Ocrevus Zunovo
b216af61-99d0-42a3-afc2-b5bf86df1ec0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
OCREVUS ZUNOVO is indicated for the treatment of: Relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults Primary progressive MS, in adults
Dosage and Administration
OCREVUS ZUNOVO should be administered by a healthcare professional ( 2.1 ) For subcutaneous use in the abdomen only ( 2.1 ) OCREVUS ZUNOVO has different dosage and administration instructions than intravenous ocrelizumab ( 2.1 ) Before initiating OCREVUS ZUNOVO, screen for Hepatitis B virus and obtain serum quantitative immunoglobulins, aminotransferases, alkaline phosphatase, and bilirubin ( 2.2 ) Pre-medicate orally with dexamethasone (or an equivalent corticosteroid) and an antihistamine (e.g., desloratadine) at least 30 minutes prior to each injection ( 2.3 ) Administer 23 mL of OCREVUS ZUNOVO (920 mg ocrelizumab and 23,000 units hyaluronidase) subcutaneously in the abdomen over approximately 10 minutes every 6 months ( 2.4 ) Monitor patients closely during all injections, for at least one hour after the initial injection, and for at least 15 minutes after subsequent injections ( 2.4 )
Contraindications
OCREVUS ZUNOVO is contraindicated in patients with: Active HBV infection [see Dosage and Administration (2.2) and Warnings and Precautions (5.2) ] A history of life-threatening administration reaction to ocrelizumab [see Warnings and Precautions (5.1) ] A history of hypersensitivity to ocrelizumab, hyaluronidase, or any component of OCREVUS ZUNOVO [see Warnings and Precautions (5.1) ] .
Warnings and Precautions
Injection Reactions: Management recommendations for injection reactions depend on the type and severity of the reaction. Permanently discontinue OCREVUS ZUNOVO if a life-threatening injection reaction occurs ( 4 , 5.1 ) Infections: Serious, including life-threatening and fatal, infections have occurred in patients treated with ocrelizumab. Delay OCREVUS ZUNOVO administration in patients with an active infection until the infection is resolved. Vaccination with live-attenuated or live vaccines is not recommended during treatment with OCREVUS ZUNOVO and after discontinuation, until B-cell repletion ( 5.2 ) Progressive Multifocal Leukoencephalopathy (PML): Withhold OCREVUS ZUNOVO at the first sign or symptom suggestive of PML ( 5.3 ) Reduction in Immunoglobulins: Monitor the level of immunoglobulins at the beginning of treatment. Monitor during and after discontinuation of treatment with OCREVUS ZUNOVO, until B-cell repletion, and especially when recurrent serious infections are suspected. Consider discontinuing OCREVUS ZUNOVO in patients with serious opportunistic or recurrent serious infections, and if prolonged hypogammaglobulinemia requires treatment with intravenous immunoglobulins ( 2.1 , 5.4 ) Malignancies: An increased risk of malignancy, including breast cancer, may exist with OCREVUS ZUNOVO ( 5.5 ) Immune-Mediated Colitis: Immune-mediated colitis has been reported in the postmarketing setting. Monitor patients for new or persistent diarrhea or other gastrointestinal symptoms, and evaluate promptly if colitis is suspected ( 5.6 ) Liver Injury: Clinically significant liver injury has occurred. Obtain serum aminotransferases, alkaline phosphatase, and bilirubin levels before initiating OCREVUS ZUNOVO, and during treatment as clinically indicated. Discontinue OCREVUS ZUNOVO in patients with evidence of liver injury in the absence of an alternative etiology ( 5.7 )
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Injection Reactions [see Warnings and Precautions (5.1) ] Infections [see Warnings and Precautions (5.2) ] Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3) ] Reduction in Immunoglobulins [see Warnings and Precautions (5.4) ] Malignancies [see Warnings and Precautions (5.5) ] Immune-Mediated Colitis [see Warnings and Precautions (5.6) ] Liver Injury [see Warnings and Precautions (5.7) ]
How Supplied
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied as a carton containing one 920 mg and 23,000 units/23 mL (40 mg and 1,000 units/mL) single-dose vial (NDC 50242-554-01).
Storage and Handling
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied as a carton containing one 920 mg and 23,000 units/23 mL (40 mg and 1,000 units/mL) single-dose vial (NDC 50242-554-01).
Description
Dosage and Administration ( 2.2 ) 8/2025 Warnings and Precautions ( 5.7 ) 8/2025
Medication Information
Warnings and Precautions
Injection Reactions: Management recommendations for injection reactions depend on the type and severity of the reaction. Permanently discontinue OCREVUS ZUNOVO if a life-threatening injection reaction occurs ( 4 , 5.1 ) Infections: Serious, including life-threatening and fatal, infections have occurred in patients treated with ocrelizumab. Delay OCREVUS ZUNOVO administration in patients with an active infection until the infection is resolved. Vaccination with live-attenuated or live vaccines is not recommended during treatment with OCREVUS ZUNOVO and after discontinuation, until B-cell repletion ( 5.2 ) Progressive Multifocal Leukoencephalopathy (PML): Withhold OCREVUS ZUNOVO at the first sign or symptom suggestive of PML ( 5.3 ) Reduction in Immunoglobulins: Monitor the level of immunoglobulins at the beginning of treatment. Monitor during and after discontinuation of treatment with OCREVUS ZUNOVO, until B-cell repletion, and especially when recurrent serious infections are suspected. Consider discontinuing OCREVUS ZUNOVO in patients with serious opportunistic or recurrent serious infections, and if prolonged hypogammaglobulinemia requires treatment with intravenous immunoglobulins ( 2.1 , 5.4 ) Malignancies: An increased risk of malignancy, including breast cancer, may exist with OCREVUS ZUNOVO ( 5.5 ) Immune-Mediated Colitis: Immune-mediated colitis has been reported in the postmarketing setting. Monitor patients for new or persistent diarrhea or other gastrointestinal symptoms, and evaluate promptly if colitis is suspected ( 5.6 ) Liver Injury: Clinically significant liver injury has occurred. Obtain serum aminotransferases, alkaline phosphatase, and bilirubin levels before initiating OCREVUS ZUNOVO, and during treatment as clinically indicated. Discontinue OCREVUS ZUNOVO in patients with evidence of liver injury in the absence of an alternative etiology ( 5.7 )
Indications and Usage
OCREVUS ZUNOVO is indicated for the treatment of: Relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults Primary progressive MS, in adults
Dosage and Administration
OCREVUS ZUNOVO should be administered by a healthcare professional ( 2.1 ) For subcutaneous use in the abdomen only ( 2.1 ) OCREVUS ZUNOVO has different dosage and administration instructions than intravenous ocrelizumab ( 2.1 ) Before initiating OCREVUS ZUNOVO, screen for Hepatitis B virus and obtain serum quantitative immunoglobulins, aminotransferases, alkaline phosphatase, and bilirubin ( 2.2 ) Pre-medicate orally with dexamethasone (or an equivalent corticosteroid) and an antihistamine (e.g., desloratadine) at least 30 minutes prior to each injection ( 2.3 ) Administer 23 mL of OCREVUS ZUNOVO (920 mg ocrelizumab and 23,000 units hyaluronidase) subcutaneously in the abdomen over approximately 10 minutes every 6 months ( 2.4 ) Monitor patients closely during all injections, for at least one hour after the initial injection, and for at least 15 minutes after subsequent injections ( 2.4 )
Contraindications
OCREVUS ZUNOVO is contraindicated in patients with: Active HBV infection [see Dosage and Administration (2.2) and Warnings and Precautions (5.2) ] A history of life-threatening administration reaction to ocrelizumab [see Warnings and Precautions (5.1) ] A history of hypersensitivity to ocrelizumab, hyaluronidase, or any component of OCREVUS ZUNOVO [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Injection Reactions [see Warnings and Precautions (5.1) ] Infections [see Warnings and Precautions (5.2) ] Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3) ] Reduction in Immunoglobulins [see Warnings and Precautions (5.4) ] Malignancies [see Warnings and Precautions (5.5) ] Immune-Mediated Colitis [see Warnings and Precautions (5.6) ] Liver Injury [see Warnings and Precautions (5.7) ]
Storage and Handling
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied as a carton containing one 920 mg and 23,000 units/23 mL (40 mg and 1,000 units/mL) single-dose vial (NDC 50242-554-01).
How Supplied
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied as a carton containing one 920 mg and 23,000 units/23 mL (40 mg and 1,000 units/mL) single-dose vial (NDC 50242-554-01).
Description
Dosage and Administration ( 2.2 ) 8/2025 Warnings and Precautions ( 5.7 ) 8/2025
Section 42229-5
Hepatitis B Virus Screening
Prior to initiating ocrelizumab treatment, perform Hepatitis B virus (HBV) screening. OCREVUS ZUNOVO is contraindicated in patients with active HBV confirmed by positive results for HBsAg and anti-HBV tests. For patients who are negative for surface antigen [HBsAg] and positive for HB core antibody [HBcAb+] or are carriers of HBV [HBsAg+], consult liver disease experts before starting and during treatment [see Warnings and Precautions (5.2)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Issued: 8/2025 | |||||||
|
MEDICATION GUIDE
OCREVUS ZUNOVO® (OH-creh-vus zoo-NOH-voh) (ocrelizumab and hyaluronidase-ocsq) injection, for subcutaneous use |
||||||||
|
What is the most important information I should know about OCREVUS ZUNOVO?
OCREVUS ZUNOVO can cause serious side effects, including:
|
||||||||
|
|
|
|
|||||
| These injection reactions can happen during or up to 24 hours after your injection. It is important that you call your healthcare provider right away if you get any of the signs or symptoms listed above after each injection. | ||||||||
|
||||||||
|
||||||||
|
|
|
||||||
| Signs of a more serious herpes infection include: | ||||||||
|
|
|
||||||
| Signs of infection can happen during treatment or after you have received your last dose of OCREVUS ZUNOVO. Tell your healthcare provider right away if you have an infection. Your healthcare provider should delay your treatment with OCREVUS ZUNOVO until your infection is gone. | ||||||||
|
||||||||
|
||||||||
|
||||||||
|
|
|||||||
|
||||||||
| See "What are the possible side effects of OCREVUS ZUNOVO?" for more information about side effects. | ||||||||
|
What is OCREVUS ZUNOVO?
OCREVUS ZUNOVO is a prescription medicine used to treat:
|
||||||||
|
Who should not receive OCREVUS ZUNOVO?
Do not receive OCREVUS ZUNOVO if you:
|
||||||||
Before receiving OCREVUS ZUNOVO, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||||
How will I receive OCREVUS ZUNOVO?
|
||||||||
|
What are the possible side effects of OCREVUS ZUNOVO? OCREVUS ZUNOVO may cause serious side effects, including:
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||||
|
General information about the safe and effective use of OCREVUS ZUNOVO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about OCREVUS ZUNOVO that is written for health professionals. |
||||||||
|
What are the ingredients in OCREVUS ZUNOVO?
Active ingredient: ocrelizumab and hyaluronidase-ocsq. Inactive ingredients: glacial acetic acid, methionine, polysorbate 20, sodium acetate, trehalose, and water for injection. Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 U.S. License No. 1048 For more information, go to www.OCREVUS.com or call 1-844-627-3887. |
Section 44425-7
Store OCREVUS ZUNOVO vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze or shake.
If necessary, OCREVUS ZUNOVO can be removed and placed back into the refrigerator. Unopened OCREVUS ZUNOVO vials may be stored in the original carton for a cumulative time of up to 12 hours without refrigeration at a temperature up to 25°C (77°F).
11 Description
Ocrelizumab is a recombinant humanized monoclonal antibody directed against CD20-expressing B-cells. Ocrelizumab is a glycosylated immunoglobulin G1 (IgG1) with a molecular mass of approximately 145 kDa.
Hyaluronidase (human recombinant) is an endoglycosidase used to increase the dispersion and absorption of co-administered drugs when administered subcutaneously. It is a glycosylated single-chain protein produced by mammalian (Chinese Hamster Ovary) cells containing a DNA plasmid encoding for a soluble fragment of human hyaluronidase (PH20). Hyaluronidase (human recombinant) has a molecular weight of approximately 61 kDa.
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied in single-dose vials. Each 23 mL of solution contains 920 mg ocrelizumab, 23,000 units of hyaluronidase (human recombinant), glacial acetic acid (5.5 mg), methionine (34.3 mg), polysorbate 20 (13.8 mg), sodium acetate (30.2 mg), trehalose (1889.5 mg), and water for injection at pH 5.3.
5.2 Infections
Serious, including life-threatening or fatal, bacterial, viral, parasitic, and fungal infections have been reported in patients receiving ocrelizumab. An increased risk of infections (including serious and fatal bacterial, fungal, and new or reactivated viral infections) has been observed in patients during and following completion of treatment with anti-CD20 B-cell depleting therapies.
A higher proportion of intravenous ocrelizumab-treated patients experienced infections compared to patients taking REBIF or placebo. In RMS trials, 58% of intravenous ocrelizumab-treated patients experienced one or more infections compared to 52% of REBIF-treated patients. In the PPMS trial, 70% of intravenous ocrelizumab-treated patients experienced one or more infections compared to 68% of patients on placebo.
Intravenous ocrelizumab was not associated with an increased risk of serious infections in MS patients in controlled trials.
Ocrelizumab increases the risk for upper respiratory tract infections, lower respiratory tract infections, skin infections, and herpes-related infections [see Adverse Reactions (6.1)].
Delay OCREVUS ZUNOVO administration in patients with an active infection until the infection is resolved.
5.5 Malignancies
An increased risk of malignancy with OCREVUS ZUNOVO may exist. In controlled trials, malignancies, including breast cancer, occurred more frequently in patients treated with intravenous ocrelizumab. Breast cancer occurred in 6 of 781 females treated with intravenous ocrelizumab and none of 668 females treated with REBIF or placebo. Patients should follow standard breast cancer screening guidelines.
5.7 Liver Injury
Clinically significant liver injury, without findings of viral hepatitis, has been reported in the postmarketing setting in patients treated with anti-CD20 B-cell depleting therapies approved for the treatment of MS, including ocrelizumab. Signs of liver injury, including markedly elevated serum hepatic enzymes with elevated total bilirubin, have occurred from weeks to months after administration.
Patients treated with OCREVUS ZUNOVO found to have an alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 3× the upper limit of normal (ULN) with serum total bilirubin greater than 2× ULN are potentially at risk for severe drug-induced liver injury.
Obtain liver function tests prior to initiating treatment with OCREVUS ZUNOVO [see Dosage and Administration (2.2)], and monitor for signs and symptoms of any hepatic injury during treatment. Measure serum aminotransferases, alkaline phosphatase, and bilirubin levels promptly in patients who report symptoms that may indicate liver injury, including new or worsening fatigue, anorexia, nausea, vomiting, right upper abdominal discomfort, dark urine, or jaundice. If liver injury is present and an alternative etiology is not identified, discontinue OCREVUS ZUNOVO.
7.2 Vaccinations
A Phase 3b randomized, open-label study examined the concomitant use of intravenous ocrelizumab and several non-live vaccines in adults 18-55 years of age with relapsing forms of MS (68 subjects undergoing treatment with intravenous ocrelizumab at the time of vaccination and 34 subjects not undergoing treatment with intravenous ocrelizumab at the time of vaccination). Concomitant exposure to intravenous ocrelizumab attenuated antibody responses to tetanus toxoid-containing vaccine, pneumococcal polysaccharide, pneumococcal conjugate vaccines, and seasonal inactivated influenza vaccines. The impact of the observed attenuation on vaccine effectiveness in this patient population is unknown. The safety and effectiveness of live or live-attenuated vaccines administered concomitantly with ocrelizumab have not been assessed [see Warnings and Precautions (5.2)].
8.4 Pediatric Use
Safety and effectiveness of OCREVUS ZUNOVO in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of OCREVUS ZUNOVO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
12.6 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. Immunogenicity data are highly dependent on the sensitivity and specificity of the test methods used. Additionally, the observed incidence of a positive result in a test method may be influenced by several factors, including sample handling, timing of sample collection, drug interference, concomitant medication, and the underlying disease. Therefore, comparison of the incidence of antibodies to OCREVUS ZUNOVO with the incidence of antibodies to other products may be misleading.
In Study 4, no patients tested positive for ocrelizumab anti-drug antibodies (ADAs) or anti-rHuPH20 (hyaluronidase [human recombinant]) antibodies.
14 Clinical Studies
Studies 1-3 (described below), which established the effectiveness of ocrelizumab for the treatment of RMS and PPMS in adults, were conducted with intravenously-administered ocrelizumab. Study 4 demonstrated comparable exposure of OCREVUS ZUNOVO relative to the ocrelizumab intravenous formulation, which established the efficacy of OCREVUS ZUNOVO [see Clinical Pharmacology (12.3)].
4 Contraindications
OCREVUS ZUNOVO is contraindicated in patients with:
- Active HBV infection [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)]
- A history of life-threatening administration reaction to ocrelizumab [see Warnings and Precautions (5.1)]
- A history of hypersensitivity to ocrelizumab, hyaluronidase, or any component of OCREVUS ZUNOVO [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Injection Reactions [see Warnings and Precautions (5.1)]
- Infections [see Warnings and Precautions (5.2)]
- Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3)]
- Reduction in Immunoglobulins [see Warnings and Precautions (5.4)]
- Malignancies [see Warnings and Precautions (5.5)]
- Immune-Mediated Colitis [see Warnings and Precautions (5.6)]
- Liver Injury [see Warnings and Precautions (5.7)]
12.2 Pharmacodynamics
For B-cell counts, assays for CD19+ B-cells are used because the presence of ocrelizumab interferes with the CD20 assay. In clinical studies with OCREVUS ZUNOVO and intravenous ocrelizumab, CD19+ B-cell counts in blood were reduced by 14 days after administration. In clinical studies with intravenous ocrelizumab, B-cell counts rose to above the lower limit of normal (LLN) or above baseline counts between infusions of ocrelizumab at least one time in 0.3% to 4.1% of patients. In a clinical study of 51 patients treated with intravenous ocrelizumab, the median time for B-cell counts to return to either baseline or LLN was 72 weeks (range 27-175 weeks) after the last ocrelizumab infusion. Within 2.5 years after the last infusion, B-cell counts rose to either baseline or LLN in 90% of patients.
12.3 Pharmacokinetics
After subcutaneous administration of 920 mg ocrelizumab, the estimated mean exposure (AUC over the 24-week dosing interval) was 3730 µg/mL∙day. The mean Cmax was 132 µg/mL and tmax was reached after approximately 4 days (range 2 – 13 days). The estimated absolute bioavailability following subcutaneous administration was 81%.
In Study 4, the differences in pharmacokinetic exposures following the administration of OCREVUS ZUNOVO subcutaneously at 920 mg/23,000 units and ocrelizumab intravenously at 600 mg in MS patients were not clinically significant.
Pharmacokinetics (PK) of ocrelizumab in MS clinical studies fit a two-compartment model with time-dependent clearance.
2.4 Recommended Dosage
The recommended dosage of OCREVUS ZUNOVO is 920 mg/23,000 units (920 mg ocrelizumab and 23,000 units of hyaluronidase) administered as a single 23 mL subcutaneous injection in the abdomen over approximately 10 minutes every 6 months.
Monitor the patient closely during injections, with access to appropriate medical support to manage severe injection reactions. For the initial dose, monitor the patient for at least one hour post-injection. For subsequent doses, monitor the patient for at least 15 minutes post-injection [see Warnings and Precautions (5.1)].
1 Indications and Usage
OCREVUS ZUNOVO is indicated for the treatment of:
- Relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults
- Primary progressive MS, in adults
5.1 Injection Reactions
OCREVUS ZUNOVO can cause injection reactions, which can be local or systemic. Common symptoms of local injection reactions reported by patients treated with OCREVUS ZUNOVO in multiple sclerosis (MS) clinical trials included erythema, pain, swelling, and pruritus. Common symptoms of systemic injection reactions reported by patients included headache and nausea. In an open-label, active-controlled trial, injection reactions were more frequently reported with the first injection; 49% of patients experienced an injection reaction with the first injection [see Adverse Reactions (6.1)].
In MS clinical trials where ocrelizumab was administered intravenously, the incidence of infusion reactions in patients [who received methylprednisolone (or an equivalent steroid) and possibly other premedication to reduce the risk of infusion reactions prior to infusion] was 34% to 40%, with the highest incidence with the first infusion. There were no fatal infusion reactions, but 0.3% of intravenous ocrelizumab-treated MS patients experienced infusion reactions that were serious, some requiring hospitalization. Symptoms of infusion reactions can include pruritus, rash, urticaria, erythema, bronchospasm, throat irritation, oropharyngeal pain, dyspnea, pharyngeal or laryngeal edema, flushing, hypotension, pyrexia, fatigue, headache, dizziness, nausea, tachycardia, and anaphylaxis.
Monitor patients during and after injections [see Dosage and Administration (2.4)]. Inform patients that injection reactions can occur during or within 24 hours of the injection.
12.1 Mechanism of Action
The precise mechanism by which ocrelizumab exerts its therapeutic effects in multiple sclerosis is unknown, but is presumed to involve binding to CD20, a cell surface antigen present on pre-B and mature B lymphocytes. Following cell surface binding to B lymphocytes, ocrelizumab results in antibody-dependent cellular cytolysis and complement-mediated lysis.
Hyaluronan is a polysaccharide found in the extracellular matrix of the subcutaneous tissue. It is depolymerized by the naturally occurring enzyme hyaluronidase. Unlike the stable structural components of the interstitial matrix, hyaluronan has a half-life of approximately 0.5 days. Hyaluronidase increases permeability of the subcutaneous tissue by depolymerizing hyaluronan. In the doses administered, hyaluronidase in OCREVUS ZUNOVO acts transiently and locally. The effects of hyaluronidase are reversible and permeability of the subcutaneous tissue is restored within 24 to 48 hours.
5 Warnings and Precautions
- Injection Reactions: Management recommendations for injection reactions depend on the type and severity of the reaction. Permanently discontinue OCREVUS ZUNOVO if a life-threatening injection reaction occurs (4, 5.1)
- Infections: Serious, including life-threatening and fatal, infections have occurred in patients treated with ocrelizumab. Delay OCREVUS ZUNOVO administration in patients with an active infection until the infection is resolved. Vaccination with live-attenuated or live vaccines is not recommended during treatment with OCREVUS ZUNOVO and after discontinuation, until B-cell repletion (5.2)
- Progressive Multifocal Leukoencephalopathy (PML): Withhold OCREVUS ZUNOVO at the first sign or symptom suggestive of PML (5.3)
- Reduction in Immunoglobulins: Monitor the level of immunoglobulins at the beginning of treatment. Monitor during and after discontinuation of treatment with OCREVUS ZUNOVO, until B-cell repletion, and especially when recurrent serious infections are suspected. Consider discontinuing OCREVUS ZUNOVO in patients with serious opportunistic or recurrent serious infections, and if prolonged hypogammaglobulinemia requires treatment with intravenous immunoglobulins (2.1, 5.4)
- Malignancies: An increased risk of malignancy, including breast cancer, may exist with OCREVUS ZUNOVO (5.5)
- Immune-Mediated Colitis: Immune-mediated colitis has been reported in the postmarketing setting. Monitor patients for new or persistent diarrhea or other gastrointestinal symptoms, and evaluate promptly if colitis is suspected (5.6)
- Liver Injury: Clinically significant liver injury has occurred. Obtain serum aminotransferases, alkaline phosphatase, and bilirubin levels before initiating OCREVUS ZUNOVO, and during treatment as clinically indicated. Discontinue OCREVUS ZUNOVO in patients with evidence of liver injury in the absence of an alternative etiology (5.7)
2 Dosage and Administration
- OCREVUS ZUNOVO should be administered by a healthcare professional (2.1)
- For subcutaneous use in the abdomen only (2.1)
- OCREVUS ZUNOVO has different dosage and administration instructions than intravenous ocrelizumab (2.1)
- Before initiating OCREVUS ZUNOVO, screen for Hepatitis B virus and obtain serum quantitative immunoglobulins, aminotransferases, alkaline phosphatase, and bilirubin (2.2)
- Pre-medicate orally with dexamethasone (or an equivalent corticosteroid) and an antihistamine (e.g., desloratadine) at least 30 minutes prior to each injection (2.3)
- Administer 23 mL of OCREVUS ZUNOVO (920 mg ocrelizumab and 23,000 units hyaluronidase) subcutaneously in the abdomen over approximately 10 minutes every 6 months (2.4)
- Monitor patients closely during all injections, for at least one hour after the initial injection, and for at least 15 minutes after subsequent injections (2.4)
2.5 Delayed Or Missed Doses
If a planned injection of OCREVUS ZUNOVO is missed, administer OCREVUS ZUNOVO as soon as possible; do not wait until the next scheduled dose. Reset the dose schedule to administer the next sequential dose 6 months after the missed dose is administered. Doses of OCREVUS ZUNOVO must be separated by at least 5 months [see Dosage and Administration (2.4)].
5.6 Immune Mediated Colitis
Immune-mediated colitis, which can present as a severe and acute-onset form of colitis, has been reported in patients receiving ocrelizumab in the postmarketing setting. Some cases of colitis were serious, requiring hospitalization, with a few patients requiring surgical intervention. Systemic corticosteroids were required in many of these patients. The time from treatment initiation to onset of symptoms in these cases ranged from a few weeks to years. Monitor patients for immune-mediated colitis during OCREVUS ZUNOVO treatment, and evaluate promptly if signs and symptoms that may indicate immune-mediated colitis, such as new or persistent diarrhea or other gastrointestinal signs and symptoms, occur.
3 Dosage Forms and Strengths
- Injection: 920 mg ocrelizumab and 23,000 units hyaluronidase per 23 mL (40 mg and 1,000 units per mL) solution in a single-dose vial (3)
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ocrelizumab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Immune-mediated colitis [see Warnings and Precautions (5.6)]
Hepatobiliary Disorders: Liver injury [see Warnings and Precautions (5.7)]
Infections and Infestations: Serious herpes infections [see Warnings and Precautions (5.2)], progressive multifocal leukoencephalopathy [see Warnings and Precautions (5.3)], and babesiosis
Skin: Pyoderma gangrenosum
8 Use in Specific Populations
- Pregnancy: Based on animal data, may cause fetal harm (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of ocrelizumab has been evaluated in active-controlled clinical trials of ocrelizumab administered intravenously in patients with relapsing forms of MS (RMS) (Study 1 and Study 2) [see Clinical Studies (14.1)] and primary progressive MS (PPMS) (Study 3) [see Clinical Studies (14.2)], and in an open-label, active-controlled trial of OCREVUS ZUNOVO administered subcutaneously in patients with RMS and PPMS (Study 4) [see Clinical Studies (14.3)].
5.4 Reduction in Immunoglobulins
As expected with any B-cell depleting therapy, decreased immunoglobulin levels are observed with ocrelizumab treatment. The pooled data of intravenous ocrelizumab clinical studies (RMS and PPMS) and their open-label extensions (up to approximately 7 years of exposure) have shown an association between decreased levels of immunoglobulin G (IgG<LLN) and increased rates of serious infections. Monitor the levels of quantitative serum immunoglobulins during OCREVUS ZUNOVO treatment and after discontinuation of treatment, until B-cell repletion, and especially in the setting of recurrent serious infections. Consider discontinuing OCREVUS ZUNOVO therapy in patients with serious opportunistic or recurrent serious infections, and if prolonged hypogammaglobulinemia requires treatment with intravenous immunoglobulins [see Adverse Reactions (6.1)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.6 Preparation and Administration
To prevent medication errors, check the vial labels to ensure that the drug being prepared and administered is OCREVUS ZUNOVO and not intravenous ocrelizumab.
Visually inspect the vial for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use the vial if particulates or discoloration are present. Do not shake. Discard any unused portion remaining in the vial.
OCREVUS ZUNOVO is compatible with polypropylene (PP), polycarbonate (PC), polyethylene (PE), stainless steel (SS), polyvinylchloride (PVC), and polyurethane (PUR).
16 How Supplied/storage and Handling
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied as a carton containing one 920 mg and 23,000 units/23 mL (40 mg and 1,000 units/mL) single-dose vial (NDC 50242-554-01).
2.1 Important Administration Information
OCREVUS ZUNOVO is for subcutaneous use in the abdomen only.
OCREVUS ZUNOVO has different dosage and administration instructions than intravenous ocrelizumab.
OCREVUS ZUNOVO should be administered via subcutaneous injection by a healthcare professional.
Principal Display Panel 23 Ml Vial Carton
NDC 50242-554-01
Ocrevus Zunovo®
(ocrelizumab and
hyaluronidase-ocsq)
Injection
920 mg and
23,000 units/23 mL
(40 mg and 1,000 units/mL)
For Subcutaneous Use Only.
For subcutaneous injection in the
abdomen over approximately 10 minutes.
Single-Dose Vial.
Discard Unused Portion.
Attention Pharmacist: Dispense the
accompanying Medication Guide to
each patient.
1 vial
Rx only
Genentech
11038285
5.3 Progressive Multifocal Leukoencephalopathy
Cases of progressive multifocal leukoencephalopathy (PML) have been reported in patients with MS treated with ocrelizumab in the postmarketing setting. PML is an opportunistic viral infection of the brain caused by the JC virus (JCV) that typically only occurs in patients who are immunocompromised, and that usually leads to death or severe disability. PML has occurred in ocrelizumab-treated patients who had not been treated previously with natalizumab (which has a known association with PML), were not taking any immunosuppressive or immunomodulatory medications associated with the risk of PML prior to or concomitantly with ocrelizumab, and did not have any known ongoing systemic medical conditions resulting in compromised immune system function.
JCV infection resulting in PML has also been observed in patients treated with other anti-CD20 antibodies and other MS therapies.
At the first sign or symptom suggestive of PML, withhold OCREVUS ZUNOVO and perform an appropriate diagnostic evaluation. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes.
MRI findings may be apparent before clinical signs or symptoms. Cases of PML, diagnosed based on MRI findings and the detection of JCV DNA in the cerebrospinal fluid in the absence of clinical signs or symptoms specific to PML, have been reported in patients treated with other MS medications associated with PML. Many of these patients subsequently became symptomatic with PML. Therefore, monitoring with MRI for signs that may be consistent with PML may be useful, and any suspicious findings should lead to further investigation to allow for an early diagnosis of PML, if present. Following discontinuation of another MS medication associated with PML, lower PML-related mortality and morbidity have been reported in patients who were initially asymptomatic at diagnosis compared to patients who had characteristic clinical signs and symptoms at diagnosis.
It is not known whether these differences are due to early detection and discontinuation of MS treatment or due to differences in disease in these patients.
If PML is confirmed, treatment with OCREVUS ZUNOVO should be discontinued.
14.3 Ocrevus Zunovo in Patients With Rms Or Ppms
Study 4 was a multicenter, randomized, open-label, parallel arm trial conducted to evaluate the comparative bioavailability, pharmacokinetics, pharmacodynamics, safety, and immunogenicity of OCREVUS ZUNOVO compared with intravenous ocrelizumab in patients with either RMS or PPMS (NCT05232825).
Study 4 enrolled 236 patients (213 with RMS, 23 with PPMS), 18-65 years of age with an EDSS between 0 to 6.5 at screening. The demographics were similar and baseline characteristics were balanced across the two treatment groups. The mean age was 40 years in both groups. In the OCREVUS ZUNOVO group, 35% of patients were male and the mean/median duration since MS diagnosis was 5.7/3.1 years, compared to 41% male and 4.8/2.4 years in the ocrelizumab IV group.
7.1 Immunosuppressive Or Immune Modulating Therapies
The concomitant use of OCREVUS ZUNOVO and other immune-modulating or immunosuppressive therapies, including immunosuppressant doses of corticosteroids, is expected to increase the risk of immunosuppression. Consider the risk of additive immune system effects when coadministering immunosuppressive therapies with OCREVUS ZUNOVO. When switching from drugs with prolonged immune effects, such as daclizumab, fingolimod, natalizumab, teriflunomide, or mitoxantrone, consider the duration and mode of action of these drugs because of additive immunosuppressive effects when initiating OCREVUS ZUNOVO [see Warnings and Precautions (5.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
OCREVUS ZUNOVO contains ocrelizumab and hyaluronidase.
No carcinogenicity studies have been performed to assess the carcinogenic potential of ocrelizumab.
No studies have been performed to assess the mutagenic potential of ocrelizumab. As an antibody, ocrelizumab is not expected to interact directly with DNA.
No effects on reproductive organs were observed in male monkeys administered ocrelizumab by intravenous injection (three loading doses of 15 or 75 mg/kg, followed by weekly doses of 20 or 100 mg/kg) for 8 weeks. There were also no effects on estrus cycle in female monkeys administered ocrelizumab over three menstrual cycles using the same dosing regimen. The doses tested in monkey are 2 and 10 times the recommended human dose of 600 mg intravenous ocrelizumab, on a mg/kg basis.
Hyaluronidases are found in most tissues of the body. Long-term animal studies have not been performed to assess the carcinogenic or mutagenic potential of hyaluronidase. In addition, when subcutaneous hyaluronidase (recombinant human) was administered to cynomolgus monkeys for 39 weeks at dose levels up to 220,000 U/kg, which is > 570 times higher than the human dose, no evidence of toxicity to the male or female reproductive system was found through periodic monitoring of in-life parameters, e.g., semen analyses, hormone levels, menstrual cycles, and also from gross pathology, histopathology and organ weight data.
14.1 Intravenous Ocrelizumab in Patients With Relapsing Forms of Multiple Sclerosis
The efficacy of intravenous ocrelizumab was demonstrated in two randomized, double-blind, double-dummy, active comparator-controlled clinical trials of identical design, in patients with relapsing forms of multiple sclerosis (RMS) treated for 96 weeks (Study 1; NCT01247324 and Study 2; NCT01412333). The dose of intravenous ocrelizumab was 600 mg every 24 weeks (initial treatment was given as two 300 mg IV infusions administered 2 weeks apart, and subsequent doses were administered as a single 600 mg IV infusion) and placebo subcutaneous injections were given 3 times per week. The dose of REBIF, the active comparator, was 44 mcg given as subcutaneous injections 3 times per week and placebo IV infusions were given every 24 weeks. Both studies included patients who had experienced at least one relapse within the prior year, or two relapses within the prior two years, and had an Expanded Disability Status Scale (EDSS) score from 0 to 5.5. Patients with primary progressive forms of multiple sclerosis (MS) were excluded. Neurological evaluations were performed every 12 weeks and at the time of a suspected relapse. Brain MRIs were performed at baseline and at Weeks 24, 48, and 96.
The primary outcome of both Study 1 and Study 2 was the annualized relapse rate (ARR). Additional outcome measures included the proportion of patients with confirmed disability progression, the mean number of MRI T1 gadolinium (Gd)-enhancing lesions at Weeks 24, 48, and 96, and new or enlarging MRI T2 hyperintense lesions. Progression of disability was defined as an increase of 1 point or more from the baseline EDSS score attributable to MS when the baseline EDSS score was 5.5 or less, or 0.5 points or more when the baseline EDSS score was above 5.5. Disability progression was considered confirmed when the increase in the EDSS was confirmed at a regularly scheduled visit 12 weeks after the initial documentation of neurological worsening. The primary population for analysis of confirmed disability progression was the pooled population from Studies 1 and 2.
In Study 1, 410 patients were randomized to intravenous ocrelizumab and 411 to REBIF; 11% of intravenous ocrelizumab-treated patients and 17% of REBIF-treated patients did not complete the 96-week double-blind treatment period. The baseline demographic and disease characteristics were balanced between the two treatment groups. At baseline, the mean age of patients was 37 years; 66% were female. The mean time from MS diagnosis to randomization was 3.8 years, the mean number of relapses in the previous year was 1.3, and the mean EDSS score was 2.8; 74% of patients had not been treated with a non-steroid therapy for MS in the 2 years prior to the study. At baseline, 40% of patients had one or more T1 Gd-enhancing lesions (mean 1.8).
In Study 2, 417 patients were randomized to intravenous ocrelizumab and 418 to REBIF; 14% of intravenous ocrelizumab-treated patients and 23% of REBIF-treated patients did not complete the 96-week double-blind treatment period. The baseline demographic and disease characteristics were balanced between the two treatment groups. At baseline, the mean age of patients was 37 years; 66% were female. The mean time from MS diagnosis to randomization was 4.1 years, the mean number of relapses in the previous year was 1.3, and the mean EDSS score was 2.8; 74% of patients had not been treated with a non-steroid therapy for MS in the 2 years prior to the study. At baseline, 40% of intravenous ocrelizumab-treated patients had one or more T1 Gd-enhancing lesions (mean 1.9).
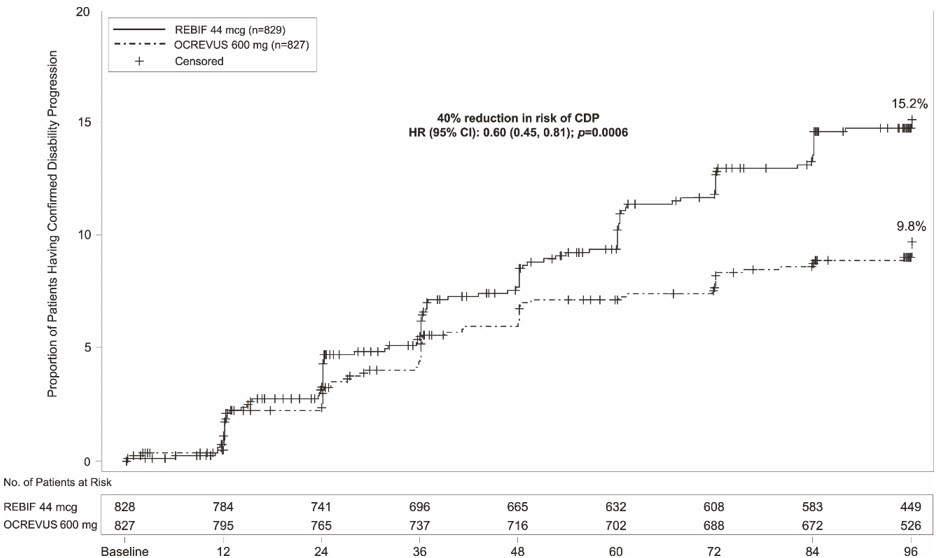
In Study 1 and Study 2, intravenous ocrelizumab significantly lowered the annualized relapse rate and the proportion of patients with disability progression confirmed at 12 weeks after onset compared to REBIF. Results for Study 1 and Study 2 are presented in Table 3 and Figure 1.
| Endpoints | Study 1 | Study 2 | ||
|---|---|---|---|---|
| Intravenous Ocrelizumab 600 mg | REBIF 44 mcg three times a week | Intravenous Ocrelizumab 600 mg | REBIF 44 mcg three times a week | |
| every 24 weeks N=410 |
N=411 | every 24 weeks N=417 |
N=418 | |
| Clinical Endpoints | ||||
| Annualized Relapse Rate (Primary Endpoint) | 0.156 | 0.292 | 0.155 | 0.290 |
| Relative Reduction | 46% (p<0.0001) | 47% (p<0.0001) | ||
| Proportion Relapse-free | 83% | 71% | 82% | 72% |
| Proportion of Patients with 12-week Confirmed Disability Progression Defined as an increase of 1.0 point or more from the baseline Expanded Disability Status Scale (EDSS) score for patients with baseline score of 5.5 or less, or 0.5 or more when the baseline score is greater than 5.5, Kaplan-Meier estimates at Week 96.
|
9.8% Intravenous Ocrelizumab vs 15.2% REBIF | |||
| Risk Reduction (Pooled Analysis Data prospectively pooled from Study 1 and Study 2. ) |
40%; p=0.0006 | |||
| MRI Endpoints | ||||
| Mean number of T1 Gd-enhancing lesions per MRI scan | 0.016 | 0.286 | 0.021 | 0.416 |
| Relative Reduction | 94% (p<0.0001) | 95% (p<0.0001) | ||
| Mean number of new and/or enlarging T2 hyperintense lesions per MRI | 0.323 | 1.413 | 0.325 | 1.904 |
| Relative Reduction | 77% (p<0.0001) | 83% (p<0.0001) |
|
Figure 1 Kaplan-Meier Plot Pre-specified pooled analysis of Study 1 and 2 of Time to Onset of Confirmed Disability Progression Sustained for at Least 12 Weeks With the Initial Event of Neurological Worsening Occurring During the Double-Blind Treatment Period in Pooled Studies 1 and 2 in Patients With RMS (Pooled ITT Population)
|
|
|
In exploratory subgroup analyses of Study 1 and Study 2, the effect of intravenous ocrelizumab on annualized relapse rate and disability progression was similar in male and female patients.
14.2 Intravenous Ocrelizumab in Patients With Primary Progressive Multiple Sclerosis
Study 3 was a randomized, double-blind, placebo-controlled clinical trial in patients with primary progressive multiple sclerosis (PPMS) (NCT01194570). Patients were randomized 2:1 to receive either intravenous ocrelizumab 600 mg or placebo as two 300 mg intravenous infusions 2 weeks apart every 24 weeks for at least 120 weeks. Selection criteria required a baseline EDSS of 3 to 6.5 and a score of 2 or greater for the EDSS pyramidal functional system due to lower extremity findings. Neurological assessments were conducted every 12 weeks. An MRI scan was obtained at baseline and at Weeks 24, 48, and 120.
In Study 3, the primary outcome was the time to onset of disability progression attributable to MS confirmed to be present at the next neurological assessment at least 12 weeks later. Disability progression occurred when the EDSS score increased by 1 point or more from the baseline EDSS if the baseline EDSS was 5.5 points or less, or by 0.5 points or more if the baseline EDSS was more than 5.5 points. In Study 3, confirmed disability progression also was deemed to have occurred if patients who had onset of disability progression discontinued participation in the study before the next assessment. Additional outcome measures included timed 25-foot walk, and percentage change in T2 hyperintense lesion volume.
Study 3 randomized 488 patients to intravenous ocrelizumab and 244 to placebo; 21% of intravenous ocrelizumab-treated patients and 34% of placebo-treated patients did not complete the trial. The baseline demographic and disease characteristics were balanced between the two treatment groups. At baseline, the mean age of patients was 45; 49% were female. The mean time since symptom onset was 6.7 years, the mean EDSS score was 4.7, and 26% had one or more T1 Gd-enhancing lesions at baseline; 88% of patients had not been treated previously with a non-steroid treatment for MS. The time to onset of disability progression confirmed at 12 weeks after onset was significantly longer for intravenous ocrelizumab-treated patients than for placebo-treated patients (see Figure 2). Results for Study 3 are presented in Table 4 and Figure 2.
| Endpoints | Study 3 | |
|---|---|---|
| Intravenous Ocrelizumab 600 mg (two 300 mg infusions two weeks apart every 24 weeks) |
Placebo | |
| N=488 | N=244 | |
| Clinical Outcomes | ||
| Proportion of patients with 12-week Confirmed Disability Progression Defined as an increase of 1.0 point or more from the baseline EDSS score for patients with baseline score of 5.5 or less, or an increase of 0.5 or more when the baseline score is more than 5.5.
|
32.9% | 39.3% |
| Risk reduction | 24%; p=0.0321 | |
| MRI Endpoints | ||
| Mean change in volume of T2 lesions, from baseline to Week 120 (cm3) | -0.39 | 0.79 |
| p<0.0001 |
|
Figure 2 Kaplan-Meier Plot of Time to Onset of Confirmed Disability Progression Sustained for at Least 12 Weeks With the Initial Event of Neurological Worsening Occurring During the Double-Blind Treatment Period in Study 3 All patients in this analysis had a minimum of 120 weeks of follow-up. The primary analysis is based on all disability progression events accrued including 21 without confirmatory EDSS at 12 weeks.
|
|
|
In the overall population in Study 3, the proportion of patients with 20 percent worsening of the timed 25-foot walk confirmed at 12 weeks was 49% in intravenous ocrelizumab-treated patients compared to 59% in placebo-treated patients (25% risk reduction).
In exploratory subgroup analyses of Study 3, the proportion of female patients with disability progression confirmed at 12 weeks after onset was similar in intravenous ocrelizumab-treated patients and placebo-treated patients (approximately 36% in each group). In male patients, the proportion of patients with disability progression confirmed at 12 weeks after onset was approximately 30% in intravenous ocrelizumab-treated patients and 43% in placebo-treated patients. Clinical and MRI endpoints that generally favored intravenous ocrelizumab numerically in the overall population, and that showed similar trends in both male and female patients, included annualized relapse rate, change in T2 lesion volume, and number of new or enlarging T2 lesions.
Structured Label Content
Section 42229-5 (42229-5)
Hepatitis B Virus Screening
Prior to initiating ocrelizumab treatment, perform Hepatitis B virus (HBV) screening. OCREVUS ZUNOVO is contraindicated in patients with active HBV confirmed by positive results for HBsAg and anti-HBV tests. For patients who are negative for surface antigen [HBsAg] and positive for HB core antibody [HBcAb+] or are carriers of HBV [HBsAg+], consult liver disease experts before starting and during treatment [see Warnings and Precautions (5.2)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Issued: 8/2025 | |||||||
|
MEDICATION GUIDE
OCREVUS ZUNOVO® (OH-creh-vus zoo-NOH-voh) (ocrelizumab and hyaluronidase-ocsq) injection, for subcutaneous use |
||||||||
|
What is the most important information I should know about OCREVUS ZUNOVO?
OCREVUS ZUNOVO can cause serious side effects, including:
|
||||||||
|
|
|
|
|||||
| These injection reactions can happen during or up to 24 hours after your injection. It is important that you call your healthcare provider right away if you get any of the signs or symptoms listed above after each injection. | ||||||||
|
||||||||
|
||||||||
|
|
|
||||||
| Signs of a more serious herpes infection include: | ||||||||
|
|
|
||||||
| Signs of infection can happen during treatment or after you have received your last dose of OCREVUS ZUNOVO. Tell your healthcare provider right away if you have an infection. Your healthcare provider should delay your treatment with OCREVUS ZUNOVO until your infection is gone. | ||||||||
|
||||||||
|
||||||||
|
||||||||
|
|
|||||||
|
||||||||
| See "What are the possible side effects of OCREVUS ZUNOVO?" for more information about side effects. | ||||||||
|
What is OCREVUS ZUNOVO?
OCREVUS ZUNOVO is a prescription medicine used to treat:
|
||||||||
|
Who should not receive OCREVUS ZUNOVO?
Do not receive OCREVUS ZUNOVO if you:
|
||||||||
Before receiving OCREVUS ZUNOVO, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||||
How will I receive OCREVUS ZUNOVO?
|
||||||||
|
What are the possible side effects of OCREVUS ZUNOVO? OCREVUS ZUNOVO may cause serious side effects, including:
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||||
|
General information about the safe and effective use of OCREVUS ZUNOVO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about OCREVUS ZUNOVO that is written for health professionals. |
||||||||
|
What are the ingredients in OCREVUS ZUNOVO?
Active ingredient: ocrelizumab and hyaluronidase-ocsq. Inactive ingredients: glacial acetic acid, methionine, polysorbate 20, sodium acetate, trehalose, and water for injection. Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 U.S. License No. 1048 For more information, go to www.OCREVUS.com or call 1-844-627-3887. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store OCREVUS ZUNOVO vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze or shake.
If necessary, OCREVUS ZUNOVO can be removed and placed back into the refrigerator. Unopened OCREVUS ZUNOVO vials may be stored in the original carton for a cumulative time of up to 12 hours without refrigeration at a temperature up to 25°C (77°F).
11 Description (11 DESCRIPTION)
Ocrelizumab is a recombinant humanized monoclonal antibody directed against CD20-expressing B-cells. Ocrelizumab is a glycosylated immunoglobulin G1 (IgG1) with a molecular mass of approximately 145 kDa.
Hyaluronidase (human recombinant) is an endoglycosidase used to increase the dispersion and absorption of co-administered drugs when administered subcutaneously. It is a glycosylated single-chain protein produced by mammalian (Chinese Hamster Ovary) cells containing a DNA plasmid encoding for a soluble fragment of human hyaluronidase (PH20). Hyaluronidase (human recombinant) has a molecular weight of approximately 61 kDa.
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied in single-dose vials. Each 23 mL of solution contains 920 mg ocrelizumab, 23,000 units of hyaluronidase (human recombinant), glacial acetic acid (5.5 mg), methionine (34.3 mg), polysorbate 20 (13.8 mg), sodium acetate (30.2 mg), trehalose (1889.5 mg), and water for injection at pH 5.3.
5.2 Infections
Serious, including life-threatening or fatal, bacterial, viral, parasitic, and fungal infections have been reported in patients receiving ocrelizumab. An increased risk of infections (including serious and fatal bacterial, fungal, and new or reactivated viral infections) has been observed in patients during and following completion of treatment with anti-CD20 B-cell depleting therapies.
A higher proportion of intravenous ocrelizumab-treated patients experienced infections compared to patients taking REBIF or placebo. In RMS trials, 58% of intravenous ocrelizumab-treated patients experienced one or more infections compared to 52% of REBIF-treated patients. In the PPMS trial, 70% of intravenous ocrelizumab-treated patients experienced one or more infections compared to 68% of patients on placebo.
Intravenous ocrelizumab was not associated with an increased risk of serious infections in MS patients in controlled trials.
Ocrelizumab increases the risk for upper respiratory tract infections, lower respiratory tract infections, skin infections, and herpes-related infections [see Adverse Reactions (6.1)].
Delay OCREVUS ZUNOVO administration in patients with an active infection until the infection is resolved.
5.5 Malignancies
An increased risk of malignancy with OCREVUS ZUNOVO may exist. In controlled trials, malignancies, including breast cancer, occurred more frequently in patients treated with intravenous ocrelizumab. Breast cancer occurred in 6 of 781 females treated with intravenous ocrelizumab and none of 668 females treated with REBIF or placebo. Patients should follow standard breast cancer screening guidelines.
5.7 Liver Injury
Clinically significant liver injury, without findings of viral hepatitis, has been reported in the postmarketing setting in patients treated with anti-CD20 B-cell depleting therapies approved for the treatment of MS, including ocrelizumab. Signs of liver injury, including markedly elevated serum hepatic enzymes with elevated total bilirubin, have occurred from weeks to months after administration.
Patients treated with OCREVUS ZUNOVO found to have an alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 3× the upper limit of normal (ULN) with serum total bilirubin greater than 2× ULN are potentially at risk for severe drug-induced liver injury.
Obtain liver function tests prior to initiating treatment with OCREVUS ZUNOVO [see Dosage and Administration (2.2)], and monitor for signs and symptoms of any hepatic injury during treatment. Measure serum aminotransferases, alkaline phosphatase, and bilirubin levels promptly in patients who report symptoms that may indicate liver injury, including new or worsening fatigue, anorexia, nausea, vomiting, right upper abdominal discomfort, dark urine, or jaundice. If liver injury is present and an alternative etiology is not identified, discontinue OCREVUS ZUNOVO.
7.2 Vaccinations
A Phase 3b randomized, open-label study examined the concomitant use of intravenous ocrelizumab and several non-live vaccines in adults 18-55 years of age with relapsing forms of MS (68 subjects undergoing treatment with intravenous ocrelizumab at the time of vaccination and 34 subjects not undergoing treatment with intravenous ocrelizumab at the time of vaccination). Concomitant exposure to intravenous ocrelizumab attenuated antibody responses to tetanus toxoid-containing vaccine, pneumococcal polysaccharide, pneumococcal conjugate vaccines, and seasonal inactivated influenza vaccines. The impact of the observed attenuation on vaccine effectiveness in this patient population is unknown. The safety and effectiveness of live or live-attenuated vaccines administered concomitantly with ocrelizumab have not been assessed [see Warnings and Precautions (5.2)].
8.4 Pediatric Use
Safety and effectiveness of OCREVUS ZUNOVO in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of OCREVUS ZUNOVO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
12.6 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. Immunogenicity data are highly dependent on the sensitivity and specificity of the test methods used. Additionally, the observed incidence of a positive result in a test method may be influenced by several factors, including sample handling, timing of sample collection, drug interference, concomitant medication, and the underlying disease. Therefore, comparison of the incidence of antibodies to OCREVUS ZUNOVO with the incidence of antibodies to other products may be misleading.
In Study 4, no patients tested positive for ocrelizumab anti-drug antibodies (ADAs) or anti-rHuPH20 (hyaluronidase [human recombinant]) antibodies.
14 Clinical Studies (14 CLINICAL STUDIES)
Studies 1-3 (described below), which established the effectiveness of ocrelizumab for the treatment of RMS and PPMS in adults, were conducted with intravenously-administered ocrelizumab. Study 4 demonstrated comparable exposure of OCREVUS ZUNOVO relative to the ocrelizumab intravenous formulation, which established the efficacy of OCREVUS ZUNOVO [see Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
OCREVUS ZUNOVO is contraindicated in patients with:
- Active HBV infection [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)]
- A history of life-threatening administration reaction to ocrelizumab [see Warnings and Precautions (5.1)]
- A history of hypersensitivity to ocrelizumab, hyaluronidase, or any component of OCREVUS ZUNOVO [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Injection Reactions [see Warnings and Precautions (5.1)]
- Infections [see Warnings and Precautions (5.2)]
- Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3)]
- Reduction in Immunoglobulins [see Warnings and Precautions (5.4)]
- Malignancies [see Warnings and Precautions (5.5)]
- Immune-Mediated Colitis [see Warnings and Precautions (5.6)]
- Liver Injury [see Warnings and Precautions (5.7)]
12.2 Pharmacodynamics
For B-cell counts, assays for CD19+ B-cells are used because the presence of ocrelizumab interferes with the CD20 assay. In clinical studies with OCREVUS ZUNOVO and intravenous ocrelizumab, CD19+ B-cell counts in blood were reduced by 14 days after administration. In clinical studies with intravenous ocrelizumab, B-cell counts rose to above the lower limit of normal (LLN) or above baseline counts between infusions of ocrelizumab at least one time in 0.3% to 4.1% of patients. In a clinical study of 51 patients treated with intravenous ocrelizumab, the median time for B-cell counts to return to either baseline or LLN was 72 weeks (range 27-175 weeks) after the last ocrelizumab infusion. Within 2.5 years after the last infusion, B-cell counts rose to either baseline or LLN in 90% of patients.
12.3 Pharmacokinetics
After subcutaneous administration of 920 mg ocrelizumab, the estimated mean exposure (AUC over the 24-week dosing interval) was 3730 µg/mL∙day. The mean Cmax was 132 µg/mL and tmax was reached after approximately 4 days (range 2 – 13 days). The estimated absolute bioavailability following subcutaneous administration was 81%.
In Study 4, the differences in pharmacokinetic exposures following the administration of OCREVUS ZUNOVO subcutaneously at 920 mg/23,000 units and ocrelizumab intravenously at 600 mg in MS patients were not clinically significant.
Pharmacokinetics (PK) of ocrelizumab in MS clinical studies fit a two-compartment model with time-dependent clearance.
2.4 Recommended Dosage
The recommended dosage of OCREVUS ZUNOVO is 920 mg/23,000 units (920 mg ocrelizumab and 23,000 units of hyaluronidase) administered as a single 23 mL subcutaneous injection in the abdomen over approximately 10 minutes every 6 months.
Monitor the patient closely during injections, with access to appropriate medical support to manage severe injection reactions. For the initial dose, monitor the patient for at least one hour post-injection. For subsequent doses, monitor the patient for at least 15 minutes post-injection [see Warnings and Precautions (5.1)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
OCREVUS ZUNOVO is indicated for the treatment of:
- Relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults
- Primary progressive MS, in adults
5.1 Injection Reactions
OCREVUS ZUNOVO can cause injection reactions, which can be local or systemic. Common symptoms of local injection reactions reported by patients treated with OCREVUS ZUNOVO in multiple sclerosis (MS) clinical trials included erythema, pain, swelling, and pruritus. Common symptoms of systemic injection reactions reported by patients included headache and nausea. In an open-label, active-controlled trial, injection reactions were more frequently reported with the first injection; 49% of patients experienced an injection reaction with the first injection [see Adverse Reactions (6.1)].
In MS clinical trials where ocrelizumab was administered intravenously, the incidence of infusion reactions in patients [who received methylprednisolone (or an equivalent steroid) and possibly other premedication to reduce the risk of infusion reactions prior to infusion] was 34% to 40%, with the highest incidence with the first infusion. There were no fatal infusion reactions, but 0.3% of intravenous ocrelizumab-treated MS patients experienced infusion reactions that were serious, some requiring hospitalization. Symptoms of infusion reactions can include pruritus, rash, urticaria, erythema, bronchospasm, throat irritation, oropharyngeal pain, dyspnea, pharyngeal or laryngeal edema, flushing, hypotension, pyrexia, fatigue, headache, dizziness, nausea, tachycardia, and anaphylaxis.
Monitor patients during and after injections [see Dosage and Administration (2.4)]. Inform patients that injection reactions can occur during or within 24 hours of the injection.
12.1 Mechanism of Action
The precise mechanism by which ocrelizumab exerts its therapeutic effects in multiple sclerosis is unknown, but is presumed to involve binding to CD20, a cell surface antigen present on pre-B and mature B lymphocytes. Following cell surface binding to B lymphocytes, ocrelizumab results in antibody-dependent cellular cytolysis and complement-mediated lysis.
Hyaluronan is a polysaccharide found in the extracellular matrix of the subcutaneous tissue. It is depolymerized by the naturally occurring enzyme hyaluronidase. Unlike the stable structural components of the interstitial matrix, hyaluronan has a half-life of approximately 0.5 days. Hyaluronidase increases permeability of the subcutaneous tissue by depolymerizing hyaluronan. In the doses administered, hyaluronidase in OCREVUS ZUNOVO acts transiently and locally. The effects of hyaluronidase are reversible and permeability of the subcutaneous tissue is restored within 24 to 48 hours.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Injection Reactions: Management recommendations for injection reactions depend on the type and severity of the reaction. Permanently discontinue OCREVUS ZUNOVO if a life-threatening injection reaction occurs (4, 5.1)
- Infections: Serious, including life-threatening and fatal, infections have occurred in patients treated with ocrelizumab. Delay OCREVUS ZUNOVO administration in patients with an active infection until the infection is resolved. Vaccination with live-attenuated or live vaccines is not recommended during treatment with OCREVUS ZUNOVO and after discontinuation, until B-cell repletion (5.2)
- Progressive Multifocal Leukoencephalopathy (PML): Withhold OCREVUS ZUNOVO at the first sign or symptom suggestive of PML (5.3)
- Reduction in Immunoglobulins: Monitor the level of immunoglobulins at the beginning of treatment. Monitor during and after discontinuation of treatment with OCREVUS ZUNOVO, until B-cell repletion, and especially when recurrent serious infections are suspected. Consider discontinuing OCREVUS ZUNOVO in patients with serious opportunistic or recurrent serious infections, and if prolonged hypogammaglobulinemia requires treatment with intravenous immunoglobulins (2.1, 5.4)
- Malignancies: An increased risk of malignancy, including breast cancer, may exist with OCREVUS ZUNOVO (5.5)
- Immune-Mediated Colitis: Immune-mediated colitis has been reported in the postmarketing setting. Monitor patients for new or persistent diarrhea or other gastrointestinal symptoms, and evaluate promptly if colitis is suspected (5.6)
- Liver Injury: Clinically significant liver injury has occurred. Obtain serum aminotransferases, alkaline phosphatase, and bilirubin levels before initiating OCREVUS ZUNOVO, and during treatment as clinically indicated. Discontinue OCREVUS ZUNOVO in patients with evidence of liver injury in the absence of an alternative etiology (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- OCREVUS ZUNOVO should be administered by a healthcare professional (2.1)
- For subcutaneous use in the abdomen only (2.1)
- OCREVUS ZUNOVO has different dosage and administration instructions than intravenous ocrelizumab (2.1)
- Before initiating OCREVUS ZUNOVO, screen for Hepatitis B virus and obtain serum quantitative immunoglobulins, aminotransferases, alkaline phosphatase, and bilirubin (2.2)
- Pre-medicate orally with dexamethasone (or an equivalent corticosteroid) and an antihistamine (e.g., desloratadine) at least 30 minutes prior to each injection (2.3)
- Administer 23 mL of OCREVUS ZUNOVO (920 mg ocrelizumab and 23,000 units hyaluronidase) subcutaneously in the abdomen over approximately 10 minutes every 6 months (2.4)
- Monitor patients closely during all injections, for at least one hour after the initial injection, and for at least 15 minutes after subsequent injections (2.4)
2.5 Delayed Or Missed Doses (2.5 Delayed or Missed Doses)
If a planned injection of OCREVUS ZUNOVO is missed, administer OCREVUS ZUNOVO as soon as possible; do not wait until the next scheduled dose. Reset the dose schedule to administer the next sequential dose 6 months after the missed dose is administered. Doses of OCREVUS ZUNOVO must be separated by at least 5 months [see Dosage and Administration (2.4)].
5.6 Immune Mediated Colitis (5.6 Immune-Mediated Colitis)
Immune-mediated colitis, which can present as a severe and acute-onset form of colitis, has been reported in patients receiving ocrelizumab in the postmarketing setting. Some cases of colitis were serious, requiring hospitalization, with a few patients requiring surgical intervention. Systemic corticosteroids were required in many of these patients. The time from treatment initiation to onset of symptoms in these cases ranged from a few weeks to years. Monitor patients for immune-mediated colitis during OCREVUS ZUNOVO treatment, and evaluate promptly if signs and symptoms that may indicate immune-mediated colitis, such as new or persistent diarrhea or other gastrointestinal signs and symptoms, occur.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Injection: 920 mg ocrelizumab and 23,000 units hyaluronidase per 23 mL (40 mg and 1,000 units per mL) solution in a single-dose vial (3)
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ocrelizumab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Immune-mediated colitis [see Warnings and Precautions (5.6)]
Hepatobiliary Disorders: Liver injury [see Warnings and Precautions (5.7)]
Infections and Infestations: Serious herpes infections [see Warnings and Precautions (5.2)], progressive multifocal leukoencephalopathy [see Warnings and Precautions (5.3)], and babesiosis
Skin: Pyoderma gangrenosum
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Based on animal data, may cause fetal harm (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of ocrelizumab has been evaluated in active-controlled clinical trials of ocrelizumab administered intravenously in patients with relapsing forms of MS (RMS) (Study 1 and Study 2) [see Clinical Studies (14.1)] and primary progressive MS (PPMS) (Study 3) [see Clinical Studies (14.2)], and in an open-label, active-controlled trial of OCREVUS ZUNOVO administered subcutaneously in patients with RMS and PPMS (Study 4) [see Clinical Studies (14.3)].
5.4 Reduction in Immunoglobulins
As expected with any B-cell depleting therapy, decreased immunoglobulin levels are observed with ocrelizumab treatment. The pooled data of intravenous ocrelizumab clinical studies (RMS and PPMS) and their open-label extensions (up to approximately 7 years of exposure) have shown an association between decreased levels of immunoglobulin G (IgG<LLN) and increased rates of serious infections. Monitor the levels of quantitative serum immunoglobulins during OCREVUS ZUNOVO treatment and after discontinuation of treatment, until B-cell repletion, and especially in the setting of recurrent serious infections. Consider discontinuing OCREVUS ZUNOVO therapy in patients with serious opportunistic or recurrent serious infections, and if prolonged hypogammaglobulinemia requires treatment with intravenous immunoglobulins [see Adverse Reactions (6.1)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.6 Preparation and Administration
To prevent medication errors, check the vial labels to ensure that the drug being prepared and administered is OCREVUS ZUNOVO and not intravenous ocrelizumab.
Visually inspect the vial for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use the vial if particulates or discoloration are present. Do not shake. Discard any unused portion remaining in the vial.
OCREVUS ZUNOVO is compatible with polypropylene (PP), polycarbonate (PC), polyethylene (PE), stainless steel (SS), polyvinylchloride (PVC), and polyurethane (PUR).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
OCREVUS ZUNOVO (ocrelizumab and hyaluronidase-ocsq) injection for subcutaneous use is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale brown solution supplied as a carton containing one 920 mg and 23,000 units/23 mL (40 mg and 1,000 units/mL) single-dose vial (NDC 50242-554-01).
2.1 Important Administration Information
OCREVUS ZUNOVO is for subcutaneous use in the abdomen only.
OCREVUS ZUNOVO has different dosage and administration instructions than intravenous ocrelizumab.
OCREVUS ZUNOVO should be administered via subcutaneous injection by a healthcare professional.
Principal Display Panel 23 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 23 mL Vial Carton)
NDC 50242-554-01
Ocrevus Zunovo®
(ocrelizumab and
hyaluronidase-ocsq)
Injection
920 mg and
23,000 units/23 mL
(40 mg and 1,000 units/mL)
For Subcutaneous Use Only.
For subcutaneous injection in the
abdomen over approximately 10 minutes.
Single-Dose Vial.
Discard Unused Portion.
Attention Pharmacist: Dispense the
accompanying Medication Guide to
each patient.
1 vial
Rx only
Genentech
11038285
5.3 Progressive Multifocal Leukoencephalopathy
Cases of progressive multifocal leukoencephalopathy (PML) have been reported in patients with MS treated with ocrelizumab in the postmarketing setting. PML is an opportunistic viral infection of the brain caused by the JC virus (JCV) that typically only occurs in patients who are immunocompromised, and that usually leads to death or severe disability. PML has occurred in ocrelizumab-treated patients who had not been treated previously with natalizumab (which has a known association with PML), were not taking any immunosuppressive or immunomodulatory medications associated with the risk of PML prior to or concomitantly with ocrelizumab, and did not have any known ongoing systemic medical conditions resulting in compromised immune system function.
JCV infection resulting in PML has also been observed in patients treated with other anti-CD20 antibodies and other MS therapies.
At the first sign or symptom suggestive of PML, withhold OCREVUS ZUNOVO and perform an appropriate diagnostic evaluation. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes.
MRI findings may be apparent before clinical signs or symptoms. Cases of PML, diagnosed based on MRI findings and the detection of JCV DNA in the cerebrospinal fluid in the absence of clinical signs or symptoms specific to PML, have been reported in patients treated with other MS medications associated with PML. Many of these patients subsequently became symptomatic with PML. Therefore, monitoring with MRI for signs that may be consistent with PML may be useful, and any suspicious findings should lead to further investigation to allow for an early diagnosis of PML, if present. Following discontinuation of another MS medication associated with PML, lower PML-related mortality and morbidity have been reported in patients who were initially asymptomatic at diagnosis compared to patients who had characteristic clinical signs and symptoms at diagnosis.
It is not known whether these differences are due to early detection and discontinuation of MS treatment or due to differences in disease in these patients.
If PML is confirmed, treatment with OCREVUS ZUNOVO should be discontinued.
14.3 Ocrevus Zunovo in Patients With Rms Or Ppms (14.3 OCREVUS ZUNOVO in Patients With RMS or PPMS)
Study 4 was a multicenter, randomized, open-label, parallel arm trial conducted to evaluate the comparative bioavailability, pharmacokinetics, pharmacodynamics, safety, and immunogenicity of OCREVUS ZUNOVO compared with intravenous ocrelizumab in patients with either RMS or PPMS (NCT05232825).
Study 4 enrolled 236 patients (213 with RMS, 23 with PPMS), 18-65 years of age with an EDSS between 0 to 6.5 at screening. The demographics were similar and baseline characteristics were balanced across the two treatment groups. The mean age was 40 years in both groups. In the OCREVUS ZUNOVO group, 35% of patients were male and the mean/median duration since MS diagnosis was 5.7/3.1 years, compared to 41% male and 4.8/2.4 years in the ocrelizumab IV group.
7.1 Immunosuppressive Or Immune Modulating Therapies (7.1 Immunosuppressive or Immune-Modulating Therapies)
The concomitant use of OCREVUS ZUNOVO and other immune-modulating or immunosuppressive therapies, including immunosuppressant doses of corticosteroids, is expected to increase the risk of immunosuppression. Consider the risk of additive immune system effects when coadministering immunosuppressive therapies with OCREVUS ZUNOVO. When switching from drugs with prolonged immune effects, such as daclizumab, fingolimod, natalizumab, teriflunomide, or mitoxantrone, consider the duration and mode of action of these drugs because of additive immunosuppressive effects when initiating OCREVUS ZUNOVO [see Warnings and Precautions (5.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
OCREVUS ZUNOVO contains ocrelizumab and hyaluronidase.
No carcinogenicity studies have been performed to assess the carcinogenic potential of ocrelizumab.
No studies have been performed to assess the mutagenic potential of ocrelizumab. As an antibody, ocrelizumab is not expected to interact directly with DNA.
No effects on reproductive organs were observed in male monkeys administered ocrelizumab by intravenous injection (three loading doses of 15 or 75 mg/kg, followed by weekly doses of 20 or 100 mg/kg) for 8 weeks. There were also no effects on estrus cycle in female monkeys administered ocrelizumab over three menstrual cycles using the same dosing regimen. The doses tested in monkey are 2 and 10 times the recommended human dose of 600 mg intravenous ocrelizumab, on a mg/kg basis.
Hyaluronidases are found in most tissues of the body. Long-term animal studies have not been performed to assess the carcinogenic or mutagenic potential of hyaluronidase. In addition, when subcutaneous hyaluronidase (recombinant human) was administered to cynomolgus monkeys for 39 weeks at dose levels up to 220,000 U/kg, which is > 570 times higher than the human dose, no evidence of toxicity to the male or female reproductive system was found through periodic monitoring of in-life parameters, e.g., semen analyses, hormone levels, menstrual cycles, and also from gross pathology, histopathology and organ weight data.
14.1 Intravenous Ocrelizumab in Patients With Relapsing Forms of Multiple Sclerosis
The efficacy of intravenous ocrelizumab was demonstrated in two randomized, double-blind, double-dummy, active comparator-controlled clinical trials of identical design, in patients with relapsing forms of multiple sclerosis (RMS) treated for 96 weeks (Study 1; NCT01247324 and Study 2; NCT01412333). The dose of intravenous ocrelizumab was 600 mg every 24 weeks (initial treatment was given as two 300 mg IV infusions administered 2 weeks apart, and subsequent doses were administered as a single 600 mg IV infusion) and placebo subcutaneous injections were given 3 times per week. The dose of REBIF, the active comparator, was 44 mcg given as subcutaneous injections 3 times per week and placebo IV infusions were given every 24 weeks. Both studies included patients who had experienced at least one relapse within the prior year, or two relapses within the prior two years, and had an Expanded Disability Status Scale (EDSS) score from 0 to 5.5. Patients with primary progressive forms of multiple sclerosis (MS) were excluded. Neurological evaluations were performed every 12 weeks and at the time of a suspected relapse. Brain MRIs were performed at baseline and at Weeks 24, 48, and 96.
The primary outcome of both Study 1 and Study 2 was the annualized relapse rate (ARR). Additional outcome measures included the proportion of patients with confirmed disability progression, the mean number of MRI T1 gadolinium (Gd)-enhancing lesions at Weeks 24, 48, and 96, and new or enlarging MRI T2 hyperintense lesions. Progression of disability was defined as an increase of 1 point or more from the baseline EDSS score attributable to MS when the baseline EDSS score was 5.5 or less, or 0.5 points or more when the baseline EDSS score was above 5.5. Disability progression was considered confirmed when the increase in the EDSS was confirmed at a regularly scheduled visit 12 weeks after the initial documentation of neurological worsening. The primary population for analysis of confirmed disability progression was the pooled population from Studies 1 and 2.
In Study 1, 410 patients were randomized to intravenous ocrelizumab and 411 to REBIF; 11% of intravenous ocrelizumab-treated patients and 17% of REBIF-treated patients did not complete the 96-week double-blind treatment period. The baseline demographic and disease characteristics were balanced between the two treatment groups. At baseline, the mean age of patients was 37 years; 66% were female. The mean time from MS diagnosis to randomization was 3.8 years, the mean number of relapses in the previous year was 1.3, and the mean EDSS score was 2.8; 74% of patients had not been treated with a non-steroid therapy for MS in the 2 years prior to the study. At baseline, 40% of patients had one or more T1 Gd-enhancing lesions (mean 1.8).
In Study 2, 417 patients were randomized to intravenous ocrelizumab and 418 to REBIF; 14% of intravenous ocrelizumab-treated patients and 23% of REBIF-treated patients did not complete the 96-week double-blind treatment period. The baseline demographic and disease characteristics were balanced between the two treatment groups. At baseline, the mean age of patients was 37 years; 66% were female. The mean time from MS diagnosis to randomization was 4.1 years, the mean number of relapses in the previous year was 1.3, and the mean EDSS score was 2.8; 74% of patients had not been treated with a non-steroid therapy for MS in the 2 years prior to the study. At baseline, 40% of intravenous ocrelizumab-treated patients had one or more T1 Gd-enhancing lesions (mean 1.9).
In Study 1 and Study 2, intravenous ocrelizumab significantly lowered the annualized relapse rate and the proportion of patients with disability progression confirmed at 12 weeks after onset compared to REBIF. Results for Study 1 and Study 2 are presented in Table 3 and Figure 1.
| Endpoints | Study 1 | Study 2 | ||
|---|---|---|---|---|
| Intravenous Ocrelizumab 600 mg | REBIF 44 mcg three times a week | Intravenous Ocrelizumab 600 mg | REBIF 44 mcg three times a week | |
| every 24 weeks N=410 |
N=411 | every 24 weeks N=417 |
N=418 | |
| Clinical Endpoints | ||||
| Annualized Relapse Rate (Primary Endpoint) | 0.156 | 0.292 | 0.155 | 0.290 |
| Relative Reduction | 46% (p<0.0001) | 47% (p<0.0001) | ||
| Proportion Relapse-free | 83% | 71% | 82% | 72% |
| Proportion of Patients with 12-week Confirmed Disability Progression Defined as an increase of 1.0 point or more from the baseline Expanded Disability Status Scale (EDSS) score for patients with baseline score of 5.5 or less, or 0.5 or more when the baseline score is greater than 5.5, Kaplan-Meier estimates at Week 96.
|
9.8% Intravenous Ocrelizumab vs 15.2% REBIF | |||
| Risk Reduction (Pooled Analysis Data prospectively pooled from Study 1 and Study 2. ) |
40%; p=0.0006 | |||
| MRI Endpoints | ||||
| Mean number of T1 Gd-enhancing lesions per MRI scan | 0.016 | 0.286 | 0.021 | 0.416 |
| Relative Reduction | 94% (p<0.0001) | 95% (p<0.0001) | ||
| Mean number of new and/or enlarging T2 hyperintense lesions per MRI | 0.323 | 1.413 | 0.325 | 1.904 |
| Relative Reduction | 77% (p<0.0001) | 83% (p<0.0001) |
|
Figure 1 Kaplan-Meier Plot Pre-specified pooled analysis of Study 1 and 2 of Time to Onset of Confirmed Disability Progression Sustained for at Least 12 Weeks With the Initial Event of Neurological Worsening Occurring During the Double-Blind Treatment Period in Pooled Studies 1 and 2 in Patients With RMS (Pooled ITT Population)
|
|
|
In exploratory subgroup analyses of Study 1 and Study 2, the effect of intravenous ocrelizumab on annualized relapse rate and disability progression was similar in male and female patients.
14.2 Intravenous Ocrelizumab in Patients With Primary Progressive Multiple Sclerosis
Study 3 was a randomized, double-blind, placebo-controlled clinical trial in patients with primary progressive multiple sclerosis (PPMS) (NCT01194570). Patients were randomized 2:1 to receive either intravenous ocrelizumab 600 mg or placebo as two 300 mg intravenous infusions 2 weeks apart every 24 weeks for at least 120 weeks. Selection criteria required a baseline EDSS of 3 to 6.5 and a score of 2 or greater for the EDSS pyramidal functional system due to lower extremity findings. Neurological assessments were conducted every 12 weeks. An MRI scan was obtained at baseline and at Weeks 24, 48, and 120.
In Study 3, the primary outcome was the time to onset of disability progression attributable to MS confirmed to be present at the next neurological assessment at least 12 weeks later. Disability progression occurred when the EDSS score increased by 1 point or more from the baseline EDSS if the baseline EDSS was 5.5 points or less, or by 0.5 points or more if the baseline EDSS was more than 5.5 points. In Study 3, confirmed disability progression also was deemed to have occurred if patients who had onset of disability progression discontinued participation in the study before the next assessment. Additional outcome measures included timed 25-foot walk, and percentage change in T2 hyperintense lesion volume.
Study 3 randomized 488 patients to intravenous ocrelizumab and 244 to placebo; 21% of intravenous ocrelizumab-treated patients and 34% of placebo-treated patients did not complete the trial. The baseline demographic and disease characteristics were balanced between the two treatment groups. At baseline, the mean age of patients was 45; 49% were female. The mean time since symptom onset was 6.7 years, the mean EDSS score was 4.7, and 26% had one or more T1 Gd-enhancing lesions at baseline; 88% of patients had not been treated previously with a non-steroid treatment for MS. The time to onset of disability progression confirmed at 12 weeks after onset was significantly longer for intravenous ocrelizumab-treated patients than for placebo-treated patients (see Figure 2). Results for Study 3 are presented in Table 4 and Figure 2.
| Endpoints | Study 3 | |
|---|---|---|
| Intravenous Ocrelizumab 600 mg (two 300 mg infusions two weeks apart every 24 weeks) |
Placebo | |
| N=488 | N=244 | |
| Clinical Outcomes | ||
| Proportion of patients with 12-week Confirmed Disability Progression Defined as an increase of 1.0 point or more from the baseline EDSS score for patients with baseline score of 5.5 or less, or an increase of 0.5 or more when the baseline score is more than 5.5.
|
32.9% | 39.3% |
| Risk reduction | 24%; p=0.0321 | |
| MRI Endpoints | ||
| Mean change in volume of T2 lesions, from baseline to Week 120 (cm3) | -0.39 | 0.79 |
| p<0.0001 |
|
Figure 2 Kaplan-Meier Plot of Time to Onset of Confirmed Disability Progression Sustained for at Least 12 Weeks With the Initial Event of Neurological Worsening Occurring During the Double-Blind Treatment Period in Study 3 All patients in this analysis had a minimum of 120 weeks of follow-up. The primary analysis is based on all disability progression events accrued including 21 without confirmatory EDSS at 12 weeks.
|
|
|
In the overall population in Study 3, the proportion of patients with 20 percent worsening of the timed 25-foot walk confirmed at 12 weeks was 49% in intravenous ocrelizumab-treated patients compared to 59% in placebo-treated patients (25% risk reduction).
In exploratory subgroup analyses of Study 3, the proportion of female patients with disability progression confirmed at 12 weeks after onset was similar in intravenous ocrelizumab-treated patients and placebo-treated patients (approximately 36% in each group). In male patients, the proportion of patients with disability progression confirmed at 12 weeks after onset was approximately 30% in intravenous ocrelizumab-treated patients and 43% in placebo-treated patients. Clinical and MRI endpoints that generally favored intravenous ocrelizumab numerically in the overall population, and that showed similar trends in both male and female patients, included annualized relapse rate, change in T2 lesion volume, and number of new or enlarging T2 lesions.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:14.937565 · Updated: 2026-03-14T22:39:42.922945