These Highlights Do Not Include All The Information Needed To Use Noxafil Safely And Effectively. See Full Prescribing Information For Noxafil.
b073b082-7b57-4423-8c06-4fd4263d6f84
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1.1 , 1.2 ) 1/2026 Dosage and Administration ( 2 ) 1/2026
Indications and Usage
Noxafil is an azole antifungal indicated as follows: Noxafil is indicated for the treatment of invasive aspergillosis as follows: ( 1.1 ) Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg Noxafil is indicated for the prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, such as hematopoietic stem cell transplant (HSCT) recipients with graft-versus-host disease (GVHD) or those with hematologic malignancies with prolonged neutropenia from chemotherapy as follows: ( 1.2 ) Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater. Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg Noxafil oral suspension: adults and pediatric patients 13 years of age and older Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg Noxafil oral suspension is indicated for the treatment of oropharyngeal candidiasis (OPC), including OPC refractory (rOPC) to itraconazole and/or fluconazole in adults and pediatric patients 13 years of age and older ( 1.3 )
Dosage and Administration
Noxafil formulations are supplied in different dose strengths of posaconazole, are approved for different indications, age groups, and weights, have different dosages and duration of therapy; and have different preparation and administration instructions. ( 2.1 ) Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations. ( 2.1 , 2.2 , 2.3 ) Noxafil injection must be administered through an in-line filter. ( 2.6 ) Administer Noxafil injection by intravenous infusion over approximately 90 minutes via a central venous line. ( 2.1 , 2.6 ) Do NOT administer Noxafil injection as an intravenous bolus injection. ( 2.1 ) Administer Noxafil delayed-release tablets with or without food. ( 2.1 ) Administer Noxafil oral suspension with a full meal. ( 2.1 ) Administer Noxafil PowderMix for delayed-release oral suspension with food. ( 2.1 ) Administer Noxafil PowderMix for delayed-release oral suspension with the provided notched tip syringes only. ( 2.1 ) See the full prescribing information for important administration instructions and preparation instructions for Noxafil (injection, delayed-release tablets, and oral suspension) and Noxafil PowderMix delayed-release oral suspension ( 2.5 , 2.6 , 2.7 , 2.8 , 2.9 , 2.10 ) For adult and pediatric patients aged 2 years of age and older, see the Full Prescribing Information for dosing recommendations for Noxafil injection, Noxafil delayed-release tablets, Noxafil oral suspension, and/or Noxafil PowderMix for delayed-release oral suspension based on the indication, age, and weight associated with the dosage form ( 1.1 , 1.2 , 1.3 , 2.1 , 2.2 , 2.3 , 2.4 )
Warnings and Precautions
Calcineurin-Inhibitor Toxicity : Noxafil increases concentrations of cyclosporine or tacrolimus; reduce dose of cyclosporine and tacrolimus and monitor concentrations frequently. ( 5.1 ) Arrhythmias and QTc Prolongation : Noxafil has been shown to prolong the QTc interval and cause cases of TdP. Administer with caution to patients with potentially proarrhythmic conditions. Do not administer with drugs known to prolong QTc interval and metabolized through CYP3A4. ( 5.2 , 7.2 ) Electrolyte Disturbances : Monitor and correct, especially those involving potassium (K + ), magnesium (Mg ++ ), and calcium (Ca ++ ), before and during Noxafil therapy. ( 5.3 ) Pseudoaldosteronism : Manifested by the onset or worsening of hypertension, and abnormal laboratory findings. Monitor blood pressure and potassium levels, and manage as necessary. ( 5.4 ) Hepatic Toxicity : Elevations in liver tests may occur. Discontinuation should be considered in patients who develop abnormal liver tests or monitor liver tests during treatment. ( 5.5 ) Renal Impairment : Noxafil injection should be avoided in patients with moderate or severe renal impairment (eGFR less than 50 mL/min/1.73 m 2 ), unless an assessment of the benefit/risk to the patient justifies the use of Noxafil injection. ( 5.6 , 8.6 ) Concomitant Use with Midazolam : Noxafil can prolong hypnotic/sedative effects. Monitor patients and benzodiazepine receptor antagonists should be available. ( 5.7 , 7.2 ) Vincristine Toxicity : Concomitant administration of azole antifungals, including Noxafil, with vincristine has been associated with neurotoxicity and other serious adverse reactions; reserve azole antifungals, including Noxafil, for patients receiving a vinca alkaloid, including vincristine, who have no alternative antifungal treatment options. ( 5.8 , 7.2 ) Risk in Patients with Hereditary Fructose Intolerance (HFI) : Noxafil PowderMix for delayed-release oral suspension contains sorbitol. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before Noxafil PowderMix for delayed-release oral suspension administration. ( 5.9 , 8.4 ) Breakthrough Fungal Infections : Monitor patients with severe diarrhea or vomiting when receiving Noxafil delayed-release tablets, Noxafil oral suspension, and Noxafil PowderMix for delayed-release oral suspension. ( 5.10 ) Venetoclax Toxicity: Concomitant administration of Noxafil with venetoclax may increase venetoclax toxicities, including the risk of tumor lysis syndrome, neutropenia, and serious infections; monitor for toxicity and reduce venetoclax dose. ( 4.6 , 5.11 , 7.2 )
Contraindications
Known hypersensitivity to posaconazole or other azole antifungal agents. ( 4.1 ) Coadministration of Noxafil with the following drugs is contraindicated; Noxafil increases concentrations and toxicities of: Sirolimus ( 4.2 , 7.2 ) CYP3A4 substrates (pimozide, quinidine): can result in QTc interval prolongation and cases of torsades de pointes (TdP) ( 4.3 , 5.2 , 7.2 ) HMG-CoA Reductase Inhibitors Primarily Metabolized through CYP3A4 ( 4.4 , 7.2 ) Ergot alkaloids ( 4.5 , 7.2 ) Venetoclax: In patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) at initiation and during the ramp-up phase ( 4.6 , 5.11 , 7.2 ) Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with known or suspected Hereditary Fructose Intolerance (HFI). ( 4.7 , 5.9 , 8.4 )
Adverse Reactions
The following serious and otherwise important adverse reactions are discussed in detail in another section of the labeling: Arrhythmias and QT Prolongation [see Warnings and Precautions (5.2) ] Electrolyte Disturbances [see Warnings and Precautions (5.3) ] Pseudoaldosteronism [see Warnings and Precautions (5.4) ] Hepatic Toxicity [see Warnings and Precautions (5.5) ]
Drug Interactions
Table 15 and Table 17 include drugs with clinically important drug interactions when administered concomitantly with Noxafil and Noxafil PowderMix and instructions for preventing or managing them. Table 16 includes important drug interactions specific to the absorption of posaconazole administered as either Noxafil oral suspension or Noxafil PowderMix. These recommendations are based on either drug interaction studies or predicted interactions due to the expected magnitude of interaction and potential for serious adverse reactions or loss of efficacy [see Clinical Pharmacology (12.3) ] . The following information was derived from data with Noxafil oral suspension or another posaconazole tablet formulation unless otherwise noted. All clinically important drug interactions with Noxafil oral suspension, except for those that affect the absorption of posaconazole (via gastric pH and motility), are considered relevant to clinically important drug interactions with Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension [see Clinical Pharmacology (12.3) ] . Consult the labeling of concomitantly used drugs to obtain further information about interactions with posaconazole.
Storage and Handling
Noxafil Injection Store Noxafil injection vial refrigerated at 2 to 8°C (36 to 46°F). Storage conditions for the diluted Noxafil infusion solution are presented in another section of the prescribing information [see Dosage and Administration (2.5) ]. Noxafil Delayed-Release Tablets Store at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature]. Noxafil Oral Suspension Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE. Noxafil PowderMix for Delayed-Release Oral Suspension Store the entire kit at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) in a clean, dry place. Do not open foil packet containing Noxafil PowderMix for delayed-release oral suspension until ready for use. Storage conditions for the reconstituted solution are presented in another section of the prescribing information [see Dosage and Administration (2.10) ].
How Supplied
Noxafil Injection Noxafil injection is a clear, colorless to yellow sterile liquid in single-dose Type I glass vials closed with bromobutyl rubber stopper and aluminum seal containing 300 mg of posaconazole in 16.7 mL of solution (18 mg of posaconazole per mL) (NDC 0085-4331-01). Noxafil Delayed-Release Tablets Noxafil delayed-release tablets are yellow, coated, oblong, debossed with "100" on one side containing 100 mg of posaconazole. Bottles with child-resistant closures of 60 delayed-release tablets (NDC 0085-4324-02). Noxafil Oral Suspension Noxafil oral suspension is a white, cherry-flavored oral suspension in 4-ounce (123 mL) amber glass bottles with child-resistant closures containing 105 mL of suspension (40 mg of posaconazole per mL).Supplied with each oral suspension bottle is a plastic dosing spoon calibrated for measuring 2.5 mL and 5 mL doses (NDC 0085-1328-01). Noxafil PowderMix for Delayed-Release Oral Suspension Noxafil PowderMix for delayed-release oral suspension is supplied as: Package A: a kit with 8 child-resistant single-use packets of Noxafil PowderMix for delayed-release oral suspension 300 mg, two 3 mL (green) notched tip syringes, two 10 mL (blue) notched tip syringes, two mixing cups, one mixing liquid bottle, one bottle adapter for the mixing liquid bottle and Instructions for Use. Package B: a box of six 3 mL (green) and six 10 mL (blue) notched tip syringes for additional supply. Packages A and B are supplied separately. NDC 0085-2224-02 unit of use carton with 8 packets. NDC 0085-2224-01 individual packet.
Medication Information
Warnings and Precautions
Calcineurin-Inhibitor Toxicity : Noxafil increases concentrations of cyclosporine or tacrolimus; reduce dose of cyclosporine and tacrolimus and monitor concentrations frequently. ( 5.1 ) Arrhythmias and QTc Prolongation : Noxafil has been shown to prolong the QTc interval and cause cases of TdP. Administer with caution to patients with potentially proarrhythmic conditions. Do not administer with drugs known to prolong QTc interval and metabolized through CYP3A4. ( 5.2 , 7.2 ) Electrolyte Disturbances : Monitor and correct, especially those involving potassium (K + ), magnesium (Mg ++ ), and calcium (Ca ++ ), before and during Noxafil therapy. ( 5.3 ) Pseudoaldosteronism : Manifested by the onset or worsening of hypertension, and abnormal laboratory findings. Monitor blood pressure and potassium levels, and manage as necessary. ( 5.4 ) Hepatic Toxicity : Elevations in liver tests may occur. Discontinuation should be considered in patients who develop abnormal liver tests or monitor liver tests during treatment. ( 5.5 ) Renal Impairment : Noxafil injection should be avoided in patients with moderate or severe renal impairment (eGFR less than 50 mL/min/1.73 m 2 ), unless an assessment of the benefit/risk to the patient justifies the use of Noxafil injection. ( 5.6 , 8.6 ) Concomitant Use with Midazolam : Noxafil can prolong hypnotic/sedative effects. Monitor patients and benzodiazepine receptor antagonists should be available. ( 5.7 , 7.2 ) Vincristine Toxicity : Concomitant administration of azole antifungals, including Noxafil, with vincristine has been associated with neurotoxicity and other serious adverse reactions; reserve azole antifungals, including Noxafil, for patients receiving a vinca alkaloid, including vincristine, who have no alternative antifungal treatment options. ( 5.8 , 7.2 ) Risk in Patients with Hereditary Fructose Intolerance (HFI) : Noxafil PowderMix for delayed-release oral suspension contains sorbitol. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before Noxafil PowderMix for delayed-release oral suspension administration. ( 5.9 , 8.4 ) Breakthrough Fungal Infections : Monitor patients with severe diarrhea or vomiting when receiving Noxafil delayed-release tablets, Noxafil oral suspension, and Noxafil PowderMix for delayed-release oral suspension. ( 5.10 ) Venetoclax Toxicity: Concomitant administration of Noxafil with venetoclax may increase venetoclax toxicities, including the risk of tumor lysis syndrome, neutropenia, and serious infections; monitor for toxicity and reduce venetoclax dose. ( 4.6 , 5.11 , 7.2 )
Indications and Usage
Noxafil is an azole antifungal indicated as follows: Noxafil is indicated for the treatment of invasive aspergillosis as follows: ( 1.1 ) Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg Noxafil is indicated for the prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, such as hematopoietic stem cell transplant (HSCT) recipients with graft-versus-host disease (GVHD) or those with hematologic malignancies with prolonged neutropenia from chemotherapy as follows: ( 1.2 ) Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater. Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg Noxafil oral suspension: adults and pediatric patients 13 years of age and older Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg Noxafil oral suspension is indicated for the treatment of oropharyngeal candidiasis (OPC), including OPC refractory (rOPC) to itraconazole and/or fluconazole in adults and pediatric patients 13 years of age and older ( 1.3 )
Dosage and Administration
Noxafil formulations are supplied in different dose strengths of posaconazole, are approved for different indications, age groups, and weights, have different dosages and duration of therapy; and have different preparation and administration instructions. ( 2.1 ) Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations. ( 2.1 , 2.2 , 2.3 ) Noxafil injection must be administered through an in-line filter. ( 2.6 ) Administer Noxafil injection by intravenous infusion over approximately 90 minutes via a central venous line. ( 2.1 , 2.6 ) Do NOT administer Noxafil injection as an intravenous bolus injection. ( 2.1 ) Administer Noxafil delayed-release tablets with or without food. ( 2.1 ) Administer Noxafil oral suspension with a full meal. ( 2.1 ) Administer Noxafil PowderMix for delayed-release oral suspension with food. ( 2.1 ) Administer Noxafil PowderMix for delayed-release oral suspension with the provided notched tip syringes only. ( 2.1 ) See the full prescribing information for important administration instructions and preparation instructions for Noxafil (injection, delayed-release tablets, and oral suspension) and Noxafil PowderMix delayed-release oral suspension ( 2.5 , 2.6 , 2.7 , 2.8 , 2.9 , 2.10 ) For adult and pediatric patients aged 2 years of age and older, see the Full Prescribing Information for dosing recommendations for Noxafil injection, Noxafil delayed-release tablets, Noxafil oral suspension, and/or Noxafil PowderMix for delayed-release oral suspension based on the indication, age, and weight associated with the dosage form ( 1.1 , 1.2 , 1.3 , 2.1 , 2.2 , 2.3 , 2.4 )
Contraindications
Known hypersensitivity to posaconazole or other azole antifungal agents. ( 4.1 ) Coadministration of Noxafil with the following drugs is contraindicated; Noxafil increases concentrations and toxicities of: Sirolimus ( 4.2 , 7.2 ) CYP3A4 substrates (pimozide, quinidine): can result in QTc interval prolongation and cases of torsades de pointes (TdP) ( 4.3 , 5.2 , 7.2 ) HMG-CoA Reductase Inhibitors Primarily Metabolized through CYP3A4 ( 4.4 , 7.2 ) Ergot alkaloids ( 4.5 , 7.2 ) Venetoclax: In patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) at initiation and during the ramp-up phase ( 4.6 , 5.11 , 7.2 ) Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with known or suspected Hereditary Fructose Intolerance (HFI). ( 4.7 , 5.9 , 8.4 )
Adverse Reactions
The following serious and otherwise important adverse reactions are discussed in detail in another section of the labeling: Arrhythmias and QT Prolongation [see Warnings and Precautions (5.2) ] Electrolyte Disturbances [see Warnings and Precautions (5.3) ] Pseudoaldosteronism [see Warnings and Precautions (5.4) ] Hepatic Toxicity [see Warnings and Precautions (5.5) ]
Drug Interactions
Table 15 and Table 17 include drugs with clinically important drug interactions when administered concomitantly with Noxafil and Noxafil PowderMix and instructions for preventing or managing them. Table 16 includes important drug interactions specific to the absorption of posaconazole administered as either Noxafil oral suspension or Noxafil PowderMix. These recommendations are based on either drug interaction studies or predicted interactions due to the expected magnitude of interaction and potential for serious adverse reactions or loss of efficacy [see Clinical Pharmacology (12.3) ] . The following information was derived from data with Noxafil oral suspension or another posaconazole tablet formulation unless otherwise noted. All clinically important drug interactions with Noxafil oral suspension, except for those that affect the absorption of posaconazole (via gastric pH and motility), are considered relevant to clinically important drug interactions with Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension [see Clinical Pharmacology (12.3) ] . Consult the labeling of concomitantly used drugs to obtain further information about interactions with posaconazole.
Storage and Handling
Noxafil Injection Store Noxafil injection vial refrigerated at 2 to 8°C (36 to 46°F). Storage conditions for the diluted Noxafil infusion solution are presented in another section of the prescribing information [see Dosage and Administration (2.5) ]. Noxafil Delayed-Release Tablets Store at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature]. Noxafil Oral Suspension Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE. Noxafil PowderMix for Delayed-Release Oral Suspension Store the entire kit at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) in a clean, dry place. Do not open foil packet containing Noxafil PowderMix for delayed-release oral suspension until ready for use. Storage conditions for the reconstituted solution are presented in another section of the prescribing information [see Dosage and Administration (2.10) ].
How Supplied
Noxafil Injection Noxafil injection is a clear, colorless to yellow sterile liquid in single-dose Type I glass vials closed with bromobutyl rubber stopper and aluminum seal containing 300 mg of posaconazole in 16.7 mL of solution (18 mg of posaconazole per mL) (NDC 0085-4331-01). Noxafil Delayed-Release Tablets Noxafil delayed-release tablets are yellow, coated, oblong, debossed with "100" on one side containing 100 mg of posaconazole. Bottles with child-resistant closures of 60 delayed-release tablets (NDC 0085-4324-02). Noxafil Oral Suspension Noxafil oral suspension is a white, cherry-flavored oral suspension in 4-ounce (123 mL) amber glass bottles with child-resistant closures containing 105 mL of suspension (40 mg of posaconazole per mL).Supplied with each oral suspension bottle is a plastic dosing spoon calibrated for measuring 2.5 mL and 5 mL doses (NDC 0085-1328-01). Noxafil PowderMix for Delayed-Release Oral Suspension Noxafil PowderMix for delayed-release oral suspension is supplied as: Package A: a kit with 8 child-resistant single-use packets of Noxafil PowderMix for delayed-release oral suspension 300 mg, two 3 mL (green) notched tip syringes, two 10 mL (blue) notched tip syringes, two mixing cups, one mixing liquid bottle, one bottle adapter for the mixing liquid bottle and Instructions for Use. Package B: a box of six 3 mL (green) and six 10 mL (blue) notched tip syringes for additional supply. Packages A and B are supplied separately. NDC 0085-2224-02 unit of use carton with 8 packets. NDC 0085-2224-01 individual packet.
Description
Indications and Usage ( 1.1 , 1.2 ) 1/2026 Dosage and Administration ( 2 ) 1/2026
Section 42229-5
Treatment of Invasive Aspergillosis in Adults and Adolescents (Noxafil injection and Noxafil Delayed-Release Tablets)
The safety of Noxafil injection and Noxafil delayed-release tablets was assessed in a randomized, double-blind, active-controlled clinical study of Noxafil injection and Noxafil delayed-release tablets versus voriconazole for treatment of invasive aspergillosis (Aspergillosis Treatment Study). A total of 575 adult and pediatric patients 14 years of age and older (288 in the Noxafil group, 287 in voriconazole group (voriconazole for injection or voriconazole tablets)) with proven, probable or possible invasive aspergillosis were included. The median duration of treatment was 67 days for Noxafil injection or Noxafil delayed-release tablets and 64 days for voriconazole. In this study, 55% to 60% of patients started intravenous treatment with Noxafil (Noxafil injection) or voriconazole (voriconazole for injection). The median duration of the first instance of intravenous treatment (before switching to oral treatment or discontinuing or completing study treatment) was 9 days for both groups. Table 7 presents adverse reactions reported at an incidence of ≥10% in either one of the treatment groups in the Aspergillosis Treatment Study.
Adverse reactions leading to treatment discontinuation were reported for 34% of patients. The most commonly reported adverse reactions (>2% of patients) leading to treatment discontinuation were septic shock, respiratory failure, and bronchopulmonary aspergillosis in the Noxafil group, and septic shock and acute myeloid leukemia in the voriconazole group. The most frequently reported adverse reactions in the Noxafil-treated group were pyrexia (28%), hypokalemia (28%), and nausea (23%).
| Adverse Reactions | Noxafil injection or Noxafil delayed-release tablets n=288 (%) |
Voriconazole for injection or Voriconazole tablets n=287 (%) |
|---|---|---|
| Percentage of Patients Reporting any Adverse Reaction | 97.6 | 97.6 |
| Hypokalemia | 28.5 | 17.1 |
| Pyrexia | 28.1 | 25.1 |
| Nausea | 22.6 | 17.8 |
| Diarrhea | 18.1 | 18.1 |
| Vomiting | 18.1 | 13.6 |
| Alanine aminotransferase increased | 14.6 | 12.9 |
| Febrile neutropenia | 14.6 | 13.2 |
| Aspartate aminotransferase increased | 13.2 | 12.5 |
| Pneumonia | 12.5 | 9.1 |
| Headache | 12.2 | 8.7 |
| Constipation | 11.1 | 8.0 |
| Edema peripheral | 11.1 | 8.4 |
| Epistaxis | 11.1 | 5.9 |
| Cough | 10.4 | 8.4 |
| Abdominal pain | 10.1 | 8.4 |
| Hypomagnesemia | 10.1 | 6.3 |
Clinical Trial Experience with Noxafil Injection for Prophylaxis of Invasive Aspergillus and Candida Infections
Administration of multiple doses of Noxafil injection via a peripheral venous catheter were associated with thrombophlebitis (60% incidence). Therefore, in subsequent studies, Noxafil injection was administered via central venous catheter [see Dosage and Administration (2.6)].
The safety of Noxafil injection has been assessed in 268 patients in a clinical trial. Patients were enrolled in a non-comparative pharmacokinetic and safety trial of Noxafil injection when given as antifungal prophylaxis (Noxafil Injection Study). Patients were immunocompromised with underlying conditions including hematological malignancy, neutropenia post-chemotherapy, GVHD, and post HSCT. This patient population was 55% male, had a mean age of 51 years (range: 18-82 years, 19% of patients were ≥65 years of age), and were 95% White and 8% Hispanic. In this study, 10 patients received a single dose of 200 mg Noxafil injection, 21 patients received 200 mg daily dosage for a median of 14 days, and 237 patients received 300 mg daily dosage for a median of 9 days (the 200 mg dosage is not a recommended dosage for prophylaxis of invasive Aspergillus and Candida infections in adults [see Dosage and Administration (2.2)]. In the 300 mg daily dosage group each patient received a loading intravenous dose of Noxafil injection 300 mg twice on Day 1, then intravenous Noxafil injection therapy, and finally Noxafil oral suspension to complete 28 days of total Noxafil therapy.
Table 8 presents adverse reactions observed in patients treated with the Noxafil injection 300 mg daily dosage group in the Noxafil Injection Study.
The most frequently reported adverse reactions with an onset during the intravenous Noxafil injection phase of dosing with 300 mg once daily were diarrhea (32%), hypokalemia (22%), pyrexia (21%), and nausea (19%). These adverse reactions were consistent with those seen in studies with Noxafil oral suspension.
| Adverse Reactions | Noxafil Injection Treatment Phase n=237 Adverse reactions reported in patients with an onset during the Noxafil intravenous dosing phase of the study.
(%) |
Noxafil Injection Treatment Phase or Subsequent Noxafil Oral Suspension Treatment Phase n=237 Adverse reactions reported with an onset at any time during the study in patients who were treated for up to 28 days of Noxafil therapy.
(%) |
|---|---|---|
| Percentage of Patients Reporting any Adverse Reaction | 93 | 99 |
| Diarrhea | 32 | 39 |
| Hypokalemia | 22 | 28 |
| Pyrexia | 21 | 31 |
| Nausea | 19 | 30 |
| Rash | 15 | 24 |
| Headache | 14 | 21 |
| Epistaxis | 14 | 17 |
| Abdominal Pain | 13 | 17 |
| Chills | 12 | 16 |
| Edema Peripheral | 12 | 15 |
| Vomiting | 12 | 19 |
| Hypomagnesemia | 11 | 13 |
| Decreased appetite | 10 | 12 |
| Cough | 9 | 13 |
| Constipation | 8 | 13 |
| Fatigue | 8 | 10 |
| Hypertension | 8 | 11 |
| Petechiae | 8 | 10 |
| Anemia | 7 | 10 |
| Dyspnea | 7 | 10 |
| Thrombocytopenia | 7 | 11 |
| Abdominal Pain Upper | 6 | 11 |
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 1/2026 | ||
|
Patient Information NOXAFIL ® (NOX-a-fil) (posaconazole) injection NOXAFIL ® (NOX-a-fil) (posaconazole) delayed-release tablets NOXAFIL ® (NOX-a-fil) (posaconazole) oral suspension NOXAFIL ® (NOX-a-fil) POWDERMIX (posaconazole) for delayed-release oral suspension |
||
|
What is Noxafil and Noxafil PowderMix?
Noxafil injection is used for:
Noxafil delayed-release tablets are used for:
Noxafil oral suspension is used for:
Noxafil PowderMix for delayed-release oral suspension is used for:
Noxafil oral suspension is also used to treat a fungal infection called “thrush” caused by Candida in your mouth or throat area. Noxafil oral suspension can be used as the first treatment for thrush, or as another treatment for thrush after itraconazole or fluconazole treatment has not worked. Noxafil oral suspension is for adults and children 13 years of age and older. It is not known if Noxafil oral suspension is safe and effective in children under 13 years of age for the treatment of thrush as the first treatment for thrush, or as another treatment for thrush after itraconazole or fluconazole treatment has not worked. It is not known if Noxafil or Noxafil PowderMix is safe and effective in children under 2 years of age. |
||
Do not take Noxafil or Noxafil PowderMix if you:
Do not start taking a new medicine without talking to your healthcare provider or pharmacist. |
||
Before you take Noxafil or Noxafil PowderMix, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Noxafil and Noxafil PowderMix can affect the way other medicines work, and other medicines can affect the way Noxafil and Noxafil PowderMix work, and can cause serious side effects.
Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure. |
||
|
How should I take Noxafil or Noxafil PowderMix?
Follow the instructions from your healthcare provider on how much Noxafil or Noxafil PowderMix you should take and when to take it. |
||
|
What are the possible side effects of Noxafil or Noxafil PowderMix? Noxafil or Noxafil PowderMix may cause serious side effects, including:
|
||
The most common side effects of Noxafil in adults include: |
||
|
|
|
|
The most common side effects of Noxafil injection and Noxafil PowderMix in children include: |
||
|
|
|
|
If you take Noxafil delayed-release tablets, Noxafil oral suspension, or Noxafil PowderMix for delayed-release oral suspension, tell your healthcare provider right away if you have diarrhea or vomiting. |
||
|
How should I store Noxafil or Noxafil PowderMix? Noxafil injection
Noxafil delayed-release tablets
Noxafil oral suspension
Noxafil PowderMix for delayed-release oral suspension
Safely throw away medicine that is out of date or no longer needed. Keep Noxafil and Noxafil PowderMix and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of Noxafil and Noxafil PowderMix. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Noxafil or Noxafil PowderMix for a condition for which it was not prescribed. Do not give Noxafil or Noxafil PowderMix to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Noxafil or Noxafil PowderMix that is written for health professionals. |
||
|
What are the ingredients in Noxafil and Noxafil PowderMix?
|
||
|
Manuf. for: Merck Sharp & Dohme LLC |
Section 59845-8
|
Important: Read This Booklet First
Noxafil PowderMix (posaconazole) for delayed-release oral suspension Instructions for Use for caregivers and healthcare providers of toddlers and children |
| Before You Start |
|
| Note: Put your child in a safe place. You will need both hands to prepare Noxafil PowderMix. Wash your hands with soap and water before preparing Noxafil PowderMix. |
| Before You Start |
- The amount of Noxafil PowderMix you give depends on your child’s weight. Your healthcare provider will tell you the right dose to give your child. Be sure to keep your healthcare provider’s appointments so you get new dosing information as your child grows.
- This booklet tells you how to:
- Make the Noxafil PowderMix into a liquid form.
- Measure the right dose using the included notched-tip
oral syringe. Only use the notched-tip syringes supplied
with the kit. Do not use other oral syringes. - Give the Noxafil PowderMix to your child.
- Clean up.
| Note before adding Noxafil PowderMix: Make sure you and your child are ready! If you do not use Noxafil PowderMix within 1 hour, you will need to throw it away and start over. |
| Note: If you have any questions, call your healthcare provider. |
|
|
||
|
|
||
|
|
||
|
|
| 2 blue (10 mL) syringes | 2 green (3 mL) syringes |
|
The kit has an extra cup and set of syringes in case one is lost or damaged.
Do not use any damaged cups or syringes. |
|
- Look for the number on the measuring scale that matches
the amount of mixing liquid or Noxafil PowderMix that
you need. - Be sure to follow the directions in this booklet to remove
air bubbles from the syringe. Air bubbles can affect the
amount of medicine that the child gets.
| Step 1. Get the mixing liquid ready. |
| Important: Noxafil PowderMix needs to be prepared using the mixing liquid. Do not mix Noxafil PowderMix with milk, juice, or water. |
When you use the mixing liquid for the first time:
- Open the bottle and remove the safety seal.
- Place the bottle adapter on top of the bottle with
the small hole facing up. - Push the bottle adapter all the way down.
- After it is in place, the bottle adapter stays in the bottle.
- Put the cap back on the bottle.
|
Step 2. Gather all your supplies and put on a clean surface. |
| Note: Put your child in a safe place. You will need both hands to prepare Noxafil PowderMix. Wash your hands with soap and water before preparing Noxafil PowderMix. |
|
1 mixing cup
(Using the tab on the mixing cup, pull open the lid.) |
1 packet of Noxafil PowderMix powder |
Mixing liquid |
2 syringes
(Have 1 of each size ready, but you may only use 1, depending on the dose.) |
Scissors
(not included with kit: use sharp household scissors or kitchen scissors) |
|
The Noxafil PowderMix box has a mixing cup holder inside to help tilt the cup when you are measuring the dose. |
|
Step 3. Add Noxafil PowderMix to the mixing cup. |
|
Note before adding Noxafil PowderMix:
Make sure you and your child are ready! If you do not use Noxafil PowderMix within 1 hour, you will need to throw it away and start over. |
|
|
|
| Step 4. Shake the mixing liquid bottle. |
|
| Step 5. Fill the blue syringe with 9 mL of mixing liquid. |
|
| Step 6. Check for air bubbles. |
|
|
|
|
Step 7. Add the 9 mL of mixing liquid to the Noxafil PowderMix. |
|
| Step 8. Mix the Noxafil PowderMix. |
|
|
|
| Step 9. Check your prescription label. |
|
| Note: The dose may change each time you go to the healthcare provider, so make sure you have all the recent information. Be sure to go to all of your child's healthcare provider appointments so your child gets the right dose. |
| Step 10. Choose the syringe you need. |
| Note: Only use the syringes provided in the kit. |
Choose the correct syringe for your child's dose:
| For 1 mL
to 3 mL Green |
For 3 mL
to 10 mL Blue |
- Then find the mL mark on the syringe that matches your
child's dose.
| Step 11. Measure the Noxafil PowderMix. |
|
| Important: You will not use all of the Noxafil PowderMix. There will be some left over in the mixing cup. |
| Step 12. Check for air bubbles. |
|
|
|
|
Step 13. Give the Noxafil PowderMix to your child. |
|
Important:
|
| Step 14. Clean the cup and syringes. |
| Note: Syringes and mixing cups should be reused. Do not throw away syringes and mixing cups provided until all the Noxafil PowderMix packets are used. |
|
|
Step 15. After all packets of Noxafil PowderMix have been used. |
- After you have used the last Noxafil PowderMix packet
in this box, you will have leftover mixing liquid in the
bottle. Throw away the leftover mixing liquid and all
components of the kit.
How should I store Noxafil PowderMix?
- Store the entire kit at room temperature between 68°F to
77°F (20°C to 25°C) in a clean, dry place. - Keep the mixing liquid at room temperature.
- Do not open the Noxafil PowderMix packet until you are
ready to mix a dose.
Keep NOXAFIL PowderMix and all medicines out of the reach of children.
For more information go to www.Noxafil.com or call
1-800-622-4477.
Distributed by:
Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent
Copyright © 2021-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
ifu-mk5592-2209r002
This Instructions for Use has been approved by the U.S. Food and
Drug Administration.
Revised: September 2022
8.8 Sex
No adjustment in the dosage of Noxafil is necessary based on sex.
8.9 Race
No adjustment in the dosage of Noxafil is necessary based on race.
8.10 Weight
Pharmacokinetic modeling suggests that patients who weigh greater than 120 kg may have lower posaconazole plasma drug exposure. Therefore, consider closely monitoring for breakthrough fungal infections particularly when using Noxafil oral suspension in patients weighing greater than 120 kg [see Clinical Pharmacology (12.3)].
10 Overdosage
There is no experience with overdosage of Noxafil injection and Noxafil delayed-release tablets.
During the clinical trials, some patients received Noxafil oral suspension up to 1,600 mg/day with no adverse reactions noted that were different from the lower doses. In addition, accidental overdose was noted in one patient who took 1200 mg twice daily Noxafil oral suspension for 3 days. No related adverse reactions were noted by the investigator.
Posaconazole is not removed by hemodialysis.
11 Description
Noxafil and Noxafil PowderMix contain posaconazole, an azole antifungal agent.
Posaconazole is designated chemically as 4-[4-[4-[4-[[ (3R,5R)-5- (2,4-difluorophenyl)tetrahydro-5- (1H-1,2,4-triazol-1-ylmethyl)-3-furanyl]methoxy]phenyl]-1-piperazinyl]phenyl]-2-[(1S,2S)-1-ethyl-2-hydroxypropyl]-2,4-dihydro-3H-1,2,4-triazol-3-one with an empirical formula of C37H42F2N8O4 and a molecular weight of 700.8. The chemical structure is:
Posaconazole is a white powder with a low aqueous solubility.
Noxafil (posaconazole) Injection
Noxafil injection, for intravenous use, is a clear colorless to yellow, without preservatives sterile liquid essentially free of foreign matter. Each vial contains 300 mg of posaconazole and the following inactive ingredients: 6.68 g Betadex Sulfobutyl Ether Sodium (SBECD), 0.0033 g edetate disodium, hydrochloric acid and sodium hydroxide to adjust the pH to 2.6, and water for injection.
Noxafil (posaconazole) Delayed-Release Tablets
Noxafil delayed-release tablet, for oral use, is yellow, coated, and oblong and contains 100 mg of posaconazole. Each delayed-release tablet contains the following inactive ingredients: croscarmellose sodium, hydroxypropylcellulose, hypromellose acetate succinate, iron oxide yellow, Macrogol/PEG 3350, magnesium stearate, microcrystalline cellulose, polyvinyl alcohol partially hydrolyzed, silicon dioxide, talc, and titanium dioxide.
Noxafil (posaconazole) Oral Suspension
Noxafil oral suspension is a white, cherry-flavored immediate-release suspension that contains 40 mg of posaconazole per mL and the following inactive ingredients: artificial cherry flavor, citric acid monohydrate, glycerin, liquid glucose, polysorbate 80, purified water, simethicone, sodium benzoate, sodium citrate dihydrate, titanium dioxide, and xanthan gum.
Noxafil PowderMix (posaconazole) for Delayed-Release Oral Suspension
Noxafil PowderMix for delayed-release oral suspension is supplied as a component of a kit. Each kit contains Noxafil as an off-white to yellowish powder for delayed-release oral suspension, a bottle of mixing liquid, two 3 mL (green) notched tip syringes, two 10 mL (blue) notched tip syringes, two mixing cups, and one bottle adapter for the mixing liquid bottle.
- Noxafil PowderMix for delayed-release oral suspension contains 300 mg of posaconazole and the following inactive ingredient: hypromellose acetate succinate.
- The mixing liquid contains: anhydrous citric acid, antifoam Af emulsion, berry citrus sweet flavor, carboxymethylcellulose sodium, carrageenan calcium sulfate trisodium phosphate, glycerin, methylparaben, microcrystalline cellulose, potassium sorbate, propylparaben, purified water, sodium citrate, sodium phosphate monobasic monohydrate, sodium saccharin, sorbitol solution, and xanthan gum.
16.1 How Supplied
Noxafil Injection
Noxafil injection is a clear, colorless to yellow sterile liquid in single-dose Type I glass vials closed with bromobutyl rubber stopper and aluminum seal containing 300 mg of posaconazole in 16.7 mL of solution (18 mg of posaconazole per mL) (NDC 0085-4331-01).
Noxafil Delayed-Release Tablets
Noxafil delayed-release tablets are yellow, coated, oblong, debossed with "100" on one side containing 100 mg of posaconazole. Bottles with child-resistant closures of 60 delayed-release tablets (NDC 0085-4324-02).
Noxafil Oral Suspension
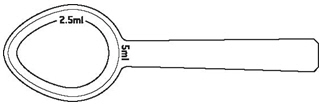
Noxafil oral suspension is a white, cherry-flavored oral suspension in 4-ounce (123 mL) amber glass bottles with child-resistant closures containing 105 mL of suspension (40 mg of posaconazole per mL).Supplied with each oral suspension bottle is a plastic dosing spoon calibrated for measuring 2.5 mL and 5 mL doses (NDC 0085-1328-01).
Noxafil PowderMix for Delayed-Release Oral Suspension
Noxafil PowderMix for delayed-release oral suspension is supplied as:
- Package A: a kit with 8 child-resistant single-use packets of Noxafil PowderMix for delayed-release oral suspension 300 mg, two 3 mL (green) notched tip syringes, two 10 mL (blue) notched tip syringes, two mixing cups, one mixing liquid bottle, one bottle adapter for the mixing liquid bottle and Instructions for Use.
- Package B: a box of six 3 mL (green) and six 10 mL (blue) notched tip syringes for additional supply.
- Packages A and B are supplied separately.
NDC 0085-2224-02 unit of use carton with 8 packets.
NDC 0085-2224-01 individual packet.
8.4 Pediatric Use
The three Noxafil dosage forms (injection, delayed-release tablets, oral suspension) and one Noxafil PowderMix (for delayed-release oral suspension) dosage form are different products; are approved for different pediatric indications, age groups, and weights; have different dosing regimens; and have different preparation and administration instructions. Therefore, select the recommended dosage form based on the pediatric indication, age group, and weight [see Dosage and Administration (2.1)].
Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with HFI. Because a diagnosis of HFI may not yet be established in pediatric patients, obtain a careful history of HFI symptoms with sorbitol/fructose/sucrose exposure prior to administration of Noxafil PowderMix for delayed-release oral suspension [see Warnings and Precautions (5.9)].
Treatment of Invasive Aspergillosis
The safety and effectiveness of Noxafil (injection and delayed-release tablets) have been established for the treatment of invasive aspergillosis in pediatric patients 2 years of age and older .
The safety and effectiveness of Noxafil PowderMix have been established for the treatment of invasive aspergillosis in pediatric patients 2 years of age and older who weigh 10 kg to 40 kg.
Use of Noxafil and Noxafil PowderMix for these pediatric indications is supported by evidence from adequate and well-controlled studies of Noxafil in adults and safety and pharmacokinetic (PK) data from two pediatric studies [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)]. The safety of Noxafil and Noxafil PowderMix in pediatric patients for these pediatric indications was consistent with the known safety profile of Noxafil in adults [see Adverse Reactions (6.1)].
Noxafil PowderMix for delayed-release oral suspension is not recommended for use in patients who weigh greater than 40 kg because the recommended dosage cannot be achieved with this dosage form.
The safety and effectiveness of Noxafil and Noxafil PowderMix have not been established in pediatric patients less than 2 years of age.
Prophylaxis of Invasive Aspergillus and Candida Infections
The safety and effectiveness of Noxafil (injection and delayed-release tablets) have been established for the prophylaxis of invasive Aspergillus and Candida infections in pediatric patients 2 years of age and older who are at high risk of developing these infections due to being severely immunocompromised.
The safety and effectiveness of Noxafil oral suspension have been established for the prophylaxis of invasive Aspergillus and Candida infections in pediatric patients 13 years of age and older who are at high risk of developing these infections due to being severely immunocompromised.
The safety and effectiveness of Noxafil PowderMix have been established for the prophylaxis of invasive Aspergillus and Candida infections in pediatric patients 2 years of age and older who weigh 10 kg to 40 kg who are at high risk of developing these infections due to being severely immunocompromised.
Use of Noxafil and Noxafil PowderMix for these pediatric indications is supported by adequate and well controlled studies of Noxafil in adults and pediatric patients aged 13 years of age and older and additional PK and safety data in pediatric patients 2 years of age and older [see Clinical Pharmacology (12.3) and Clinical studies (14)].
Noxafil PowderMix is not recommended for use in patients who weigh greater than 40 kg because the recommended dosage cannot be achieved with this dosage form.
The safety and effectiveness of Noxafil and Noxafil PowderMix have not been established in pediatric patients less than 2 years of age.
Treatment of Oropharyngeal Candidiasis, including Refractory to Itraconazole and/or Fluconazole
The safety and effectiveness of Noxafil oral suspension have been established for the treatment of oropharyngeal candidiasis (OPC), including OPC refractory (rOPC) to itraconazole and/or fluconazole in pediatric patients 13 years of age and older.
Use of Noxafil oral suspension for this pediatric indication is supported by adequate and well controlled studies in adults and pediatric patients 13 years of age and older [see Clinical studies (14.4)].
The Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix products are not approved for the treatment of oropharyngeal candidiasis in pediatric patients. Noxafil Oral Suspension is the only dosage form approved for the treatment of OPC and rOPC in pediatric patients [see Dosage and Administration (2.4)].
The safety and effectiveness of Noxafil oral suspension for the treatment of OPC and rOPC have not been established in pediatric patients less than 13 years of age.
8.5 Geriatric Use
No overall differences in the safety or effectiveness of Noxafil injection, Noxafil delayed-release tablets, and Noxafil oral suspension have been observed between geriatric patients and younger adult patients in the clinical trials; therefore, the recommended dosage in geriatric patients is the same as that for younger adult patients. No clinically meaningful differences in posaconazole pharmacokinetics were observed in Noxafil-treated geriatric patients compared to Noxafil-treated younger adult patients during clinical trials [see Clinical Pharmacology (12.3)].
- Of the 279 patients treated with Noxafil injection in the Noxafil Injection Study (prophylaxis of invasive Aspergillus and Candida infections in those at high risk of developing these infections due to being severely immunocompromised), 52 (19%) patients were >65 years of age.
- Of the 230 patients treated with Noxafil delayed-release tablets, 38 (17%) patients were >65 years of age.
- Of the 605 patients treated with Noxafil oral suspension in Noxafil Oral Suspension Study 1 and Study 2 (prophylaxis of invasive Aspergillus and Candida infections in those at high risk of developing these infections due to being severely immunocompromised), 63 (10%) patients were ≥65 years of age.
- In studies of Noxafil for an unapproved indication, 48 patients treated with Noxafil oral suspension (greater than or equal to 800 mg/day (eight times the maximum recommended maintenance dosage for the treatment of OPC)) were ≥65 years of age.
- Of the 288 patients treated with Noxafil injection or Noxafil delayed-release tablets in the Aspergillosis Treatment Study, 85 (29%) patients were ≥65 years of age.
4 Contraindications
- Known hypersensitivity to posaconazole or other azole antifungal agents. (4.1)
- Coadministration of Noxafil with the following drugs is contraindicated; Noxafil increases concentrations and toxicities of:
- Sirolimus (4.2, 7.2)
- CYP3A4 substrates (pimozide, quinidine): can result in QTc interval prolongation and cases of torsades de pointes (TdP) (4.3, 5.2, 7.2)
- HMG-CoA Reductase Inhibitors Primarily Metabolized through CYP3A4 (4.4, 7.2)
- Ergot alkaloids (4.5, 7.2)
- Venetoclax: In patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) at initiation and during the ramp-up phase (4.6, 5.11, 7.2)
- Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with known or suspected Hereditary Fructose Intolerance (HFI). (4.7, 5.9, 8.4)
6 Adverse Reactions
The following serious and otherwise important adverse reactions are discussed in detail in another section of the labeling:
- Arrhythmias and QT Prolongation [see Warnings and Precautions (5.2)]
- Electrolyte Disturbances [see Warnings and Precautions (5.3)]
- Pseudoaldosteronism [see Warnings and Precautions (5.4)]
- Hepatic Toxicity [see Warnings and Precautions (5.5)]
7 Drug Interactions
Table 15 and Table 17 include drugs with clinically important drug interactions when administered concomitantly with Noxafil and Noxafil PowderMix and instructions for preventing or managing them. Table 16 includes important drug interactions specific to the absorption of posaconazole administered as either Noxafil oral suspension or Noxafil PowderMix.
These recommendations are based on either drug interaction studies or predicted interactions due to the expected magnitude of interaction and potential for serious adverse reactions or loss of efficacy [see Clinical Pharmacology (12.3)].
The following information was derived from data with Noxafil oral suspension or another posaconazole tablet formulation unless otherwise noted. All clinically important drug interactions with Noxafil oral suspension, except for those that affect the absorption of posaconazole (via gastric pH and motility), are considered relevant to clinically important drug interactions with Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension [see Clinical Pharmacology (12.3)].
Consult the labeling of concomitantly used drugs to obtain further information about interactions with posaconazole.
4.1 Hypersensitivity
Noxafil is contraindicated in persons with known hypersensitivity to posaconazole or other azole antifungal agents.
5.5 Hepatic Toxicity
Hepatic reactions (e.g., mild to moderate elevations in alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase, total bilirubin, and/or clinical hepatitis) have been reported in clinical trials. The elevations in liver tests were generally reversible on discontinuation of therapy, and in some instances these tests normalized without drug interruption. Cases of more severe hepatic reactions including cholestasis or hepatic failure including deaths have been reported in patients with serious underlying medical conditions (e.g., hematologic malignancy) during treatment with Noxafil. These severe hepatic reactions were seen primarily in subjects receiving the Noxafil oral suspension 800 mg daily (400 mg twice daily or 200 mg four times a day) in clinical trials.
Liver tests should be evaluated at the start of and during the course of Noxafil therapy. Patients who develop abnormal liver tests during Noxafil therapy should be monitored for the development of more severe hepatic injury. Patient management should include laboratory evaluation of hepatic function (particularly liver tests and bilirubin). Discontinuation of Noxafil must be considered if clinical signs and symptoms consistent with liver disease develop that may be attributable to Noxafil.
5.6 Renal Impairment
Due to the variability in exposure with Noxafil delayed-release tablets, Noxafil oral suspension, and Noxafil PowderMix for delayed-release oral suspension, patients with severe renal impairment should be monitored closely for breakthrough fungal infections [see Dosage and Administration (2.11) and Use in Specific Populations (8.6)].
Noxafil injection should be avoided in patients with moderate or severe renal impairment (eGFR <50 mL/min/1.73 m2), unless an assessment of the benefit/risk to the patient justifies the use of Noxafil injection. In patients with moderate or severe renal impairment (eGFR <50 mL/min/1.73 m2), receiving the Noxafil injection, accumulation of the intravenous vehicle, SBECD, is expected to occur. Serum creatinine levels should be closely monitored in these patients, and, if increases occur, consideration should be given to changing to oral Noxafil therapy [see Dosage and Administration (2.11) and Use in Specific Populations (8.6)].
8.6 Renal Impairment
Noxafil Oral Suspension and Noxafil Delayed-Release Tablets
No dosage adjustment is required for patients with eGFR 20 mL/minute/1.73 m2 or higher.
Due to variability in posaconazole exposure, closely monitor patients with eGFR less than 20 mL/minute/1.73 m2 for breakthrough fungal infections. [see Clinical Pharmacology (12.3)].
Noxafil Injection
Avoid use of Noxafil injection in patients with eGFR less than 50 mL/minute/1.73 m2 unless the benefit/risk to the patient justifies its use. The inactive ingredient in Noxafil injection, Betadex Sulfobutyl Ether Sodium (SBECD) is expected to accumulate in patients with reduced renal function. Safety and effectiveness of Noxafil injection have not been established in patients with less than 50 mL/minute/1.73 m2.
If treatment with Noxafil injection is unavoidable in patients with eGFR less than 50 mL/minute/1.73 m2, monitor serum creatinine levels. If serum creatinine increases, consider changing to oral Noxafil therapy.
4.2 Use With Sirolimus
Noxafil is contraindicated with sirolimus. Concomitant administration of Noxafil with sirolimus increases the sirolimus blood concentrations by approximately 9-fold and can result in sirolimus toxicity [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
5.7 Midazolam Toxicity
Concomitant administration of Noxafil with midazolam increases the midazolam plasma concentrations by approximately 5-fold. Increased plasma midazolam concentrations could potentiate and prolong hypnotic and sedative effects. Patients must be monitored closely for adverse effects associated with high plasma concentrations of midazolam and benzodiazepine receptor antagonists must be available to reverse these effects [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dosage adjustment is recommended for Noxafil oral suspension, Noxafil delayed-release tablets, Noxafil PowderMix for delayed-release oral suspension, and Noxafil injection in patients with mild, moderate, or severe hepatic impairment (Child-Pugh Class A, B, or C, respectively) [see Clinical Pharmacology (12.3)].
However, a specific hepatic impairment study has not been conducted with the Noxafil delayed-release tablets, Noxafil PowderMix for delayed-release oral suspension, or Noxafil injection.
1 Indications and Usage
Noxafil is an azole antifungal indicated as follows:
- Noxafil is indicated for the treatment of invasive aspergillosis as follows: (1.1)
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg
-
Noxafil is indicated for the prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, such as hematopoietic stem cell transplant (HSCT) recipients with graft-versus-host disease (GVHD) or those with hematologic malignancies with prolonged neutropenia from chemotherapy as follows: (1.2)
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater.
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil oral suspension: adults and pediatric patients 13 years of age and older
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg
- Noxafil oral suspension is indicated for the treatment of oropharyngeal candidiasis (OPC), including OPC refractory (rOPC) to itraconazole and/or fluconazole in adults and pediatric patients 13 years of age and older (1.3)
4.6 Use With Venetoclax
Coadministration of Noxafil with venetoclax at initiation and during the ramp-up phase is contraindicated in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) due to the potential for increased risk of tumor lysis syndrome [see Warnings and Precautions (5.11) and Drug Interactions (7.2)].
5.4 Pseudoaldosteronism
Pseudoaldosteronism, manifested by the onset of hypertension or worsening of hypertension, and abnormal laboratory findings (hypokalemia, low serum renin and aldosterone, and elevated 11-deoxycortisol), has been reported with posaconazole use in the postmarket setting. Monitor blood pressure and potassium levels and manage as necessary. Management of pseudoaldosteronism may include discontinuation of Noxafil, substitution with an appropriate antifungal drug that is not associated with pseudoaldosteronism, or use of aldosterone receptor antagonists.
12.1 Mechanism of Action
Posaconazole is an azole antifungal agent [see Clinical Pharmacology (12.4)].
5.11 Venetoclax Toxicity
Concomitant administration of Noxafil, a strong CYP3A4 inhibitor, with venetoclax may increase venetoclax toxicities, including the risk of tumor lysis syndrome (TLS), neutropenia, and serious infections. In patients with CLL/SLL, administration of Noxafil during initiation and the ramp-up phase of venetoclax is contraindicated [see Contraindications (4.6)]. Refer to the venetoclax labeling for safety monitoring and dose reduction in the steady daily dosing phase in CLL/SLL patients.
For patients with acute myeloid leukemia (AML), dose reduction and safety monitoring are recommended across all dosing phases when coadministering Noxafil with venetoclax [see Drug Interactions (7.2)]. Refer to the venetoclax prescribing information for dosing instructions.
5.8 Vincristine Toxicity
Concomitant administration of azole antifungals, including Noxafil, with vincristine has been associated with neurotoxicity and other serious adverse reactions, including seizures, peripheral neuropathy, syndrome of inappropriate antidiuretic hormone secretion, and paralytic ileus. Reserve azole antifungals, including Noxafil, for patients receiving a vinca alkaloid, including vincristine, who have no alternative antifungal treatment options [see Drug Interactions (7.2)].
16.2 Storage and Handling
Noxafil Injection
Store Noxafil injection vial refrigerated at 2 to 8°C (36 to 46°F). Storage conditions for the diluted Noxafil infusion solution are presented in another section of the prescribing information [see Dosage and Administration (2.5)].
Noxafil Delayed-Release Tablets
Store at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature].
Noxafil Oral Suspension
Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
Noxafil PowderMix for Delayed-Release Oral Suspension
Store the entire kit at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) in a clean, dry place. Do not open foil packet containing Noxafil PowderMix for delayed-release oral suspension until ready for use. Storage conditions for the reconstituted solution are presented in another section of the prescribing information [see Dosage and Administration (2.10)].
5 Warnings and Precautions
- Calcineurin-Inhibitor Toxicity: Noxafil increases concentrations of cyclosporine or tacrolimus; reduce dose of cyclosporine and tacrolimus and monitor concentrations frequently. (5.1)
- Arrhythmias and QTc Prolongation: Noxafil has been shown to prolong the QTc interval and cause cases of TdP. Administer with caution to patients with potentially proarrhythmic conditions. Do not administer with drugs known to prolong QTc interval and metabolized through CYP3A4. (5.2, 7.2)
- Electrolyte Disturbances: Monitor and correct, especially those involving potassium (K+), magnesium (Mg++), and calcium (Ca++), before and during Noxafil therapy. (5.3)
- Pseudoaldosteronism: Manifested by the onset or worsening of hypertension, and abnormal laboratory findings. Monitor blood pressure and potassium levels, and manage as necessary. (5.4)
- Hepatic Toxicity: Elevations in liver tests may occur. Discontinuation should be considered in patients who develop abnormal liver tests or monitor liver tests during treatment. (5.5)
- Renal Impairment: Noxafil injection should be avoided in patients with moderate or severe renal impairment (eGFR less than 50 mL/min/1.73 m2), unless an assessment of the benefit/risk to the patient justifies the use of Noxafil injection. (5.6, 8.6)
- Concomitant Use with Midazolam: Noxafil can prolong hypnotic/sedative effects. Monitor patients and benzodiazepine receptor antagonists should be available. (5.7, 7.2)
- Vincristine Toxicity: Concomitant administration of azole antifungals, including Noxafil, with vincristine has been associated with neurotoxicity and other serious adverse reactions; reserve azole antifungals, including Noxafil, for patients receiving a vinca alkaloid, including vincristine, who have no alternative antifungal treatment options. (5.8, 7.2)
- Risk in Patients with Hereditary Fructose Intolerance (HFI): Noxafil PowderMix for delayed-release oral suspension contains sorbitol. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before Noxafil PowderMix for delayed-release oral suspension administration. (5.9, 8.4)
- Breakthrough Fungal Infections: Monitor patients with severe diarrhea or vomiting when receiving Noxafil delayed-release tablets, Noxafil oral suspension, and Noxafil PowderMix for delayed-release oral suspension. (5.10)
- Venetoclax Toxicity: Concomitant administration of Noxafil with venetoclax may increase venetoclax toxicities, including the risk of tumor lysis syndrome, neutropenia, and serious infections; monitor for toxicity and reduce venetoclax dose. (4.6, 5.11, 7.2)
2 Dosage and Administration
- Noxafil formulations are supplied in different dose strengths of posaconazole, are approved for different indications, age groups, and weights, have different dosages and duration of therapy; and have different preparation and administration instructions. (2.1)
- Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations. (2.1, 2.2, 2.3)
- Noxafil injection must be administered through an in-line filter. (2.6)
- Administer Noxafil injection by intravenous infusion over approximately 90 minutes via a central venous line. (2.1, 2.6)
- Do NOT administer Noxafil injection as an intravenous bolus injection. (2.1)
- Administer Noxafil delayed-release tablets with or without food. (2.1)
- Administer Noxafil oral suspension with a full meal. (2.1)
- Administer Noxafil PowderMix for delayed-release oral suspension with food. (2.1)
- Administer Noxafil PowderMix for delayed-release oral suspension with the provided notched tip syringes only. (2.1)
- See the full prescribing information for important administration instructions and preparation instructions for Noxafil (injection, delayed-release tablets, and oral suspension) and Noxafil PowderMix delayed-release oral suspension (2.5, 2.6, 2.7, 2.8, 2.9, 2.10)
- For adult and pediatric patients aged 2 years of age and older, see the Full Prescribing Information for dosing recommendations for Noxafil injection, Noxafil delayed-release tablets, Noxafil oral suspension, and/or Noxafil PowderMix for delayed-release oral suspension based on the indication, age, and weight associated with the dosage form (1.1, 1.2, 1.3, 2.1, 2.2, 2.3, 2.4)
3 Dosage Forms and Strengths
Noxafil injection
300 mg/16.7 mL (18 mg/mL) of posaconazole: Clear, colorless to yellow sterile liquid in a single-dose vial.
Noxafil Delayed-Release Tablets
100 mg of posaconazole: Yellow, coated, oblong tablets, debossed with "100" on one side.
Noxafil Oral Suspension
4,200 mg/105 mL (40 mg/mL) of posaconazole: White, cherry-flavored suspension in amber glass bottles with child-resistant closures.
Noxafil PowderMix for Delayed-Release Oral Suspension
300 mg: Off-white to yellowish powder for delayed-release oral suspension and a mixing liquid in a kit. The kit contains (1) Package A that contains single-use packets of Noxafil PowderMix, green notched tip syringes, blue notched tip syringes, mixing cups, mixing liquid bottle, and one bottle adapter for the mixing liquid bottle; and (2) Package B that contains green and blue notched tip syringes for additional supply [see How Supplied/Storage and Handling (16.1)].
4.5 Use With Ergot Alkaloids
Noxafil may increase the plasma concentrations of ergot alkaloids (ergotamine and dihydroergotamine) which may lead to ergotism [see Drug Interactions (7.2)].
5.3 Electrolyte Disturbances
Electrolyte disturbances, especially those involving potassium, magnesium or calcium levels, should be monitored and corrected as necessary before and during Noxafil therapy.
6.2 Postmarketing Experience
The following adverse reaction has been identified during the post-approval use of Noxafil. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of Noxafil cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
14.2 Prophylaxis of Aspergillus
Two randomized, controlled studies were conducted using Noxafil as prophylaxis for the prevention of invasive fungal infections (IFIs) among patients at high risk due to severely compromised immune systems.
The first study (Noxafil Oral Suspension Study 1) was a randomized, double-blind trial that compared Noxafil oral suspension (200 mg three times a day) with fluconazole capsules (400 mg once daily) as prophylaxis against invasive fungal infections in allogeneic hematopoietic stem cell transplant (HSCT) recipients with Graft versus Host Disease (GVHD). Efficacy of prophylaxis was evaluated using a composite endpoint of proven/probable IFIs, death, or treatment with systemic antifungal therapy (patients may have met more than one of these criteria). This assessed all patients while on study therapy plus 7 days and at 16 weeks post-randomization. The mean duration of therapy was comparable between the 2 treatment groups (80 days, Noxafil oral suspension; 77 days, fluconazole). Table 34 contains the results from Noxafil Oral Suspension Study 1.
| Posaconazole n=301 |
Fluconazole n=299 |
|
|---|---|---|
| On therapy plus 7 days | ||
|
Clinical Failure Patients may have met more than one criterion defining failure.
|
50 (17%) | 55 (18%) |
| Failure due to: | ||
| Proven/Probable IFI | 7 (2%) | 22 (7%) |
| (Aspergillus) | 3 (1%) | 17 (6%) |
| (Candida) | 1 (<1%) | 3 (1%) |
| (Other) | 3 (1%) | 2 (1%) |
| All Deaths | 22 (7%) | 24 (8%) |
| Proven/probable fungal infection prior to death | 2 (<1%) | 6 (2%) |
| SAF Use of systemic antifungal therapy (SAF) criterion is based on protocol definitions (empiric/IFI usage >4 consecutive days).
|
27 (9%) | 25 (8%) |
| Through 16 weeks | ||
|
Clinical Failure
,
95% confidence interval (posaconazole-fluconazole) = (-11.5%, + 3.7%).
|
99 (33%) | 110 (37%) |
| Failure due to: | ||
| Proven/Probable IFI | 16 (5%) | 27 (9%) |
| (Aspergillus) | 7 (2%) | 21 (7%) |
| (Candida) | 4 (1%) | 4 (1%) |
| (Other) | 5 (2%) | 2 (1%) |
| All Deaths | 58 (19%) | 59 (20%) |
| Proven/probable fungal infection prior to death | 10 (3%) | 16 (5%) |
| SAF | 26 (9%) | 30 (10%) |
| Event free lost to follow-up Patients who are lost to follow-up (not observed for 112 days), and who did not meet another clinical failure endpoint. These patients were considered failures.
|
24 (8%) | 30 (10%) |
The second study (Noxafil Oral Suspension Study 2) was a randomized, open-label study that compared Noxafil oral suspension (200 mg 3 times a day) with fluconazole suspension (400 mg once daily) or itraconazole oral solution (200 mg twice a day) as prophylaxis against IFIs in neutropenic patients who were receiving cytotoxic chemotherapy for AML or MDS. As in Noxafil Oral Suspension Study 1, efficacy of prophylaxis was evaluated using a composite endpoint of proven/probable IFIs, death, or treatment with systemic antifungal therapy (Patients might have met more than one of these criteria). This study assessed patients while on treatment plus 7 days and 100 days post-randomization. The mean duration of therapy was comparable between the 2 treatment groups (29 days, posaconazole; 25 days, fluconazole or itraconazole). Table 35 contains the results from Noxafil Oral Suspension Study 2.
| Posaconazole n=304 |
Fluconazole/Itraconazole n=298 |
|
|---|---|---|
| On therapy plus 7 days | ||
|
Clinical Failure 95% confidence interval (posaconazole-fluconazole/itraconazole) = (-22.9%, -7.8%).
,
Patients may have met more than one criterion defining failure.
|
82 (27%) | 126 (42%) |
| Failure due to: | ||
| Proven/Probable IFI | 7 (2%) | 25 (8%) |
| (Aspergillus) | 2 (1%) | 20 (7%) |
| (Candida) | 3 (1%) | 2 (1%) |
| (Other) | 2 (1%) | 3 (1%) |
| All Deaths | 17 (6%) | 25 (8%) |
| Proven/probable fungal infection prior to death | 1 (<1%) | 2 (1%) |
| SAF Use of systemic antifungal therapy (SAF) criterion is based on protocol definitions (empiric/IFI usage >3 consecutive days).
|
67 (22%) | 98 (33%) |
| Through 100 days post-randomization | ||
| Clinical Failure | 158 (52%) | 191 (64%) |
| Failure due to: | ||
| Proven/Probable IFI | 14 (5%) | 33 (11%) |
| (Aspergillus) | 2 (1%) | 26 (9%) |
| (Candida) | 10 (3%) | 4 (1%) |
| (Other) | 2 (1%) | 3 (1%) |
| All Deaths | 44 (14%) | 64 (21%) |
| Proven/probable fungal infection prior to death | 2 (1%) | 16 (5%) |
| SAF | 98 (32%) | 125 (42%) |
| Event free lost to follow-up Patients who are lost to follow-up (not observed for 100 days), and who did not meet another clinical failure endpoint. These patients were considered failures.
|
34 (11%) | 24 (8%) |
In summary, 2 clinical studies of prophylaxis were conducted with the Noxafil oral suspension. As seen in the accompanying tables (Table 34 and Table 35), clinical failure represented a composite endpoint of breakthrough IFI, mortality and use of systemic antifungal therapy. In Noxafil Oral Suspension Study 1 (Table 34), the clinical failure rate of posaconazole (33%) was similar to fluconazole (37%), (95% CI for the difference posaconazole–comparator -11.5% to 3.7%) while in Noxafil Oral Suspension Study 2 (Table 35) clinical failure was lower for patients treated with posaconazole (27%) when compared to patients treated with fluconazole or itraconazole (42%), (95% CI for the difference posaconazole–comparator -22.9% to -7.8%).
All-cause mortality was similar at 16 weeks for both treatment arms in Noxafil Oral Suspension Study 1 [POS 58/301 (19%) vs. FLU 59/299 (20%)]; all-cause mortality was lower at 100 days for Noxafil-treated patients in Noxafil Oral Suspension Study 2 [POS 44/304 (14%) vs. FLU/ITZ 64/298 (21%)]. Both studies demonstrated fewer breakthrough infections caused by Aspergillus species in patients receiving Noxafil prophylaxis when compared to patients receiving fluconazole or itraconazole.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Important Administration Instructions
Noxafil Delayed-Release Tablets
Advise patients that Noxafil delayed-release tablets must be swallowed whole and not divided, crushed, or chewed.
Instruct patients that if they miss a dose, they should take it as soon as they remember. If they do not remember until it is within 12 hours of the next dose, they should be instructed to skip the missed dose and go back to the regular schedule. Patients should not double their next dose or take more than the prescribed dose.
Noxafil Oral Suspension
Advise patients to take each dose of Noxafil oral suspension during or immediately (i.e., within 20 minutes) following a full meal. In patients who cannot eat a full meal, each dose of Noxafil oral suspension should be administered with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale) in order to enhance absorption.
Instruct patients that if they miss a dose, they should take it as soon as they remember. However, if it is almost time for the next dose, they should be instructed to skip the missed dose and go back to the regular schedule. Patients should not double their next dose or take more than the prescribed dose.
Noxafil PowderMix for Delayed-Release Oral Suspension
Instruct parents and/or caregivers that ONLY the provided notched tip syringes can be used to administer Noxafil PowderMix for delayed-release oral suspension to pediatric patients.
Advise patients to take Noxafil PowderMix for delayed-release oral suspension with food.
Drug Interactions
Advise patients to inform their physician immediately if they:
- develop severe diarrhea or vomiting.
- are currently taking drugs that are known to prolong the QTc interval and are metabolized through CYP3A4.
- are currently taking a cyclosporine or tacrolimus, or they notice swelling in an arm or leg or shortness of breath.
- are taking other drugs or before they begin taking other drugs as certain drugs can decrease or increase the plasma concentrations of posaconazole.
Serious and Potentially Serious Adverse Reactions
Advise patients to inform their physician immediately if they:
- notice a change in heart rate or heart rhythm or have a heart condition or circulatory disease. Noxafil can be administered with caution to patients with potentially proarrhythmic conditions.
- are pregnant, plan to become pregnant, or are nursing.
- have liver disease or develop itching, nausea or vomiting, their eyes or skin turn yellow, they feel more tired than usual or feel like they have the flu.
- have ever had an allergic reaction to other antifungal medicines such as ketoconazole, fluconazole, itraconazole, or voriconazole.
Hereditary Fructose Intolerance (HFI)
Inform patients and caregivers that Noxafil PowderMix for delayed-release oral suspension contains sorbitol and can be life-threatening when administered to patients with hereditary fructose intolerance (HFI) [see Warnings and Precautions (5.9)]. Inquire for symptoms of sorbitol/fructose and/or sucrose intolerance before administration.
5.1 Calcineurin Inhibitor Toxicity
Concomitant administration of Noxafil with cyclosporine or tacrolimus increases the whole blood trough concentrations of these calcineurin-inhibitors [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)]. Nephrotoxicity and leukoencephalopathy (including deaths) have been reported in clinical efficacy studies in patients with elevated cyclosporine or tacrolimus concentrations. Frequent monitoring of tacrolimus or cyclosporine whole blood trough concentrations should be performed during and at discontinuation of Noxafil treatment and the tacrolimus or cyclosporine dose adjusted accordingly.
5.10 Breakthrough Fungal Infections
Patients who have severe diarrhea or vomiting should be monitored closely for breakthrough fungal infections when receiving Noxafil delayed-release tablets, Noxafil oral suspension, or Noxafil PowderMix for delayed-release oral suspension.
5.2 Arrhythmias and Qt Prolongation
Some azoles, including Noxafil, have been associated with prolongation of the QT interval on the electrocardiogram. In addition, cases of torsades de pointes have been reported in patients taking Noxafil.
Results from a multiple time-matched ECG analysis in healthy volunteers did not show any increase in the mean of the QTc interval. Multiple, time-matched ECGs collected over a 12-hour period were recorded at baseline and steady-state from 173 healthy male and female volunteers (18-85 years of age) administered Noxafil oral suspension 400 mg twice daily with a high-fat meal. In this pooled analysis, the mean QTc (Fridericia) interval change from baseline was –5 msec following administration of the recommended clinical dose. A decrease in the QTc(F) interval (–3 msec) was also observed in a small number of subjects (n=16) administered placebo. The placebo-adjusted mean maximum QTc(F) interval change from baseline was <0 msec (–8 msec). No healthy subject administered Noxafil had a QTc(F) interval ≥500 msec or an increase ≥60 msec in their QTc(F) interval from baseline.
Noxafil should be administered with caution to patients with potentially proarrhythmic conditions. Do not administer with drugs that are known to prolong the QTc interval and are metabolized through CYP3A4 [see Contraindications (4.3) and Drug Interactions (7.2)].
2.5 Preparation of Noxafil Injection
Preparation of Noxafil Injection:
- Remove the vial of Noxafil injection from the refrigerator and allow to equilibrate to room temperature prior to use.
-
To prepare the required dose, aseptically transfer one vial of Noxafil injection (containing 300 mg of posaconazole in 16.7 mL of solution) to an intravenous bag or bottle of one of the following compatible diluents to achieve a final posaconazole concentration between 1 mg/mL and 2 mg/mL:
- 0.45% Sodium Chloride Injection
- 0.9% Sodium Chloride Injection
- 5% Dextrose Injection
- 5% Dextrose and 0.45% Sodium Chloride Injection
- 5% Dextrose and 0.9% Sodium Chloride Injection
- 5% Dextrose and 20 mEq Potassium Chloride Injection
Use of other diluents is not recommended because they may result in particulate formation.
- Discard any unused Noxafil injection from the vial.
- Parenteral drug products should be inspected visually for particulate matter prior to administration, whenever solution and container permit. Once admixed, the diluted Noxafil infusion solution ranges from colorless to yellow (variations of color within this range do not affect the quality of the product).
- Immediately use the diluted Noxafil infusion solution, once admixed. If not used immediately, refrigerate (2 to 8°C (36 to 46°F)) the diluted Noxafil infusion solution up to 24 hours. Discard any unused portion.
Incompatible Diluents
Co-administration of drug products besides the infusion solutions or products stated above are not recommended because this may result in particulate formation. The following diluents were determined to be incompatible with Noxafil injection; thus, do not dilute Noxafil injection with them:
- Lactated Ringer’s Injection
- Lactated Ringer's and 5% Dextrose Injection
- 4.2% Sodium Bicarbonate Injection
1.1 Treatment of Invasive Aspergillosis
Noxafil is indicated for the treatment of invasive aspergillosis as follows:
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 to 40 kg
1.2 Prophylaxis of Invasive Aspergillus
Noxafil is indicated for the prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, such as hematopoietic stem cell transplant (HSCT) recipients with graft-versus-host disease (GVHD) or those with hematologic malignancies with prolonged neutropenia from chemotherapy as follows:
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil oral suspension: adults and pediatric patients 13 years of age and older
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg
Principal Display Panel 300 Mg Carton
NDC 0085-2224-02
Noxafil® PowderMix
(posaconazole) for
Delayed-Release Oral Suspension
300 mg
For Pediatric Use
For Oral Administration Only
Each packet contains 300 mg posaconazole.
Attention: Noxafil PowderMix for Delayed-Release Oral Suspension is NOT
substitutable with Noxafil Oral Suspension due to differences in the
dosing of each formulation.
Use only the notched-tip syringes and mixing liquid provided with this kit.
Rx only
8 packets
Multi-Dose Kit A
Additional notched-tip syringes are provided in
Package B with this kit.
DIRECTIONS FOR USE:
See instructions for use booklet and prescribing
information for additional information.
2.1 Important Administration Instructions
Noxafil injection, Noxafil delayed-release tablets, Noxafil oral suspension and Noxafil PowderMix for delayed-release oral suspension are supplied in different dose strengths of posaconazole, are approved for different indications, age groups and weights; have different dosages and duration of therapy; and have different preparation and administration instructions.
Therefore, select the recommended dosage form based on the indication, age group, and weight and carefully follow the recommended dosage, preparation and administration instructions described for each product [see Dosage and Administration (2.2 to 2.11)], and the following important administration instructions described below.
Non-substitutable
Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations [see Dosage and Administration (2.2, 2.3)].
Noxafil injection
- Administer via a central venous line, including a central venous catheter or peripherally inserted central catheter (PICC), by slow intravenous infusion over approximately 90 minutes [see Dosage and Administration (2.6)].
- Do NOT administer Noxafil injection as an intravenous bolus injection.
Noxafil delayed-release tablets
- Swallow tablets whole. Do not divide, crush, or chew.
- Administer with or without food [see Dosage and Administration (2.7) and Clinical Pharmacology (12.3)].
- For patients who cannot eat a full meal, Noxafil delayed-release tablets should be used instead of Noxafil oral suspension for the prophylaxis indication. Noxafil delayed-release tablets generally provide higher plasma drug exposures than Noxafil oral suspension under both fed and fasted conditions [see Dosage and Administration (2.8)].
Noxafil oral suspension
- Administer with a full meal or with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale) in patients who cannot eat a full meal [see Dosage and Administration (2.8)].
Noxafil PowderMix for delayed-release oral suspension
- Administer with food [see Clinical Pharmacology (12.3)].
- To ensure delivery of the correct dose, ONLY the provided notched tip syringes must be used for preparation and administration. The design of the notched tip syringe prevents aggregation of the suspension during preparation and administration [see Dosage and Administration (2.10)].
Principal Display Panel 16.7 Ml Vial Carton
NDC 0085-4331-01
Noxafil®
(posaconazole)
Injection
300 mg/16.7 mL
(18 mg/mL)
For Intravenous Use Only
Requires further dilution prior to infusion.
Rx only
Sterile Single-Dose Vial
Discard Unused Portion
Principal Display Panel 105 Ml Bottle Carton
NDC 0085-1328-01
105 mL
NOXAFIL®
(posaconazole)
Oral Suspension
200 mg/5 mL
Each mL contains: 40 mg posaconazole.
Attention: Noxafil Oral Suspension is
NOT substitutable with Noxafil
Delayed-Release Tablets or Noxafil PowderMix
for Delayed-Release Oral Suspension due to
differences in the dosing of each formulation.
SHAKE WELL BEFORE EACH USE.
Take with a meal, or a nutritional supplement,
or an acidic carbonated beverage.
Carton contains measured dosing spoon.
Rx only
Principal Display Panel 60 Tablet Bottle Label
NDC 0085-4324-02
Noxafil®
(posaconazole)
delayed-release tablets
100 mg
Each tablet contains 100 mg posaconazole.
Attention: Noxafil Oral Suspension and Delayed-
Release Tablets are NOT substitutable due to
differences in the dosing of each formulation.
Rx only
60 Tablets
2.2 Recommended Dosage of Noxafil in Adult Patients
The recommended dosage of Noxafil (injection, delayed-release tablets, and oral suspension) in adult patients for the treatment of invasive aspergillosis, prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, or for the treatment of oropharyngeal candidiasis (OPC) is shown in Table 1 [see Dosage and Administration (2.5, 2.6, 2.7,2.8, 2.9) and Clinical Pharmacology (12.3)].
Noxafil PowderMix for delayed-release oral suspension is not recommended for use in adults [see Indications and Usage (1.1, 1.2)].
| Dosage | Duration of Therapy |
|---|---|
|
Treatment of Invasive Aspergillosis
Switching between the Noxafil injection and delayed-release tablets is acceptable. A loading dose is not required when switching between dosage forms.
|
|
|
Noxafil Injection:
Loading dose:
Maintenance dose:
Noxafil Delayed-Release Tablets:
|
Loading dose:
Maintenance dose:
|
| Prophylaxis of Invasive Aspergillus and Candida Infections | |
|
Noxafil Injection:
Loading dose: 300 mg Noxafil injection intravenously twice a day on the first day. Maintenance dose: 300 mg Noxafil injection intravenously once a day thereafter. Noxafil Delayed-Release Tablets: Loading dose: 300 mg (three 100 mg delayed-release tablets) twice a day on the first day. Maintenance dose: 300 mg (three 100 mg delayed-release tablets) once a day, starting on the second day. Noxafil Oral Suspension: 200 mg (5 mL) three times a day. |
Loading dose: 1 day Maintenance dose: Duration of therapy is based on recovery from neutropenia or immunosuppression |
| Oropharyngeal Candidiasis (OPC) | |
|
Noxafil Oral Suspension:
Loading dose: 100 mg (2.5 mL) twice a day on the first day. Maintenance dose: 100 mg (2.5 mL) once a day thereafter. |
Loading dose: 1 day Maintenance dose: 13 days |
| OPC Refractory (rOPC) to Itraconazole and/or Fluconazole | |
| Noxafil Oral Suspension: 400 mg (10 mL) twice a day. | Duration of therapy is based on the severity of the patient’s underlying disease and clinical response. |
2.6 Administration of Diluted Noxafil Infusion Solution
See Dosage and Administration (2.5) for the preparation instructions for the diluted Noxafil Solution.
Important Administration Instructions for the Diluted Noxafil Infusion Solution
- Must administer diluted Noxafil Infusion Solution through a 0.22-micron polyethersulfone (PES) or polyvinylidene difluoride (PVDF) filter.
- Administer diluted Noxafil infusion solution via a central venous line, including a central venous catheter (CVC) or peripherally inserted central catheter (PICC), by slow intravenous infusion over approximately 90 minutes [see Adverse Reactions (6.1)].
- If a CVC or PICC are not available, may administer diluted Noxafil solution once through a peripheral venous catheter by intravenous infusion over approximately 30 minutes to bridge the period during which a CVC or PICC are replaced, inserted, or unavailable for use (e.g., the CVC is being used for intravenous treatment with another product). However, do not administer diluted Noxafil infusion solution more than once via peripheral venous catheter because in clinical trials, multiple peripheral infusions given through the same vein resulted in infusion site reactions [see Adverse Reactions (6.1)].
- When multiple dosing is required, the infusion should be done via a central venous line.
Additional Administration Instructions for the Diluted Noxafil Infusion Solution
-
Administer diluted Noxafil infusion solution intravenously through the same intravenous line (or cannula) with the following compatible infusion solutions:
- 0.45% Sodium Chloride Injection
- 0.9% Sodium Chloride Injection
- 5% Dextrose Injection
- 5% Dextrose and 0.45% Sodium Chloride Injection
- 5% Dextrose and 0.9% Sodium Chloride Injection
- 5% Dextrose and 20 mEq potassium chloride Injection
-
Administer diluted Noxafil infusion solution intravenously at the same time through the same intravenous line (or cannula) with the following intravenous drug products prepared in 5% Dextrose Injection or 0.9% Sodium Chloride Injection:
- Amikacin Sulfate Injection
- Caspofungin Acetate for Injection
- Ciprofloxacin Injection
- Daptomycin for Injection
- Dobutamine Injection
- Famotidine Injection
- Filgrastim Injection
- Gentamicin Injection
- Hydromorphone Hydrochloride Injection
- Levofloxacin Injection
- Lorazepam Injection
- Meropenem for Injection
- Micafungin for Injection
- Morphine Sulfate Injection
- Norepinephrine Bitartrate Injection
- Potassium Chloride Injection
- Vancomycin Hydrochloride for Injection
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No drug-related neoplasms were recorded in rats or mice treated with posaconazole for 2 years at doses higher than the clinical dose. In a 2 year carcinogenicity study, rats were given posaconazole orally at doses up to 20 mg/kg (females), or 30 mg/kg (males). These doses are equivalent to 3.9- or 3.5 times the exposure achieved with a 400 mg twice daily oral suspension regimen, respectively, based on steady-state AUC in healthy volunteers administered a high-fat meal (400 mg twice daily oral suspension regimen). In the mouse study, mice were treated at oral doses up to 60 mg/kg/day or 4.8 times the exposure achieved with a 400 mg twice daily oral suspension regimen.
Mutagenesis
Posaconazole was not genotoxic or clastogenic when evaluated in bacterial mutagenicity (Ames), a chromosome aberration study in human peripheral blood lymphocytes, a Chinese hamster ovary cell mutagenicity study, and a mouse bone marrow micronucleus study.
Impairment of Fertility
Posaconazole had no effect on fertility of male rats at a dose up to 180 mg/kg (1.7 x the 400 mg twice daily oral suspension regimen based on steady-state plasma concentrations in healthy volunteers) or female rats at a dose up to 45 mg/kg (2.2 x the 400 mg twice daily oral suspension regimen).
2.11 Dosage Modifications in Patients With Renal Impairment
The recommended dosage of Noxafil oral suspension, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension is the same in patients with renal impairment compared to those with normal renal function.
Avoid the use of Noxafil injection in patients with eGFR less than 50 mL/minute/1.73 m2, unless an assessment of the benefit/risk to the patient justifies its use. If the decision is made to use Noxafil injection in patients with eGFR less than 50 mL/minute/1.73 m2, closely monitor serum creatinine levels, and, if increases occur, consider changing to oral Noxafil therapy. The recommended dosage of Noxafil injection in patients with eGFR 50 to 90 mL/minute/1.73 m2 is the same as those with normal renal function.
2.8 Administration Instructions for Noxafil Oral Suspension
Administer Noxafil oral suspension with a full meal or with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale) in patients who cannot eat a full meal.
For patients who cannot eat a full meal, use Noxafil delayed-release tablets instead of the Noxafil oral suspension for the prophylaxis of invasive Aspergillus and Candida infections in those who are at high risk of developing these infections due to being severely immunocompromised. This is because Noxafil delayed-release tablets provide higher plasma drug exposures than Noxafil oral suspension under fasted condition [see Dosage and Administration (2.1)].
For those patients using the Noxafil oral suspension:
- Shake Noxafil oral suspension well before use.
- Administer with measured dosing spoon provided in the package (see Figure 1).
| Figure 1: Measured dosing spoon provided in the package marked for doses of 2.5 mL and 5 mL. |
- Administer each dose of Noxafil oral suspension during or immediately (i.e., within 20 minutes) following a full meal [see Clinical Pharmacology (12.3)].
-
In patients who cannot eat a full meal and for whom Noxafil delayed-release tablets or Noxafil injection are not options, administer each dose of Noxafil oral suspension with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale). If these patients cannot tolerate an oral nutritional supplement or an acidic carbonated beverage either use:
- An alternative antifungal therapy, or
- Noxafil oral suspension and closely monitor patients for breakthrough fungal infections.
- Rinse the spoon with water after each administration and before storage.
7.2 Effects of Noxafil and Noxafil Powdermix On Other Drugs
Posaconazole is a strong CYP3A4 inhibitor. Therefore, concomitant use of Noxafil may increase plasma concentrations of drugs that are CYP3A4 substrates [see Clinical Pharmacology (12.3)].
| Digoxin | ||
|---|---|---|
| Clinical Effect(s) | Increased digoxin plasma concentrations have been reported in patients who received concomitant posaconazole and digoxin. | |
| Prevention or Management | Monitor digoxin plasma concentrations during concomitant use of posaconazole. | |
| Glipizide | ||
| Clinical Effect(s) | No dosage modification of glipizide is needed when used concomitantly with Noxafil. However, glucose concentrations decrease in some patients concomitantly administered posaconazole and glipizide. | |
| Prevention or Management | Increase monitoring of glucose concentrations when used concomitantly. | |
| CYP3A Substrates | ||
| Immunosuppressants that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Posaconazole is a strong CYP3A4 inhibitor. Therefore, plasma concentrations of CYP3A4 substrates may be increased by posaconazole use [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Sirolimus | Posaconazole is contraindicated with sirolimus [see Clinical Pharmacology (12.3)]. |
| Tacrolimus | • At initiation of posaconazole treatment, reduce the tacrolimus dosage to approximately one-third of the original tacrolimus dosage. • Frequent monitoring of tacrolimus whole blood trough concentrations should be performed during and at discontinuation of posaconazole treatment and the tacrolimus dosage should be modified accordingly [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)]. |
|
| Cyclosporine | • At initiation of posaconazole treatment reduce the cyclosporine dosage to approximately three-fourths of the original dosage. • Frequent monitoring of cyclosporine whole blood trough concentrations should be performed during and at discontinuation of posaconazole treatment and the cyclosporine dosage should be modified accordingly [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)]. |
|
| CYP3A4 Substrates that Prolong QTc Interval | ||
| Mechanism and Clinical Effect(s) | Concomitant use of posaconazole with CYP3A4 substrates such as pimozide and quinidine may result in increased plasma concentrations of the CYP3A4 substrates leading to QTc interval prolongation and torsades de pointes [see Warnings and Precautions (5.2)]. | |
| Prevention or Management | Pimozide | Concomitant use with posaconazole is contraindicated. |
| Quinidine | ||
| HMG-CoA Reductase Inhibitors (Statins) that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Concomitant use of posaconazole with simvastatin increased simvastatin plasma concentrations which can lead to rhabdomyolysis [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Atorvastatin, Lovastatin, Simvastatin | Concomitant use with posaconazole is contraindicated. |
| Benzodiazepines that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Concomitant use of posaconazole with midazolam increased midazolam plasma concentrations which could potentiate and prolong hypnotic and sedative effects [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Midazolam, Alprazolam, Triazolam | Closely monitor for adverse reactions associated with high plasma concentrations of benzodiazepines that are CYP3A4 substrates during concomitant use, and a benzodiazepine receptor antagonist should be available to reverse effects [see Warnings and Precautions (5.7)]. |
| Calcium Channel Blockers that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Posaconazole may increase the plasma concentrations of calcium channel blockers that are substrates of CYP3A4. | |
| Prevention or Management | Verapamil, Diltiazem, Nifedipine, Nicardipine, Felodipine | Monitor frequently for adverse reactions and toxicity with concomitant use of posaconazole with calcium channel blockers that are CYP3A4 substrates. Dosage reduction of the calcium channel blocker may be needed. |
| Anti-HIV Drugs that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Ritonavir and atazanavir are CYP3A4 substrates and posaconazole increased plasma concentrations of these drugs [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Ritonavir and Atazanavir | Monitor frequently for adverse reactions and toxicity of ritonavir and atazanavir during concomitant use. |
| Antineoplastic Drugs that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Posaconazole may increase plasma concentrations of oncology drugs that are CYP3A4 substrates, which may increase the risk of serious adverse reactions. | |
| Prevention or Management | Venetoclax |
CLL/SLL patients
: Concomitant use of posaconazole with venetoclax during initiation and ramp-up phase is contraindicated. AML patients : With concomitant use, venetoclax dosage reduction and safety monitoring is recommended across all dosing phases [see Warnings and Precautions (5.11)]. |
| Vinca alkaloids (e.g., vincristine, vinblastine) | Reserve concomitant use for patients with no alternative antifungal treatment options [see Warnings and Precautions (5.8)]. | |
| Ergot Alkaloids | ||
| Mechanism and Clinical Effect(s) | Most of the ergot alkaloids are CYP3A4 substrates. Posaconazole may increase the plasma concentrations of ergot alkaloids (ergotamine and dihydroergotamine) which may lead to ergotism. | |
| Prevention or Management | Ergotamine, Dihydroergotamine | Concomitant use with posaconazole is contraindicated. |
| Phenytoin | ||
| Mechanism and Clinical Effect(s) | Phenytoin is a CYP3A4 substrate. Concomitant use of posaconazole with phenytoin increased phenytoin plasma concentrations [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Avoid concomitant use of posaconazole with phenytoin unless the benefit outweighs the risk. frequently monitor phenytoin concentrations and consider a dosage reduction of phenytoin. See Table 15 for additional monitoring considerations when phenytoin affects posaconazole via UDP-glucuronosyltransferase inhibition. | |
| Rifabutin | ||
| Mechanism and Clinical Effect(s) | Rifabutin is a CYP3A4 substrate. Concomitant use of posaconazole with rifabutin increased rifabutin plasma concentrations [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Avoid concomitant use of posaconazole with rifabutin unless the benefit outweighs the risk. Frequent monitoring of full blood counts and adverse reactions due to increased rifabutin plasma concentrations (e.g., uveitis, leukopenia) during concomitant use are recommended. See Table 15 for additional monitoring considerations when rifabutin affects posaconazole via UDP-glucuronosyltransferase inhibition. |
4.3 Qt Prolongation With Concomitant Use With Cyp3a4 Substrates
Noxafil is contraindicated with CYP3A4 substrates that prolong the QT interval. Concomitant administration of Noxafil with the CYP3A4 substrates, pimozide and quinidine may result in increased plasma concentrations of these drugs, leading to QTc prolongation and cases of torsades de pointes [see Warnings and Precautions (5.2) and Drug Interactions (7.2)].
5.9 Risk in Patients With Hereditary Fructose Intolerance (hfi)
Noxafil PowderMix for delayed-release oral suspension contains sorbitol, an inactive ingredient, and may precipitate a metabolic crisis that may include, but is not limited to life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure in patients with HFI. Obtain careful history of HFI symptoms (nausea, vomiting, abdominal pain) with sorbitol/fructose/sucrose exposure prior to Noxafil PowderMix for delayed-release oral suspension administration because a diagnosis of HFI may not yet be established in pediatric patients [see Contraindications (4) and Use in Specific Populations (8.4)].
2.7 Administration Instructions for Noxafil Delayed Release Tablets
- Swallow the Noxafil delayed-release tablets whole. Do not divide, crush, or chew.
- Administer Noxafil delayed-release tablets orally with or without food [see Clinical Pharmacology (12.3)].
4.4 Hmg Coa Reductase Inhibitors Primarily Metabolized Through Cyp3a4
Coadministration with the HMG-CoA reductase inhibitors that are primarily metabolized through CYP3A4 (e.g., atorvastatin, lovastatin, and simvastatin) is contraindicated since increased plasma concentration of these drugs can lead to rhabdomyolysis [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
14.3 Treatment of Oropharyngeal Candidiasis With Noxafil Oral Suspension
Noxafil Oral Suspension Study 3 was a randomized, controlled, evaluator-blinded study in HIV-infected patients with oropharyngeal candidiasis. Patients were treated with Noxafil or fluconazole oral suspension (both Noxafil and fluconazole were given as follows: 100 mg twice a day for 1 day followed by 100 mg once a day for 13 days).
Clinical and mycological outcomes were assessed after 14 days of treatment and at 4 weeks after the end of treatment. Patients who received at least 1 dose of study medication and had a positive oral swish culture of Candida species at baseline were included in the analyses (see Table 36). The majority of the subjects had C. albicans as the baseline pathogen.
Clinical success at Day 14 (complete or partial resolution of all ulcers and/or plaques and symptoms) and clinical relapse rates (recurrence of signs or symptoms after initial cure or improvement) 4 weeks after the end of treatment were similar between the treatment arms (see Table 36).
Mycologic eradication rates (absence of colony forming units in quantitative culture at the end of therapy, Day 14), as well as mycologic relapse rates (4 weeks after the end of treatment) were also similar between the treatment arms (see Table 36).
| Noxafil | Fluconazole | |
|---|---|---|
| Clinical Success at End of Therapy (Day 14) | 155/169 (91.7%) | 148/160 (92.5%) |
| Clinical Relapse (4 Weeks after End of Therapy) | 45/155 (29.0%) | 52/148 (35.1%) |
| Mycological Eradication (absence of CFU) at End of Therapy (Day 14) | 88/169 (52.1%) | 80/160 (50.0%) |
| Mycological Relapse (4 Weeks after End of Treatment) | 49/88 (55.6%) | 51/80 (63.7%) |
Mycologic response rates, using a criterion for success as a posttreatment quantitative culture with ≤20 colony forming units (CFU/mL) were also similar between the two groups (Noxafil 68.0%, fluconazole 68.1%). The clinical significance of this finding is unknown.
2.9 Non Substitutability Between Noxafil Oral Suspension and Other Formulations
Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations [see Dosage and Administration (2.2, 2.3)].
7.3 Absence of Clinically Important Interaction With Noxafil and Noxafil Powdermix
Additional clinical studies demonstrated that no clinically important effects on zidovudine, lamivudine, indinavir, or caffeine were observed when administered with Noxafil 200 mg once daily; therefore, no dose adjustments are required for these drugs when coadministered with Noxafil 200 mg once daily.
No clinically relevant effects on the pharmacokinetics of Noxafil delayed-release tablets were observed during concomitant use with antacids, H2-receptor antagonists and proton pump inhibitors, and metoclopramide [see Clinical Pharmacology (12.3)]. No dosage adjustment of Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension is required during concomitant use with these drugs.
No clinically relevant effects on the pharmacokinetics of Noxafil oral suspension were observed during concomitant use with antacids, H2-receptor antagonists (other than cimetidine), and loperamide [see Clinical Pharmacology (12.3)]. No dosage adjustment of Noxafil oral suspension is required during concomitant use with these drugs (other than cimetidine).
14.1 Treatment of Invasive Aspergillosis With Noxafil Injection and Noxafil Delayed Release Tablets
Aspergillosis Treatment Study (NCT01782131) was a randomized, double-blind, controlled trial which evaluated the safety and efficacy of Noxafil injection and Noxafil delayed-release tablets versus voriconazole for primary treatment of invasive fungal disease caused by Aspergillus species. Eligible patients had proven, probable, or possible invasive fungal infections per the European Organization for Research and Treatment of Cancer/Mycoses Study Group, EORTC/MSG criteria. Patients were stratified by risk for mortality or poor outcome where high risk included a history of allogeneic bone marrow transplant, liver transplant, or relapsed leukemia undergoing salvage chemotherapy. The median age of patients was 57 years (range: 14-91 years), with 27.8% of patients aged ≥65 years; 5 patients were pediatric patients 14-16 years of age, of whom 3 were treated with Noxafil and 2 with voriconazole. The majority of patients were male (59.8%) and white (67.1%). With regard to risk factors for invasive aspergillosis, approximately two-thirds of the patients in the study had a recent history of neutropenia, while approximately 20% with a history of an allogeneic stem cell transplant. Over 80% of subjects in each treatment group had infection limited to the lower respiratory tract (primarily lung), while approximately 11% to 13% also had infection in another organ. Invasive aspergillosis was proven or probable in 58.1% of patients as classified by independent adjudicators blinded to study treatment assignment. At least one Aspergillus species was identified in 21% of the patients; A. fumigatus and A. flavus were the most common pathogens identified.
Patients randomized to receive Noxafil were given a dose of 300 mg once daily (twice daily on Day 1) IV or tablet. Patients randomized to receive voriconazole were given a dose of 6 mg/kg twice daily Day 1 followed by 4 mg/kg twice daily IV, or oral 300 mg twice daily Day 1 followed by 200 mg twice daily. The recommended initial route of administration was IV; however, patients could begin oral therapy if clinically stable and able to tolerate oral dosing. The transition from IV to oral therapy occurred when the patient was clinically stable. The protocol recommended duration of therapy was 84 days with a maximum allowed duration of 98 days. Median treatment duration was 67 days for Noxafil patients and 64 days for voriconazole patients. Overall, 55% to 60% of patients began treatment with the IV formulation with a median duration of 9 days for the initial IV dosing.
The Intent to Treat (ITT) population included all patients randomized and receiving at least one dose of study treatment. All-cause mortality through Day 42 in the overall population (ITT) was 15.3% for Noxafil patients compared to 20.6% for voriconazole patients for an adjusted treatment difference of -5.3% with a 95% confidence interval of -11.6 to 1.0%. Consistent results were seen in patients with proven or probable invasive aspergillosis per EORTC criteria (see Table 32).
| Noxafil Injection and Delayed-Release Tablets | Voriconazole | ||||
| Population | N | n (%) | N | n (%) | Difference Adjusted treatment difference based on Miettinen and Nurminen’s method stratified by randomization factor (risk for mortality/poor outcome), using Cochran-Mantel-Haenszel weighting scheme. (95% CI) |
| Intent to Treat | 288 | 44 (15.3) | 287 | 59 (20.6) | -5.3 (-11.6, 1.0) |
| Proven/Probable Invasive Aspergillosis | 163 | 31 (19.0) | 171 | 32 (18.7) | 0.3 (-8.2, 8.8) |
Global clinical response at Week 6 was assessed by a blinded, independent adjudication committee based upon prespecified clinical, radiologic, and mycologic criteria. In the subgroup of patients with proven or probable invasive aspergillosis per EORTC criteria, the global clinical response of success (complete or partial response) at Week 6 was seen in 44.8% for Noxafil-treated patients compared to 45.6% for voriconazole-treated patients (see Table 33).
| Posaconazole | Voriconazole | ||||
| Population | N | Success | N | Success | Difference Adjusted treatment difference based on Miettinen and Nurminen’s method stratified by randomization factor (risk for mortality/poor outcome), using Cochran-Mantel-Haenszel weighting scheme. (95% CI) |
| Proven/Probable Invasive Aspergillosis | 163 | 73 (44.8) | 171 | 78 (45.6) | -0.6 (-11.2, 10.1) |
2.10 Preparation and Administration Instructions for Noxafil Powdermix for Delayed Release Oral Suspension
For details on preparation and administration of Noxafil PowderMix for delayed-release oral suspension, see Instructions for Use.
Preparation Instructions for Noxafil PowderMix for Delayed-Release Oral Suspension
- Do not open the Noxafil PowderMix packet until ready to prepare the drug.
- Remove cap from the mixing liquid and push the bottle adapter into the neck of the bottle. Once in place, the bottle adapter stays in the bottle.
- Remove 9 mL of mixing liquid using the provided blue syringe. Put the cap back on the bottle. Only use the mixing liquid in the kit to prepare Noxafil PowderMix.
- Using the provided mixing cup, combine 9 mL of mixing liquid and the entire contents of one packet in the Noxafil PowderMix kit and mix (containing 300 mg of posaconazole).
- Shake the mixing cup vigorously for 45 seconds to mix the powder and mixing liquid from the Noxafil PowderMix kit. The final concentration of the reconstituted Noxafil PowderMix delayed-release suspension is approximately 30 mg/mL of posaconazole. Check to make sure the powder is mixed (the mixture should look cloudy and free of clumps).
- Must use the reconstituted Noxafil PowderMix delayed-release suspension within 1 hour of reconstitution. Discard unused portion of the reconstituted Noxafil PowderMix delayed-release suspension.
Administration Instructions for Noxafil PowderMix Delayed-Release Reconstituted Suspension
- To ensure delivery of the correct reconstituted Noxafil PowderMix Delayed-release dose, only use the provided notched tip syringes for preparation and administration because its design reduces the risk of aggregation of the product during preparation and administration.
-
Choose the correct syringe based on the prescribed Noxafil PowderMix dose:
- Use 3 mL (green) notched tip syringe (provided with the kit) if dose is 3 mL or less.
- Use 10 mL (blue) notched tip syringe (provided with the kit) if dose is more than 3 mL.
- Administer reconstituted Noxafil PowderMix suspension orally with food within 1 hour of reconstitution [see Clinical Pharmacology (12.3)].
- The maximum dose that can be accurately withdrawn from the mixing cup after reconstitution is 240 mg (8 mL).
Discarding Unused Reconstituted Noxafil PowderMix Suspension and Reuse of Syringes
- Not all the reconstituted Noxafil PowderMix suspension in the mixing cup will be used; there will be some left over in the mixing cup.
- Discard any remaining reconstituted Noxafil PowderMix suspension.
- The mixing cup may be hand washed and reused. Alternatively, the mixing cup may be discarded, and a similar mixing cup with a lid may be used for subsequent doses.
- The notched tip syringes may be hand washed and reused.
4.7 Use of Noxafil Powdermix for Delayed Release Oral Suspension in Patients With Hereditary Fructose Intolerance
Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with known or suspected hereditary fructose intolerance (HFI) [see Warnings and Precautions (5.9) and Use in Specific Populations (8.4)].
2.3 Recommended Dosage of Noxafil for the Treatment of Invasive Aspergillosis and Prophylaxis of Invasive Aspergillus
Noxafil injection and Noxafil delayed-release tablets
The recommended dosage of (1) Noxafil injection in pediatric patients 2 years of age and older who weigh 10 kg or greater, and (2) Noxafil delayed-release tablets in pediatric patients 2 years of age and older who weigh greater than 40 kg for the treatment of invasive aspergillosis and prophylaxis of invasive Aspergillus and Candida infections is shown in Table 2 [see Dosage and Administration (2.5, 2.6, 2.7, 2.9) and Clinical Pharmacology (12.3)].
Noxafil delayed-release tablets are not recommended for use in pediatric patients who weigh 40 kg or less because the recommended dosage cannot be achieved with this dosage form.
| Recommended Pediatric Dosage by Formulation | Duration of Therapy |
|---|---|
|
Noxafil Injection (patients weighing 10 kg or greater):
Loading dose:
Maintenance dose:
Noxafil Delayed-Release Tablets (patients weighing greater than 40 kg):
|
Treatment of invasive aspergillosis:
Prophylaxis of invasive Aspergillus and Candida infections:
|
Noxafil Oral Suspension
The recommended dosage of Noxafil oral suspension in pediatric patients 13 years of age and older for the prophylaxis of invasive Aspergillus and Candida Infections is shown in Table 3.
| Recommended Pediatric Dosage of Noxafil Oral Suspension | Duration of Therapy |
|---|---|
|
200 mg (5 mL) three times a day |
Duration of therapy is based on recovery from neutropenia or immunosuppression. |
Noxafil PowderMix
The recommended dosage of Noxafil PowderMix for delayed-release oral suspension in pediatric patients 2 years of age and older who weigh 10 kg to 40 kg, for the treatment of invasive aspergillosis, and the prophylaxis of invasive Aspergillus and Candida infections, is shown in Table 4 and Table 5. The dosing for these indications is the same, except for patients weighing 10 to less than 12 kg [see Dosage and Administration (2.9, 2.10) and Clinical Pharmacology (12.3)].
Noxafil PowderMix for delayed-release oral suspension is not recommended for use in pediatric patients who weigh greater than 40 kg because the recommended dosage cannot be achieved with this dosage form.
| Weight (kg) | Recommended Pediatric Dosage of Noxafil PowderMix for Delayed-Release Oral Suspension | Duration of Therapy |
|---|---|---|
| 10 to less than 17 |
Loading Dose:
120 mg (4 mL) twice daily on the first day Maintenance Dose: 120 mg (4 mL) once daily |
Recommended total duration of therapy is 6 to 12 weeks. |
| 17 to less than 21 |
Loading Dose:
150 mg (5 mL) twice daily on the first day Maintenance Dose: 150 mg (5 mL) once daily |
|
| 21 to less than 26 |
Loading Dose:
180 mg (6 mL) twice daily on the first day Maintenance Dose: 180 mg (6 mL) once daily |
|
| 26 to less than 36 |
Loading Dose:
210 mg (7 mL) twice daily on the first day Maintenance Dose: 210 mg (7 mL) once daily |
|
| 36 to 40 |
Loading Dose:
240 mg (8 mL) twice daily on the first day Maintenance Dose: 240 mg (8 mL) once daily |
| Weight (kg) | Recommended Pediatric Dosage of Noxafil PowderMix for Delayed-Release Oral Suspension | Duration of Therapy |
|---|---|---|
| 10 to less than 12 |
Loading Dose:
90 mg (3 mL) twice daily on the first day Maintenance Dose: 90 mg (3 mL) once daily |
Duration of therapy is based on recovery from neutropenia or immunosuppression. |
| 12 to less than 17 |
Loading Dose:
120 mg (4 mL) twice daily on the first day Maintenance Dose: 120 mg (4 mL) once daily |
|
| 17 to less than 21 |
Loading Dose:
150 mg (5 mL) twice daily on the first day Maintenance Dose: 150 mg (5 mL) once daily |
|
| 21 to less than 26 |
Loading Dose:
180 mg (6 mL) twice daily on the first day Maintenance Dose: 180 mg (6 mL) once daily |
|
| 26 to less than 36 |
Loading Dose:
210 mg (7 mL) twice daily on the first day Maintenance Dose: 210 mg (7 mL) once daily |
|
| 36 to 40 |
Loading Dose:
240 mg (8 mL) twice daily on the first day Maintenance Dose: 240 mg (8 mL) once daily |
1.3 Treatment of Oropharyngeal Candidiasis Including Oropharyngeal Candidiasis Refractory to Itraconazole And/or Fluconazole
Noxafil oral suspension is indicated for the treatment of oropharyngeal candidiasis, including oropharyngeal candidiasis refractory to itraconazole and/or fluconazole in adults and pediatric patients 13 years of age and older.
14.4 Noxafil Oral Suspension Treatment of Oropharyngeal Candidiasis Refractory to Treatment With Fluconazole Or Itraconazole
Noxafil Oral Suspension Study 4 was a noncomparative study of Noxafil oral suspension in HIV-infected subjects with OPC that was refractory to treatment with fluconazole or itraconazole. An episode of OPC was considered refractory if there was failure to improve or worsening of OPC after a standard course of therapy with fluconazole greater than or equal to 100 mg/day for at least 10 consecutive days or itraconazole 200 mg/day for at least 10 consecutive days and treatment with either fluconazole or itraconazole had not been discontinued for more than 14 days prior to treatment with Noxafil. Of the 199 subjects enrolled in this study, 89 subjects met these strict criteria for refractory infection.
Forty-five subjects with refractory OPC were treated with Noxafil oral suspension 400 mg twice daily for 3 days, followed by 400 mg once daily for 25 days with an option for further treatment during a 3 month maintenance period. Following a dosing amendment, a further 44 subjects were treated with posaconazole 400 mg twice daily for 28 days. The efficacy of Noxafil was assessed by the clinical success (cure or improvement) rate after 4 weeks of treatment. The clinical success rate was 74.2% (66/89). The clinical success rates for both the original and the amended dosing regimens were similar (73.3% and 75.0%, respectively).
2.4 Recommended Dosage of Noxafil Oral Suspension for the Treatment of Oropharyngeal Candidiasis in Pediatric Patients 13 Years of Age and Older
The recommended dosage of Noxafil oral suspension for the treatment of oropharyngeal candidiasis (OPC) and OPC refractory (rOPC) to itraconazole and/or fluconazole in pediatric patients 13 years of age and older is shown in Table 6.
The Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension products are not approved for the treatment of oropharyngeal candidiasis in pediatric patients.
| Recommended Pediatric Dosage of Noxafil Oral Suspension | Duration of Therapy |
|---|---|
| Oropharyngeal Candidiasis (OPC) | |
|
Loading Dose:
Maintenance Dose:
|
Loading dose:
Maintenance dose:
|
| OPC Refractory (rOPC) to Itraconazole and/or Fluconazole | |
|
400 mg (10 mL) twice daily |
Duration of therapy is based on the severity of the patient’s underlying disease and clinical response. |
Structured Label Content
Section 42229-5 (42229-5)
Treatment of Invasive Aspergillosis in Adults and Adolescents (Noxafil injection and Noxafil Delayed-Release Tablets)
The safety of Noxafil injection and Noxafil delayed-release tablets was assessed in a randomized, double-blind, active-controlled clinical study of Noxafil injection and Noxafil delayed-release tablets versus voriconazole for treatment of invasive aspergillosis (Aspergillosis Treatment Study). A total of 575 adult and pediatric patients 14 years of age and older (288 in the Noxafil group, 287 in voriconazole group (voriconazole for injection or voriconazole tablets)) with proven, probable or possible invasive aspergillosis were included. The median duration of treatment was 67 days for Noxafil injection or Noxafil delayed-release tablets and 64 days for voriconazole. In this study, 55% to 60% of patients started intravenous treatment with Noxafil (Noxafil injection) or voriconazole (voriconazole for injection). The median duration of the first instance of intravenous treatment (before switching to oral treatment or discontinuing or completing study treatment) was 9 days for both groups. Table 7 presents adverse reactions reported at an incidence of ≥10% in either one of the treatment groups in the Aspergillosis Treatment Study.
Adverse reactions leading to treatment discontinuation were reported for 34% of patients. The most commonly reported adverse reactions (>2% of patients) leading to treatment discontinuation were septic shock, respiratory failure, and bronchopulmonary aspergillosis in the Noxafil group, and septic shock and acute myeloid leukemia in the voriconazole group. The most frequently reported adverse reactions in the Noxafil-treated group were pyrexia (28%), hypokalemia (28%), and nausea (23%).
| Adverse Reactions | Noxafil injection or Noxafil delayed-release tablets n=288 (%) |
Voriconazole for injection or Voriconazole tablets n=287 (%) |
|---|---|---|
| Percentage of Patients Reporting any Adverse Reaction | 97.6 | 97.6 |
| Hypokalemia | 28.5 | 17.1 |
| Pyrexia | 28.1 | 25.1 |
| Nausea | 22.6 | 17.8 |
| Diarrhea | 18.1 | 18.1 |
| Vomiting | 18.1 | 13.6 |
| Alanine aminotransferase increased | 14.6 | 12.9 |
| Febrile neutropenia | 14.6 | 13.2 |
| Aspartate aminotransferase increased | 13.2 | 12.5 |
| Pneumonia | 12.5 | 9.1 |
| Headache | 12.2 | 8.7 |
| Constipation | 11.1 | 8.0 |
| Edema peripheral | 11.1 | 8.4 |
| Epistaxis | 11.1 | 5.9 |
| Cough | 10.4 | 8.4 |
| Abdominal pain | 10.1 | 8.4 |
| Hypomagnesemia | 10.1 | 6.3 |
Clinical Trial Experience with Noxafil Injection for Prophylaxis of Invasive Aspergillus and Candida Infections
Administration of multiple doses of Noxafil injection via a peripheral venous catheter were associated with thrombophlebitis (60% incidence). Therefore, in subsequent studies, Noxafil injection was administered via central venous catheter [see Dosage and Administration (2.6)].
The safety of Noxafil injection has been assessed in 268 patients in a clinical trial. Patients were enrolled in a non-comparative pharmacokinetic and safety trial of Noxafil injection when given as antifungal prophylaxis (Noxafil Injection Study). Patients were immunocompromised with underlying conditions including hematological malignancy, neutropenia post-chemotherapy, GVHD, and post HSCT. This patient population was 55% male, had a mean age of 51 years (range: 18-82 years, 19% of patients were ≥65 years of age), and were 95% White and 8% Hispanic. In this study, 10 patients received a single dose of 200 mg Noxafil injection, 21 patients received 200 mg daily dosage for a median of 14 days, and 237 patients received 300 mg daily dosage for a median of 9 days (the 200 mg dosage is not a recommended dosage for prophylaxis of invasive Aspergillus and Candida infections in adults [see Dosage and Administration (2.2)]. In the 300 mg daily dosage group each patient received a loading intravenous dose of Noxafil injection 300 mg twice on Day 1, then intravenous Noxafil injection therapy, and finally Noxafil oral suspension to complete 28 days of total Noxafil therapy.
Table 8 presents adverse reactions observed in patients treated with the Noxafil injection 300 mg daily dosage group in the Noxafil Injection Study.
The most frequently reported adverse reactions with an onset during the intravenous Noxafil injection phase of dosing with 300 mg once daily were diarrhea (32%), hypokalemia (22%), pyrexia (21%), and nausea (19%). These adverse reactions were consistent with those seen in studies with Noxafil oral suspension.
| Adverse Reactions | Noxafil Injection Treatment Phase n=237 Adverse reactions reported in patients with an onset during the Noxafil intravenous dosing phase of the study.
(%) |
Noxafil Injection Treatment Phase or Subsequent Noxafil Oral Suspension Treatment Phase n=237 Adverse reactions reported with an onset at any time during the study in patients who were treated for up to 28 days of Noxafil therapy.
(%) |
|---|---|---|
| Percentage of Patients Reporting any Adverse Reaction | 93 | 99 |
| Diarrhea | 32 | 39 |
| Hypokalemia | 22 | 28 |
| Pyrexia | 21 | 31 |
| Nausea | 19 | 30 |
| Rash | 15 | 24 |
| Headache | 14 | 21 |
| Epistaxis | 14 | 17 |
| Abdominal Pain | 13 | 17 |
| Chills | 12 | 16 |
| Edema Peripheral | 12 | 15 |
| Vomiting | 12 | 19 |
| Hypomagnesemia | 11 | 13 |
| Decreased appetite | 10 | 12 |
| Cough | 9 | 13 |
| Constipation | 8 | 13 |
| Fatigue | 8 | 10 |
| Hypertension | 8 | 11 |
| Petechiae | 8 | 10 |
| Anemia | 7 | 10 |
| Dyspnea | 7 | 10 |
| Thrombocytopenia | 7 | 11 |
| Abdominal Pain Upper | 6 | 11 |
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 1/2026 | ||
|
Patient Information NOXAFIL ® (NOX-a-fil) (posaconazole) injection NOXAFIL ® (NOX-a-fil) (posaconazole) delayed-release tablets NOXAFIL ® (NOX-a-fil) (posaconazole) oral suspension NOXAFIL ® (NOX-a-fil) POWDERMIX (posaconazole) for delayed-release oral suspension |
||
|
What is Noxafil and Noxafil PowderMix?
Noxafil injection is used for:
Noxafil delayed-release tablets are used for:
Noxafil oral suspension is used for:
Noxafil PowderMix for delayed-release oral suspension is used for:
Noxafil oral suspension is also used to treat a fungal infection called “thrush” caused by Candida in your mouth or throat area. Noxafil oral suspension can be used as the first treatment for thrush, or as another treatment for thrush after itraconazole or fluconazole treatment has not worked. Noxafil oral suspension is for adults and children 13 years of age and older. It is not known if Noxafil oral suspension is safe and effective in children under 13 years of age for the treatment of thrush as the first treatment for thrush, or as another treatment for thrush after itraconazole or fluconazole treatment has not worked. It is not known if Noxafil or Noxafil PowderMix is safe and effective in children under 2 years of age. |
||
Do not take Noxafil or Noxafil PowderMix if you:
Do not start taking a new medicine without talking to your healthcare provider or pharmacist. |
||
Before you take Noxafil or Noxafil PowderMix, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Noxafil and Noxafil PowderMix can affect the way other medicines work, and other medicines can affect the way Noxafil and Noxafil PowderMix work, and can cause serious side effects.
Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure. |
||
|
How should I take Noxafil or Noxafil PowderMix?
Follow the instructions from your healthcare provider on how much Noxafil or Noxafil PowderMix you should take and when to take it. |
||
|
What are the possible side effects of Noxafil or Noxafil PowderMix? Noxafil or Noxafil PowderMix may cause serious side effects, including:
|
||
The most common side effects of Noxafil in adults include: |
||
|
|
|
|
The most common side effects of Noxafil injection and Noxafil PowderMix in children include: |
||
|
|
|
|
If you take Noxafil delayed-release tablets, Noxafil oral suspension, or Noxafil PowderMix for delayed-release oral suspension, tell your healthcare provider right away if you have diarrhea or vomiting. |
||
|
How should I store Noxafil or Noxafil PowderMix? Noxafil injection
Noxafil delayed-release tablets
Noxafil oral suspension
Noxafil PowderMix for delayed-release oral suspension
Safely throw away medicine that is out of date or no longer needed. Keep Noxafil and Noxafil PowderMix and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of Noxafil and Noxafil PowderMix. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Noxafil or Noxafil PowderMix for a condition for which it was not prescribed. Do not give Noxafil or Noxafil PowderMix to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Noxafil or Noxafil PowderMix that is written for health professionals. |
||
|
What are the ingredients in Noxafil and Noxafil PowderMix?
|
||
|
Manuf. for: Merck Sharp & Dohme LLC |
Section 43683-2 (43683-2)
Section 59845-8 (59845-8)
|
Important: Read This Booklet First
Noxafil PowderMix (posaconazole) for delayed-release oral suspension Instructions for Use for caregivers and healthcare providers of toddlers and children |
| Before You Start |
|
| Note: Put your child in a safe place. You will need both hands to prepare Noxafil PowderMix. Wash your hands with soap and water before preparing Noxafil PowderMix. |
| Before You Start |
- The amount of Noxafil PowderMix you give depends on your child’s weight. Your healthcare provider will tell you the right dose to give your child. Be sure to keep your healthcare provider’s appointments so you get new dosing information as your child grows.
- This booklet tells you how to:
- Make the Noxafil PowderMix into a liquid form.
- Measure the right dose using the included notched-tip
oral syringe. Only use the notched-tip syringes supplied
with the kit. Do not use other oral syringes. - Give the Noxafil PowderMix to your child.
- Clean up.
| Note before adding Noxafil PowderMix: Make sure you and your child are ready! If you do not use Noxafil PowderMix within 1 hour, you will need to throw it away and start over. |
| Note: If you have any questions, call your healthcare provider. |
|
|
||
|
|
||
|
|
||
|
|
| 2 blue (10 mL) syringes | 2 green (3 mL) syringes |
|
The kit has an extra cup and set of syringes in case one is lost or damaged.
Do not use any damaged cups or syringes. |
|
- Look for the number on the measuring scale that matches
the amount of mixing liquid or Noxafil PowderMix that
you need. - Be sure to follow the directions in this booklet to remove
air bubbles from the syringe. Air bubbles can affect the
amount of medicine that the child gets.
| Step 1. Get the mixing liquid ready. |
| Important: Noxafil PowderMix needs to be prepared using the mixing liquid. Do not mix Noxafil PowderMix with milk, juice, or water. |
When you use the mixing liquid for the first time:
- Open the bottle and remove the safety seal.
- Place the bottle adapter on top of the bottle with
the small hole facing up. - Push the bottle adapter all the way down.
- After it is in place, the bottle adapter stays in the bottle.
- Put the cap back on the bottle.
|
Step 2. Gather all your supplies and put on a clean surface. |
| Note: Put your child in a safe place. You will need both hands to prepare Noxafil PowderMix. Wash your hands with soap and water before preparing Noxafil PowderMix. |
|
1 mixing cup
(Using the tab on the mixing cup, pull open the lid.) |
1 packet of Noxafil PowderMix powder |
Mixing liquid |
2 syringes
(Have 1 of each size ready, but you may only use 1, depending on the dose.) |
Scissors
(not included with kit: use sharp household scissors or kitchen scissors) |
|
The Noxafil PowderMix box has a mixing cup holder inside to help tilt the cup when you are measuring the dose. |
|
Step 3. Add Noxafil PowderMix to the mixing cup. |
|
Note before adding Noxafil PowderMix:
Make sure you and your child are ready! If you do not use Noxafil PowderMix within 1 hour, you will need to throw it away and start over. |
|
|
|
| Step 4. Shake the mixing liquid bottle. |
|
| Step 5. Fill the blue syringe with 9 mL of mixing liquid. |
|
| Step 6. Check for air bubbles. |
|
|
|
|
Step 7. Add the 9 mL of mixing liquid to the Noxafil PowderMix. |
|
| Step 8. Mix the Noxafil PowderMix. |
|
|
|
| Step 9. Check your prescription label. |
|
| Note: The dose may change each time you go to the healthcare provider, so make sure you have all the recent information. Be sure to go to all of your child's healthcare provider appointments so your child gets the right dose. |
| Step 10. Choose the syringe you need. |
| Note: Only use the syringes provided in the kit. |
Choose the correct syringe for your child's dose:
| For 1 mL
to 3 mL Green |
For 3 mL
to 10 mL Blue |
- Then find the mL mark on the syringe that matches your
child's dose.
| Step 11. Measure the Noxafil PowderMix. |
|
| Important: You will not use all of the Noxafil PowderMix. There will be some left over in the mixing cup. |
| Step 12. Check for air bubbles. |
|
|
|
|
Step 13. Give the Noxafil PowderMix to your child. |
|
Important:
|
| Step 14. Clean the cup and syringes. |
| Note: Syringes and mixing cups should be reused. Do not throw away syringes and mixing cups provided until all the Noxafil PowderMix packets are used. |
|
|
Step 15. After all packets of Noxafil PowderMix have been used. |
- After you have used the last Noxafil PowderMix packet
in this box, you will have leftover mixing liquid in the
bottle. Throw away the leftover mixing liquid and all
components of the kit.
How should I store Noxafil PowderMix?
- Store the entire kit at room temperature between 68°F to
77°F (20°C to 25°C) in a clean, dry place. - Keep the mixing liquid at room temperature.
- Do not open the Noxafil PowderMix packet until you are
ready to mix a dose.
Keep NOXAFIL PowderMix and all medicines out of the reach of children.
For more information go to www.Noxafil.com or call
1-800-622-4477.
Distributed by:
Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent
Copyright © 2021-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
ifu-mk5592-2209r002
This Instructions for Use has been approved by the U.S. Food and
Drug Administration.
Revised: September 2022
8.8 Sex
No adjustment in the dosage of Noxafil is necessary based on sex.
8.9 Race
No adjustment in the dosage of Noxafil is necessary based on race.
8.10 Weight
Pharmacokinetic modeling suggests that patients who weigh greater than 120 kg may have lower posaconazole plasma drug exposure. Therefore, consider closely monitoring for breakthrough fungal infections particularly when using Noxafil oral suspension in patients weighing greater than 120 kg [see Clinical Pharmacology (12.3)].
10 Overdosage (10 OVERDOSAGE)
There is no experience with overdosage of Noxafil injection and Noxafil delayed-release tablets.
During the clinical trials, some patients received Noxafil oral suspension up to 1,600 mg/day with no adverse reactions noted that were different from the lower doses. In addition, accidental overdose was noted in one patient who took 1200 mg twice daily Noxafil oral suspension for 3 days. No related adverse reactions were noted by the investigator.
Posaconazole is not removed by hemodialysis.
11 Description (11 DESCRIPTION)
Noxafil and Noxafil PowderMix contain posaconazole, an azole antifungal agent.
Posaconazole is designated chemically as 4-[4-[4-[4-[[ (3R,5R)-5- (2,4-difluorophenyl)tetrahydro-5- (1H-1,2,4-triazol-1-ylmethyl)-3-furanyl]methoxy]phenyl]-1-piperazinyl]phenyl]-2-[(1S,2S)-1-ethyl-2-hydroxypropyl]-2,4-dihydro-3H-1,2,4-triazol-3-one with an empirical formula of C37H42F2N8O4 and a molecular weight of 700.8. The chemical structure is:
Posaconazole is a white powder with a low aqueous solubility.
Noxafil (posaconazole) Injection
Noxafil injection, for intravenous use, is a clear colorless to yellow, without preservatives sterile liquid essentially free of foreign matter. Each vial contains 300 mg of posaconazole and the following inactive ingredients: 6.68 g Betadex Sulfobutyl Ether Sodium (SBECD), 0.0033 g edetate disodium, hydrochloric acid and sodium hydroxide to adjust the pH to 2.6, and water for injection.
Noxafil (posaconazole) Delayed-Release Tablets
Noxafil delayed-release tablet, for oral use, is yellow, coated, and oblong and contains 100 mg of posaconazole. Each delayed-release tablet contains the following inactive ingredients: croscarmellose sodium, hydroxypropylcellulose, hypromellose acetate succinate, iron oxide yellow, Macrogol/PEG 3350, magnesium stearate, microcrystalline cellulose, polyvinyl alcohol partially hydrolyzed, silicon dioxide, talc, and titanium dioxide.
Noxafil (posaconazole) Oral Suspension
Noxafil oral suspension is a white, cherry-flavored immediate-release suspension that contains 40 mg of posaconazole per mL and the following inactive ingredients: artificial cherry flavor, citric acid monohydrate, glycerin, liquid glucose, polysorbate 80, purified water, simethicone, sodium benzoate, sodium citrate dihydrate, titanium dioxide, and xanthan gum.
Noxafil PowderMix (posaconazole) for Delayed-Release Oral Suspension
Noxafil PowderMix for delayed-release oral suspension is supplied as a component of a kit. Each kit contains Noxafil as an off-white to yellowish powder for delayed-release oral suspension, a bottle of mixing liquid, two 3 mL (green) notched tip syringes, two 10 mL (blue) notched tip syringes, two mixing cups, and one bottle adapter for the mixing liquid bottle.
- Noxafil PowderMix for delayed-release oral suspension contains 300 mg of posaconazole and the following inactive ingredient: hypromellose acetate succinate.
- The mixing liquid contains: anhydrous citric acid, antifoam Af emulsion, berry citrus sweet flavor, carboxymethylcellulose sodium, carrageenan calcium sulfate trisodium phosphate, glycerin, methylparaben, microcrystalline cellulose, potassium sorbate, propylparaben, purified water, sodium citrate, sodium phosphate monobasic monohydrate, sodium saccharin, sorbitol solution, and xanthan gum.
16.1 How Supplied
Noxafil Injection
Noxafil injection is a clear, colorless to yellow sterile liquid in single-dose Type I glass vials closed with bromobutyl rubber stopper and aluminum seal containing 300 mg of posaconazole in 16.7 mL of solution (18 mg of posaconazole per mL) (NDC 0085-4331-01).
Noxafil Delayed-Release Tablets
Noxafil delayed-release tablets are yellow, coated, oblong, debossed with "100" on one side containing 100 mg of posaconazole. Bottles with child-resistant closures of 60 delayed-release tablets (NDC 0085-4324-02).
Noxafil Oral Suspension
Noxafil oral suspension is a white, cherry-flavored oral suspension in 4-ounce (123 mL) amber glass bottles with child-resistant closures containing 105 mL of suspension (40 mg of posaconazole per mL).Supplied with each oral suspension bottle is a plastic dosing spoon calibrated for measuring 2.5 mL and 5 mL doses (NDC 0085-1328-01).
Noxafil PowderMix for Delayed-Release Oral Suspension
Noxafil PowderMix for delayed-release oral suspension is supplied as:
- Package A: a kit with 8 child-resistant single-use packets of Noxafil PowderMix for delayed-release oral suspension 300 mg, two 3 mL (green) notched tip syringes, two 10 mL (blue) notched tip syringes, two mixing cups, one mixing liquid bottle, one bottle adapter for the mixing liquid bottle and Instructions for Use.
- Package B: a box of six 3 mL (green) and six 10 mL (blue) notched tip syringes for additional supply.
- Packages A and B are supplied separately.
NDC 0085-2224-02 unit of use carton with 8 packets.
NDC 0085-2224-01 individual packet.
8.4 Pediatric Use
The three Noxafil dosage forms (injection, delayed-release tablets, oral suspension) and one Noxafil PowderMix (for delayed-release oral suspension) dosage form are different products; are approved for different pediatric indications, age groups, and weights; have different dosing regimens; and have different preparation and administration instructions. Therefore, select the recommended dosage form based on the pediatric indication, age group, and weight [see Dosage and Administration (2.1)].
Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with HFI. Because a diagnosis of HFI may not yet be established in pediatric patients, obtain a careful history of HFI symptoms with sorbitol/fructose/sucrose exposure prior to administration of Noxafil PowderMix for delayed-release oral suspension [see Warnings and Precautions (5.9)].
Treatment of Invasive Aspergillosis
The safety and effectiveness of Noxafil (injection and delayed-release tablets) have been established for the treatment of invasive aspergillosis in pediatric patients 2 years of age and older .
The safety and effectiveness of Noxafil PowderMix have been established for the treatment of invasive aspergillosis in pediatric patients 2 years of age and older who weigh 10 kg to 40 kg.
Use of Noxafil and Noxafil PowderMix for these pediatric indications is supported by evidence from adequate and well-controlled studies of Noxafil in adults and safety and pharmacokinetic (PK) data from two pediatric studies [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)]. The safety of Noxafil and Noxafil PowderMix in pediatric patients for these pediatric indications was consistent with the known safety profile of Noxafil in adults [see Adverse Reactions (6.1)].
Noxafil PowderMix for delayed-release oral suspension is not recommended for use in patients who weigh greater than 40 kg because the recommended dosage cannot be achieved with this dosage form.
The safety and effectiveness of Noxafil and Noxafil PowderMix have not been established in pediatric patients less than 2 years of age.
Prophylaxis of Invasive Aspergillus and Candida Infections
The safety and effectiveness of Noxafil (injection and delayed-release tablets) have been established for the prophylaxis of invasive Aspergillus and Candida infections in pediatric patients 2 years of age and older who are at high risk of developing these infections due to being severely immunocompromised.
The safety and effectiveness of Noxafil oral suspension have been established for the prophylaxis of invasive Aspergillus and Candida infections in pediatric patients 13 years of age and older who are at high risk of developing these infections due to being severely immunocompromised.
The safety and effectiveness of Noxafil PowderMix have been established for the prophylaxis of invasive Aspergillus and Candida infections in pediatric patients 2 years of age and older who weigh 10 kg to 40 kg who are at high risk of developing these infections due to being severely immunocompromised.
Use of Noxafil and Noxafil PowderMix for these pediatric indications is supported by adequate and well controlled studies of Noxafil in adults and pediatric patients aged 13 years of age and older and additional PK and safety data in pediatric patients 2 years of age and older [see Clinical Pharmacology (12.3) and Clinical studies (14)].
Noxafil PowderMix is not recommended for use in patients who weigh greater than 40 kg because the recommended dosage cannot be achieved with this dosage form.
The safety and effectiveness of Noxafil and Noxafil PowderMix have not been established in pediatric patients less than 2 years of age.
Treatment of Oropharyngeal Candidiasis, including Refractory to Itraconazole and/or Fluconazole
The safety and effectiveness of Noxafil oral suspension have been established for the treatment of oropharyngeal candidiasis (OPC), including OPC refractory (rOPC) to itraconazole and/or fluconazole in pediatric patients 13 years of age and older.
Use of Noxafil oral suspension for this pediatric indication is supported by adequate and well controlled studies in adults and pediatric patients 13 years of age and older [see Clinical studies (14.4)].
The Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix products are not approved for the treatment of oropharyngeal candidiasis in pediatric patients. Noxafil Oral Suspension is the only dosage form approved for the treatment of OPC and rOPC in pediatric patients [see Dosage and Administration (2.4)].
The safety and effectiveness of Noxafil oral suspension for the treatment of OPC and rOPC have not been established in pediatric patients less than 13 years of age.
8.5 Geriatric Use
No overall differences in the safety or effectiveness of Noxafil injection, Noxafil delayed-release tablets, and Noxafil oral suspension have been observed between geriatric patients and younger adult patients in the clinical trials; therefore, the recommended dosage in geriatric patients is the same as that for younger adult patients. No clinically meaningful differences in posaconazole pharmacokinetics were observed in Noxafil-treated geriatric patients compared to Noxafil-treated younger adult patients during clinical trials [see Clinical Pharmacology (12.3)].
- Of the 279 patients treated with Noxafil injection in the Noxafil Injection Study (prophylaxis of invasive Aspergillus and Candida infections in those at high risk of developing these infections due to being severely immunocompromised), 52 (19%) patients were >65 years of age.
- Of the 230 patients treated with Noxafil delayed-release tablets, 38 (17%) patients were >65 years of age.
- Of the 605 patients treated with Noxafil oral suspension in Noxafil Oral Suspension Study 1 and Study 2 (prophylaxis of invasive Aspergillus and Candida infections in those at high risk of developing these infections due to being severely immunocompromised), 63 (10%) patients were ≥65 years of age.
- In studies of Noxafil for an unapproved indication, 48 patients treated with Noxafil oral suspension (greater than or equal to 800 mg/day (eight times the maximum recommended maintenance dosage for the treatment of OPC)) were ≥65 years of age.
- Of the 288 patients treated with Noxafil injection or Noxafil delayed-release tablets in the Aspergillosis Treatment Study, 85 (29%) patients were ≥65 years of age.
4 Contraindications (4 CONTRAINDICATIONS)
- Known hypersensitivity to posaconazole or other azole antifungal agents. (4.1)
- Coadministration of Noxafil with the following drugs is contraindicated; Noxafil increases concentrations and toxicities of:
- Sirolimus (4.2, 7.2)
- CYP3A4 substrates (pimozide, quinidine): can result in QTc interval prolongation and cases of torsades de pointes (TdP) (4.3, 5.2, 7.2)
- HMG-CoA Reductase Inhibitors Primarily Metabolized through CYP3A4 (4.4, 7.2)
- Ergot alkaloids (4.5, 7.2)
- Venetoclax: In patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) at initiation and during the ramp-up phase (4.6, 5.11, 7.2)
- Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with known or suspected Hereditary Fructose Intolerance (HFI). (4.7, 5.9, 8.4)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious and otherwise important adverse reactions are discussed in detail in another section of the labeling:
- Arrhythmias and QT Prolongation [see Warnings and Precautions (5.2)]
- Electrolyte Disturbances [see Warnings and Precautions (5.3)]
- Pseudoaldosteronism [see Warnings and Precautions (5.4)]
- Hepatic Toxicity [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 15 and Table 17 include drugs with clinically important drug interactions when administered concomitantly with Noxafil and Noxafil PowderMix and instructions for preventing or managing them. Table 16 includes important drug interactions specific to the absorption of posaconazole administered as either Noxafil oral suspension or Noxafil PowderMix.
These recommendations are based on either drug interaction studies or predicted interactions due to the expected magnitude of interaction and potential for serious adverse reactions or loss of efficacy [see Clinical Pharmacology (12.3)].
The following information was derived from data with Noxafil oral suspension or another posaconazole tablet formulation unless otherwise noted. All clinically important drug interactions with Noxafil oral suspension, except for those that affect the absorption of posaconazole (via gastric pH and motility), are considered relevant to clinically important drug interactions with Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension [see Clinical Pharmacology (12.3)].
Consult the labeling of concomitantly used drugs to obtain further information about interactions with posaconazole.
4.1 Hypersensitivity
Noxafil is contraindicated in persons with known hypersensitivity to posaconazole or other azole antifungal agents.
5.5 Hepatic Toxicity
Hepatic reactions (e.g., mild to moderate elevations in alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase, total bilirubin, and/or clinical hepatitis) have been reported in clinical trials. The elevations in liver tests were generally reversible on discontinuation of therapy, and in some instances these tests normalized without drug interruption. Cases of more severe hepatic reactions including cholestasis or hepatic failure including deaths have been reported in patients with serious underlying medical conditions (e.g., hematologic malignancy) during treatment with Noxafil. These severe hepatic reactions were seen primarily in subjects receiving the Noxafil oral suspension 800 mg daily (400 mg twice daily or 200 mg four times a day) in clinical trials.
Liver tests should be evaluated at the start of and during the course of Noxafil therapy. Patients who develop abnormal liver tests during Noxafil therapy should be monitored for the development of more severe hepatic injury. Patient management should include laboratory evaluation of hepatic function (particularly liver tests and bilirubin). Discontinuation of Noxafil must be considered if clinical signs and symptoms consistent with liver disease develop that may be attributable to Noxafil.
5.6 Renal Impairment
Due to the variability in exposure with Noxafil delayed-release tablets, Noxafil oral suspension, and Noxafil PowderMix for delayed-release oral suspension, patients with severe renal impairment should be monitored closely for breakthrough fungal infections [see Dosage and Administration (2.11) and Use in Specific Populations (8.6)].
Noxafil injection should be avoided in patients with moderate or severe renal impairment (eGFR <50 mL/min/1.73 m2), unless an assessment of the benefit/risk to the patient justifies the use of Noxafil injection. In patients with moderate or severe renal impairment (eGFR <50 mL/min/1.73 m2), receiving the Noxafil injection, accumulation of the intravenous vehicle, SBECD, is expected to occur. Serum creatinine levels should be closely monitored in these patients, and, if increases occur, consideration should be given to changing to oral Noxafil therapy [see Dosage and Administration (2.11) and Use in Specific Populations (8.6)].
8.6 Renal Impairment
Noxafil Oral Suspension and Noxafil Delayed-Release Tablets
No dosage adjustment is required for patients with eGFR 20 mL/minute/1.73 m2 or higher.
Due to variability in posaconazole exposure, closely monitor patients with eGFR less than 20 mL/minute/1.73 m2 for breakthrough fungal infections. [see Clinical Pharmacology (12.3)].
Noxafil Injection
Avoid use of Noxafil injection in patients with eGFR less than 50 mL/minute/1.73 m2 unless the benefit/risk to the patient justifies its use. The inactive ingredient in Noxafil injection, Betadex Sulfobutyl Ether Sodium (SBECD) is expected to accumulate in patients with reduced renal function. Safety and effectiveness of Noxafil injection have not been established in patients with less than 50 mL/minute/1.73 m2.
If treatment with Noxafil injection is unavoidable in patients with eGFR less than 50 mL/minute/1.73 m2, monitor serum creatinine levels. If serum creatinine increases, consider changing to oral Noxafil therapy.
4.2 Use With Sirolimus (4.2 Use with Sirolimus)
Noxafil is contraindicated with sirolimus. Concomitant administration of Noxafil with sirolimus increases the sirolimus blood concentrations by approximately 9-fold and can result in sirolimus toxicity [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
5.7 Midazolam Toxicity
Concomitant administration of Noxafil with midazolam increases the midazolam plasma concentrations by approximately 5-fold. Increased plasma midazolam concentrations could potentiate and prolong hypnotic and sedative effects. Patients must be monitored closely for adverse effects associated with high plasma concentrations of midazolam and benzodiazepine receptor antagonists must be available to reverse these effects [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dosage adjustment is recommended for Noxafil oral suspension, Noxafil delayed-release tablets, Noxafil PowderMix for delayed-release oral suspension, and Noxafil injection in patients with mild, moderate, or severe hepatic impairment (Child-Pugh Class A, B, or C, respectively) [see Clinical Pharmacology (12.3)].
However, a specific hepatic impairment study has not been conducted with the Noxafil delayed-release tablets, Noxafil PowderMix for delayed-release oral suspension, or Noxafil injection.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Noxafil is an azole antifungal indicated as follows:
- Noxafil is indicated for the treatment of invasive aspergillosis as follows: (1.1)
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg
-
Noxafil is indicated for the prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, such as hematopoietic stem cell transplant (HSCT) recipients with graft-versus-host disease (GVHD) or those with hematologic malignancies with prolonged neutropenia from chemotherapy as follows: (1.2)
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater.
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil oral suspension: adults and pediatric patients 13 years of age and older
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg
- Noxafil oral suspension is indicated for the treatment of oropharyngeal candidiasis (OPC), including OPC refractory (rOPC) to itraconazole and/or fluconazole in adults and pediatric patients 13 years of age and older (1.3)
4.6 Use With Venetoclax (4.6 Use with Venetoclax)
Coadministration of Noxafil with venetoclax at initiation and during the ramp-up phase is contraindicated in patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) due to the potential for increased risk of tumor lysis syndrome [see Warnings and Precautions (5.11) and Drug Interactions (7.2)].
5.4 Pseudoaldosteronism
Pseudoaldosteronism, manifested by the onset of hypertension or worsening of hypertension, and abnormal laboratory findings (hypokalemia, low serum renin and aldosterone, and elevated 11-deoxycortisol), has been reported with posaconazole use in the postmarket setting. Monitor blood pressure and potassium levels and manage as necessary. Management of pseudoaldosteronism may include discontinuation of Noxafil, substitution with an appropriate antifungal drug that is not associated with pseudoaldosteronism, or use of aldosterone receptor antagonists.
12.1 Mechanism of Action
Posaconazole is an azole antifungal agent [see Clinical Pharmacology (12.4)].
5.11 Venetoclax Toxicity
Concomitant administration of Noxafil, a strong CYP3A4 inhibitor, with venetoclax may increase venetoclax toxicities, including the risk of tumor lysis syndrome (TLS), neutropenia, and serious infections. In patients with CLL/SLL, administration of Noxafil during initiation and the ramp-up phase of venetoclax is contraindicated [see Contraindications (4.6)]. Refer to the venetoclax labeling for safety monitoring and dose reduction in the steady daily dosing phase in CLL/SLL patients.
For patients with acute myeloid leukemia (AML), dose reduction and safety monitoring are recommended across all dosing phases when coadministering Noxafil with venetoclax [see Drug Interactions (7.2)]. Refer to the venetoclax prescribing information for dosing instructions.
5.8 Vincristine Toxicity
Concomitant administration of azole antifungals, including Noxafil, with vincristine has been associated with neurotoxicity and other serious adverse reactions, including seizures, peripheral neuropathy, syndrome of inappropriate antidiuretic hormone secretion, and paralytic ileus. Reserve azole antifungals, including Noxafil, for patients receiving a vinca alkaloid, including vincristine, who have no alternative antifungal treatment options [see Drug Interactions (7.2)].
16.2 Storage and Handling
Noxafil Injection
Store Noxafil injection vial refrigerated at 2 to 8°C (36 to 46°F). Storage conditions for the diluted Noxafil infusion solution are presented in another section of the prescribing information [see Dosage and Administration (2.5)].
Noxafil Delayed-Release Tablets
Store at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature].
Noxafil Oral Suspension
Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
Noxafil PowderMix for Delayed-Release Oral Suspension
Store the entire kit at 20 to 25°C (68 to 77°F), excursions permitted to 15 to 30°C (59 to 86°F) in a clean, dry place. Do not open foil packet containing Noxafil PowderMix for delayed-release oral suspension until ready for use. Storage conditions for the reconstituted solution are presented in another section of the prescribing information [see Dosage and Administration (2.10)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Calcineurin-Inhibitor Toxicity: Noxafil increases concentrations of cyclosporine or tacrolimus; reduce dose of cyclosporine and tacrolimus and monitor concentrations frequently. (5.1)
- Arrhythmias and QTc Prolongation: Noxafil has been shown to prolong the QTc interval and cause cases of TdP. Administer with caution to patients with potentially proarrhythmic conditions. Do not administer with drugs known to prolong QTc interval and metabolized through CYP3A4. (5.2, 7.2)
- Electrolyte Disturbances: Monitor and correct, especially those involving potassium (K+), magnesium (Mg++), and calcium (Ca++), before and during Noxafil therapy. (5.3)
- Pseudoaldosteronism: Manifested by the onset or worsening of hypertension, and abnormal laboratory findings. Monitor blood pressure and potassium levels, and manage as necessary. (5.4)
- Hepatic Toxicity: Elevations in liver tests may occur. Discontinuation should be considered in patients who develop abnormal liver tests or monitor liver tests during treatment. (5.5)
- Renal Impairment: Noxafil injection should be avoided in patients with moderate or severe renal impairment (eGFR less than 50 mL/min/1.73 m2), unless an assessment of the benefit/risk to the patient justifies the use of Noxafil injection. (5.6, 8.6)
- Concomitant Use with Midazolam: Noxafil can prolong hypnotic/sedative effects. Monitor patients and benzodiazepine receptor antagonists should be available. (5.7, 7.2)
- Vincristine Toxicity: Concomitant administration of azole antifungals, including Noxafil, with vincristine has been associated with neurotoxicity and other serious adverse reactions; reserve azole antifungals, including Noxafil, for patients receiving a vinca alkaloid, including vincristine, who have no alternative antifungal treatment options. (5.8, 7.2)
- Risk in Patients with Hereditary Fructose Intolerance (HFI): Noxafil PowderMix for delayed-release oral suspension contains sorbitol. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before Noxafil PowderMix for delayed-release oral suspension administration. (5.9, 8.4)
- Breakthrough Fungal Infections: Monitor patients with severe diarrhea or vomiting when receiving Noxafil delayed-release tablets, Noxafil oral suspension, and Noxafil PowderMix for delayed-release oral suspension. (5.10)
- Venetoclax Toxicity: Concomitant administration of Noxafil with venetoclax may increase venetoclax toxicities, including the risk of tumor lysis syndrome, neutropenia, and serious infections; monitor for toxicity and reduce venetoclax dose. (4.6, 5.11, 7.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Noxafil formulations are supplied in different dose strengths of posaconazole, are approved for different indications, age groups, and weights, have different dosages and duration of therapy; and have different preparation and administration instructions. (2.1)
- Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations. (2.1, 2.2, 2.3)
- Noxafil injection must be administered through an in-line filter. (2.6)
- Administer Noxafil injection by intravenous infusion over approximately 90 minutes via a central venous line. (2.1, 2.6)
- Do NOT administer Noxafil injection as an intravenous bolus injection. (2.1)
- Administer Noxafil delayed-release tablets with or without food. (2.1)
- Administer Noxafil oral suspension with a full meal. (2.1)
- Administer Noxafil PowderMix for delayed-release oral suspension with food. (2.1)
- Administer Noxafil PowderMix for delayed-release oral suspension with the provided notched tip syringes only. (2.1)
- See the full prescribing information for important administration instructions and preparation instructions for Noxafil (injection, delayed-release tablets, and oral suspension) and Noxafil PowderMix delayed-release oral suspension (2.5, 2.6, 2.7, 2.8, 2.9, 2.10)
- For adult and pediatric patients aged 2 years of age and older, see the Full Prescribing Information for dosing recommendations for Noxafil injection, Noxafil delayed-release tablets, Noxafil oral suspension, and/or Noxafil PowderMix for delayed-release oral suspension based on the indication, age, and weight associated with the dosage form (1.1, 1.2, 1.3, 2.1, 2.2, 2.3, 2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Noxafil injection
300 mg/16.7 mL (18 mg/mL) of posaconazole: Clear, colorless to yellow sterile liquid in a single-dose vial.
Noxafil Delayed-Release Tablets
100 mg of posaconazole: Yellow, coated, oblong tablets, debossed with "100" on one side.
Noxafil Oral Suspension
4,200 mg/105 mL (40 mg/mL) of posaconazole: White, cherry-flavored suspension in amber glass bottles with child-resistant closures.
Noxafil PowderMix for Delayed-Release Oral Suspension
300 mg: Off-white to yellowish powder for delayed-release oral suspension and a mixing liquid in a kit. The kit contains (1) Package A that contains single-use packets of Noxafil PowderMix, green notched tip syringes, blue notched tip syringes, mixing cups, mixing liquid bottle, and one bottle adapter for the mixing liquid bottle; and (2) Package B that contains green and blue notched tip syringes for additional supply [see How Supplied/Storage and Handling (16.1)].
4.5 Use With Ergot Alkaloids (4.5 Use with Ergot Alkaloids)
Noxafil may increase the plasma concentrations of ergot alkaloids (ergotamine and dihydroergotamine) which may lead to ergotism [see Drug Interactions (7.2)].
5.3 Electrolyte Disturbances
Electrolyte disturbances, especially those involving potassium, magnesium or calcium levels, should be monitored and corrected as necessary before and during Noxafil therapy.
6.2 Postmarketing Experience
The following adverse reaction has been identified during the post-approval use of Noxafil. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of Noxafil cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
14.2 Prophylaxis of Aspergillus
Two randomized, controlled studies were conducted using Noxafil as prophylaxis for the prevention of invasive fungal infections (IFIs) among patients at high risk due to severely compromised immune systems.
The first study (Noxafil Oral Suspension Study 1) was a randomized, double-blind trial that compared Noxafil oral suspension (200 mg three times a day) with fluconazole capsules (400 mg once daily) as prophylaxis against invasive fungal infections in allogeneic hematopoietic stem cell transplant (HSCT) recipients with Graft versus Host Disease (GVHD). Efficacy of prophylaxis was evaluated using a composite endpoint of proven/probable IFIs, death, or treatment with systemic antifungal therapy (patients may have met more than one of these criteria). This assessed all patients while on study therapy plus 7 days and at 16 weeks post-randomization. The mean duration of therapy was comparable between the 2 treatment groups (80 days, Noxafil oral suspension; 77 days, fluconazole). Table 34 contains the results from Noxafil Oral Suspension Study 1.
| Posaconazole n=301 |
Fluconazole n=299 |
|
|---|---|---|
| On therapy plus 7 days | ||
|
Clinical Failure Patients may have met more than one criterion defining failure.
|
50 (17%) | 55 (18%) |
| Failure due to: | ||
| Proven/Probable IFI | 7 (2%) | 22 (7%) |
| (Aspergillus) | 3 (1%) | 17 (6%) |
| (Candida) | 1 (<1%) | 3 (1%) |
| (Other) | 3 (1%) | 2 (1%) |
| All Deaths | 22 (7%) | 24 (8%) |
| Proven/probable fungal infection prior to death | 2 (<1%) | 6 (2%) |
| SAF Use of systemic antifungal therapy (SAF) criterion is based on protocol definitions (empiric/IFI usage >4 consecutive days).
|
27 (9%) | 25 (8%) |
| Through 16 weeks | ||
|
Clinical Failure
,
95% confidence interval (posaconazole-fluconazole) = (-11.5%, + 3.7%).
|
99 (33%) | 110 (37%) |
| Failure due to: | ||
| Proven/Probable IFI | 16 (5%) | 27 (9%) |
| (Aspergillus) | 7 (2%) | 21 (7%) |
| (Candida) | 4 (1%) | 4 (1%) |
| (Other) | 5 (2%) | 2 (1%) |
| All Deaths | 58 (19%) | 59 (20%) |
| Proven/probable fungal infection prior to death | 10 (3%) | 16 (5%) |
| SAF | 26 (9%) | 30 (10%) |
| Event free lost to follow-up Patients who are lost to follow-up (not observed for 112 days), and who did not meet another clinical failure endpoint. These patients were considered failures.
|
24 (8%) | 30 (10%) |
The second study (Noxafil Oral Suspension Study 2) was a randomized, open-label study that compared Noxafil oral suspension (200 mg 3 times a day) with fluconazole suspension (400 mg once daily) or itraconazole oral solution (200 mg twice a day) as prophylaxis against IFIs in neutropenic patients who were receiving cytotoxic chemotherapy for AML or MDS. As in Noxafil Oral Suspension Study 1, efficacy of prophylaxis was evaluated using a composite endpoint of proven/probable IFIs, death, or treatment with systemic antifungal therapy (Patients might have met more than one of these criteria). This study assessed patients while on treatment plus 7 days and 100 days post-randomization. The mean duration of therapy was comparable between the 2 treatment groups (29 days, posaconazole; 25 days, fluconazole or itraconazole). Table 35 contains the results from Noxafil Oral Suspension Study 2.
| Posaconazole n=304 |
Fluconazole/Itraconazole n=298 |
|
|---|---|---|
| On therapy plus 7 days | ||
|
Clinical Failure 95% confidence interval (posaconazole-fluconazole/itraconazole) = (-22.9%, -7.8%).
,
Patients may have met more than one criterion defining failure.
|
82 (27%) | 126 (42%) |
| Failure due to: | ||
| Proven/Probable IFI | 7 (2%) | 25 (8%) |
| (Aspergillus) | 2 (1%) | 20 (7%) |
| (Candida) | 3 (1%) | 2 (1%) |
| (Other) | 2 (1%) | 3 (1%) |
| All Deaths | 17 (6%) | 25 (8%) |
| Proven/probable fungal infection prior to death | 1 (<1%) | 2 (1%) |
| SAF Use of systemic antifungal therapy (SAF) criterion is based on protocol definitions (empiric/IFI usage >3 consecutive days).
|
67 (22%) | 98 (33%) |
| Through 100 days post-randomization | ||
| Clinical Failure | 158 (52%) | 191 (64%) |
| Failure due to: | ||
| Proven/Probable IFI | 14 (5%) | 33 (11%) |
| (Aspergillus) | 2 (1%) | 26 (9%) |
| (Candida) | 10 (3%) | 4 (1%) |
| (Other) | 2 (1%) | 3 (1%) |
| All Deaths | 44 (14%) | 64 (21%) |
| Proven/probable fungal infection prior to death | 2 (1%) | 16 (5%) |
| SAF | 98 (32%) | 125 (42%) |
| Event free lost to follow-up Patients who are lost to follow-up (not observed for 100 days), and who did not meet another clinical failure endpoint. These patients were considered failures.
|
34 (11%) | 24 (8%) |
In summary, 2 clinical studies of prophylaxis were conducted with the Noxafil oral suspension. As seen in the accompanying tables (Table 34 and Table 35), clinical failure represented a composite endpoint of breakthrough IFI, mortality and use of systemic antifungal therapy. In Noxafil Oral Suspension Study 1 (Table 34), the clinical failure rate of posaconazole (33%) was similar to fluconazole (37%), (95% CI for the difference posaconazole–comparator -11.5% to 3.7%) while in Noxafil Oral Suspension Study 2 (Table 35) clinical failure was lower for patients treated with posaconazole (27%) when compared to patients treated with fluconazole or itraconazole (42%), (95% CI for the difference posaconazole–comparator -22.9% to -7.8%).
All-cause mortality was similar at 16 weeks for both treatment arms in Noxafil Oral Suspension Study 1 [POS 58/301 (19%) vs. FLU 59/299 (20%)]; all-cause mortality was lower at 100 days for Noxafil-treated patients in Noxafil Oral Suspension Study 2 [POS 44/304 (14%) vs. FLU/ITZ 64/298 (21%)]. Both studies demonstrated fewer breakthrough infections caused by Aspergillus species in patients receiving Noxafil prophylaxis when compared to patients receiving fluconazole or itraconazole.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Important Administration Instructions
Noxafil Delayed-Release Tablets
Advise patients that Noxafil delayed-release tablets must be swallowed whole and not divided, crushed, or chewed.
Instruct patients that if they miss a dose, they should take it as soon as they remember. If they do not remember until it is within 12 hours of the next dose, they should be instructed to skip the missed dose and go back to the regular schedule. Patients should not double their next dose or take more than the prescribed dose.
Noxafil Oral Suspension
Advise patients to take each dose of Noxafil oral suspension during or immediately (i.e., within 20 minutes) following a full meal. In patients who cannot eat a full meal, each dose of Noxafil oral suspension should be administered with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale) in order to enhance absorption.
Instruct patients that if they miss a dose, they should take it as soon as they remember. However, if it is almost time for the next dose, they should be instructed to skip the missed dose and go back to the regular schedule. Patients should not double their next dose or take more than the prescribed dose.
Noxafil PowderMix for Delayed-Release Oral Suspension
Instruct parents and/or caregivers that ONLY the provided notched tip syringes can be used to administer Noxafil PowderMix for delayed-release oral suspension to pediatric patients.
Advise patients to take Noxafil PowderMix for delayed-release oral suspension with food.
Drug Interactions
Advise patients to inform their physician immediately if they:
- develop severe diarrhea or vomiting.
- are currently taking drugs that are known to prolong the QTc interval and are metabolized through CYP3A4.
- are currently taking a cyclosporine or tacrolimus, or they notice swelling in an arm or leg or shortness of breath.
- are taking other drugs or before they begin taking other drugs as certain drugs can decrease or increase the plasma concentrations of posaconazole.
Serious and Potentially Serious Adverse Reactions
Advise patients to inform their physician immediately if they:
- notice a change in heart rate or heart rhythm or have a heart condition or circulatory disease. Noxafil can be administered with caution to patients with potentially proarrhythmic conditions.
- are pregnant, plan to become pregnant, or are nursing.
- have liver disease or develop itching, nausea or vomiting, their eyes or skin turn yellow, they feel more tired than usual or feel like they have the flu.
- have ever had an allergic reaction to other antifungal medicines such as ketoconazole, fluconazole, itraconazole, or voriconazole.
Hereditary Fructose Intolerance (HFI)
Inform patients and caregivers that Noxafil PowderMix for delayed-release oral suspension contains sorbitol and can be life-threatening when administered to patients with hereditary fructose intolerance (HFI) [see Warnings and Precautions (5.9)]. Inquire for symptoms of sorbitol/fructose and/or sucrose intolerance before administration.
5.1 Calcineurin Inhibitor Toxicity (5.1 Calcineurin-Inhibitor Toxicity)
Concomitant administration of Noxafil with cyclosporine or tacrolimus increases the whole blood trough concentrations of these calcineurin-inhibitors [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)]. Nephrotoxicity and leukoencephalopathy (including deaths) have been reported in clinical efficacy studies in patients with elevated cyclosporine or tacrolimus concentrations. Frequent monitoring of tacrolimus or cyclosporine whole blood trough concentrations should be performed during and at discontinuation of Noxafil treatment and the tacrolimus or cyclosporine dose adjusted accordingly.
5.10 Breakthrough Fungal Infections
Patients who have severe diarrhea or vomiting should be monitored closely for breakthrough fungal infections when receiving Noxafil delayed-release tablets, Noxafil oral suspension, or Noxafil PowderMix for delayed-release oral suspension.
5.2 Arrhythmias and Qt Prolongation (5.2 Arrhythmias and QT Prolongation)
Some azoles, including Noxafil, have been associated with prolongation of the QT interval on the electrocardiogram. In addition, cases of torsades de pointes have been reported in patients taking Noxafil.
Results from a multiple time-matched ECG analysis in healthy volunteers did not show any increase in the mean of the QTc interval. Multiple, time-matched ECGs collected over a 12-hour period were recorded at baseline and steady-state from 173 healthy male and female volunteers (18-85 years of age) administered Noxafil oral suspension 400 mg twice daily with a high-fat meal. In this pooled analysis, the mean QTc (Fridericia) interval change from baseline was –5 msec following administration of the recommended clinical dose. A decrease in the QTc(F) interval (–3 msec) was also observed in a small number of subjects (n=16) administered placebo. The placebo-adjusted mean maximum QTc(F) interval change from baseline was <0 msec (–8 msec). No healthy subject administered Noxafil had a QTc(F) interval ≥500 msec or an increase ≥60 msec in their QTc(F) interval from baseline.
Noxafil should be administered with caution to patients with potentially proarrhythmic conditions. Do not administer with drugs that are known to prolong the QTc interval and are metabolized through CYP3A4 [see Contraindications (4.3) and Drug Interactions (7.2)].
2.5 Preparation of Noxafil Injection
Preparation of Noxafil Injection:
- Remove the vial of Noxafil injection from the refrigerator and allow to equilibrate to room temperature prior to use.
-
To prepare the required dose, aseptically transfer one vial of Noxafil injection (containing 300 mg of posaconazole in 16.7 mL of solution) to an intravenous bag or bottle of one of the following compatible diluents to achieve a final posaconazole concentration between 1 mg/mL and 2 mg/mL:
- 0.45% Sodium Chloride Injection
- 0.9% Sodium Chloride Injection
- 5% Dextrose Injection
- 5% Dextrose and 0.45% Sodium Chloride Injection
- 5% Dextrose and 0.9% Sodium Chloride Injection
- 5% Dextrose and 20 mEq Potassium Chloride Injection
Use of other diluents is not recommended because they may result in particulate formation.
- Discard any unused Noxafil injection from the vial.
- Parenteral drug products should be inspected visually for particulate matter prior to administration, whenever solution and container permit. Once admixed, the diluted Noxafil infusion solution ranges from colorless to yellow (variations of color within this range do not affect the quality of the product).
- Immediately use the diluted Noxafil infusion solution, once admixed. If not used immediately, refrigerate (2 to 8°C (36 to 46°F)) the diluted Noxafil infusion solution up to 24 hours. Discard any unused portion.
Incompatible Diluents
Co-administration of drug products besides the infusion solutions or products stated above are not recommended because this may result in particulate formation. The following diluents were determined to be incompatible with Noxafil injection; thus, do not dilute Noxafil injection with them:
- Lactated Ringer’s Injection
- Lactated Ringer's and 5% Dextrose Injection
- 4.2% Sodium Bicarbonate Injection
1.1 Treatment of Invasive Aspergillosis
Noxafil is indicated for the treatment of invasive aspergillosis as follows:
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 to 40 kg
1.2 Prophylaxis of Invasive Aspergillus
Noxafil is indicated for the prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, such as hematopoietic stem cell transplant (HSCT) recipients with graft-versus-host disease (GVHD) or those with hematologic malignancies with prolonged neutropenia from chemotherapy as follows:
- Noxafil injection: adults and pediatric patients 2 years of age and older who weigh 10 kg or greater
- Noxafil delayed-release tablets: adults and pediatric patients 2 years of age and older who weigh greater than 40 kg
- Noxafil oral suspension: adults and pediatric patients 13 years of age and older
- Noxafil PowderMix for delayed-release oral suspension: pediatric patients 2 years of age and older who weigh 10 kg to 40 kg
Principal Display Panel 300 Mg Carton (PRINCIPAL DISPLAY PANEL - 300 mg Carton)
NDC 0085-2224-02
Noxafil® PowderMix
(posaconazole) for
Delayed-Release Oral Suspension
300 mg
For Pediatric Use
For Oral Administration Only
Each packet contains 300 mg posaconazole.
Attention: Noxafil PowderMix for Delayed-Release Oral Suspension is NOT
substitutable with Noxafil Oral Suspension due to differences in the
dosing of each formulation.
Use only the notched-tip syringes and mixing liquid provided with this kit.
Rx only
8 packets
Multi-Dose Kit A
Additional notched-tip syringes are provided in
Package B with this kit.
DIRECTIONS FOR USE:
See instructions for use booklet and prescribing
information for additional information.
2.1 Important Administration Instructions
Noxafil injection, Noxafil delayed-release tablets, Noxafil oral suspension and Noxafil PowderMix for delayed-release oral suspension are supplied in different dose strengths of posaconazole, are approved for different indications, age groups and weights; have different dosages and duration of therapy; and have different preparation and administration instructions.
Therefore, select the recommended dosage form based on the indication, age group, and weight and carefully follow the recommended dosage, preparation and administration instructions described for each product [see Dosage and Administration (2.2 to 2.11)], and the following important administration instructions described below.
Non-substitutable
Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations [see Dosage and Administration (2.2, 2.3)].
Noxafil injection
- Administer via a central venous line, including a central venous catheter or peripherally inserted central catheter (PICC), by slow intravenous infusion over approximately 90 minutes [see Dosage and Administration (2.6)].
- Do NOT administer Noxafil injection as an intravenous bolus injection.
Noxafil delayed-release tablets
- Swallow tablets whole. Do not divide, crush, or chew.
- Administer with or without food [see Dosage and Administration (2.7) and Clinical Pharmacology (12.3)].
- For patients who cannot eat a full meal, Noxafil delayed-release tablets should be used instead of Noxafil oral suspension for the prophylaxis indication. Noxafil delayed-release tablets generally provide higher plasma drug exposures than Noxafil oral suspension under both fed and fasted conditions [see Dosage and Administration (2.8)].
Noxafil oral suspension
- Administer with a full meal or with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale) in patients who cannot eat a full meal [see Dosage and Administration (2.8)].
Noxafil PowderMix for delayed-release oral suspension
- Administer with food [see Clinical Pharmacology (12.3)].
- To ensure delivery of the correct dose, ONLY the provided notched tip syringes must be used for preparation and administration. The design of the notched tip syringe prevents aggregation of the suspension during preparation and administration [see Dosage and Administration (2.10)].
Principal Display Panel 16.7 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 16.7 mL Vial Carton)
NDC 0085-4331-01
Noxafil®
(posaconazole)
Injection
300 mg/16.7 mL
(18 mg/mL)
For Intravenous Use Only
Requires further dilution prior to infusion.
Rx only
Sterile Single-Dose Vial
Discard Unused Portion
Principal Display Panel 105 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 105 mL Bottle Carton)
NDC 0085-1328-01
105 mL
NOXAFIL®
(posaconazole)
Oral Suspension
200 mg/5 mL
Each mL contains: 40 mg posaconazole.
Attention: Noxafil Oral Suspension is
NOT substitutable with Noxafil
Delayed-Release Tablets or Noxafil PowderMix
for Delayed-Release Oral Suspension due to
differences in the dosing of each formulation.
SHAKE WELL BEFORE EACH USE.
Take with a meal, or a nutritional supplement,
or an acidic carbonated beverage.
Carton contains measured dosing spoon.
Rx only
Principal Display Panel 60 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 60 Tablet Bottle Label)
NDC 0085-4324-02
Noxafil®
(posaconazole)
delayed-release tablets
100 mg
Each tablet contains 100 mg posaconazole.
Attention: Noxafil Oral Suspension and Delayed-
Release Tablets are NOT substitutable due to
differences in the dosing of each formulation.
Rx only
60 Tablets
2.2 Recommended Dosage of Noxafil in Adult Patients
The recommended dosage of Noxafil (injection, delayed-release tablets, and oral suspension) in adult patients for the treatment of invasive aspergillosis, prophylaxis of invasive Aspergillus and Candida infections in patients who are at high risk of developing these infections due to being severely immunocompromised, or for the treatment of oropharyngeal candidiasis (OPC) is shown in Table 1 [see Dosage and Administration (2.5, 2.6, 2.7,2.8, 2.9) and Clinical Pharmacology (12.3)].
Noxafil PowderMix for delayed-release oral suspension is not recommended for use in adults [see Indications and Usage (1.1, 1.2)].
| Dosage | Duration of Therapy |
|---|---|
|
Treatment of Invasive Aspergillosis
Switching between the Noxafil injection and delayed-release tablets is acceptable. A loading dose is not required when switching between dosage forms.
|
|
|
Noxafil Injection:
Loading dose:
Maintenance dose:
Noxafil Delayed-Release Tablets:
|
Loading dose:
Maintenance dose:
|
| Prophylaxis of Invasive Aspergillus and Candida Infections | |
|
Noxafil Injection:
Loading dose: 300 mg Noxafil injection intravenously twice a day on the first day. Maintenance dose: 300 mg Noxafil injection intravenously once a day thereafter. Noxafil Delayed-Release Tablets: Loading dose: 300 mg (three 100 mg delayed-release tablets) twice a day on the first day. Maintenance dose: 300 mg (three 100 mg delayed-release tablets) once a day, starting on the second day. Noxafil Oral Suspension: 200 mg (5 mL) three times a day. |
Loading dose: 1 day Maintenance dose: Duration of therapy is based on recovery from neutropenia or immunosuppression |
| Oropharyngeal Candidiasis (OPC) | |
|
Noxafil Oral Suspension:
Loading dose: 100 mg (2.5 mL) twice a day on the first day. Maintenance dose: 100 mg (2.5 mL) once a day thereafter. |
Loading dose: 1 day Maintenance dose: 13 days |
| OPC Refractory (rOPC) to Itraconazole and/or Fluconazole | |
| Noxafil Oral Suspension: 400 mg (10 mL) twice a day. | Duration of therapy is based on the severity of the patient’s underlying disease and clinical response. |
2.6 Administration of Diluted Noxafil Infusion Solution
See Dosage and Administration (2.5) for the preparation instructions for the diluted Noxafil Solution.
Important Administration Instructions for the Diluted Noxafil Infusion Solution
- Must administer diluted Noxafil Infusion Solution through a 0.22-micron polyethersulfone (PES) or polyvinylidene difluoride (PVDF) filter.
- Administer diluted Noxafil infusion solution via a central venous line, including a central venous catheter (CVC) or peripherally inserted central catheter (PICC), by slow intravenous infusion over approximately 90 minutes [see Adverse Reactions (6.1)].
- If a CVC or PICC are not available, may administer diluted Noxafil solution once through a peripheral venous catheter by intravenous infusion over approximately 30 minutes to bridge the period during which a CVC or PICC are replaced, inserted, or unavailable for use (e.g., the CVC is being used for intravenous treatment with another product). However, do not administer diluted Noxafil infusion solution more than once via peripheral venous catheter because in clinical trials, multiple peripheral infusions given through the same vein resulted in infusion site reactions [see Adverse Reactions (6.1)].
- When multiple dosing is required, the infusion should be done via a central venous line.
Additional Administration Instructions for the Diluted Noxafil Infusion Solution
-
Administer diluted Noxafil infusion solution intravenously through the same intravenous line (or cannula) with the following compatible infusion solutions:
- 0.45% Sodium Chloride Injection
- 0.9% Sodium Chloride Injection
- 5% Dextrose Injection
- 5% Dextrose and 0.45% Sodium Chloride Injection
- 5% Dextrose and 0.9% Sodium Chloride Injection
- 5% Dextrose and 20 mEq potassium chloride Injection
-
Administer diluted Noxafil infusion solution intravenously at the same time through the same intravenous line (or cannula) with the following intravenous drug products prepared in 5% Dextrose Injection or 0.9% Sodium Chloride Injection:
- Amikacin Sulfate Injection
- Caspofungin Acetate for Injection
- Ciprofloxacin Injection
- Daptomycin for Injection
- Dobutamine Injection
- Famotidine Injection
- Filgrastim Injection
- Gentamicin Injection
- Hydromorphone Hydrochloride Injection
- Levofloxacin Injection
- Lorazepam Injection
- Meropenem for Injection
- Micafungin for Injection
- Morphine Sulfate Injection
- Norepinephrine Bitartrate Injection
- Potassium Chloride Injection
- Vancomycin Hydrochloride for Injection
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No drug-related neoplasms were recorded in rats or mice treated with posaconazole for 2 years at doses higher than the clinical dose. In a 2 year carcinogenicity study, rats were given posaconazole orally at doses up to 20 mg/kg (females), or 30 mg/kg (males). These doses are equivalent to 3.9- or 3.5 times the exposure achieved with a 400 mg twice daily oral suspension regimen, respectively, based on steady-state AUC in healthy volunteers administered a high-fat meal (400 mg twice daily oral suspension regimen). In the mouse study, mice were treated at oral doses up to 60 mg/kg/day or 4.8 times the exposure achieved with a 400 mg twice daily oral suspension regimen.
Mutagenesis
Posaconazole was not genotoxic or clastogenic when evaluated in bacterial mutagenicity (Ames), a chromosome aberration study in human peripheral blood lymphocytes, a Chinese hamster ovary cell mutagenicity study, and a mouse bone marrow micronucleus study.
Impairment of Fertility
Posaconazole had no effect on fertility of male rats at a dose up to 180 mg/kg (1.7 x the 400 mg twice daily oral suspension regimen based on steady-state plasma concentrations in healthy volunteers) or female rats at a dose up to 45 mg/kg (2.2 x the 400 mg twice daily oral suspension regimen).
2.11 Dosage Modifications in Patients With Renal Impairment (2.11 Dosage Modifications in Patients with Renal Impairment)
The recommended dosage of Noxafil oral suspension, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension is the same in patients with renal impairment compared to those with normal renal function.
Avoid the use of Noxafil injection in patients with eGFR less than 50 mL/minute/1.73 m2, unless an assessment of the benefit/risk to the patient justifies its use. If the decision is made to use Noxafil injection in patients with eGFR less than 50 mL/minute/1.73 m2, closely monitor serum creatinine levels, and, if increases occur, consider changing to oral Noxafil therapy. The recommended dosage of Noxafil injection in patients with eGFR 50 to 90 mL/minute/1.73 m2 is the same as those with normal renal function.
2.8 Administration Instructions for Noxafil Oral Suspension
Administer Noxafil oral suspension with a full meal or with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale) in patients who cannot eat a full meal.
For patients who cannot eat a full meal, use Noxafil delayed-release tablets instead of the Noxafil oral suspension for the prophylaxis of invasive Aspergillus and Candida infections in those who are at high risk of developing these infections due to being severely immunocompromised. This is because Noxafil delayed-release tablets provide higher plasma drug exposures than Noxafil oral suspension under fasted condition [see Dosage and Administration (2.1)].
For those patients using the Noxafil oral suspension:
- Shake Noxafil oral suspension well before use.
- Administer with measured dosing spoon provided in the package (see Figure 1).
| Figure 1: Measured dosing spoon provided in the package marked for doses of 2.5 mL and 5 mL. |
- Administer each dose of Noxafil oral suspension during or immediately (i.e., within 20 minutes) following a full meal [see Clinical Pharmacology (12.3)].
-
In patients who cannot eat a full meal and for whom Noxafil delayed-release tablets or Noxafil injection are not options, administer each dose of Noxafil oral suspension with a liquid nutritional supplement or an acidic carbonated beverage (e.g., ginger ale). If these patients cannot tolerate an oral nutritional supplement or an acidic carbonated beverage either use:
- An alternative antifungal therapy, or
- Noxafil oral suspension and closely monitor patients for breakthrough fungal infections.
- Rinse the spoon with water after each administration and before storage.
7.2 Effects of Noxafil and Noxafil Powdermix On Other Drugs (7.2 Effects of Noxafil and Noxafil PowderMix on Other Drugs)
Posaconazole is a strong CYP3A4 inhibitor. Therefore, concomitant use of Noxafil may increase plasma concentrations of drugs that are CYP3A4 substrates [see Clinical Pharmacology (12.3)].
| Digoxin | ||
|---|---|---|
| Clinical Effect(s) | Increased digoxin plasma concentrations have been reported in patients who received concomitant posaconazole and digoxin. | |
| Prevention or Management | Monitor digoxin plasma concentrations during concomitant use of posaconazole. | |
| Glipizide | ||
| Clinical Effect(s) | No dosage modification of glipizide is needed when used concomitantly with Noxafil. However, glucose concentrations decrease in some patients concomitantly administered posaconazole and glipizide. | |
| Prevention or Management | Increase monitoring of glucose concentrations when used concomitantly. | |
| CYP3A Substrates | ||
| Immunosuppressants that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Posaconazole is a strong CYP3A4 inhibitor. Therefore, plasma concentrations of CYP3A4 substrates may be increased by posaconazole use [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Sirolimus | Posaconazole is contraindicated with sirolimus [see Clinical Pharmacology (12.3)]. |
| Tacrolimus | • At initiation of posaconazole treatment, reduce the tacrolimus dosage to approximately one-third of the original tacrolimus dosage. • Frequent monitoring of tacrolimus whole blood trough concentrations should be performed during and at discontinuation of posaconazole treatment and the tacrolimus dosage should be modified accordingly [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)]. |
|
| Cyclosporine | • At initiation of posaconazole treatment reduce the cyclosporine dosage to approximately three-fourths of the original dosage. • Frequent monitoring of cyclosporine whole blood trough concentrations should be performed during and at discontinuation of posaconazole treatment and the cyclosporine dosage should be modified accordingly [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)]. |
|
| CYP3A4 Substrates that Prolong QTc Interval | ||
| Mechanism and Clinical Effect(s) | Concomitant use of posaconazole with CYP3A4 substrates such as pimozide and quinidine may result in increased plasma concentrations of the CYP3A4 substrates leading to QTc interval prolongation and torsades de pointes [see Warnings and Precautions (5.2)]. | |
| Prevention or Management | Pimozide | Concomitant use with posaconazole is contraindicated. |
| Quinidine | ||
| HMG-CoA Reductase Inhibitors (Statins) that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Concomitant use of posaconazole with simvastatin increased simvastatin plasma concentrations which can lead to rhabdomyolysis [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Atorvastatin, Lovastatin, Simvastatin | Concomitant use with posaconazole is contraindicated. |
| Benzodiazepines that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Concomitant use of posaconazole with midazolam increased midazolam plasma concentrations which could potentiate and prolong hypnotic and sedative effects [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Midazolam, Alprazolam, Triazolam | Closely monitor for adverse reactions associated with high plasma concentrations of benzodiazepines that are CYP3A4 substrates during concomitant use, and a benzodiazepine receptor antagonist should be available to reverse effects [see Warnings and Precautions (5.7)]. |
| Calcium Channel Blockers that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Posaconazole may increase the plasma concentrations of calcium channel blockers that are substrates of CYP3A4. | |
| Prevention or Management | Verapamil, Diltiazem, Nifedipine, Nicardipine, Felodipine | Monitor frequently for adverse reactions and toxicity with concomitant use of posaconazole with calcium channel blockers that are CYP3A4 substrates. Dosage reduction of the calcium channel blocker may be needed. |
| Anti-HIV Drugs that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Ritonavir and atazanavir are CYP3A4 substrates and posaconazole increased plasma concentrations of these drugs [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Ritonavir and Atazanavir | Monitor frequently for adverse reactions and toxicity of ritonavir and atazanavir during concomitant use. |
| Antineoplastic Drugs that are CYP3A4 Substrates | ||
| Mechanism and Clinical Effect(s) | Posaconazole may increase plasma concentrations of oncology drugs that are CYP3A4 substrates, which may increase the risk of serious adverse reactions. | |
| Prevention or Management | Venetoclax |
CLL/SLL patients
: Concomitant use of posaconazole with venetoclax during initiation and ramp-up phase is contraindicated. AML patients : With concomitant use, venetoclax dosage reduction and safety monitoring is recommended across all dosing phases [see Warnings and Precautions (5.11)]. |
| Vinca alkaloids (e.g., vincristine, vinblastine) | Reserve concomitant use for patients with no alternative antifungal treatment options [see Warnings and Precautions (5.8)]. | |
| Ergot Alkaloids | ||
| Mechanism and Clinical Effect(s) | Most of the ergot alkaloids are CYP3A4 substrates. Posaconazole may increase the plasma concentrations of ergot alkaloids (ergotamine and dihydroergotamine) which may lead to ergotism. | |
| Prevention or Management | Ergotamine, Dihydroergotamine | Concomitant use with posaconazole is contraindicated. |
| Phenytoin | ||
| Mechanism and Clinical Effect(s) | Phenytoin is a CYP3A4 substrate. Concomitant use of posaconazole with phenytoin increased phenytoin plasma concentrations [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Avoid concomitant use of posaconazole with phenytoin unless the benefit outweighs the risk. frequently monitor phenytoin concentrations and consider a dosage reduction of phenytoin. See Table 15 for additional monitoring considerations when phenytoin affects posaconazole via UDP-glucuronosyltransferase inhibition. | |
| Rifabutin | ||
| Mechanism and Clinical Effect(s) | Rifabutin is a CYP3A4 substrate. Concomitant use of posaconazole with rifabutin increased rifabutin plasma concentrations [see Clinical Pharmacology (12.3)]. | |
| Prevention or Management | Avoid concomitant use of posaconazole with rifabutin unless the benefit outweighs the risk. Frequent monitoring of full blood counts and adverse reactions due to increased rifabutin plasma concentrations (e.g., uveitis, leukopenia) during concomitant use are recommended. See Table 15 for additional monitoring considerations when rifabutin affects posaconazole via UDP-glucuronosyltransferase inhibition. |
4.3 Qt Prolongation With Concomitant Use With Cyp3a4 Substrates (4.3 QT Prolongation with Concomitant Use with CYP3A4 Substrates)
Noxafil is contraindicated with CYP3A4 substrates that prolong the QT interval. Concomitant administration of Noxafil with the CYP3A4 substrates, pimozide and quinidine may result in increased plasma concentrations of these drugs, leading to QTc prolongation and cases of torsades de pointes [see Warnings and Precautions (5.2) and Drug Interactions (7.2)].
5.9 Risk in Patients With Hereditary Fructose Intolerance (hfi) (5.9 Risk in Patients with Hereditary Fructose Intolerance (HFI))
Noxafil PowderMix for delayed-release oral suspension contains sorbitol, an inactive ingredient, and may precipitate a metabolic crisis that may include, but is not limited to life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure in patients with HFI. Obtain careful history of HFI symptoms (nausea, vomiting, abdominal pain) with sorbitol/fructose/sucrose exposure prior to Noxafil PowderMix for delayed-release oral suspension administration because a diagnosis of HFI may not yet be established in pediatric patients [see Contraindications (4) and Use in Specific Populations (8.4)].
2.7 Administration Instructions for Noxafil Delayed Release Tablets (2.7 Administration Instructions for Noxafil Delayed-Release Tablets)
- Swallow the Noxafil delayed-release tablets whole. Do not divide, crush, or chew.
- Administer Noxafil delayed-release tablets orally with or without food [see Clinical Pharmacology (12.3)].
4.4 Hmg Coa Reductase Inhibitors Primarily Metabolized Through Cyp3a4 (4.4 HMG-CoA Reductase Inhibitors Primarily Metabolized Through CYP3A4)
Coadministration with the HMG-CoA reductase inhibitors that are primarily metabolized through CYP3A4 (e.g., atorvastatin, lovastatin, and simvastatin) is contraindicated since increased plasma concentration of these drugs can lead to rhabdomyolysis [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
14.3 Treatment of Oropharyngeal Candidiasis With Noxafil Oral Suspension (14.3 Treatment of Oropharyngeal Candidiasis with Noxafil Oral Suspension)
Noxafil Oral Suspension Study 3 was a randomized, controlled, evaluator-blinded study in HIV-infected patients with oropharyngeal candidiasis. Patients were treated with Noxafil or fluconazole oral suspension (both Noxafil and fluconazole were given as follows: 100 mg twice a day for 1 day followed by 100 mg once a day for 13 days).
Clinical and mycological outcomes were assessed after 14 days of treatment and at 4 weeks after the end of treatment. Patients who received at least 1 dose of study medication and had a positive oral swish culture of Candida species at baseline were included in the analyses (see Table 36). The majority of the subjects had C. albicans as the baseline pathogen.
Clinical success at Day 14 (complete or partial resolution of all ulcers and/or plaques and symptoms) and clinical relapse rates (recurrence of signs or symptoms after initial cure or improvement) 4 weeks after the end of treatment were similar between the treatment arms (see Table 36).
Mycologic eradication rates (absence of colony forming units in quantitative culture at the end of therapy, Day 14), as well as mycologic relapse rates (4 weeks after the end of treatment) were also similar between the treatment arms (see Table 36).
| Noxafil | Fluconazole | |
|---|---|---|
| Clinical Success at End of Therapy (Day 14) | 155/169 (91.7%) | 148/160 (92.5%) |
| Clinical Relapse (4 Weeks after End of Therapy) | 45/155 (29.0%) | 52/148 (35.1%) |
| Mycological Eradication (absence of CFU) at End of Therapy (Day 14) | 88/169 (52.1%) | 80/160 (50.0%) |
| Mycological Relapse (4 Weeks after End of Treatment) | 49/88 (55.6%) | 51/80 (63.7%) |
Mycologic response rates, using a criterion for success as a posttreatment quantitative culture with ≤20 colony forming units (CFU/mL) were also similar between the two groups (Noxafil 68.0%, fluconazole 68.1%). The clinical significance of this finding is unknown.
2.9 Non Substitutability Between Noxafil Oral Suspension and Other Formulations (2.9 Non-substitutability between Noxafil Oral Suspension and Other Formulations)
Noxafil oral suspension is not substitutable with Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension due to the differences in the dosing of each formulation. Therefore, follow the specific dosage recommendations for each of the formulations [see Dosage and Administration (2.2, 2.3)].
7.3 Absence of Clinically Important Interaction With Noxafil and Noxafil Powdermix (7.3 Absence of Clinically Important Interaction with Noxafil and Noxafil PowderMix)
Additional clinical studies demonstrated that no clinically important effects on zidovudine, lamivudine, indinavir, or caffeine were observed when administered with Noxafil 200 mg once daily; therefore, no dose adjustments are required for these drugs when coadministered with Noxafil 200 mg once daily.
No clinically relevant effects on the pharmacokinetics of Noxafil delayed-release tablets were observed during concomitant use with antacids, H2-receptor antagonists and proton pump inhibitors, and metoclopramide [see Clinical Pharmacology (12.3)]. No dosage adjustment of Noxafil delayed-release tablets or Noxafil PowderMix for delayed-release oral suspension is required during concomitant use with these drugs.
No clinically relevant effects on the pharmacokinetics of Noxafil oral suspension were observed during concomitant use with antacids, H2-receptor antagonists (other than cimetidine), and loperamide [see Clinical Pharmacology (12.3)]. No dosage adjustment of Noxafil oral suspension is required during concomitant use with these drugs (other than cimetidine).
14.1 Treatment of Invasive Aspergillosis With Noxafil Injection and Noxafil Delayed Release Tablets (14.1 Treatment of Invasive Aspergillosis with Noxafil Injection and Noxafil Delayed-Release Tablets)
Aspergillosis Treatment Study (NCT01782131) was a randomized, double-blind, controlled trial which evaluated the safety and efficacy of Noxafil injection and Noxafil delayed-release tablets versus voriconazole for primary treatment of invasive fungal disease caused by Aspergillus species. Eligible patients had proven, probable, or possible invasive fungal infections per the European Organization for Research and Treatment of Cancer/Mycoses Study Group, EORTC/MSG criteria. Patients were stratified by risk for mortality or poor outcome where high risk included a history of allogeneic bone marrow transplant, liver transplant, or relapsed leukemia undergoing salvage chemotherapy. The median age of patients was 57 years (range: 14-91 years), with 27.8% of patients aged ≥65 years; 5 patients were pediatric patients 14-16 years of age, of whom 3 were treated with Noxafil and 2 with voriconazole. The majority of patients were male (59.8%) and white (67.1%). With regard to risk factors for invasive aspergillosis, approximately two-thirds of the patients in the study had a recent history of neutropenia, while approximately 20% with a history of an allogeneic stem cell transplant. Over 80% of subjects in each treatment group had infection limited to the lower respiratory tract (primarily lung), while approximately 11% to 13% also had infection in another organ. Invasive aspergillosis was proven or probable in 58.1% of patients as classified by independent adjudicators blinded to study treatment assignment. At least one Aspergillus species was identified in 21% of the patients; A. fumigatus and A. flavus were the most common pathogens identified.
Patients randomized to receive Noxafil were given a dose of 300 mg once daily (twice daily on Day 1) IV or tablet. Patients randomized to receive voriconazole were given a dose of 6 mg/kg twice daily Day 1 followed by 4 mg/kg twice daily IV, or oral 300 mg twice daily Day 1 followed by 200 mg twice daily. The recommended initial route of administration was IV; however, patients could begin oral therapy if clinically stable and able to tolerate oral dosing. The transition from IV to oral therapy occurred when the patient was clinically stable. The protocol recommended duration of therapy was 84 days with a maximum allowed duration of 98 days. Median treatment duration was 67 days for Noxafil patients and 64 days for voriconazole patients. Overall, 55% to 60% of patients began treatment with the IV formulation with a median duration of 9 days for the initial IV dosing.
The Intent to Treat (ITT) population included all patients randomized and receiving at least one dose of study treatment. All-cause mortality through Day 42 in the overall population (ITT) was 15.3% for Noxafil patients compared to 20.6% for voriconazole patients for an adjusted treatment difference of -5.3% with a 95% confidence interval of -11.6 to 1.0%. Consistent results were seen in patients with proven or probable invasive aspergillosis per EORTC criteria (see Table 32).
| Noxafil Injection and Delayed-Release Tablets | Voriconazole | ||||
| Population | N | n (%) | N | n (%) | Difference Adjusted treatment difference based on Miettinen and Nurminen’s method stratified by randomization factor (risk for mortality/poor outcome), using Cochran-Mantel-Haenszel weighting scheme. (95% CI) |
| Intent to Treat | 288 | 44 (15.3) | 287 | 59 (20.6) | -5.3 (-11.6, 1.0) |
| Proven/Probable Invasive Aspergillosis | 163 | 31 (19.0) | 171 | 32 (18.7) | 0.3 (-8.2, 8.8) |
Global clinical response at Week 6 was assessed by a blinded, independent adjudication committee based upon prespecified clinical, radiologic, and mycologic criteria. In the subgroup of patients with proven or probable invasive aspergillosis per EORTC criteria, the global clinical response of success (complete or partial response) at Week 6 was seen in 44.8% for Noxafil-treated patients compared to 45.6% for voriconazole-treated patients (see Table 33).
| Posaconazole | Voriconazole | ||||
| Population | N | Success | N | Success | Difference Adjusted treatment difference based on Miettinen and Nurminen’s method stratified by randomization factor (risk for mortality/poor outcome), using Cochran-Mantel-Haenszel weighting scheme. (95% CI) |
| Proven/Probable Invasive Aspergillosis | 163 | 73 (44.8) | 171 | 78 (45.6) | -0.6 (-11.2, 10.1) |
2.10 Preparation and Administration Instructions for Noxafil Powdermix for Delayed Release Oral Suspension (2.10 Preparation and Administration Instructions for Noxafil PowderMix for Delayed-Release Oral Suspension)
For details on preparation and administration of Noxafil PowderMix for delayed-release oral suspension, see Instructions for Use.
Preparation Instructions for Noxafil PowderMix for Delayed-Release Oral Suspension
- Do not open the Noxafil PowderMix packet until ready to prepare the drug.
- Remove cap from the mixing liquid and push the bottle adapter into the neck of the bottle. Once in place, the bottle adapter stays in the bottle.
- Remove 9 mL of mixing liquid using the provided blue syringe. Put the cap back on the bottle. Only use the mixing liquid in the kit to prepare Noxafil PowderMix.
- Using the provided mixing cup, combine 9 mL of mixing liquid and the entire contents of one packet in the Noxafil PowderMix kit and mix (containing 300 mg of posaconazole).
- Shake the mixing cup vigorously for 45 seconds to mix the powder and mixing liquid from the Noxafil PowderMix kit. The final concentration of the reconstituted Noxafil PowderMix delayed-release suspension is approximately 30 mg/mL of posaconazole. Check to make sure the powder is mixed (the mixture should look cloudy and free of clumps).
- Must use the reconstituted Noxafil PowderMix delayed-release suspension within 1 hour of reconstitution. Discard unused portion of the reconstituted Noxafil PowderMix delayed-release suspension.
Administration Instructions for Noxafil PowderMix Delayed-Release Reconstituted Suspension
- To ensure delivery of the correct reconstituted Noxafil PowderMix Delayed-release dose, only use the provided notched tip syringes for preparation and administration because its design reduces the risk of aggregation of the product during preparation and administration.
-
Choose the correct syringe based on the prescribed Noxafil PowderMix dose:
- Use 3 mL (green) notched tip syringe (provided with the kit) if dose is 3 mL or less.
- Use 10 mL (blue) notched tip syringe (provided with the kit) if dose is more than 3 mL.
- Administer reconstituted Noxafil PowderMix suspension orally with food within 1 hour of reconstitution [see Clinical Pharmacology (12.3)].
- The maximum dose that can be accurately withdrawn from the mixing cup after reconstitution is 240 mg (8 mL).
Discarding Unused Reconstituted Noxafil PowderMix Suspension and Reuse of Syringes
- Not all the reconstituted Noxafil PowderMix suspension in the mixing cup will be used; there will be some left over in the mixing cup.
- Discard any remaining reconstituted Noxafil PowderMix suspension.
- The mixing cup may be hand washed and reused. Alternatively, the mixing cup may be discarded, and a similar mixing cup with a lid may be used for subsequent doses.
- The notched tip syringes may be hand washed and reused.
4.7 Use of Noxafil Powdermix for Delayed Release Oral Suspension in Patients With Hereditary Fructose Intolerance (4.7 Use of Noxafil PowderMix for Delayed-Release Oral Suspension in Patients with Hereditary Fructose Intolerance)
Noxafil PowderMix for delayed-release oral suspension is contraindicated in patients with known or suspected hereditary fructose intolerance (HFI) [see Warnings and Precautions (5.9) and Use in Specific Populations (8.4)].
2.3 Recommended Dosage of Noxafil for the Treatment of Invasive Aspergillosis and Prophylaxis of Invasive Aspergillus
Noxafil injection and Noxafil delayed-release tablets
The recommended dosage of (1) Noxafil injection in pediatric patients 2 years of age and older who weigh 10 kg or greater, and (2) Noxafil delayed-release tablets in pediatric patients 2 years of age and older who weigh greater than 40 kg for the treatment of invasive aspergillosis and prophylaxis of invasive Aspergillus and Candida infections is shown in Table 2 [see Dosage and Administration (2.5, 2.6, 2.7, 2.9) and Clinical Pharmacology (12.3)].
Noxafil delayed-release tablets are not recommended for use in pediatric patients who weigh 40 kg or less because the recommended dosage cannot be achieved with this dosage form.
| Recommended Pediatric Dosage by Formulation | Duration of Therapy |
|---|---|
|
Noxafil Injection (patients weighing 10 kg or greater):
Loading dose:
Maintenance dose:
Noxafil Delayed-Release Tablets (patients weighing greater than 40 kg):
|
Treatment of invasive aspergillosis:
Prophylaxis of invasive Aspergillus and Candida infections:
|
Noxafil Oral Suspension
The recommended dosage of Noxafil oral suspension in pediatric patients 13 years of age and older for the prophylaxis of invasive Aspergillus and Candida Infections is shown in Table 3.
| Recommended Pediatric Dosage of Noxafil Oral Suspension | Duration of Therapy |
|---|---|
|
200 mg (5 mL) three times a day |
Duration of therapy is based on recovery from neutropenia or immunosuppression. |
Noxafil PowderMix
The recommended dosage of Noxafil PowderMix for delayed-release oral suspension in pediatric patients 2 years of age and older who weigh 10 kg to 40 kg, for the treatment of invasive aspergillosis, and the prophylaxis of invasive Aspergillus and Candida infections, is shown in Table 4 and Table 5. The dosing for these indications is the same, except for patients weighing 10 to less than 12 kg [see Dosage and Administration (2.9, 2.10) and Clinical Pharmacology (12.3)].
Noxafil PowderMix for delayed-release oral suspension is not recommended for use in pediatric patients who weigh greater than 40 kg because the recommended dosage cannot be achieved with this dosage form.
| Weight (kg) | Recommended Pediatric Dosage of Noxafil PowderMix for Delayed-Release Oral Suspension | Duration of Therapy |
|---|---|---|
| 10 to less than 17 |
Loading Dose:
120 mg (4 mL) twice daily on the first day Maintenance Dose: 120 mg (4 mL) once daily |
Recommended total duration of therapy is 6 to 12 weeks. |
| 17 to less than 21 |
Loading Dose:
150 mg (5 mL) twice daily on the first day Maintenance Dose: 150 mg (5 mL) once daily |
|
| 21 to less than 26 |
Loading Dose:
180 mg (6 mL) twice daily on the first day Maintenance Dose: 180 mg (6 mL) once daily |
|
| 26 to less than 36 |
Loading Dose:
210 mg (7 mL) twice daily on the first day Maintenance Dose: 210 mg (7 mL) once daily |
|
| 36 to 40 |
Loading Dose:
240 mg (8 mL) twice daily on the first day Maintenance Dose: 240 mg (8 mL) once daily |
| Weight (kg) | Recommended Pediatric Dosage of Noxafil PowderMix for Delayed-Release Oral Suspension | Duration of Therapy |
|---|---|---|
| 10 to less than 12 |
Loading Dose:
90 mg (3 mL) twice daily on the first day Maintenance Dose: 90 mg (3 mL) once daily |
Duration of therapy is based on recovery from neutropenia or immunosuppression. |
| 12 to less than 17 |
Loading Dose:
120 mg (4 mL) twice daily on the first day Maintenance Dose: 120 mg (4 mL) once daily |
|
| 17 to less than 21 |
Loading Dose:
150 mg (5 mL) twice daily on the first day Maintenance Dose: 150 mg (5 mL) once daily |
|
| 21 to less than 26 |
Loading Dose:
180 mg (6 mL) twice daily on the first day Maintenance Dose: 180 mg (6 mL) once daily |
|
| 26 to less than 36 |
Loading Dose:
210 mg (7 mL) twice daily on the first day Maintenance Dose: 210 mg (7 mL) once daily |
|
| 36 to 40 |
Loading Dose:
240 mg (8 mL) twice daily on the first day Maintenance Dose: 240 mg (8 mL) once daily |
1.3 Treatment of Oropharyngeal Candidiasis Including Oropharyngeal Candidiasis Refractory to Itraconazole And/or Fluconazole (1.3 Treatment of Oropharyngeal Candidiasis Including Oropharyngeal Candidiasis Refractory to Itraconazole and/or Fluconazole)
Noxafil oral suspension is indicated for the treatment of oropharyngeal candidiasis, including oropharyngeal candidiasis refractory to itraconazole and/or fluconazole in adults and pediatric patients 13 years of age and older.
14.4 Noxafil Oral Suspension Treatment of Oropharyngeal Candidiasis Refractory to Treatment With Fluconazole Or Itraconazole (14.4 Noxafil Oral Suspension Treatment of Oropharyngeal Candidiasis Refractory to Treatment with Fluconazole or Itraconazole)
Noxafil Oral Suspension Study 4 was a noncomparative study of Noxafil oral suspension in HIV-infected subjects with OPC that was refractory to treatment with fluconazole or itraconazole. An episode of OPC was considered refractory if there was failure to improve or worsening of OPC after a standard course of therapy with fluconazole greater than or equal to 100 mg/day for at least 10 consecutive days or itraconazole 200 mg/day for at least 10 consecutive days and treatment with either fluconazole or itraconazole had not been discontinued for more than 14 days prior to treatment with Noxafil. Of the 199 subjects enrolled in this study, 89 subjects met these strict criteria for refractory infection.
Forty-five subjects with refractory OPC were treated with Noxafil oral suspension 400 mg twice daily for 3 days, followed by 400 mg once daily for 25 days with an option for further treatment during a 3 month maintenance period. Following a dosing amendment, a further 44 subjects were treated with posaconazole 400 mg twice daily for 28 days. The efficacy of Noxafil was assessed by the clinical success (cure or improvement) rate after 4 weeks of treatment. The clinical success rate was 74.2% (66/89). The clinical success rates for both the original and the amended dosing regimens were similar (73.3% and 75.0%, respectively).
2.4 Recommended Dosage of Noxafil Oral Suspension for the Treatment of Oropharyngeal Candidiasis in Pediatric Patients 13 Years of Age and Older
The recommended dosage of Noxafil oral suspension for the treatment of oropharyngeal candidiasis (OPC) and OPC refractory (rOPC) to itraconazole and/or fluconazole in pediatric patients 13 years of age and older is shown in Table 6.
The Noxafil injection, Noxafil delayed-release tablets, and Noxafil PowderMix for delayed-release oral suspension products are not approved for the treatment of oropharyngeal candidiasis in pediatric patients.
| Recommended Pediatric Dosage of Noxafil Oral Suspension | Duration of Therapy |
|---|---|
| Oropharyngeal Candidiasis (OPC) | |
|
Loading Dose:
Maintenance Dose:
|
Loading dose:
Maintenance dose:
|
| OPC Refractory (rOPC) to Itraconazole and/or Fluconazole | |
|
400 mg (10 mL) twice daily |
Duration of therapy is based on the severity of the patient’s underlying disease and clinical response. |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:41.283262 · Updated: 2026-03-14T22:51:41.355943