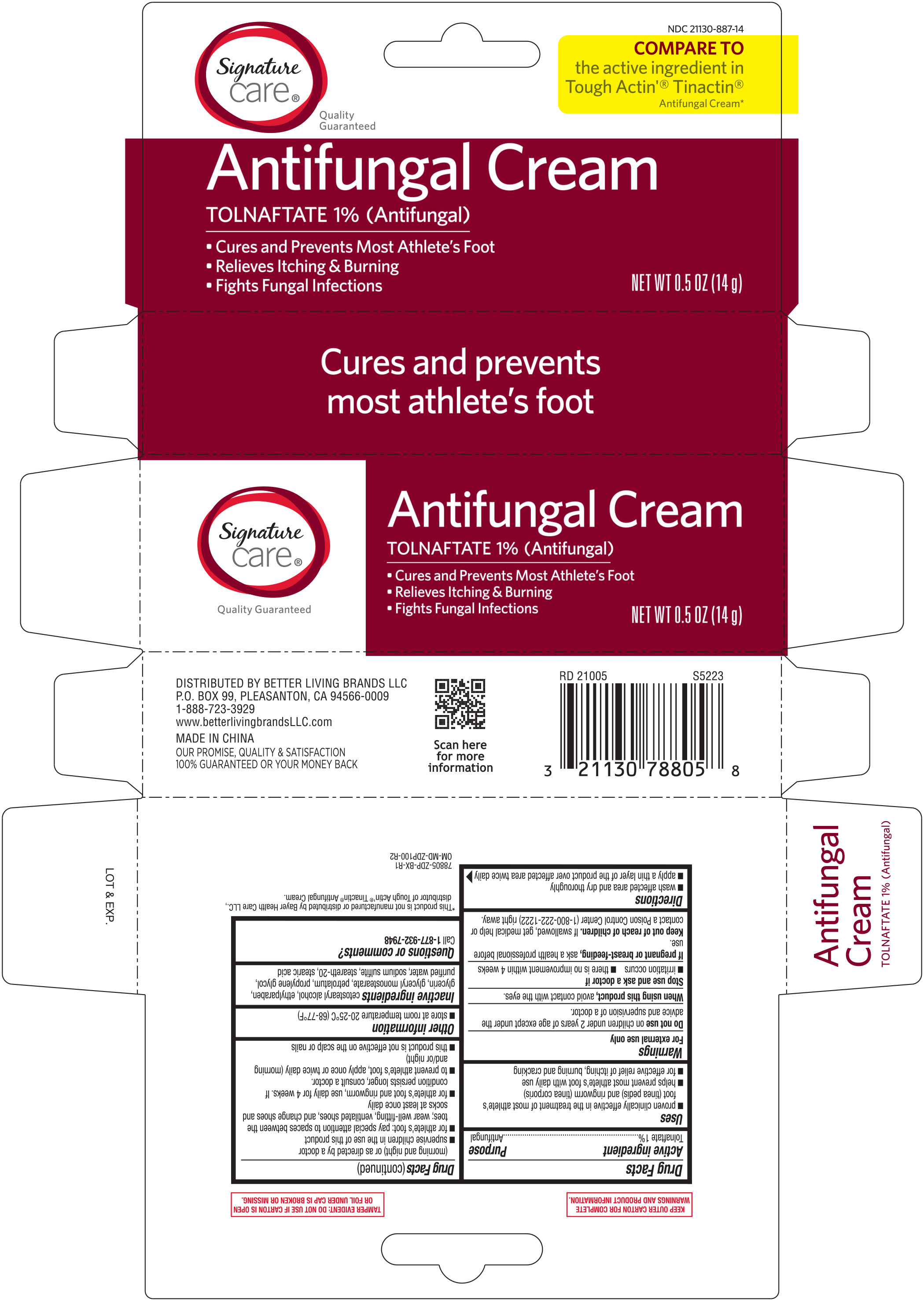
Signature Care Antifungal Cream 0.5 Oz Tolnaftate 1% 78805 (2021) Zdp
b02b81d6-8db4-927b-e053-2995a90a3958
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Purpose Tolnaftate 1%.............................................................Antifungal
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Directions
- wash affected area and dry thoroughly
- apply a thin layer of the product over affected area twice daily (morning and night) or as directed by a doctor
- supervise children in the use of this product
- for athlete's foot: pay special attention to spaces between the toes; wear well-fitting, ventilated shoes, and change shoes and socks at least once daily
- for athlete's foot and ringworm, use daily for 4 weeks. If conditions persists longer, consult a doctor.
- to prevent athlete's foot, apply once or twice daily (morning and/or night)
- this product is not effective on the scalp or nails
Dosage and Administration
Distributed by Better Living Brands LLC
P.O. Box 99, Pleasanton, CA 94566
1-888-723-3929
Made in China
Description
Active ingredient Purpose Tolnaftate 1%.............................................................Antifungal
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9
Stop use and ask a doctor if
- irritation occurs
- there is no improvement within 4 weeks
Section 50567-7
When using this product, avoid contact with the eyes
Section 50570-1
Do not use on children under 2 years of age, except under the advice and supervision of a doctor.
Section 51727-6
Inactive ingredients cetostearyl alcohol, ethylparaben, glycerin, glyceryl, monostearate, petrolatum, propylene glycol, purified water, sodium sulfite, steareth-20, stearic acid
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Uses
- proven clinically effective in the treatment of most athlete's foot (tinea pedis) and ringworm (tinea corporis)
- helps prevent most athlete's foot with daily use
- for effective relief of itching, burning and cracking
Section 55106-9
Active ingredient Purpose
Tolnaftate 1%.............................................................Antifungal
Structured Label Content
Indications and Usage (34067-9)
Directions
- wash affected area and dry thoroughly
- apply a thin layer of the product over affected area twice daily (morning and night) or as directed by a doctor
- supervise children in the use of this product
- for athlete's foot: pay special attention to spaces between the toes; wear well-fitting, ventilated shoes, and change shoes and socks at least once daily
- for athlete's foot and ringworm, use daily for 4 weeks. If conditions persists longer, consult a doctor.
- to prevent athlete's foot, apply once or twice daily (morning and/or night)
- this product is not effective on the scalp or nails
Dosage and Administration (34068-7)
Distributed by Better Living Brands LLC
P.O. Box 99, Pleasanton, CA 94566
1-888-723-3929
Made in China
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- irritation occurs
- there is no improvement within 4 weeks
Section 50567-7 (50567-7)
When using this product, avoid contact with the eyes
Section 50570-1 (50570-1)
Do not use on children under 2 years of age, except under the advice and supervision of a doctor.
Section 51727-6 (51727-6)
Inactive ingredients cetostearyl alcohol, ethylparaben, glycerin, glyceryl, monostearate, petrolatum, propylene glycol, purified water, sodium sulfite, steareth-20, stearic acid
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Uses
- proven clinically effective in the treatment of most athlete's foot (tinea pedis) and ringworm (tinea corporis)
- helps prevent most athlete's foot with daily use
- for effective relief of itching, burning and cracking
Section 55106-9 (55106-9)
Active ingredient Purpose
Tolnaftate 1%.............................................................Antifungal
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:58.709121 · Updated: 2026-03-14T23:13:11.905913