Miclara Lq
b1e0f143-e192-4bab-9163-ae21c765b765
34390-5
HUMAN OTC DRUG LABEL
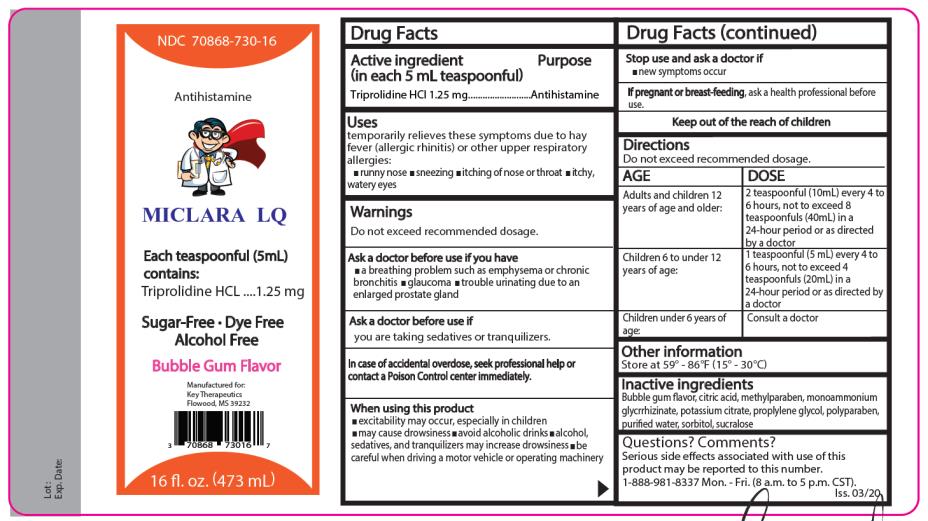
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Miclara LQ- t riprolidine hydrochloride liquid Key Therapeutics Disclaimer: Most OTC drugs are not reviewed and approved by FDA; however, they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ----------
Purpose
Antihistamine
Medication Information
Purpose
Antihistamine
Description
Miclara LQ- t riprolidine hydrochloride liquid Key Therapeutics Disclaimer: Most OTC drugs are not reviewed and approved by FDA; however, they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ----------
Uses
temporarily relieves these symptoms due to hay fever (allergic rhinitis) or other upper respiratory allergies:
- runny nose
- sneezing
- itching of the nose or throat itchy,
- watery eyes
Section 42229-5
Miclara LQ- t riprolidine hydrochloride liquid
Key Therapeutics
Disclaimer: Most OTC drugs are not reviewed and approved by FDA; however, they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Miclara
Drug Facts
Warnings
Do not exceed recommended dosage.
Do not use this product if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Directions
Do not exceed recommended dosage.
Adults and children
12 years of age and older:
Children 6 to under 12 years of age:
2 teaspoonfuls (10 mL) every 4 to 6 hours, not to exceed
8 teaspoonfuls (40mL) in 24-hour
period or as directed by a doctor.
1 teaspoonful (5 mL) every 4 to 6 hours, not to exceed 4 teaspoonfuls (20mL) in a 24-hour period or as directed by a doctor.
Children under 6 years of age: Consult a doctor
Other Information
Store at 59° - 86°F (15° - 30°C)
Active Ingredients
(in each 5 mL teaspoonful) Triprolidine HCl 1.25 mg
Inactive Ingredients
Bubble gum flavor, citric acid, methylparaben, monoammonium glycyrrhizinate, potassium citrate, propylene glycol, propylparaben, purified water, sorbitol, sucralose.
Questions? Comments?
Serious side effects associated with use of this product May be reported to this number. Call 1-888-981-8337
Mon - Fri (8 a.m. to 5 p.m. CST)
Ask A Doctor Before Use
if you are taking sedatives or tranquilizers.
Principal Display Panel
NDC 70868 - 7 3 0 -16 Miclara LQ
Antihistamine
Each 5 mL (1 teaspoonful) contains: Triprolidine HCl 1.25 mg………Antihistamine
Bubble Gum Flavor
Dye Free - Sugar Free - Alcohol Free
16 fl oz. (473 mL)
Distributed by:
Key Therapeutics, LLC
Flowood, MS 39232
Iss. 03/20
When Using This Product
- may cause excitability especially in children
- may cause drowsiness
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase the drowsiness effect
- be careful when driving a motor vehicle or operating machinery
Stop Use and Ask A Doctor If
- new symptoms occur
If pregnant or breast-feeding, ask a health professional before use.
Keep Out of the Reach of Children.
In case of accidental overdose seek professional help or contact a Poison Control Center immediately.
Structured Label Content
Uses
temporarily relieves these symptoms due to hay fever (allergic rhinitis) or other upper respiratory allergies:
- runny nose
- sneezing
- itching of the nose or throat itchy,
- watery eyes
Section 42229-5 (42229-5)
Miclara LQ- t riprolidine hydrochloride liquid
Key Therapeutics
Disclaimer: Most OTC drugs are not reviewed and approved by FDA; however, they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Miclara (MICLARA)
Drug Facts
Purpose
Antihistamine
Warnings
Do not exceed recommended dosage.
Do not use this product if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Directions
Do not exceed recommended dosage.
Adults and children
12 years of age and older:
Children 6 to under 12 years of age:
2 teaspoonfuls (10 mL) every 4 to 6 hours, not to exceed
8 teaspoonfuls (40mL) in 24-hour
period or as directed by a doctor.
1 teaspoonful (5 mL) every 4 to 6 hours, not to exceed 4 teaspoonfuls (20mL) in a 24-hour period or as directed by a doctor.
Children under 6 years of age: Consult a doctor
Other Information (Other information)
Store at 59° - 86°F (15° - 30°C)
Active Ingredients (Active ingredients)
(in each 5 mL teaspoonful) Triprolidine HCl 1.25 mg
Inactive Ingredients (Inactive ingredients)
Bubble gum flavor, citric acid, methylparaben, monoammonium glycyrrhizinate, potassium citrate, propylene glycol, propylparaben, purified water, sorbitol, sucralose.
Questions? Comments?
Serious side effects associated with use of this product May be reported to this number. Call 1-888-981-8337
Mon - Fri (8 a.m. to 5 p.m. CST)
Ask A Doctor Before Use (Ask a doctor before use)
if you are taking sedatives or tranquilizers.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 70868 - 7 3 0 -16 Miclara LQ
Antihistamine
Each 5 mL (1 teaspoonful) contains: Triprolidine HCl 1.25 mg………Antihistamine
Bubble Gum Flavor
Dye Free - Sugar Free - Alcohol Free
16 fl oz. (473 mL)
Distributed by:
Key Therapeutics, LLC
Flowood, MS 39232
Iss. 03/20
When Using This Product (When using this product)
- may cause excitability especially in children
- may cause drowsiness
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase the drowsiness effect
- be careful when driving a motor vehicle or operating machinery
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- new symptoms occur
If pregnant or breast-feeding, ask a health professional before use.
Keep Out of the Reach of Children. (Keep out of the reach of children.)
In case of accidental overdose seek professional help or contact a Poison Control Center immediately.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:05.426461 · Updated: 2026-03-14T23:00:25.149305