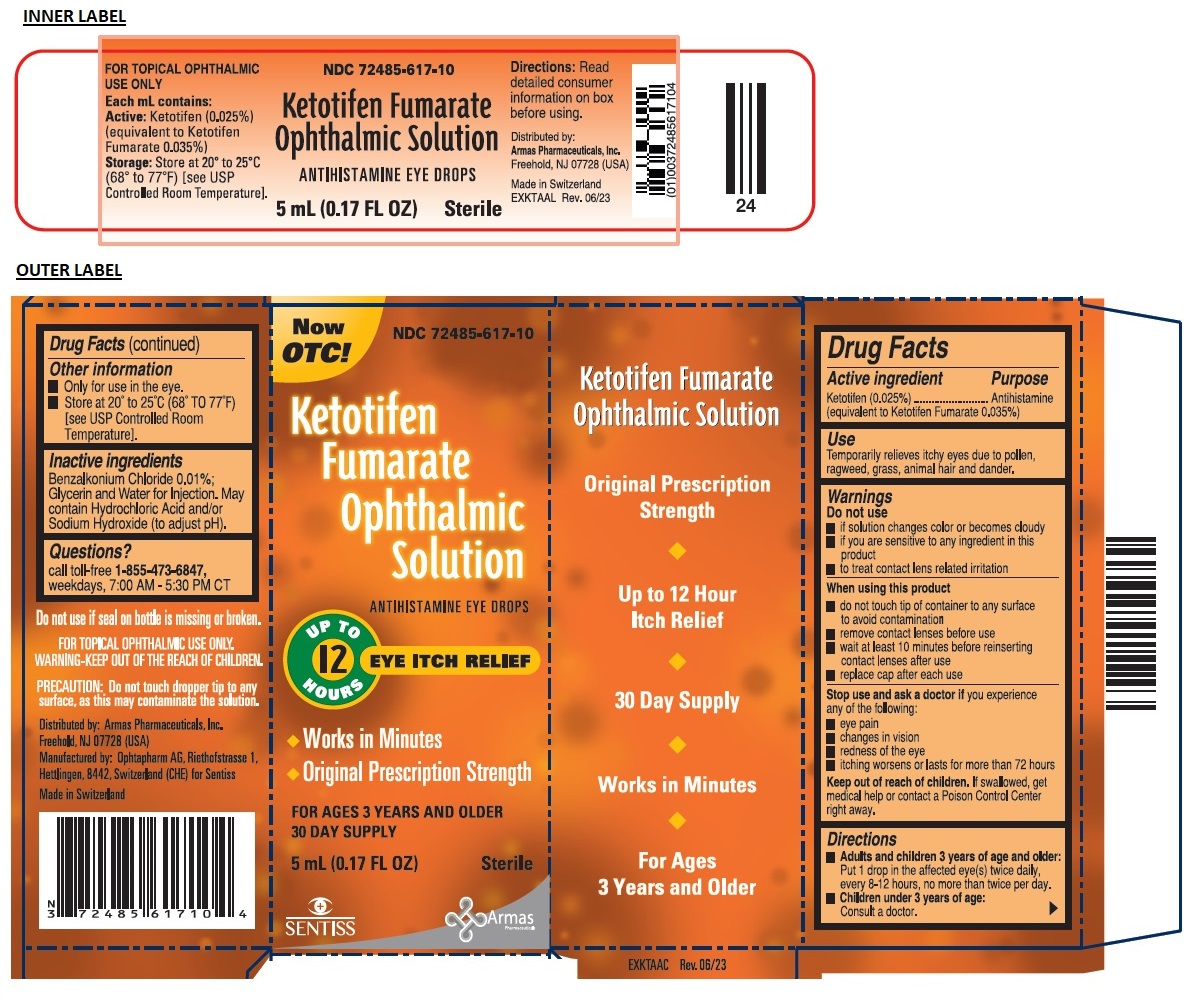
ketotifen fumarate ophthalmic solution
b1ea15a5-5354-4086-a22d-cc1b3e0e1eb3
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antihistamine
Description
Drug Facts
Medication Information
Warnings
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
- to treat contact lens related irritation
When using this product
- do not touch tip of container to any surface to avoid contamination
- remove contact lenses before use
- wait at least 10 minutes before reinserting contact lenses after use
- replace cap after each use
Stop use and ask a doctor if you experience any of the following:
- eye pain
- changes in vision
- redness of the eye
- itching worsens or lasts for more than 72 hours
Purpose
Antihistamine
Directions
- Adults and children 3 years of age and older: Put 1 drop in the affected eye(s) twice daily, every 8-12 hours, no more than twice per day.
- Children under 3 years of age: Consult a doctor.
Other Information
- Only for use in the eye.
- Store at 20° to 25°C (68° TO 77°F) [see USP Controlled Room Temperature].
Inactive Ingredients
Benzalkonium Chloride 0.01%; Glycerin and Water for Injection. May contain Hydrochloric Acid and/or Sodium Hydroxide (to adjust pH).
Description
Drug Facts
Questions?
call toll-free 1-855-473-6847,weekdays, 7:00 AM - 5:30 PM CT
Use
Temporarily relieves itchy eyes due to pollen, ragweed, grass, animal hair and dander.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Packaging
Active Ingredient
Ketotifen (0.025%)
(equivalent to Ketotifen Fumarate 0.035%)
Structured Label Content
Warnings
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
- to treat contact lens related irritation
When using this product
- do not touch tip of container to any surface to avoid contamination
- remove contact lenses before use
- wait at least 10 minutes before reinserting contact lenses after use
- replace cap after each use
Stop use and ask a doctor if you experience any of the following:
- eye pain
- changes in vision
- redness of the eye
- itching worsens or lasts for more than 72 hours
Purpose
Antihistamine
Directions
- Adults and children 3 years of age and older: Put 1 drop in the affected eye(s) twice daily, every 8-12 hours, no more than twice per day.
- Children under 3 years of age: Consult a doctor.
Other Information (Other information)
- Only for use in the eye.
- Store at 20° to 25°C (68° TO 77°F) [see USP Controlled Room Temperature].
Inactive Ingredients (Inactive ingredients)
Benzalkonium Chloride 0.01%; Glycerin and Water for Injection. May contain Hydrochloric Acid and/or Sodium Hydroxide (to adjust pH).
Questions?
call toll-free 1-855-473-6847,weekdays, 7:00 AM - 5:30 PM CT
Use
Temporarily relieves itchy eyes due to pollen, ragweed, grass, animal hair and dander.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Packaging
Active Ingredient (Active ingredient)
Ketotifen (0.025%)
(equivalent to Ketotifen Fumarate 0.035%)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:38.096295 · Updated: 2026-03-14T23:02:18.218337