These Highlights Do Not Include All The Information Needed To Use Rivastigmine Transdermal System Safely And Effectively.
b201fe68-b5e5-4a6f-8cf1-069543f03e76
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rivastigmine Transdermal System is an acetylcholinesterase inhibitor indicated for treatment of: Mild, moderate, and severe dementia of the Alzheimer's type (AD) ( 1.1 ) Mild-to-moderate dementia associated with Parkinson's disease (PD) ( 1.2 )
Indications and Usage
Rivastigmine Transdermal System is an acetylcholinesterase inhibitor indicated for treatment of: Mild, moderate, and severe dementia of the Alzheimer's type (AD) ( 1.1 ) Mild-to-moderate dementia associated with Parkinson's disease (PD) ( 1.2 )
Dosage and Administration
Apply patch on intact skin for a 24-hour period; replace with a new patch every 24 hours. ( 2.1 , 2.4 ) Initial Dose : Initiate treatment with 4.6 mg/24 hours Rivastigmine Transdermal System. ( 2.1 ) Dose Titration ( 2.1 ) : After a minimum of 4 weeks, if tolerated, increase dose to 9.5 mg/24 hours, which is the minimum effective dose. Following a minimum additional 4 weeks, may increase dosage to maximum dosage of 13.3 mg/24 hours. Mild-to-Moderate Alzheimer's Disease and Parkinson's Disease Dementia : Rivastigmine Transdermal System 9.5 mg/24 hours or 13.3 mg/24 hours once daily. ( 2.1 ) Severe Alzheimer's Disease : Rivastigmine Transdermal System 13.3 mg/24 hours once daily. ( 2.1 ) For treatment interruption longer than 3 days, retitrate dosage starting at 4.6 mg per 24 hours. ( 2.1 ) Consider dose adjustments in patients with ( 2.2 ): Mild-to-moderate hepatic impairment ( 8.6 ) Low (less than 50 kg) body weight ( 8.7 )
Warnings and Precautions
Hospitalization and, rarely, death have been reported due to application of multiple patches at same time. Ensure patients or caregivers receive instruction on proper dosing and administration. ( 5.1 ) Gastrointestinal Adverse Reactions : May include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. ( 5.2 ) Application site reactions may occur with the patch form of rivastigmine. Discontinue treatment if application site reactions spread beyond the patch size, if there is evidence of a more intense local reaction (e.g., increasing erythema, edema, papules, vesicles), and if symptoms do not significantly improve within 48 hours after patch removal. ( 5.3 )
Contraindications
Rivastigmine Transdermal System is contraindicated in patients with: known hypersensitivity to rivastigmine, other carbamate derivatives, or other components of the formulation [see Description (11) ] . previous history of application site reactions with rivastigmine transdermal patch suggestive of allergic contact dermatitis [see Warnings and Precautions (5.3) ] . Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2) ] .
Adverse Reactions
Rivastigmine transdermal system can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1) ] . For this reason, initiate treatment with Rivastigmine Transdermal System at a dose of 4.6 mg/24 hours and titrate to a dose of 9.5 mg/24 hours and then to a dose of 13.3 mg/24 hours, if appropriate [see Dosage and Administration (2.1) ] . If treatment is interrupted for more than 3 days because of intolerance, reinitiate Rivastigmine Transdermal System with the 4.6 mg/24 hours dose to reduce the possibility of severe vomiting and its potentially serious sequelae. A postmarketing report described a case of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment of an oral formulation of rivastigmine without retitration after 8 weeks of treatment interruption. Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
Drug Interactions
Concomitant use with metoclopramide, beta-blockers, or cholinomimetics and anticholinergic medications is not recommended. ( 7.1 , 7.2 , 7.3 )
Patient Information
Rivastigmine (RIV-a-STIG-meen) Transdermal System What is the most important information I should know about Rivastigmine Transdermal System? Rivastigmine Transdermal System is for skin use only. What is Rivastigmine Transdermal System? Rivastigmine Transdermal System is a prescription medicine used to treat: Mild, moderate, and severe memory problems (dementia) associated with Alzheimer's disease. Mild-to-moderate memory problems (dementia) associated with Parkinson's disease (PD). Based on clinical trials conducted over 6 to 12 months, Rivastigmine Transdermal System was shown to help with cognition which includes (memory, understanding communication and reasoning) and with doing daily tasks. Rivastigmine Transdermal System does not work the same in all people. Some people treated with Rivastigmine Transdermal System may: Seem much better Get better in small ways or stay the same Get worse but slower than expected Not change and then get worse as expected Some patients will not benefit from treatment with Rivastigmine Transdermal System. Rivastigmine Transdermal System does not cure Alzheimer's disease. All patients with Alzheimer's disease get worse over time. Rivastigmine Transdermal System comes as a transdermal system that delivers rivastigmine (the medicine in Rivastigmine Transdermal System) through the skin. It is not known if Rivastigmine Transdermal System is safe or effective in children under 18 years of age. Who should not use Rivastigmine Transdermal System? Do not use Rivastigmine Transdermal System if you: are allergic to rivastigmine, carbamate derivatives, or any of the ingredients in Rivastigmine Transdermal System. See the end of this leaflet for a complete list of ingredients in Rivastigmine Transdermal System. have had a skin reaction that: spread beyond the Rivastigmine Transdermal System size had blisters, increased skin redness, or swelling did not get better within 48 hours after you removed the Rivastigmine Transdermal System Ask your healthcare provider if you are not sure if you should use Rivastigmine Transdermal System. What should I tell my healthcare provider before using Rivastigmine Transdermal System? Before you use Rivastigmine Transdermal System, tell your healthcare provider if you: have or have had a stomach ulcer are planning to have surgery have or have had problems with your heart have problems passing urine have or have had seizures have problems with movement (tremors) have asthma or breathing problems have a loss of appetite or are losing weight have had a skin reaction to rivastigmine (the medicine in Rivastigmine Transdermal System) in the past. have any other medical conditions are pregnant or plan to become pregnant. It is not known if the medicine in Rivastigmine Transdermal System will harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant. are breastfeeding or plan to breastfeed. It is not known if the medicine in Rivastigmine Transdermal System passes into your breast milk. Talk to your doctor about the best way to feed your baby if you take Rivastigmine Transdermal System. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take: a medicine used to treat inflammation [nonsteroidal anti-inflammatory drugs (NSAIDs)] other medicines used to treat Alzheimer's or Parkinson's disease an anticholinergic medicine, such as an allergy or cold medicine, a medicine to treat bladder or bowel spasms, or certain asthma medicines, or certain medicines to prevent motion or travel sickness metoclopramide, a drug given to relieve symptoms of nausea, gastroesophageal reflux disease(GERD), or nausea and vomiting after surgery or chemotherapy treatment If you are undergoing surgery while using Rivastigmine Transdermal System, inform your doctor because Rivastigmine Transdermal System may exaggerate the effects of anesthesia, or the effects of a betablocker, a type of medicine given for high blood pressure, heart disease, and other medical conditions Ask your healthcare provider if you are not sure if your medicine is one listed above. Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine. How should I use Rivastigmine Transdermal System? Use Rivastigmine Transdermal System exactly as your healthcare provider tells you to use it. Rivastigmine Transdermal Systems come in 3 different dosage strengths. Your healthcare provider may change your dose as needed Wear only 1 Rivastigmine Transdermal System at a time. Rivastigmine Transdermal System is for skin use only. Only apply Rivastigmine Transdermal System to healthy skin that is clean, dry, hairless, and free of redness, irritation, burns or cuts. Avoid applying Rivastigmine Transdermal System to areas on your body that will be rubbed against tight clothing. Do not apply Rivastigmine Transdermal System to skin that has cream, lotion, or powder on it. Change your Rivastigmine Transdermal System every 24 hours at the same time of day. You may write the date and time you put on the Rivastigmine Transdermal System with a ballpoint pen before applying the patch to help you remember when to remove it. Change your application site every day to avoid skin irritation. You can use the same area, but do not use the exact same spot for at least 14 days after your last application. Check to see if the Rivastigmine Transdermal System has become loose when you are bathing, swimming, or showering. Rivastigmine Transdermal System is designed to deliver medication during the time it is worn. If your Rivastigmine Transdermal System falls off before its usual replacement time, put on a new rivastigmine transdermal system right away. Replace the new patch the next day at the same time as usual. Do not use overlays, bandages, or tape to secure an Rivastigmine Transdermal System that has become loose or try to reapply an Rivastigmine Transdermal System that has fallen off. If you miss a dose or forget to change your Rivastigmine Transdermal System apply your next Rivastigmine Transdermal System as soon as you remember. Do not apply 2 rivastigmine transdermal systems to make up for the missed dose. If you miss more than 3 doses of applying Rivastigmine Transdermal System, call your healthcare provider before putting on a new Rivastigmine Transdermal System. You may need to restart Rivastigmine Transdermal System at a lower dose. Always remove the old Rivastigmine Transdermal System from the previous day before you apply a new one. Having more than 1 Rivastigmine Transdermal System on your body at the same time can cause you to get too much medicine. If you accidentally use more than 1 Rivastigmine Transdermal System at a time, call your healthcare provider right away. If you are unable to reach your healthcare provider, call your local Poison Control Center at 1-800-222-1222 or go to the nearest hospital emergency room right away. What should I avoid while using Rivastigmine Transdermal System? Do not touch your eyes after you touch the Rivastigmine Transdermal System. In case of accidental contact with your eyes or if your eyes become red after handling the patch, rinse immediately with plenty of water and seek medical advice if symptoms do not resolve. Rivastigmine Transdermal System can cause drowsiness, dizziness, weakness, or fainting. Do not drive, operate heavy machinery, or do other dangerous activities until you know how Rivastigmine Transdermal System affects you. Avoid exposure to heat sources such as excessive sunlight, saunas, or sunrooms for long periods of time. What are the possible side effects of Rivastigmine Transdermal System? Rivastigmine Transdermal System may cause serious side effects, including: Medication overdose. Hospitalization and rarely death may happen when people accidently wear more than 1 patch at the same time. It is important that the old Rivastigmine Transdermal System be removed before you apply a new one. Do not wear more than 1 Rivastigmine Transdermal System at a time. Stomach or bowel (intestinal) problems, including: nausea vomiting diarrhea dehydration loss of appetite weight loss bleeding in your stomach (ulcers) Skin reactions. Some people have had a serious skin reaction called allergic contact dermatitis (ACD) when using Rivastigmine Transdermal System. Stop using Rivastigmine Transdermal System and call your healthcare provider right away if you experience reactions that spread beyond the patch size, are intense in nature and do not improve within 48 hours after the patch is removed. Symptoms of ACD may be intense and include: itching, redness, swelling, warmth or tenderness of the skin peeling or blistering of the skin that may ooze, drain or crust over heart problems seizures problems with movement (tremors) The most common side effects of Rivastigmine Transdermal System include: depression headache anxiety dizziness stomach pain urinary tract infections muscle weakness tiredness trouble sleeping Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Rivastigmine Transdermal System. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088. How should I store Rivastigmine Transdermal System? Store Rivastigmine Transdermal System between 68°F to 77°F (20°C to 25°C). Keep Rivastigmine Transdermal System in the sealed pouch until ready to use. Keep Rivastigmine Transdermal System and all medicines out of the reach of children. General information about the safe and effective use of Rivastigmine Transdermal System. Medicines are sometimes prescribed for purposes other than those listed in the Patient Information leaflet. Do not use Rivastigmine Transdermal System for a condition for which it was not prescribed. Do not give Rivastigmine Transdermal System to other people, even if they have the same symptoms you have. It may harm them. This Patient Information leaflet summarizes the most important information about Rivastigmine Transdermal System . If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Rivastigmine Transdermal System that is written for health professionals. For more information, go to www.breckenridgepharma.com or call 1-800-367-3395. What are the ingredients of Rivastigmine Transdermal System? Active ingredient: rivastigmine Excipients include: acrylic adhesive (applied to a flexible polymer backing film), acrylates copolymer, silicone adhesive. Revised: 10/2024
Medication Information
Warnings and Precautions
Hospitalization and, rarely, death have been reported due to application of multiple patches at same time. Ensure patients or caregivers receive instruction on proper dosing and administration. ( 5.1 ) Gastrointestinal Adverse Reactions : May include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. ( 5.2 ) Application site reactions may occur with the patch form of rivastigmine. Discontinue treatment if application site reactions spread beyond the patch size, if there is evidence of a more intense local reaction (e.g., increasing erythema, edema, papules, vesicles), and if symptoms do not significantly improve within 48 hours after patch removal. ( 5.3 )
Indications and Usage
Rivastigmine Transdermal System is an acetylcholinesterase inhibitor indicated for treatment of: Mild, moderate, and severe dementia of the Alzheimer's type (AD) ( 1.1 ) Mild-to-moderate dementia associated with Parkinson's disease (PD) ( 1.2 )
Dosage and Administration
Apply patch on intact skin for a 24-hour period; replace with a new patch every 24 hours. ( 2.1 , 2.4 ) Initial Dose : Initiate treatment with 4.6 mg/24 hours Rivastigmine Transdermal System. ( 2.1 ) Dose Titration ( 2.1 ) : After a minimum of 4 weeks, if tolerated, increase dose to 9.5 mg/24 hours, which is the minimum effective dose. Following a minimum additional 4 weeks, may increase dosage to maximum dosage of 13.3 mg/24 hours. Mild-to-Moderate Alzheimer's Disease and Parkinson's Disease Dementia : Rivastigmine Transdermal System 9.5 mg/24 hours or 13.3 mg/24 hours once daily. ( 2.1 ) Severe Alzheimer's Disease : Rivastigmine Transdermal System 13.3 mg/24 hours once daily. ( 2.1 ) For treatment interruption longer than 3 days, retitrate dosage starting at 4.6 mg per 24 hours. ( 2.1 ) Consider dose adjustments in patients with ( 2.2 ): Mild-to-moderate hepatic impairment ( 8.6 ) Low (less than 50 kg) body weight ( 8.7 )
Contraindications
Rivastigmine Transdermal System is contraindicated in patients with: known hypersensitivity to rivastigmine, other carbamate derivatives, or other components of the formulation [see Description (11) ] . previous history of application site reactions with rivastigmine transdermal patch suggestive of allergic contact dermatitis [see Warnings and Precautions (5.3) ] . Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2) ] .
Adverse Reactions
Rivastigmine transdermal system can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1) ] . For this reason, initiate treatment with Rivastigmine Transdermal System at a dose of 4.6 mg/24 hours and titrate to a dose of 9.5 mg/24 hours and then to a dose of 13.3 mg/24 hours, if appropriate [see Dosage and Administration (2.1) ] . If treatment is interrupted for more than 3 days because of intolerance, reinitiate Rivastigmine Transdermal System with the 4.6 mg/24 hours dose to reduce the possibility of severe vomiting and its potentially serious sequelae. A postmarketing report described a case of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment of an oral formulation of rivastigmine without retitration after 8 weeks of treatment interruption. Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
Drug Interactions
Concomitant use with metoclopramide, beta-blockers, or cholinomimetics and anticholinergic medications is not recommended. ( 7.1 , 7.2 , 7.3 )
Patient Information
Rivastigmine (RIV-a-STIG-meen)
Transdermal System
What is the most important information I should know about Rivastigmine Transdermal System?
Rivastigmine Transdermal System is for skin use only.
What is Rivastigmine Transdermal System?
Rivastigmine Transdermal System is a prescription medicine used to treat:
- Mild, moderate, and severe memory problems (dementia) associated with Alzheimer's disease.
- Mild-to-moderate memory problems (dementia) associated with Parkinson's disease (PD).
Based on clinical trials conducted over 6 to 12 months, Rivastigmine Transdermal System was shown to help with cognition which includes (memory, understanding communication and reasoning) and with doing daily tasks. Rivastigmine Transdermal System does not work the same in all people. Some people treated with Rivastigmine Transdermal System may:
- Seem much better
- Get better in small ways or stay the same
- Get worse but slower than expected
- Not change and then get worse as expected
Some patients will not benefit from treatment with Rivastigmine Transdermal System. Rivastigmine Transdermal System does not cure Alzheimer's disease. All patients with Alzheimer's disease get worse over time.
Rivastigmine Transdermal System comes as a transdermal system that delivers rivastigmine (the medicine in Rivastigmine Transdermal System) through the skin.
It is not known if Rivastigmine Transdermal System is safe or effective in children under 18 years of age.
Who should not use Rivastigmine Transdermal System?
Do not use Rivastigmine Transdermal System if you:
- are allergic to rivastigmine, carbamate derivatives, or any of the ingredients in Rivastigmine Transdermal System. See the end of this leaflet for a complete list of ingredients in Rivastigmine Transdermal System.
- have had a skin reaction that:
- spread beyond the Rivastigmine Transdermal System size
- had blisters, increased skin redness, or swelling
- did not get better within 48 hours after you removed the Rivastigmine Transdermal System
Ask your healthcare provider if you are not sure if you should use Rivastigmine Transdermal System.
What should I tell my healthcare provider before using Rivastigmine Transdermal System?
Before you use Rivastigmine Transdermal System, tell your healthcare provider if you:
- have or have had a stomach ulcer
- are planning to have surgery
- have or have had problems with your heart
- have problems passing urine
- have or have had seizures
- have problems with movement (tremors)
- have asthma or breathing problems
- have a loss of appetite or are losing weight
- have had a skin reaction to rivastigmine (the medicine in Rivastigmine Transdermal System) in the past.
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if the medicine in Rivastigmine Transdermal System will harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
- are breastfeeding or plan to breastfeed. It is not known if the medicine in Rivastigmine Transdermal System passes into your breast milk. Talk to your doctor about the best way to feed your baby if you take Rivastigmine Transdermal System.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Especially tell your healthcare provider if you take:
- a medicine used to treat inflammation [nonsteroidal anti-inflammatory drugs (NSAIDs)]
- other medicines used to treat Alzheimer's or Parkinson's disease
- an anticholinergic medicine, such as an allergy or cold medicine, a medicine to treat bladder or bowel spasms, or certain asthma medicines, or certain medicines to prevent motion or travel sickness
- metoclopramide, a drug given to relieve symptoms of nausea, gastroesophageal reflux disease(GERD), or nausea and vomiting after surgery or chemotherapy treatment
- If you are undergoing surgery while using Rivastigmine Transdermal System, inform your doctor because Rivastigmine Transdermal System may exaggerate the effects of anesthesia, or the effects of a betablocker, a type of medicine given for high blood pressure, heart disease, and other medical conditions
Ask your healthcare provider if you are not sure if your medicine is one listed above.
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I use Rivastigmine Transdermal System?
- Use Rivastigmine Transdermal System exactly as your healthcare provider tells you to use it.
- Rivastigmine Transdermal Systems come in 3 different dosage strengths.
- Your healthcare provider may change your dose as needed
- Wear only 1 Rivastigmine Transdermal System at a time.
- Rivastigmine Transdermal System is for skin use only.
- Only apply Rivastigmine Transdermal System to healthy skin that is clean, dry, hairless, and free of redness, irritation, burns or cuts.
- Avoid applying Rivastigmine Transdermal System to areas on your body that will be rubbed against tight clothing.
- Do not apply Rivastigmine Transdermal System to skin that has cream, lotion, or powder on it.
- Change your Rivastigmine Transdermal System every 24 hours at the same time of day. You may write the date and time you put on the Rivastigmine Transdermal System with a ballpoint pen before applying the patch to help you remember when to remove it.
- Change your application site every day to avoid skin irritation. You can use the same area, but do not use the exact same spot for at least 14 days after your last application.
- Check to see if the Rivastigmine Transdermal System has become loose when you are bathing, swimming, or showering.
- Rivastigmine Transdermal System is designed to deliver medication during the time it is worn. If your Rivastigmine Transdermal System falls off before its usual replacement time, put on a new rivastigmine transdermal system right away. Replace the new patch the next day at the same time as usual. Do not use overlays, bandages, or tape to secure an Rivastigmine Transdermal System that has become loose or try to reapply an Rivastigmine Transdermal System that has fallen off.
- If you miss a dose or forget to change your Rivastigmine Transdermal System apply your next Rivastigmine Transdermal System as soon as you remember. Do not apply 2 rivastigmine transdermal systems to make up for the missed dose.
- If you miss more than 3 doses of applying Rivastigmine Transdermal System, call your healthcare provider before putting on a new Rivastigmine Transdermal System. You may need to restart Rivastigmine
Transdermal System at a lower dose.
- Always remove the old Rivastigmine Transdermal System from the previous day before you apply a new one.
- Having more than 1 Rivastigmine Transdermal System on your body at the same time can cause you to get too much medicine. If you accidentally use more than 1 Rivastigmine Transdermal System at a time, call your healthcare provider right away. If you are unable to reach your healthcare provider, call your local Poison Control Center at 1-800-222-1222 or go to the nearest hospital emergency room right away.
What should I avoid while using Rivastigmine Transdermal System?
- Do not touch your eyes after you touch the Rivastigmine Transdermal System. In case of accidental contact with your eyes or if your eyes become red after handling the patch, rinse immediately with plenty of water and seek medical advice if symptoms do not resolve.
- Rivastigmine Transdermal System can cause drowsiness, dizziness, weakness, or fainting. Do not drive, operate heavy machinery, or do other dangerous activities until you know how Rivastigmine Transdermal System affects you.
- Avoid exposure to heat sources such as excessive sunlight, saunas, or sunrooms for long periods of time.
What are the possible side effects of Rivastigmine Transdermal System?
Rivastigmine Transdermal System may cause serious side effects, including:
- Medication overdose. Hospitalization and rarely death may happen when people accidently wear more than 1 patch at the same time. It is important that the old Rivastigmine Transdermal System be removed before you apply a new one. Do not wear more than 1 Rivastigmine Transdermal System at a time.
-
Stomach or bowel (intestinal) problems, including:
- nausea
- vomiting
- diarrhea
- dehydration
- loss of appetite
- weight loss
- bleeding in your stomach (ulcers)
-
Skin reactions. Some people have had a serious skin reaction called allergic contact dermatitis (ACD) when using Rivastigmine Transdermal System. Stop using Rivastigmine Transdermal System and call your healthcare provider right away if you experience reactions that spread beyond the patch size, are intense in nature and do not improve within 48 hours after the patch is removed. Symptoms of ACD may be intense and include:
- itching, redness, swelling, warmth or tenderness of the skin
- peeling or blistering of the skin that may ooze, drain or crust over
- heart problems
- seizures
- problems with movement (tremors)
The most common side effects of Rivastigmine Transdermal System include:
|
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of Rivastigmine Transdermal System. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
How should I store Rivastigmine Transdermal System?
- Store Rivastigmine Transdermal System between 68°F to 77°F (20°C to 25°C).
- Keep Rivastigmine Transdermal System in the sealed pouch until ready to use.
Keep Rivastigmine Transdermal System and all medicines out of the reach of children.
General information about the safe and effective use of Rivastigmine Transdermal System.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information leaflet. Do not use Rivastigmine Transdermal System for a condition for which it was not prescribed. Do not give Rivastigmine Transdermal System to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Rivastigmine Transdermal System . If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Rivastigmine Transdermal System that is written for health professionals.
For more information, go to www.breckenridgepharma.com or call 1-800-367-3395.
What are the ingredients of Rivastigmine Transdermal System?
Active ingredient: rivastigmine
Excipients include: acrylic adhesive (applied to a flexible polymer backing film), acrylates copolymer, silicone adhesive.
Revised: 10/2024
Description
Rivastigmine Transdermal System is an acetylcholinesterase inhibitor indicated for treatment of: Mild, moderate, and severe dementia of the Alzheimer's type (AD) ( 1.1 ) Mild-to-moderate dementia associated with Parkinson's disease (PD) ( 1.2 )
Section 42229-5
Initial Dose
Initiate treatment with one 4.6 mg/24 hours Rivastigmine Transdermal System applied to the skin once daily [see Dosage and Administration (2.4)].
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep Rivastigmine Transdermal System in the individual sealed pouch until use. Each pouch contains 1 patch. Used systems should be folded, with the adhesive surfaces pressed together, and discarded safely.
10 Overdosage
Overdose with Rivastigmine Transdermal System has been reported in the postmarketing setting [see Warnings and Precautions (5.1)]. Overdoses have occurred from application of more than one patch at one time and not removing the previous day's patch before applying a new patch. The symptoms reported in these overdose cases are similar to those seen in cases of overdose associated with rivastigmine oral formulations.
Because strategies for the management of overdose are continually evolving, it is advisable to contact a Poison Control Center to determine the latest recommendations for the management of an overdose of any drug. As rivastigmine has a plasma half-life of about 3.4 hours after patch administration and a duration of acetylcholinesterase inhibition of about 9 hours, it is recommended that in cases of asymptomatic overdose the patch should be immediately removed and no further patch should be applied for the next 24 hours.
As in any case of overdose, general supportive measures should be utilized.
Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Atypical responses in blood pressure and heart rate have been reported with other drugs that increase cholinergic activity when coadministered with quaternary anticholinergics such as glycopyrrolate. Additional symptoms associated with rivastigmine overdose are diarrhea, abdominal pain, dizziness, tremor, headache, somnolence, confusional state, hyperhidrosis, hypertension, hallucinations and malaise. Due to the short plasma elimination half-life of rivastigmine after patch administration, dialysis (hemodialysis, peritoneal dialysis, or hemofiltration) would not be clinically indicated in the event of an overdose.
In overdose accompanied by severe nausea and vomiting, the use of antiemetics should be considered. A fatal outcome has rarely been reported with rivastigmine overdose.
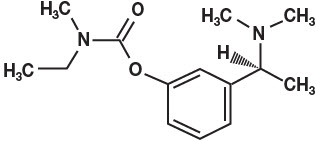
11 Description
Rivastigmine Transdermal System contains rivastigmine, a reversible cholinesterase inhibitor known chemically as (S)-3-[1-(dimethylamino) ethyl]phenyl ethylmethylcarbamate. It has an empirical formula of C14H22N2O2 as the base and a molecular weight of 250.34 g/mol (as the base). Rivastigmine is a viscous, clear, and colorless to yellow to very slightly brown liquid that is sparingly soluble in water and very soluble in ethanol, acetonitrile, n-octanol and ethyl acetate. The distribution coefficient at 37°C in n-octanol/phosphate buffer solution pH 7 is 4.27.
Rivastigmine Transdermal System is for transdermal administration. The patch is a 4-layer laminate containing the backing layer, drug matrix, adhesive matrix and release liner (see Figure 1 ). The release liner is removed and discarded prior to use.
| Layer 1: Backing Film Layer 2: Drug Product (Acrylic) Matrix Layer 3: Adhesive (Silicone) Matrix Layer 4: Release Liner (removed at time of use) |
Excipients within the formulation include acrylic adhesive (applied to a flexible polymer backing film), acrylates copolymer, silicone adhesive.
7.3 Beta Blockers
Additive bradycardic effects resulting in syncope may occur when rivastigmine is used concomitantly with beta-blockers, especially cardioselective beta-blockers (including atenolol). Concomitant use is not recommended when signs of bradycardia including syncope are present.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. The use of Rivastigmine Transdermal System in pediatric patients (below 18 years of age) is not recommended.
8.5 Geriatric Use
Of the total number of patients in clinical studies of Rivastigmine Transdermal System, 88% were 65 years and over, while 55% were 75 years. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.3 Skin Reactions
Skin application site reactions may occur with Rivastigmine Transdermal System. These reactions are not in themselves an indication of sensitization. However, use of Rivastigmine Transdermal System may lead to allergic contact dermatitis.
Allergic contact dermatitis should be suspected if application site reactions spread beyond the patch size, if there is evidence of a more intense local reaction (e.g. increasing erythema, edema, papules, vesicles) and if symptoms do not significantly improve within 48 hours after patch removal. In these cases, treatment should be discontinued [see Contraindications (4)].
In patients who develop application site reactions to Rivastigmine Transdermal System suggestive of allergic contact dermatitis and who still require rivastigmine, treatment should be switched to oral rivastigmine only after negative allergy testing and under close medical supervision. It is possible that some patients sensitized to rivastigmine by exposure to Rivastigmine Transdermal System may not be able to take rivastigmine in any form.
There have been isolated postmarketing reports of patients experiencing disseminated allergic dermatitis when administered rivastigmine irrespective of the route of administration (oral or transdermal). In these cases, treatment should be discontinued [see Contraindications (4)]. Patients and caregivers should be instructed accordingly.
7.1 Metoclopramide
Due to the risk of additive extra-pyramidal adverse reactions, the concomitant use of metoclopramide and Rivastigmine Transdermal System is not recommended.
14 Clinical Studies
The effectiveness of the Rivastigmine Transdermal System in dementia of the Alzheimer's type and dementia associated with Parkinson's disease was based on the results of 3 controlled trials of Rivastigmine Transdermal System in patients with Alzheimer's disease (Studies 1, 2, and 3) (see below); 3 controlled trials of oral rivastigmine in patients with dementia of the Alzheimer's type; and 1 controlled trial of oral rivastigmine in patients with dementia associated with Parkinson's disease. See the prescribing information for oral rivastigmine for details of the four studies of oral rivastigmine.
4 Contraindications
Rivastigmine Transdermal System is contraindicated in patients with:
- known hypersensitivity to rivastigmine, other carbamate derivatives, or other components of the formulation [see Description (11)].
- previous history of application site reactions with rivastigmine transdermal patch suggestive of allergic contact dermatitis [see Warnings and Precautions (5.3)].
Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following adverse reactions are described below and elsewhere in the labeling:
- Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.2)].
- Skin Reactions [see Warnings and Precautions (5.3)].
- Other Adverse Reactions from Increased Cholinergic Activity [see Warnings and Precautions (5.4)].
7 Drug Interactions
Instructions for Use
You will need the following supplies (See Figure A):
Rivastigmine Transdermal System is supplied in cartons containing 30 patches (See Figure A)
- Rivastigmine Transdermal System is a thin, transparent, plastic patch that sticks to the skin. Each Rivastigmine Transdermal System is sealed in a pouch that protects it until you are ready to put it on (See Figure A).
- Only 1 Rivastigmine Transdermal System should be worn at a time. Do not apply more than 1 Rivastigmine Transdermal System at a time to the body.
- Do not open the pouch or remove the Rivastigmine Transdermal System until you are ready to apply it.
Using Rivastigmine Transdermal System:
Step 1. Choose an area to apply the Rivastigmine Transdermal System (See Figure B).
- Instructions for Caregivers: Apply Rivastigmine Transdermal System to the upper or lower back if it is likely that the patient will remove it. If this is not a concern, the Rivastigmine Transdermal System can be applied instead to the upper arm or chest. Do not apply the Rivastigmine Transdermal System to areas where it can be rubbed off by tight clothing or belts.
- Only apply the Rivastigmine Transdermal System to healthy skin that is clean, dry, hairless, and free of redness, irritation, burns or cuts.
The diagram represents areas on the body where Rivastigmine Transdermal System may be applied.
Only 1 patch should be worn at a time. Do not apply multiple patches to the body.
Step 2. Remove the Rivastigmine Transdermal System from the pouch (See Figure C).
Carefully cut the pouch along the dotted line to open and remove the Rivastigmine Transdermal System. Save the pouch for later use.
- Do not cut or fold the Rivastigmine Transdermal System itself.
Step 3. Remove 1 side of the adhesive liner (See Figure D).
- A protective liner covers the sticky (adhesive) side of the Rivastigmine Transdermal System. Peel off 1 side of the protective cover. Do not touch the sticky part of the Rivastigmine Transdermal System with your fingers.
Step 4. Apply the Rivastigmine Transdermal System to your skin (See Figure E).
- Apply the sticky (adhesive) side of the Rivastigmine Transdermal System to your chosen area of skin and then peel off the other side of the protective cover.
- Press down on the Rivastigmine Transdermal System firmly for 30 seconds to make sure that the edges stick to your skin (See Figure F).
Step 5. Wash your hands with soap and water right away.
Note:
- If your Rivastigmine Transdermal System falls off, select a new area, and repeat Steps 2 to 5 to apply a new Rivastigmine Transdermal System.
- Be sure to replace the new Rivastigmine Transdermal System the next day at the same time as usual.
Removing your Rivastigmine Transdermal System:
Step 6. Remove the Rivastigmine Transdermal System from the skin (See Figure G).
- Gently pull on 1 edge of the Rivastigmine Transdermal System to remove it from your skin.
Throwing away the used Rivastigmine Transdermal System:
Step 7. Throw away the used Rivastigmine Transdermal System (See Figure H).
- Fold the used Rivastigmine Transdermal System in half (with the sticky sides together) and put it back into the pouch that you saved.
- Throw away the used Rivastigmine Transdermal System safely and out of the reach of children and pets.
- Some medicine stays in the patch for 24 hours after you use it and should be folded together (sticky side together) and safely thrown away. Do not try to re-use Rivastigmine Transdermal Systems.
Step 8. Wash your hands with soap and water right away.
- After you remove the Rivastigmine Transdermal System, if any adhesive remains on your skin, you can use soap and water or an oil-based substance (such as baby oil) to remove the adhesive. Alcohol or other dissolving liquids (such as nail polish remover) should not be used.
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Distributed by:
Breckenridge Pharmaceutical, Inc.
Berkeley Heights, NJ 07922
Manufactured by:
AdhexPharma
21300 Chenôve
France
Revised: 12/2023
12.2 Pharmacodynamics
After a 6-mg oral dose of rivastigmine in humans, anticholinesterase activity is present in cerebrospinal fluid for about 10 hours, with a maximum inhibition of about 60% 5 hours after dosing.
In vitro and in vivo studies demonstrate that the inhibition of cholinesterase by rivastigmine is not affected by the concomitant administration of memantine, an N-methyl-D-aspartate receptor antagonist.
8.6 Hepatic Impairment
Increased exposure to rivastigmine was observed in patients with mild or moderate hepatic impairment with oral rivastigmine. Patients with mild or moderate hepatic impairment may be able to only tolerate lower doses [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)]. No data are available on the use of rivastigmine in patients with severe hepatic impairment.
1 Indications and Usage
1.1 Alzheimer's Disease
Rivastigmine Transdermal System is indicated for the treatment of dementia of the Alzheimer's type (AD). Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
12.1 Mechanism of Action
Although the precise mechanism of action of rivastigmine is unknown, it is thought to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by cholinesterase. The effect of rivastigmine may lessen as the disease process advances and fewer cholinergic neurons remain functionally intact. There is no evidence that rivastigmine alters the course of the underlying dementing process.
5 Warnings and Precautions
- Hospitalization and, rarely, death have been reported due to application of multiple patches at same time. Ensure patients or caregivers receive instruction on proper dosing and administration. (5.1)
- Gastrointestinal Adverse Reactions: May include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. (5.2)
- Application site reactions may occur with the patch form of rivastigmine. Discontinue treatment if application site reactions spread beyond the patch size, if there is evidence of a more intense local reaction (e.g., increasing erythema, edema, papules, vesicles), and if symptoms do not significantly improve within 48 hours after patch removal. (5.3)
2 Dosage and Administration
- Apply patch on intact skin for a 24-hour period; replace with a new patch every 24 hours. (2.1, 2.4)
- Initial Dose: Initiate treatment with 4.6 mg/24 hours Rivastigmine Transdermal System. (2.1)
- Dose Titration (2.1): After a minimum of 4 weeks, if tolerated, increase dose to 9.5 mg/24 hours, which is the minimum effective dose. Following a minimum additional 4 weeks, may increase dosage to maximum dosage of 13.3 mg/24 hours.
- Mild-to-Moderate Alzheimer's Disease and Parkinson's Disease Dementia: Rivastigmine Transdermal System 9.5 mg/24 hours or 13.3 mg/24 hours once daily. (2.1)
- Severe Alzheimer's Disease: Rivastigmine Transdermal System 13.3 mg/24 hours once daily. (2.1)
- For treatment interruption longer than 3 days, retitrate dosage starting at 4.6 mg per 24 hours. (2.1)
- Consider dose adjustments in patients with (2.2):
8.7 Low Or High Body Weight
Because rivastigmine blood levels vary with weight, careful titration and monitoring should be performed in patients with low or high body weights [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths
Rivastigmine Transdermal System is available in 3 strengths. Each patch has a transparent backing layer labeled as either:
- Rivastigmine 4.6 mg/24 hr.
- Rivastigmine 9.5 mg/24 hr.
- Rivastigmine 13.3 mg/24 hr.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of rivastigmine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: Tachycardia
Hepatobiliary Disorders: Abnormal liver function tests, hepatitis
Nervous System Disorders: Parkinson's disease (worsening), seizure, tremor
Psychiatric Disorders: nightmares
Skin and Subcutaneous Tissue Disorders: Allergic dermatitis, application site hypersensitivity, blister, disseminated allergic dermatitis, Stevens-Johnson syndrome, urticaria
Vascular Disorders: Hypertension
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Rivastigmine Transdermal System has been administered to 4516 patients with Alzheimer's disease during clinical trials worldwide. Of these, 3005 patients have been treated for at least 26 weeks, 1771 patients have been treated for at least 52 weeks, 974 patients have been treated for at least 78 weeks, and 24 patients have been treated for at least 104 weeks.
1.2 Parkinson's Disease Dementia
Rivastigmine Transdermal System is indicated for the treatment of mild-to-moderate dementia associated with Parkinson's disease (PDD).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.2 Gastrointestinal Adverse Reactions
Rivastigmine transdermal system can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1)]. For this reason, initiate treatment with Rivastigmine Transdermal System at a dose of 4.6 mg/24 hours and titrate to a dose of 9.5 mg/24 hours and then to a dose of 13.3 mg/24 hours, if appropriate [see Dosage and Administration (2.1)].
If treatment is interrupted for more than 3 days because of intolerance, reinitiate Rivastigmine Transdermal System with the 4.6 mg/24 hours dose to reduce the possibility of severe vomiting and its potentially serious sequelae. A postmarketing report described a case of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment of an oral formulation of rivastigmine without retitration after 8 weeks of treatment interruption.
Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
2.4 Important Administration Instructions
Rivastigmine Transdermal System is for transdermal use on intact skin.
-
(a)Do not use the patch if the pouch seal is broken or the patch is cut, damaged, or changed in any way.
-
(b)Apply the Rivastigmine Transdermal System once a day
- Press down firmly for 30 seconds until the edges stick well when applying to clean, dry, hairless, intact healthy skin in a place that will not be rubbed against by tight clothing.
- Use the upper or lower back as the site of application because the patch is less likely to be removed by the patient. If sites on the back are not accessible, apply the patch to the upper arm or chest.
- Do not apply to a skin area where cream, lotion, or powder has recently been applied.
-
(c)Do not apply to skin that is red, irritated, or cut.
-
(d)Replace the Rivastigmine Transdermal System with a new patch every 24 hours. Instruct patients to only wear 1 patch at a time (remove the previous day's patch before applying a new patch) [see Warnings and Precautions (5.1) and Overdosage (10)]. If a patch falls off or if a dose is missed, apply a new patch immediately and then replace this patch the following day at the usual application time.
-
(e)Change the site of patch application daily to minimize potential irritation, although a new patch can be applied to the same general anatomic site (e.g., another spot on the upper back) on consecutive days. Do not apply a new patch to the same location for at least 14 days.
-
(f)May wear the patch during bathing and in hot weather. But avoid long exposure to external heat sources (excessive sunlight, saunas, solariums).
-
(g)Place used patches in the previously saved pouch and discard in the trash, away from pets or children.
-
(h)Wash hands with soap and water after removing the patch. In case of contact with eyes or if the eyes become red after handling the patch, rinse immediately with plenty of water and seek medical advice if symptoms do not resolve.
5.1 Medication Errors Resulting in Overdose
Medication errors with Rivastigmine Transdermal System have resulted in serious adverse reactions; some cases have required hospitalization, and rarely, led to death. The majority of medication errors have involved not removing the old patch when putting on a new one and the use of multiple patches at one time. Instruct patients and their caregivers on important administration instructions for Rivastigmine Transdermal System [see Dosage and Administration (2.4)].
5.5 Impairment in Driving Or Use of Machinery
Dementia may cause gradual impairment of driving performance or compromise the ability to use machinery. The administration of rivastigmine may also result in adverse reactions that are detrimental to these functions. During treatment with the Rivastigmine Transdermal System, routinely evaluate the patient's ability to continue driving or operating machinery.
7.2 Cholinomimetic and Anticholinergic Medications
Rivastigmine Transdermal System may increase the cholinergic effects of other cholinomimetic medications and may also interfere with the activity of anticholinergic medications (e.g., oxybutynin, tolterodine). Concomitant use of Rivastigmine Transdermal System with medications having these pharmacologic effects is not recommended unless deemed clinically necessary [see Warnings and Precautions (5.4)].
Principal Display Panel 4.6 Mg Patch Pouch Carton
NDC 51991-897-30
Rivastigmine Transdermal
System
Each System Delivers 4.6 mg/24 hours
For Transdermal Use Only
breckenridge
A Towa
Company
Rx Only
Contains 30 Systems
Principal Display Panel 9.5 Mg Patch Pouch Carton
NDC 51991-898-30
Rivastigmine Transdermal
System
Each System Delivers 9.5 mg/24 hours
For Transdermal Use Only
breckenridge
A Towa
Company
Rx Only
Contains 30 Systems
Principal Display Panel 13.3 Mg Patch Pouch Carton
NDC 51991-899-30
Rivastigmine Transdermal
System
Each System Delivers 13.3 mg/24 hours
For Transdermal Use Only
breckenridge
A Towa
Company
Rx Only
Contains 30 Systems
2.3 Switching to Rivastigmine Transdermal System From Rivastigmine Capsules Or Rivastigmine Oral Solution
Patients treated with Rivastigmine capsules or oral solution may be switched to Rivastigmine Transdermal System as follows:
- A patient who is on a total daily dose of less than 6 mg of oral rivastigmine can be switched to the 4.6 mg/24 hours Rivastigmine Transdermal System.
- A patient who is on a total daily dose of 6 mg to 12 mg of oral rivastigmine can be switched to the 9.5 mg/24 hours Rivastigmine Transdermal System.
Instruct patients or caregivers to apply the first patch on the day following the last oral dose.
Structured Label Content
Section 42229-5 (42229-5)
Initial Dose
Initiate treatment with one 4.6 mg/24 hours Rivastigmine Transdermal System applied to the skin once daily [see Dosage and Administration (2.4)].
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep Rivastigmine Transdermal System in the individual sealed pouch until use. Each pouch contains 1 patch. Used systems should be folded, with the adhesive surfaces pressed together, and discarded safely.
10 Overdosage (10 OVERDOSAGE)
Overdose with Rivastigmine Transdermal System has been reported in the postmarketing setting [see Warnings and Precautions (5.1)]. Overdoses have occurred from application of more than one patch at one time and not removing the previous day's patch before applying a new patch. The symptoms reported in these overdose cases are similar to those seen in cases of overdose associated with rivastigmine oral formulations.
Because strategies for the management of overdose are continually evolving, it is advisable to contact a Poison Control Center to determine the latest recommendations for the management of an overdose of any drug. As rivastigmine has a plasma half-life of about 3.4 hours after patch administration and a duration of acetylcholinesterase inhibition of about 9 hours, it is recommended that in cases of asymptomatic overdose the patch should be immediately removed and no further patch should be applied for the next 24 hours.
As in any case of overdose, general supportive measures should be utilized.
Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Atypical responses in blood pressure and heart rate have been reported with other drugs that increase cholinergic activity when coadministered with quaternary anticholinergics such as glycopyrrolate. Additional symptoms associated with rivastigmine overdose are diarrhea, abdominal pain, dizziness, tremor, headache, somnolence, confusional state, hyperhidrosis, hypertension, hallucinations and malaise. Due to the short plasma elimination half-life of rivastigmine after patch administration, dialysis (hemodialysis, peritoneal dialysis, or hemofiltration) would not be clinically indicated in the event of an overdose.
In overdose accompanied by severe nausea and vomiting, the use of antiemetics should be considered. A fatal outcome has rarely been reported with rivastigmine overdose.
11 Description (11 DESCRIPTION)
Rivastigmine Transdermal System contains rivastigmine, a reversible cholinesterase inhibitor known chemically as (S)-3-[1-(dimethylamino) ethyl]phenyl ethylmethylcarbamate. It has an empirical formula of C14H22N2O2 as the base and a molecular weight of 250.34 g/mol (as the base). Rivastigmine is a viscous, clear, and colorless to yellow to very slightly brown liquid that is sparingly soluble in water and very soluble in ethanol, acetonitrile, n-octanol and ethyl acetate. The distribution coefficient at 37°C in n-octanol/phosphate buffer solution pH 7 is 4.27.
Rivastigmine Transdermal System is for transdermal administration. The patch is a 4-layer laminate containing the backing layer, drug matrix, adhesive matrix and release liner (see Figure 1 ). The release liner is removed and discarded prior to use.
| Layer 1: Backing Film Layer 2: Drug Product (Acrylic) Matrix Layer 3: Adhesive (Silicone) Matrix Layer 4: Release Liner (removed at time of use) |
Excipients within the formulation include acrylic adhesive (applied to a flexible polymer backing film), acrylates copolymer, silicone adhesive.
7.3 Beta Blockers (7.3 Beta-blockers)
Additive bradycardic effects resulting in syncope may occur when rivastigmine is used concomitantly with beta-blockers, especially cardioselective beta-blockers (including atenolol). Concomitant use is not recommended when signs of bradycardia including syncope are present.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. The use of Rivastigmine Transdermal System in pediatric patients (below 18 years of age) is not recommended.
8.5 Geriatric Use
Of the total number of patients in clinical studies of Rivastigmine Transdermal System, 88% were 65 years and over, while 55% were 75 years. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.3 Skin Reactions
Skin application site reactions may occur with Rivastigmine Transdermal System. These reactions are not in themselves an indication of sensitization. However, use of Rivastigmine Transdermal System may lead to allergic contact dermatitis.
Allergic contact dermatitis should be suspected if application site reactions spread beyond the patch size, if there is evidence of a more intense local reaction (e.g. increasing erythema, edema, papules, vesicles) and if symptoms do not significantly improve within 48 hours after patch removal. In these cases, treatment should be discontinued [see Contraindications (4)].
In patients who develop application site reactions to Rivastigmine Transdermal System suggestive of allergic contact dermatitis and who still require rivastigmine, treatment should be switched to oral rivastigmine only after negative allergy testing and under close medical supervision. It is possible that some patients sensitized to rivastigmine by exposure to Rivastigmine Transdermal System may not be able to take rivastigmine in any form.
There have been isolated postmarketing reports of patients experiencing disseminated allergic dermatitis when administered rivastigmine irrespective of the route of administration (oral or transdermal). In these cases, treatment should be discontinued [see Contraindications (4)]. Patients and caregivers should be instructed accordingly.
7.1 Metoclopramide
Due to the risk of additive extra-pyramidal adverse reactions, the concomitant use of metoclopramide and Rivastigmine Transdermal System is not recommended.
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of the Rivastigmine Transdermal System in dementia of the Alzheimer's type and dementia associated with Parkinson's disease was based on the results of 3 controlled trials of Rivastigmine Transdermal System in patients with Alzheimer's disease (Studies 1, 2, and 3) (see below); 3 controlled trials of oral rivastigmine in patients with dementia of the Alzheimer's type; and 1 controlled trial of oral rivastigmine in patients with dementia associated with Parkinson's disease. See the prescribing information for oral rivastigmine for details of the four studies of oral rivastigmine.
4 Contraindications (4 CONTRAINDICATIONS)
Rivastigmine Transdermal System is contraindicated in patients with:
- known hypersensitivity to rivastigmine, other carbamate derivatives, or other components of the formulation [see Description (11)].
- previous history of application site reactions with rivastigmine transdermal patch suggestive of allergic contact dermatitis [see Warnings and Precautions (5.3)].
Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described below and elsewhere in the labeling:
- Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.2)].
- Skin Reactions [see Warnings and Precautions (5.3)].
- Other Adverse Reactions from Increased Cholinergic Activity [see Warnings and Precautions (5.4)].
7 Drug Interactions (7 DRUG INTERACTIONS)
Patient Information
Rivastigmine (RIV-a-STIG-meen)
Transdermal System
What is the most important information I should know about Rivastigmine Transdermal System?
Rivastigmine Transdermal System is for skin use only.
What is Rivastigmine Transdermal System?
Rivastigmine Transdermal System is a prescription medicine used to treat:
- Mild, moderate, and severe memory problems (dementia) associated with Alzheimer's disease.
- Mild-to-moderate memory problems (dementia) associated with Parkinson's disease (PD).
Based on clinical trials conducted over 6 to 12 months, Rivastigmine Transdermal System was shown to help with cognition which includes (memory, understanding communication and reasoning) and with doing daily tasks. Rivastigmine Transdermal System does not work the same in all people. Some people treated with Rivastigmine Transdermal System may:
- Seem much better
- Get better in small ways or stay the same
- Get worse but slower than expected
- Not change and then get worse as expected
Some patients will not benefit from treatment with Rivastigmine Transdermal System. Rivastigmine Transdermal System does not cure Alzheimer's disease. All patients with Alzheimer's disease get worse over time.
Rivastigmine Transdermal System comes as a transdermal system that delivers rivastigmine (the medicine in Rivastigmine Transdermal System) through the skin.
It is not known if Rivastigmine Transdermal System is safe or effective in children under 18 years of age.
Who should not use Rivastigmine Transdermal System?
Do not use Rivastigmine Transdermal System if you:
- are allergic to rivastigmine, carbamate derivatives, or any of the ingredients in Rivastigmine Transdermal System. See the end of this leaflet for a complete list of ingredients in Rivastigmine Transdermal System.
- have had a skin reaction that:
- spread beyond the Rivastigmine Transdermal System size
- had blisters, increased skin redness, or swelling
- did not get better within 48 hours after you removed the Rivastigmine Transdermal System
Ask your healthcare provider if you are not sure if you should use Rivastigmine Transdermal System.
What should I tell my healthcare provider before using Rivastigmine Transdermal System?
Before you use Rivastigmine Transdermal System, tell your healthcare provider if you:
- have or have had a stomach ulcer
- are planning to have surgery
- have or have had problems with your heart
- have problems passing urine
- have or have had seizures
- have problems with movement (tremors)
- have asthma or breathing problems
- have a loss of appetite or are losing weight
- have had a skin reaction to rivastigmine (the medicine in Rivastigmine Transdermal System) in the past.
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if the medicine in Rivastigmine Transdermal System will harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
- are breastfeeding or plan to breastfeed. It is not known if the medicine in Rivastigmine Transdermal System passes into your breast milk. Talk to your doctor about the best way to feed your baby if you take Rivastigmine Transdermal System.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Especially tell your healthcare provider if you take:
- a medicine used to treat inflammation [nonsteroidal anti-inflammatory drugs (NSAIDs)]
- other medicines used to treat Alzheimer's or Parkinson's disease
- an anticholinergic medicine, such as an allergy or cold medicine, a medicine to treat bladder or bowel spasms, or certain asthma medicines, or certain medicines to prevent motion or travel sickness
- metoclopramide, a drug given to relieve symptoms of nausea, gastroesophageal reflux disease(GERD), or nausea and vomiting after surgery or chemotherapy treatment
- If you are undergoing surgery while using Rivastigmine Transdermal System, inform your doctor because Rivastigmine Transdermal System may exaggerate the effects of anesthesia, or the effects of a betablocker, a type of medicine given for high blood pressure, heart disease, and other medical conditions
Ask your healthcare provider if you are not sure if your medicine is one listed above.
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I use Rivastigmine Transdermal System?
- Use Rivastigmine Transdermal System exactly as your healthcare provider tells you to use it.
- Rivastigmine Transdermal Systems come in 3 different dosage strengths.
- Your healthcare provider may change your dose as needed
- Wear only 1 Rivastigmine Transdermal System at a time.
- Rivastigmine Transdermal System is for skin use only.
- Only apply Rivastigmine Transdermal System to healthy skin that is clean, dry, hairless, and free of redness, irritation, burns or cuts.
- Avoid applying Rivastigmine Transdermal System to areas on your body that will be rubbed against tight clothing.
- Do not apply Rivastigmine Transdermal System to skin that has cream, lotion, or powder on it.
- Change your Rivastigmine Transdermal System every 24 hours at the same time of day. You may write the date and time you put on the Rivastigmine Transdermal System with a ballpoint pen before applying the patch to help you remember when to remove it.
- Change your application site every day to avoid skin irritation. You can use the same area, but do not use the exact same spot for at least 14 days after your last application.
- Check to see if the Rivastigmine Transdermal System has become loose when you are bathing, swimming, or showering.
- Rivastigmine Transdermal System is designed to deliver medication during the time it is worn. If your Rivastigmine Transdermal System falls off before its usual replacement time, put on a new rivastigmine transdermal system right away. Replace the new patch the next day at the same time as usual. Do not use overlays, bandages, or tape to secure an Rivastigmine Transdermal System that has become loose or try to reapply an Rivastigmine Transdermal System that has fallen off.
- If you miss a dose or forget to change your Rivastigmine Transdermal System apply your next Rivastigmine Transdermal System as soon as you remember. Do not apply 2 rivastigmine transdermal systems to make up for the missed dose.
- If you miss more than 3 doses of applying Rivastigmine Transdermal System, call your healthcare provider before putting on a new Rivastigmine Transdermal System. You may need to restart Rivastigmine
Transdermal System at a lower dose.
- Always remove the old Rivastigmine Transdermal System from the previous day before you apply a new one.
- Having more than 1 Rivastigmine Transdermal System on your body at the same time can cause you to get too much medicine. If you accidentally use more than 1 Rivastigmine Transdermal System at a time, call your healthcare provider right away. If you are unable to reach your healthcare provider, call your local Poison Control Center at 1-800-222-1222 or go to the nearest hospital emergency room right away.
What should I avoid while using Rivastigmine Transdermal System?
- Do not touch your eyes after you touch the Rivastigmine Transdermal System. In case of accidental contact with your eyes or if your eyes become red after handling the patch, rinse immediately with plenty of water and seek medical advice if symptoms do not resolve.
- Rivastigmine Transdermal System can cause drowsiness, dizziness, weakness, or fainting. Do not drive, operate heavy machinery, or do other dangerous activities until you know how Rivastigmine Transdermal System affects you.
- Avoid exposure to heat sources such as excessive sunlight, saunas, or sunrooms for long periods of time.
What are the possible side effects of Rivastigmine Transdermal System?
Rivastigmine Transdermal System may cause serious side effects, including:
- Medication overdose. Hospitalization and rarely death may happen when people accidently wear more than 1 patch at the same time. It is important that the old Rivastigmine Transdermal System be removed before you apply a new one. Do not wear more than 1 Rivastigmine Transdermal System at a time.
-
Stomach or bowel (intestinal) problems, including:
- nausea
- vomiting
- diarrhea
- dehydration
- loss of appetite
- weight loss
- bleeding in your stomach (ulcers)
-
Skin reactions. Some people have had a serious skin reaction called allergic contact dermatitis (ACD) when using Rivastigmine Transdermal System. Stop using Rivastigmine Transdermal System and call your healthcare provider right away if you experience reactions that spread beyond the patch size, are intense in nature and do not improve within 48 hours after the patch is removed. Symptoms of ACD may be intense and include:
- itching, redness, swelling, warmth or tenderness of the skin
- peeling or blistering of the skin that may ooze, drain or crust over
- heart problems
- seizures
- problems with movement (tremors)
The most common side effects of Rivastigmine Transdermal System include:
|
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of Rivastigmine Transdermal System. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
How should I store Rivastigmine Transdermal System?
- Store Rivastigmine Transdermal System between 68°F to 77°F (20°C to 25°C).
- Keep Rivastigmine Transdermal System in the sealed pouch until ready to use.
Keep Rivastigmine Transdermal System and all medicines out of the reach of children.
General information about the safe and effective use of Rivastigmine Transdermal System.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information leaflet. Do not use Rivastigmine Transdermal System for a condition for which it was not prescribed. Do not give Rivastigmine Transdermal System to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Rivastigmine Transdermal System . If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Rivastigmine Transdermal System that is written for health professionals.
For more information, go to www.breckenridgepharma.com or call 1-800-367-3395.
What are the ingredients of Rivastigmine Transdermal System?
Active ingredient: rivastigmine
Excipients include: acrylic adhesive (applied to a flexible polymer backing film), acrylates copolymer, silicone adhesive.
Revised: 10/2024
Instructions for Use
You will need the following supplies (See Figure A):
Rivastigmine Transdermal System is supplied in cartons containing 30 patches (See Figure A)
- Rivastigmine Transdermal System is a thin, transparent, plastic patch that sticks to the skin. Each Rivastigmine Transdermal System is sealed in a pouch that protects it until you are ready to put it on (See Figure A).
- Only 1 Rivastigmine Transdermal System should be worn at a time. Do not apply more than 1 Rivastigmine Transdermal System at a time to the body.
- Do not open the pouch or remove the Rivastigmine Transdermal System until you are ready to apply it.
Using Rivastigmine Transdermal System:
Step 1. Choose an area to apply the Rivastigmine Transdermal System (See Figure B).
- Instructions for Caregivers: Apply Rivastigmine Transdermal System to the upper or lower back if it is likely that the patient will remove it. If this is not a concern, the Rivastigmine Transdermal System can be applied instead to the upper arm or chest. Do not apply the Rivastigmine Transdermal System to areas where it can be rubbed off by tight clothing or belts.
- Only apply the Rivastigmine Transdermal System to healthy skin that is clean, dry, hairless, and free of redness, irritation, burns or cuts.
The diagram represents areas on the body where Rivastigmine Transdermal System may be applied.
Only 1 patch should be worn at a time. Do not apply multiple patches to the body.
Step 2. Remove the Rivastigmine Transdermal System from the pouch (See Figure C).
Carefully cut the pouch along the dotted line to open and remove the Rivastigmine Transdermal System. Save the pouch for later use.
- Do not cut or fold the Rivastigmine Transdermal System itself.
Step 3. Remove 1 side of the adhesive liner (See Figure D).
- A protective liner covers the sticky (adhesive) side of the Rivastigmine Transdermal System. Peel off 1 side of the protective cover. Do not touch the sticky part of the Rivastigmine Transdermal System with your fingers.
Step 4. Apply the Rivastigmine Transdermal System to your skin (See Figure E).
- Apply the sticky (adhesive) side of the Rivastigmine Transdermal System to your chosen area of skin and then peel off the other side of the protective cover.
- Press down on the Rivastigmine Transdermal System firmly for 30 seconds to make sure that the edges stick to your skin (See Figure F).
Step 5. Wash your hands with soap and water right away.
Note:
- If your Rivastigmine Transdermal System falls off, select a new area, and repeat Steps 2 to 5 to apply a new Rivastigmine Transdermal System.
- Be sure to replace the new Rivastigmine Transdermal System the next day at the same time as usual.
Removing your Rivastigmine Transdermal System:
Step 6. Remove the Rivastigmine Transdermal System from the skin (See Figure G).
- Gently pull on 1 edge of the Rivastigmine Transdermal System to remove it from your skin.
Throwing away the used Rivastigmine Transdermal System:
Step 7. Throw away the used Rivastigmine Transdermal System (See Figure H).
- Fold the used Rivastigmine Transdermal System in half (with the sticky sides together) and put it back into the pouch that you saved.
- Throw away the used Rivastigmine Transdermal System safely and out of the reach of children and pets.
- Some medicine stays in the patch for 24 hours after you use it and should be folded together (sticky side together) and safely thrown away. Do not try to re-use Rivastigmine Transdermal Systems.
Step 8. Wash your hands with soap and water right away.
- After you remove the Rivastigmine Transdermal System, if any adhesive remains on your skin, you can use soap and water or an oil-based substance (such as baby oil) to remove the adhesive. Alcohol or other dissolving liquids (such as nail polish remover) should not be used.
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Distributed by:
Breckenridge Pharmaceutical, Inc.
Berkeley Heights, NJ 07922
Manufactured by:
AdhexPharma
21300 Chenôve
France
Revised: 12/2023
12.2 Pharmacodynamics
After a 6-mg oral dose of rivastigmine in humans, anticholinesterase activity is present in cerebrospinal fluid for about 10 hours, with a maximum inhibition of about 60% 5 hours after dosing.
In vitro and in vivo studies demonstrate that the inhibition of cholinesterase by rivastigmine is not affected by the concomitant administration of memantine, an N-methyl-D-aspartate receptor antagonist.
8.6 Hepatic Impairment
Increased exposure to rivastigmine was observed in patients with mild or moderate hepatic impairment with oral rivastigmine. Patients with mild or moderate hepatic impairment may be able to only tolerate lower doses [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)]. No data are available on the use of rivastigmine in patients with severe hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
1.1 Alzheimer's Disease
Rivastigmine Transdermal System is indicated for the treatment of dementia of the Alzheimer's type (AD). Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
12.1 Mechanism of Action
Although the precise mechanism of action of rivastigmine is unknown, it is thought to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by cholinesterase. The effect of rivastigmine may lessen as the disease process advances and fewer cholinergic neurons remain functionally intact. There is no evidence that rivastigmine alters the course of the underlying dementing process.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hospitalization and, rarely, death have been reported due to application of multiple patches at same time. Ensure patients or caregivers receive instruction on proper dosing and administration. (5.1)
- Gastrointestinal Adverse Reactions: May include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. (5.2)
- Application site reactions may occur with the patch form of rivastigmine. Discontinue treatment if application site reactions spread beyond the patch size, if there is evidence of a more intense local reaction (e.g., increasing erythema, edema, papules, vesicles), and if symptoms do not significantly improve within 48 hours after patch removal. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Apply patch on intact skin for a 24-hour period; replace with a new patch every 24 hours. (2.1, 2.4)
- Initial Dose: Initiate treatment with 4.6 mg/24 hours Rivastigmine Transdermal System. (2.1)
- Dose Titration (2.1): After a minimum of 4 weeks, if tolerated, increase dose to 9.5 mg/24 hours, which is the minimum effective dose. Following a minimum additional 4 weeks, may increase dosage to maximum dosage of 13.3 mg/24 hours.
- Mild-to-Moderate Alzheimer's Disease and Parkinson's Disease Dementia: Rivastigmine Transdermal System 9.5 mg/24 hours or 13.3 mg/24 hours once daily. (2.1)
- Severe Alzheimer's Disease: Rivastigmine Transdermal System 13.3 mg/24 hours once daily. (2.1)
- For treatment interruption longer than 3 days, retitrate dosage starting at 4.6 mg per 24 hours. (2.1)
- Consider dose adjustments in patients with (2.2):
8.7 Low Or High Body Weight (8.7 Low or High Body Weight)
Because rivastigmine blood levels vary with weight, careful titration and monitoring should be performed in patients with low or high body weights [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Rivastigmine Transdermal System is available in 3 strengths. Each patch has a transparent backing layer labeled as either:
- Rivastigmine 4.6 mg/24 hr.
- Rivastigmine 9.5 mg/24 hr.
- Rivastigmine 13.3 mg/24 hr.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of rivastigmine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: Tachycardia
Hepatobiliary Disorders: Abnormal liver function tests, hepatitis
Nervous System Disorders: Parkinson's disease (worsening), seizure, tremor
Psychiatric Disorders: nightmares
Skin and Subcutaneous Tissue Disorders: Allergic dermatitis, application site hypersensitivity, blister, disseminated allergic dermatitis, Stevens-Johnson syndrome, urticaria
Vascular Disorders: Hypertension
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Rivastigmine Transdermal System has been administered to 4516 patients with Alzheimer's disease during clinical trials worldwide. Of these, 3005 patients have been treated for at least 26 weeks, 1771 patients have been treated for at least 52 weeks, 974 patients have been treated for at least 78 weeks, and 24 patients have been treated for at least 104 weeks.
1.2 Parkinson's Disease Dementia
Rivastigmine Transdermal System is indicated for the treatment of mild-to-moderate dementia associated with Parkinson's disease (PDD).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.2 Gastrointestinal Adverse Reactions
Rivastigmine transdermal system can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1)]. For this reason, initiate treatment with Rivastigmine Transdermal System at a dose of 4.6 mg/24 hours and titrate to a dose of 9.5 mg/24 hours and then to a dose of 13.3 mg/24 hours, if appropriate [see Dosage and Administration (2.1)].
If treatment is interrupted for more than 3 days because of intolerance, reinitiate Rivastigmine Transdermal System with the 4.6 mg/24 hours dose to reduce the possibility of severe vomiting and its potentially serious sequelae. A postmarketing report described a case of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment of an oral formulation of rivastigmine without retitration after 8 weeks of treatment interruption.
Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
2.4 Important Administration Instructions
Rivastigmine Transdermal System is for transdermal use on intact skin.
-
(a)Do not use the patch if the pouch seal is broken or the patch is cut, damaged, or changed in any way.
-
(b)Apply the Rivastigmine Transdermal System once a day
- Press down firmly for 30 seconds until the edges stick well when applying to clean, dry, hairless, intact healthy skin in a place that will not be rubbed against by tight clothing.
- Use the upper or lower back as the site of application because the patch is less likely to be removed by the patient. If sites on the back are not accessible, apply the patch to the upper arm or chest.
- Do not apply to a skin area where cream, lotion, or powder has recently been applied.
-
(c)Do not apply to skin that is red, irritated, or cut.
-
(d)Replace the Rivastigmine Transdermal System with a new patch every 24 hours. Instruct patients to only wear 1 patch at a time (remove the previous day's patch before applying a new patch) [see Warnings and Precautions (5.1) and Overdosage (10)]. If a patch falls off or if a dose is missed, apply a new patch immediately and then replace this patch the following day at the usual application time.
-
(e)Change the site of patch application daily to minimize potential irritation, although a new patch can be applied to the same general anatomic site (e.g., another spot on the upper back) on consecutive days. Do not apply a new patch to the same location for at least 14 days.
-
(f)May wear the patch during bathing and in hot weather. But avoid long exposure to external heat sources (excessive sunlight, saunas, solariums).
-
(g)Place used patches in the previously saved pouch and discard in the trash, away from pets or children.
-
(h)Wash hands with soap and water after removing the patch. In case of contact with eyes or if the eyes become red after handling the patch, rinse immediately with plenty of water and seek medical advice if symptoms do not resolve.
5.1 Medication Errors Resulting in Overdose
Medication errors with Rivastigmine Transdermal System have resulted in serious adverse reactions; some cases have required hospitalization, and rarely, led to death. The majority of medication errors have involved not removing the old patch when putting on a new one and the use of multiple patches at one time. Instruct patients and their caregivers on important administration instructions for Rivastigmine Transdermal System [see Dosage and Administration (2.4)].
5.5 Impairment in Driving Or Use of Machinery (5.5 Impairment in Driving or Use of Machinery)
Dementia may cause gradual impairment of driving performance or compromise the ability to use machinery. The administration of rivastigmine may also result in adverse reactions that are detrimental to these functions. During treatment with the Rivastigmine Transdermal System, routinely evaluate the patient's ability to continue driving or operating machinery.
7.2 Cholinomimetic and Anticholinergic Medications
Rivastigmine Transdermal System may increase the cholinergic effects of other cholinomimetic medications and may also interfere with the activity of anticholinergic medications (e.g., oxybutynin, tolterodine). Concomitant use of Rivastigmine Transdermal System with medications having these pharmacologic effects is not recommended unless deemed clinically necessary [see Warnings and Precautions (5.4)].
Principal Display Panel 4.6 Mg Patch Pouch Carton (PRINCIPAL DISPLAY PANEL - 4.6 mg Patch Pouch Carton)
NDC 51991-897-30
Rivastigmine Transdermal
System
Each System Delivers 4.6 mg/24 hours
For Transdermal Use Only
breckenridge
A Towa
Company
Rx Only
Contains 30 Systems
Principal Display Panel 9.5 Mg Patch Pouch Carton (PRINCIPAL DISPLAY PANEL - 9.5 mg Patch Pouch Carton)
NDC 51991-898-30
Rivastigmine Transdermal
System
Each System Delivers 9.5 mg/24 hours
For Transdermal Use Only
breckenridge
A Towa
Company
Rx Only
Contains 30 Systems
Principal Display Panel 13.3 Mg Patch Pouch Carton (PRINCIPAL DISPLAY PANEL - 13.3 mg Patch Pouch Carton)
NDC 51991-899-30
Rivastigmine Transdermal
System
Each System Delivers 13.3 mg/24 hours
For Transdermal Use Only
breckenridge
A Towa
Company
Rx Only
Contains 30 Systems
2.3 Switching to Rivastigmine Transdermal System From Rivastigmine Capsules Or Rivastigmine Oral Solution (2.3 Switching to Rivastigmine Transdermal System from Rivastigmine Capsules or Rivastigmine Oral Solution)
Patients treated with Rivastigmine capsules or oral solution may be switched to Rivastigmine Transdermal System as follows:
- A patient who is on a total daily dose of less than 6 mg of oral rivastigmine can be switched to the 4.6 mg/24 hours Rivastigmine Transdermal System.
- A patient who is on a total daily dose of 6 mg to 12 mg of oral rivastigmine can be switched to the 9.5 mg/24 hours Rivastigmine Transdermal System.
Instruct patients or caregivers to apply the first patch on the day following the last oral dose.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:01.478446 · Updated: 2026-03-14T22:21:43.238921