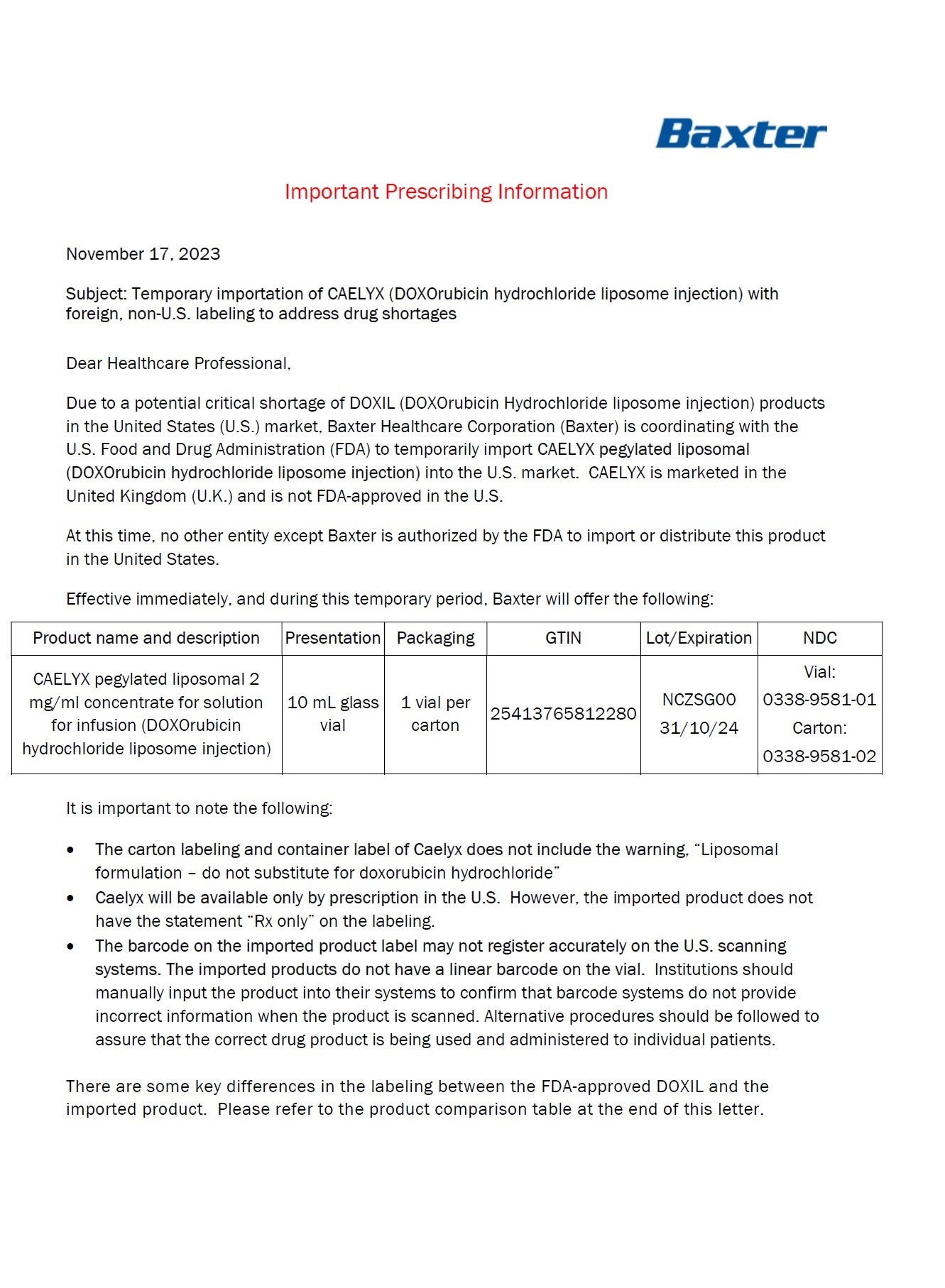
Caelyx ® pegylated liposomal 2 mg/ml sterile concentrate doxorubicin hydrochloride 20 mg/10 mL IV after dilution. Caelyx ® pegylated liposomal 2 mg/ml concentrate for solution for infusion doxorubicin hydrochloride 20 mg/10 mL Intravenous use after dilution . Do not use interchangeably with other formulations of doxorubicin hydrochloride. Cytotoxic 1 vial Baxter Logo One ml of Caelyx pegylated liposomal contains 2 mg doxorubicin hydrochloride. Excipients: α-(2-[1,2-distearoyl- sn- glycero(3) phosphooxy]ethylcarbamoyl) -ϖ-methoxypoly(oxyethylen)-40 sodium salt, fully hydrogenated soy phosphatidylcholine, cholesterol, ammonium sulphate, sucrose, histidine, water for injections, hydrochloric acid and sodium hydroxide. Keep out of the sight and reach of children. Store in a refrigerator. Do not freeze. Read the package leaflet before use. Baxter Healthcare Limited Caxton Way, Thetford, Norfolk, IP24 3SE, United Kingdom PLGB 00116/0255
Medication Information
Description
Caelyx ® pegylated liposomal 2 mg/ml sterile concentrate doxorubicin hydrochloride 20 mg/10 mL IV after dilution. Caelyx ® pegylated liposomal 2 mg/ml concentrate for solution for infusion doxorubicin hydrochloride 20 mg/10 mL Intravenous use after dilution . Do not use interchangeably with other formulations of doxorubicin hydrochloride. Cytotoxic 1 vial Baxter Logo One ml of Caelyx pegylated liposomal contains 2 mg doxorubicin hydrochloride. Excipients: α-(2-[1,2-distearoyl- sn- glycero(3) phosphooxy]ethylcarbamoyl) -ϖ-methoxypoly(oxyethylen)-40 sodium salt, fully hydrogenated soy phosphatidylcholine, cholesterol, ammonium sulphate, sucrose, histidine, water for injections, hydrochloric acid and sodium hydroxide. Keep out of the sight and reach of children. Store in a refrigerator. Do not freeze. Read the package leaflet before use. Baxter Healthcare Limited Caxton Way, Thetford, Norfolk, IP24 3SE, United Kingdom PLGB 00116/0255
Health Care Provider Letter
Package/label Principal Display Panel – 20 Mg/10 Ml
Caelyx® pegylated liposomal
2 mg/ml
sterile concentrate
doxorubicin hydrochloride
20 mg/10 mL
IV after dilution.
Caelyx®
pegylated liposomal
2 mg/ml
concentrate for
solution for infusion
doxorubicin hydrochloride
20 mg/10 mL
Intravenous use after
dilution.
Do not use interchangeably
with other formulations of
doxorubicin hydrochloride.
Cytotoxic
1 vial
Baxter Logo
One ml of Caelyx pegylated
liposomal contains 2 mg
doxorubicin hydrochloride.
Excipients: α-(2-[1,2-distearoyl-sn-glycero(3)
phosphooxy]ethylcarbamoyl)
-ϖ-methoxypoly(oxyethylen)-40
sodium salt, fully hydrogenated
soy phosphatidylcholine,
cholesterol, ammonium
sulphate, sucrose, histidine,
water for injections,
hydrochloric acid and sodium
hydroxide.
Keep out of the sight and
reach of children.
Store in a refrigerator.
Do not freeze.
Read the package leaflet
before use.
Baxter Healthcare Limited
Caxton Way,
Thetford,
Norfolk,
IP24 3SE,
United Kingdom
PLGB 00116/0255
Structured Label Content
Health Care Provider Letter (HEALTH CARE PROVIDER LETTER)
Package/label Principal Display Panel – 20 Mg/10 Ml (PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 20 mg/10 mL)
Caelyx® pegylated liposomal
2 mg/ml
sterile concentrate
doxorubicin hydrochloride
20 mg/10 mL
IV after dilution.
Caelyx®
pegylated liposomal
2 mg/ml
concentrate for
solution for infusion
doxorubicin hydrochloride
20 mg/10 mL
Intravenous use after
dilution.
Do not use interchangeably
with other formulations of
doxorubicin hydrochloride.
Cytotoxic
1 vial
Baxter Logo
One ml of Caelyx pegylated
liposomal contains 2 mg
doxorubicin hydrochloride.
Excipients: α-(2-[1,2-distearoyl-sn-glycero(3)
phosphooxy]ethylcarbamoyl)
-ϖ-methoxypoly(oxyethylen)-40
sodium salt, fully hydrogenated
soy phosphatidylcholine,
cholesterol, ammonium
sulphate, sucrose, histidine,
water for injections,
hydrochloric acid and sodium
hydroxide.
Keep out of the sight and
reach of children.
Store in a refrigerator.
Do not freeze.
Read the package leaflet
before use.
Baxter Healthcare Limited
Caxton Way,
Thetford,
Norfolk,
IP24 3SE,
United Kingdom
PLGB 00116/0255
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:12.849400 · Updated: 2026-03-14T22:39:36.817935