b0a2a1f7-354e-48bd-9e98-9207bf9f53fd
34390-5
HUMAN OTC DRUG LABEL
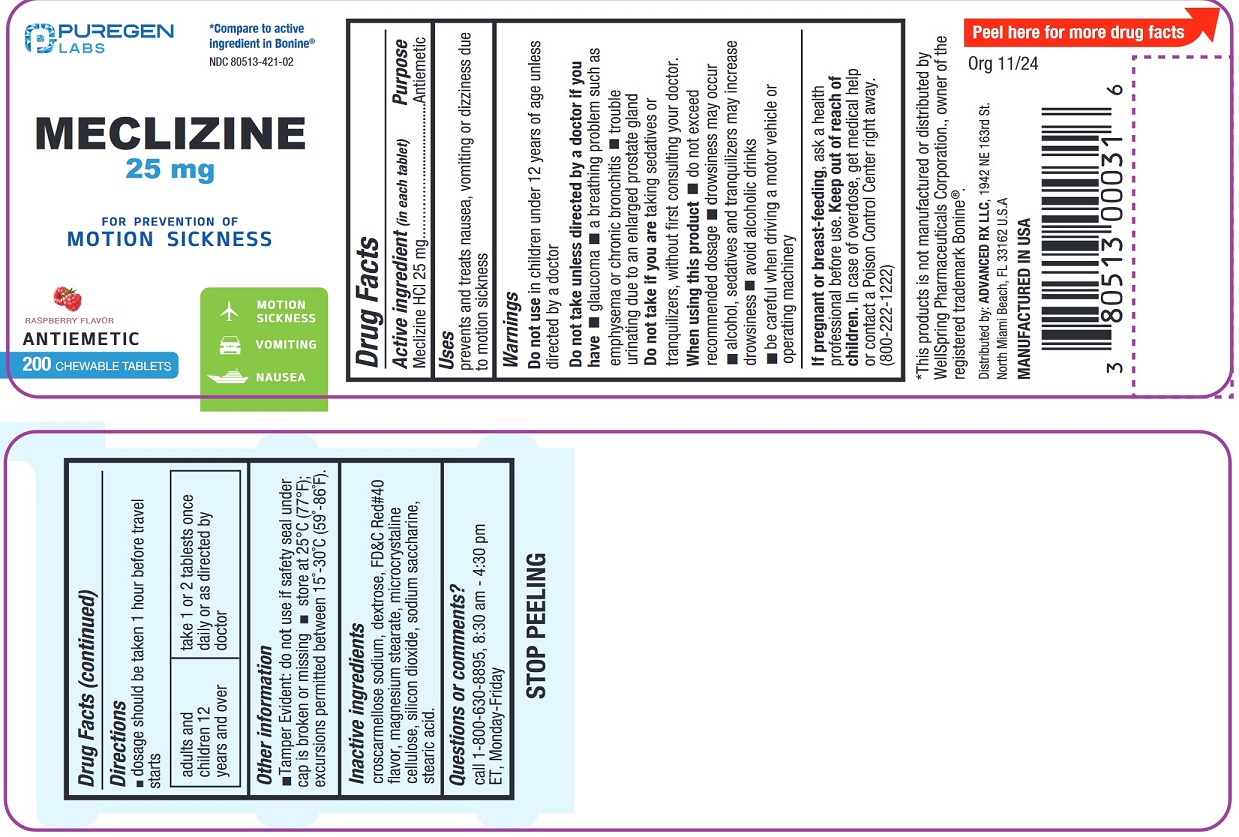
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Meclizine HCl 25 mg
Purpose
Antiemetic
Medication Information
Purpose
Antiemetic
Description
Meclizine HCl 25 mg
Uses
prevents and treats nausea, vomiting or dizziness associated with motion sickness
Section 51945-4
NDC 80513-421-02
*Compare to the active ingredient in Bonine ®
Meclizine HCl 25 mg
Raspberry Flavor
Antiemetic
• Motion Sickness
• Vomiting
• Nausea
200 CHEWABLE TABLETS
*This product is not manufactured or distributed by WellSpring Pharmaceuticals Corporation., owner of the registered trademark Bonine ®
Distributed by:
ADVANCED RX LLC,
1942 NE 163rd St
North Miami Beach, FL 33162 U.S.A.
Manufactured in the USA
Warnings
Do not use in children under 12 years of age unless directed by a doctor.
Do not take unless directed by a doctor if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Do not take this product if you aretaking sedatives or tranquilizers, without first consulting your doctor.
Directions
- doage should be taken 1 hour before travel starts
| adults and children 12 years and over |
take 1 or 2 tablets once daily or as directed by doctor |
Other Information
- Tamper Evident:do not use if safety seal under cap is broken or missing
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F).
Inactive Ingredients
croscarmellose sodium, dextrose, FD&C Red#40, flavor, magnesium stearate, microcrystalline cellulose, silicon dioxide, sodium saccharine, stearic acid.
Questions Or Comments?
call 1-800-630-8895, 8:30 am - 4.30 pm ET, Monday-Friday
When Using This Product
- do not exceed recommended dosage
- drowsiness may occures
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- be careful when driving a motor vehicle or operating machinery
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222)
Active Ingredient (in Each Tablet)
Meclizine HCl 25 mg
Structured Label Content
Uses
prevents and treats nausea, vomiting or dizziness associated with motion sickness
Section 51945-4 (51945-4)
NDC 80513-421-02
*Compare to the active ingredient in Bonine ®
Meclizine HCl 25 mg
Raspberry Flavor
Antiemetic
• Motion Sickness
• Vomiting
• Nausea
200 CHEWABLE TABLETS
*This product is not manufactured or distributed by WellSpring Pharmaceuticals Corporation., owner of the registered trademark Bonine ®
Distributed by:
ADVANCED RX LLC,
1942 NE 163rd St
North Miami Beach, FL 33162 U.S.A.
Manufactured in the USA
Purpose
Antiemetic
Warnings
Do not use in children under 12 years of age unless directed by a doctor.
Do not take unless directed by a doctor if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Do not take this product if you aretaking sedatives or tranquilizers, without first consulting your doctor.
Directions
- doage should be taken 1 hour before travel starts
| adults and children 12 years and over |
take 1 or 2 tablets once daily or as directed by doctor |
Other Information (Other information)
- Tamper Evident:do not use if safety seal under cap is broken or missing
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F).
Inactive Ingredients (Inactive ingredients)
croscarmellose sodium, dextrose, FD&C Red#40, flavor, magnesium stearate, microcrystalline cellulose, silicon dioxide, sodium saccharine, stearic acid.
Questions Or Comments? (Questions or comments?)
call 1-800-630-8895, 8:30 am - 4.30 pm ET, Monday-Friday
When Using This Product (When using this product)
- do not exceed recommended dosage
- drowsiness may occures
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- be careful when driving a motor vehicle or operating machinery
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222)
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Meclizine HCl 25 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:34.393733 · Updated: 2026-03-14T23:05:25.370894