Adderall Xr
aff45863-ffe1-4d4f-8acf-c7081512a6c0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
ADDERALL XR, a CNS stimulant, is indicated for the treatment of attention deficit hyperactivity disorder (ADHD) in adults and pediatric patients 6 years and older. ( 1 ) Limitations of Use The use of ADDERALL XR is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage. ( 5.5 , 8.4 )
Dosage and Administration
Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 years of age is 30 mg once daily. ( 2.2 , 2.3 , 2.4 ) Adults: 20 mg once daily in the morning. ( 2.5 ) Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. ( 2.6 , 8.6 ) Adults with severe renal impairment: 15 mg once daily in the morning. ( 2.6 , 8.6 ) Patients with end stage renal disease (ESRD): Not recommended. ( 2.6 , 8.6 )
Contraindications
ADDERALL XR administration is contraindicated in patients: known to be hypersensitive to amphetamine, or other components of ADDERALL XR. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions ( 6.2 )] . taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions ( 5.8 ), Drug Interactions ( 7.1 )].
Warnings and Precautions
Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. ( 5.2 ) Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse at appropriate intervals. ( 5.3 ) Psychiatric Adverse Reactions: Prior to initiating ADDERALL XR, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing ADDERALL XR. ( 5.4 ) Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. ( 5.5 ) Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. ( 5.6 ) Peripheral Vasculopathy, including Raynaud’s Phenomenon: Careful observation for digital changes is necessary during ADDERALL XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. ( 5.7 ) Serotonin Syndrome: Increased risk when coadministered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue ADDERALL XR and initiate supportive treatment. ( 4 , 5.8 , 10 ) Motor and Verbal Tics, and Worsening of Tourette’s Syndrome: Before initiating ADDERALL XR, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette’s syndrome. Discontinue treatment if clinically appropriate. ( 5.9 )
Adverse Reactions
The following adverse reactions have been identified during postapproval use of amphetamine, ADDERALL XR, or Adderall. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Allergic: Urticaria, rash, hypersensitivity reactions including angioedema and anaphylaxis. Serious skin rashes, including Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported. Cardiovascular: Palpitations. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use. Central Nervous System: Psychotic episodes at recommended doses, overstimulation, restlessness, irritability, euphoria, dyskinesia, dysphoria, depression, tremor, motor and verbal tics, aggression, anger, logorrhea, dermatillomania, paresthesia (including formication), and bruxism. Endocrine: Impotence, changes in libido, frequent or prolonged erections. Eye Disorders: Vision blurred, mydriasis. Gastrointestinal: Unpleasant taste, constipation, intestinal ischemia, and other gastrointestinal disturbances. Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis. Skin: Alopecia. Vascular Disorders: Raynaud’s phenomenon.
Drug Interactions
Alkalinizing agents (GI antacids and urinary): These agents increase blood levels of amphetamine. ( 2.7 , 7.1 ) Acidifying agents (GI and urinary): These agents reduce blood levels of amphetamine. ( 7.1 )
How Supplied
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg), bottles of 100, NDC 54092-381-01 ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg), bottles of 100, NDC 54092-383-01 ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg), bottles of 100, NDC 54092-385-01 ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg), bottles of 100, NDC 54092-387-01 ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg), bottles of 100, NDC 54092-389-01 ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg), bottles of 100, NDC 54092-391-01
Storage and Handling
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg), bottles of 100, NDC 54092-381-01 ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg), bottles of 100, NDC 54092-383-01 ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg), bottles of 100, NDC 54092-385-01 ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg), bottles of 100, NDC 54092-387-01 ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg), bottles of 100, NDC 54092-389-01 ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg), bottles of 100, NDC 54092-391-01
Description
ADDERALL XR has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage ( 10 )] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection. Before prescribing ADDERALL XR, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout ADDERALL XR treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions ( 5.1 ), Drug Abuse and Dependence ( 9.2 )] .
Medication Information
Warnings and Precautions
Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. ( 5.2 ) Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse at appropriate intervals. ( 5.3 ) Psychiatric Adverse Reactions: Prior to initiating ADDERALL XR, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing ADDERALL XR. ( 5.4 ) Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. ( 5.5 ) Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. ( 5.6 ) Peripheral Vasculopathy, including Raynaud’s Phenomenon: Careful observation for digital changes is necessary during ADDERALL XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. ( 5.7 ) Serotonin Syndrome: Increased risk when coadministered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue ADDERALL XR and initiate supportive treatment. ( 4 , 5.8 , 10 ) Motor and Verbal Tics, and Worsening of Tourette’s Syndrome: Before initiating ADDERALL XR, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette’s syndrome. Discontinue treatment if clinically appropriate. ( 5.9 )
Indications and Usage
ADDERALL XR, a CNS stimulant, is indicated for the treatment of attention deficit hyperactivity disorder (ADHD) in adults and pediatric patients 6 years and older. ( 1 ) Limitations of Use The use of ADDERALL XR is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage. ( 5.5 , 8.4 )
Dosage and Administration
Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 years of age is 30 mg once daily. ( 2.2 , 2.3 , 2.4 ) Adults: 20 mg once daily in the morning. ( 2.5 ) Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. ( 2.6 , 8.6 ) Adults with severe renal impairment: 15 mg once daily in the morning. ( 2.6 , 8.6 ) Patients with end stage renal disease (ESRD): Not recommended. ( 2.6 , 8.6 )
Contraindications
ADDERALL XR administration is contraindicated in patients: known to be hypersensitive to amphetamine, or other components of ADDERALL XR. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions ( 6.2 )] . taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions ( 5.8 ), Drug Interactions ( 7.1 )].
Adverse Reactions
The following adverse reactions have been identified during postapproval use of amphetamine, ADDERALL XR, or Adderall. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Allergic: Urticaria, rash, hypersensitivity reactions including angioedema and anaphylaxis. Serious skin rashes, including Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported. Cardiovascular: Palpitations. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use. Central Nervous System: Psychotic episodes at recommended doses, overstimulation, restlessness, irritability, euphoria, dyskinesia, dysphoria, depression, tremor, motor and verbal tics, aggression, anger, logorrhea, dermatillomania, paresthesia (including formication), and bruxism. Endocrine: Impotence, changes in libido, frequent or prolonged erections. Eye Disorders: Vision blurred, mydriasis. Gastrointestinal: Unpleasant taste, constipation, intestinal ischemia, and other gastrointestinal disturbances. Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis. Skin: Alopecia. Vascular Disorders: Raynaud’s phenomenon.
Drug Interactions
Alkalinizing agents (GI antacids and urinary): These agents increase blood levels of amphetamine. ( 2.7 , 7.1 ) Acidifying agents (GI and urinary): These agents reduce blood levels of amphetamine. ( 7.1 )
Storage and Handling
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg), bottles of 100, NDC 54092-381-01 ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg), bottles of 100, NDC 54092-383-01 ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg), bottles of 100, NDC 54092-385-01 ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg), bottles of 100, NDC 54092-387-01 ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg), bottles of 100, NDC 54092-389-01 ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg), bottles of 100, NDC 54092-391-01
How Supplied
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg), bottles of 100, NDC 54092-381-01 ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg), bottles of 100, NDC 54092-383-01 ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg), bottles of 100, NDC 54092-385-01 ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg), bottles of 100, NDC 54092-387-01 ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg), bottles of 100, NDC 54092-389-01 ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg), bottles of 100, NDC 54092-391-01
Description
ADDERALL XR has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage ( 10 )] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection. Before prescribing ADDERALL XR, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout ADDERALL XR treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions ( 5.1 ), Drug Abuse and Dependence ( 9.2 )] .
Section 42229-5
Adverse Reactions Leading to Discontinuation of Treatment
In two placebo-controlled studies of up to 5 weeks duration among children with ADHD, 2.4% (10/425) of ADDERALL XR-treated patients discontinued due to adverse reactions (including three patients with loss of appetite, one of whom also reported insomnia) compared to 2.7% (7/259) receiving placebo.
The most frequent adverse reactions leading to discontinuation of ADDERALL XR in controlled and uncontrolled, multiple-dose clinical trials of children (N=595) were anorexia (loss of appetite) (2.9%), insomnia (1.5%), weight loss (1.2%), emotional lability (1%), and depression (0.7%). Over half of these patients were exposed to ADDERALL XR for 12 months or more.
In a separate placebo-controlled 4 week study in adolescents with ADHD, five patients (2.1%) discontinued treatment due to adverse events among ADDERALL XR-treated patients (N=233) compared to none who received placebo (N=54). The most frequent adverse event leading to discontinuation and considered to be drug-related (i.e., leading to discontinuation in at least 1% of ADDERALL XR-treated patients and at a rate at least twice that of placebo) was insomnia (1.3%, n=3).
In one placebo-controlled 4 week study among adults with ADHD with doses 20 to 60 mg, 23 patients (12.0%) discontinued treatment due to adverse events among ADDERALL XR-treated patients (N=191) compared to one patient (1.6%) who received placebo (N=64). The most frequent adverse events leading to discontinuation and considered to be drug-related (i.e., leading to discontinuation in at least 1% of ADDERALL XR-treated patients and at a rate at least twice that of placebo) were insomnia (5.2%, n=10), anxiety (2.1%, n=4), nervousness (1.6%, n=3), dry mouth (1.6%, n=3), anorexia (1.6%, n=3), tachycardia (1.6%, n=3), headache (1.6%, n=3), and asthenia (1.0%, n=2).
Section 42231-1
| MEDICATION GUIDE ADDERALL XR® (ADD-ur-all X-R) (mixed salts of a single-entity amphetamine product) extended-release capsules, CII |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ADL364 Revised: 09/2025 | |
|
What is the most important information I should know about ADDERALL XR? ADDERALL XR may cause serious side effects, including: Abuse, misuse, and addiction. ADDERALL XR has a high chance for abuse and misuse and may lead to substance use problems, including addiction. Misuse and abuse of ADDERALL XR, other amphetamine containing medicines, and methylphenidate containing medicines, can lead to overdose and death. The risk of overdose and death is increased with higher doses of ADDERALL XR or when it is used in ways that are not approved, such as snorting or injection.
|
||
Tell your healthcare provider if you or your child have ever abused or been dependent on alcohol, prescription medicines, or street drugs.
|
||
| Your healthcare provider should check you or your child carefully for heart problems before starting treatment with ADDERALL XR. Tell your healthcare provider if you or your child have any heart problems, heart disease, or heart defects. | ||
Call your healthcare provider or go to the nearest hospital emergency room right away if you or your child have any signs of heart problems such as chest pain, shortness of breath, or fainting during treatment with ADDERALL XR.
|
||
| Tell your healthcare provider about any mental problems you or your child have or about a family history of suicide, bipolar illness, or depression. | ||
| Call your healthcare provider right away if you or your child have any new or worsening mental symptoms or problems during treatment with ADDERALL XR, especially hearing voices, seeing or believing things that are not real, or new manic symptoms. | ||
| What is ADDERALL XR? | ||
| ADDERALL XR is a central nervous system (CNS) stimulant prescription medicine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in adults and children 6 years of age and older. ADDERALL XR may help increase attention and decrease impulsiveness and hyperactivity in people with ADHD. | ||
| ADDERALL XR is not recommended for use in children under 6 years of age with ADHD. | ||
| ADDERALL XR is a federally controlled substance (CII) because it contains amphetamine that can be a target for people who abuse prescription medicines or street drugs. Keep ADDERALL XR in a safe place to protect it from theft. Never give your ADDERALL XR to anyone else because it may cause death or harm them. Selling or giving away ADDERALL XR may harm others and is against the law. | ||
Do not take ADDERALL XR if you or your child:
|
||
Before taking ADDERALL XR tell your healthcare provider about all of your or your child’s medical conditions, including if you or your child:
|
||
| Tell your healthcare provider about all of the medicines that you or your child take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | ||
| ADDERALL XR and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be changed during treatment with ADDERALL XR. | ||
| Your healthcare provider will decide if ADDERALL XR can be taken with other medicines. | ||
| Especially tell your healthcare provider if you or your child take: | ||
|
|
|
| Know the medicines that you or your child take. Keep a list of your or your child’s medicines with you to show your healthcare provider and pharmacist when you or your child get a new medicine. | ||
| Do not start any new medicine during treatment with ADDERALL XR without talking to your healthcare provider first. | ||
How should ADDERALL XR be taken?
|
||
|
What are the possible side effects of ADDERALL XR? ADDERALL XR may cause serious side effects, including:
|
||
|
||
|
|
|
|
||
| The most common side effects of ADDERALL XR in children ages 6 to 12 include: | ||
|
|
|
| The most common side effects of ADDERALL XR in adolescents ages 13 to 17 include: | ||
|
|
|
| The most common side effects of ADDERALL XR in adults include: | ||
|
|
|
| These are not all the possible side effects of ADDERALL XR. | ||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
How should I store ADDERALL XR?
|
||
|
General information about the safe and effective use of ADDERALL XR.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ADDERALL XR for a condition for which it was not prescribed. Do not give ADDERALL XR to other people, even if they have the same condition. It may harm them and it is against the law. |
||
| You can ask your healthcare provider or pharmacist for information about ADDERALL XR that is written for healthcare professionals. | ||
| What are the ingredients in ADDERALL XR? | ||
| Active ingredient: dextroamphetamine sulfate, amphetamine sulfate, dextroamphetamine saccharate, and amphetamine aspartate monohydrate | ||
| Inactive ingredients: gelatin capsules, hydroxypropyl methylcellulose, methacrylic acid copolymer, Opadry® beige, sugar spheres, talc, and triethyl citrate. Gelatin capsules contain edible inks, kosher gelatin, and titanium dioxide. The 5 mg, 10 mg, and 15 mg capsules also contain FD&C Blue #2. The 20 mg, 25 mg, and 30 mg capsules also contain red iron oxide and yellow iron oxide | ||
|
Distributed by: Takeda Pharmaceuticals America, Inc, Cambridge, MA 02142 |
||
| ADDERALL XR is a registered trademark of Takeda Pharmaceuticals U.S.A., Inc. OPADRY is a registered trademark of BPSI Holdings, LLC. © 2025 Takeda Pharmaceuticals U.S.A., Inc. All rights reserved. |
||
| For more information, you may also contact Takeda Pharmaceuticals (the maker of ADDERALL XR) at 1-877-825-3327 or visit the website at http://www.adderallxr.com. |
Section 44425-7
Dispense in a tight, light-resistant container as defined in the USP.
Store at room temperature, 20 to 25ºC (68 to 77ºF). Excursions permitted to 15 to 30ºC (59 to 86ºF) [see USP Controlled Room Temperature].
9.2 Abuse
ADDERALL XR has a high potential for abuse and misuse which can lead to the development of a substance use disorder, including addiction [see Warnings and Precautions (5.1)]. ADDERALL XR can be diverted for non-medical use into illicit channels or distribution.
Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a healthcare provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of amphetamine may cause increased heart rate, respiratory rate, or blood pressure; sweating; dilated pupils; hyperactivity; restlessness; insomnia; decreased appetite; loss of coordination; tremors; flushed skin; vomiting; and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed with CNS stimulants abuse and/or misuse. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
5.6 Seizures
There is some clinical evidence that stimulants may lower the convulsive threshold in patients with prior history of seizures, in patients with prior EEG abnormalities in the absence of seizures, and very rarely, in patients without a history of seizures and no prior EEG evidence of seizures. In the presence of seizures, ADDERALL XR should be discontinued.
10 Overdosage
Clinical Effects of Overdose
Overdose of CNS stimulants is characterized by the following sympathomimetic effects:
- Cardiovascular effects including tachyarrhythmias, and hypertension or hypotension. Vasospasm, myocardial infarction, or aortic dissection may precipitate sudden cardiac death. Takotsubo cardiomyopathy may develop.
- CNS effects including psychomotor agitation, confusion, and hallucinations. Serotonin syndrome, seizures, cerebral vascular accidents, and coma may occur.
- Life-threatening hyperthermia (temperatures greater than 104°F) and rhabdomyolysis may develop.
Overdose Management
Consider the possibility of multiple drug ingestion. The pharmacokinetic profile of ADDERALL XR should be considered when treating patients with overdose. D-amphetamine is not dialyzable. Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description
ADDERALL XR extended-release capsules contain mixed salts of a single-entity amphetamine, a CNS stimulant. ADDERALL XR contains equal amounts (by weight) of four salts: dextroamphetamine sulfate, amphetamine sulfate, dextroamphetamine saccharate and amphetamine (D, L)-aspartate monohydrate. This results in a 3.1:1 mixture of dextro- to levo-amphetamine base equivalent.
The 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, and 30 mg strength extended-release capsules are for oral administration. ADDERALL XR contains two types of drug-containing beads (immediate-release and delayed-release) which prolong the release of amphetamine compared to the Adderall (immediate-release) tablet formulation.
Each capsule contains:
| Capsule Strength | 5 mg | 10 mg | 15 mg | 20 mg | 25 mg | 30 mg |
|---|---|---|---|---|---|---|
| Dextroamphetamine Saccharate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine (D,L)-Aspartate Monohydrate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Dextroamphetamine Sulfate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine Sulfate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Total amphetamine base equivalence | 3.1 mg | 6.3 mg | 9.4 mg | 12.5 mg | 15.6 mg | 18.8 mg |
| d-amphetamine base equivalence | 2.4 mg | 4.7 mg | 7.1 mg | 9.5 mg | 11.9 mg | 14.2 mg |
| l-amphetamine base equivalence | 0.75 mg | 1.5 mg | 2.3 mg | 3.0 mg | 3.8 mg | 4.5 mg |
9.3 Dependence
Physical Dependence
ADDERALL XR may produce physical dependence. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
Withdrawal signs and symptoms after abrupt discontinuation or dose reduction following prolonged use of CNS stimulants including ADDERALL XR include dysphoric mood; depression; fatigue; vivid, unpleasant dreams; insomnia or hypersomnia; increased appetite; and psychomotor retardation or agitation.
Tolerance
ADDERALL XR may produce tolerance. Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose).
8.4 Pediatric Use
The safety and effectiveness of ADDERALL XR have not been established in pediatric patients less than 6 years of age.
The safety and effectiveness of ADDERALL XR have been established in pediatric patients with ADHD 6 years of age and older.
In studies evaluating extended-release amphetamine products, patients 4 to <6 years of age had higher systemic amphetamine exposures than those observed in older pediatric patients at the same dosage. Pediatric patients 4 to <6 years of age also had a higher incidence of adverse reactions, including weight loss.
8.5 Geriatric Use
ADDERALL XR has not been studied in the geriatric population.
4 Contraindications
ADDERALL XR administration is contraindicated in patients:
- known to be hypersensitive to amphetamine, or other components of ADDERALL XR. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions (6.2)].
- taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions (5.8), Drug Interactions (7.1)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Abuse, Misuse, and Addiction [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)]
- Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2)]
- Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4)]
- Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.5)]
- Seizures [see Warnings and Precautions (5.6)]
- Peripheral Vasculopathy, including Raynaud’s Phenomenon [see Warnings and Precautions (5.7)]
- Serotonin Syndrome [see Warnings and Precautions (5.8)]
- Motor and Verbal Tics, and Worsening of Tourette’s Syndrome [see Warnings and Precautions (5.9)]
7 Drug Interactions
8.6 Renal Impairment
Due to reduced clearance of amphetamines in patients with severe renal impairment (GFR 15 to <30 mL/min/1.73 m2), the recommended dose should be reduced. ADDERALL XR is not recommended in patients with ESRD (GFR <15 mL/min/1.73 m2) [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
d-Amphetamine is not dialyzable.
12.2 Pharmacodynamics
Amphetamines block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
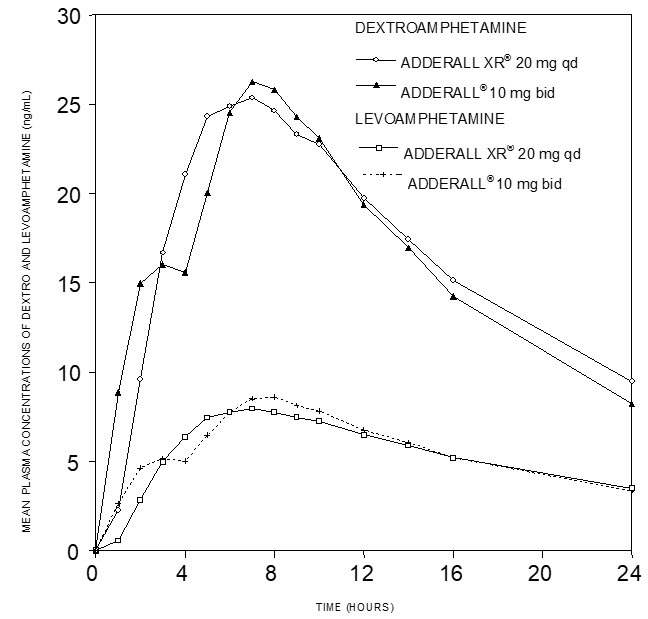
Pharmacokinetic studies of ADDERALL XR have been conducted in healthy adult and pediatric (children aged 6 to 12 yrs) subjects, adolescent (13 to 17 yrs), and children with ADHD. Both Adderall (immediate-release) tablets and ADDERALL XR extended-release capsules contain d-amphetamine and l-amphetamine salts in the ratio of 3:1. Following administration of Adderall (immediate-release), the peak plasma concentrations occurred in about 3 hours for both d-amphetamine and l-amphetamine.
The time to reach maximum plasma concentration (Tmax) for ADDERALL XR is about 7 hours, which is about 4 hours longer compared to Adderall (immediate-release). This is consistent with the extended-release nature of the product.
Figure 1: Mean d-amphetamine and l-amphetamine Plasma Concentrations Following Administration of ADDERALL XR 20 mg (8 am) and Adderall (immediate-release) 10 mg Twice Daily (8 am and 12 noon) in the Fed State.
A single dose of ADDERALL XR 20 mg extended-release capsules provided comparable plasma concentration profiles of both d-amphetamine and l-amphetamine to Adderall (immediate-release) 10 mg twice daily administered 4 hours apart.
The mean elimination half-life for d-amphetamine is 10 hours in adults; 11 hours in adolescents aged 13 to 17 years and weighing less than or equal to 75 kg/165 lbs; and 9 hours in children aged 6 to 12 years. For the l-amphetamine, the mean elimination half-life in adults is 13 hours; 13 to 14 hours in adolescents; and 11 hours in children aged 6 to 12 years. On a mg/kg body weight basis, children have a higher clearance than adolescents or adults (see Special Populations).
ADDERALL XR demonstrates linear pharmacokinetics over the dose range of 20 to 60 mg in adults and adolescents weighing greater than 75 kg/165 lbs, over the dose range of 10 to 40 mg in adolescents weighing less than or equal to 75 kg/165 lbs, and 5 to 30 mg in children aged 6 to 12 years. There is no unexpected accumulation at steady state in children.
Food does not affect the extent of absorption of d-amphetamine and l-amphetamine, but prolongs Tmax by 2.5 hours (from 5.2 hrs at fasted state to 7.7 hrs after a high-fat meal) for d-amphetamine and 2.7 hours (from 5.6 hrs at fasted state to 8.3 hrs after a high-fat meal) for l-amphetamine after administration of ADDERALL XR 30 mg. Opening the capsule and sprinkling the contents on applesauce results in comparable absorption to the intact capsule taken in the fasted state. Equal doses of ADDERALL XR strengths are bioequivalent.
5.8 Serotonin Syndrome
Serotonin syndrome, a potentially life-threatening reaction, may occur when amphetamines are used in combination with other drugs that affect the serotonergic neurotransmitter systems such as MAOIs, selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John’s Wort [see Drug Interactions (7.1)]. Amphetamines and amphetamine derivatives are known to be metabolized, to some degree, by cytochrome P450 2D6 (CYP2D6) and display minor inhibition of CYP2D6 metabolism [see Clinical Pharmacology (12.3)]. The potential for a pharmacokinetic interaction exists with the coadministration of CYP2D6 inhibitors which may increase the risk with increased exposure to ADDERALL XR. In these situations, consider an alternative nonserotonergic drug or an alternative drug that does not inhibit CYP2D6 [see Drug Interactions (7.1)]. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
Concomitant use of ADDERALL XR with MAOI drugs is contraindicated [see Contraindications (4)].
Discontinue treatment with ADDERALL XR and any concomitant serotonergic agents immediately if symptoms of serotonin syndrome occur, and initiate supportive symptomatic treatment. Concomitant use of ADDERALL XR with other serotonergic drugs or CYP2D6 inhibitors should be used only if the potential benefit justifies the potential risk. If clinically warranted, consider initiating ADDERALL XR with lower doses, monitoring patients for the emergence of serotonin syndrome during drug initiation or titration, and informing patients of the increased risk for serotonin syndrome.
1 Indications and Usage
ADDERALL XR, a CNS stimulant, is indicated for the treatment of attention deficit hyperactivity disorder (ADHD) in adults and pediatric patients 6 years and older. (1)
Limitations of Use
The use of ADDERALL XR is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage. (5.5, 8.4)
12.1 Mechanism of Action
Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
ADDERALL XR contains amphetamine, a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to treating patients with ADDERALL XR, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2)].
- the family history and clinically evaluate patients for motor or verbal tics or Tourette’s syndrome before initiating ADDERALL XR [see Warnings and Precautions (5.9)].
5 Warnings and Precautions
- Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. (5.2)
- Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse at appropriate intervals. (5.3)
- Psychiatric Adverse Reactions: Prior to initiating ADDERALL XR, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing ADDERALL XR. (5.4)
- Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. (5.5)
- Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. (5.6)
- Peripheral Vasculopathy, including Raynaud’s Phenomenon: Careful observation for digital changes is necessary during ADDERALL XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. (5.7)
- Serotonin Syndrome: Increased risk when coadministered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue ADDERALL XR and initiate supportive treatment. (4, 5.8, 10)
- Motor and Verbal Tics, and Worsening of Tourette’s Syndrome: Before initiating ADDERALL XR, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette’s syndrome. Discontinue treatment if clinically appropriate. (5.9)
2 Dosage and Administration
- Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 years of age is 30 mg once daily. (2.2, 2.3, 2.4)
- Adults: 20 mg once daily in the morning. (2.5)
- Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. (2.6, 8.6)
- Adults with severe renal impairment: 15 mg once daily in the morning. (2.6, 8.6)
- Patients with end stage renal disease (ESRD): Not recommended. (2.6, 8.6)
3 Dosage Forms and Strengths
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg)
ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg)
ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg)
ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg)
ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg)
ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg)
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The premarketing development program for ADDERALL XR included exposures in a total of 1,315 participants in clinical trials (635 pediatric patients, 350 adolescent patients, 248 adult patients, and 82 healthy adult subjects). Of these, 635 patients (ages 6 to 12) were evaluated in two controlled clinical studies, one open-label clinical study, and two single-dose clinical pharmacology studies (N=40). Safety data on all patients are included in the discussion that follows. Adverse reactions were assessed by collecting adverse reactions, results of physical examinations, vital signs, weights, laboratory analyses, and ECGs.
Adverse reactions during exposure were obtained primarily by general inquiry and recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse reactions without first grouping similar types of reactions into a smaller number of standardized event categories. In the tables and listings that follow, COSTART terminology has been used to classify reported adverse reactions.
The stated frequencies of adverse reactions represent the proportion of individuals who experienced, at least once, a treatment-emergent adverse event of the type listed.
2.5 Recommended Dosage in Adults
In adults with ADHD who are either starting treatment for the first time or switching from another medication, the recommended dose is 20 mg/day.
5.1 Abuse, Misuse, and Addiction
ADDERALL XR has a high potential for abuse and misuse. The use of ADDERALL XR exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. ADDERALL XR can be diverted for non-medical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2)]. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing ADDERALL XR, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store ADDERALL XR in a safe place, preferably locked, and instruct patients to not give ADDERALL XR to anyone else. Throughout ADDERALL XR treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Abuse, Misuse, and Addiction
Educate patients and their families about the risks of abuse, misuse, and addiction of ADDERALL XR, which can lead to overdose and death, and proper disposal of any unused drug [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2), Overdosage (10)]. Advise patients to store ADDERALL XR in a safe place, preferably locked, and instruct patients to not give ADDERALL XR to anyone else.
Risks to Patients with Serious Cardiac Disease
Advise patients that there are potential risks to patients with serious cardiac disease, including sudden death, with ADDERALL XR use. Instruct patients to contact a healthcare provider immediately if they develop symptoms such as exertional chest pain, unexplained syncope, or other symptoms suggestive of cardiac disease [see Warnings and Precautions (5.2)].
Increased Blood Pressure and Heart Rate
Advise patients that ADDERALL XR can cause elevations in blood pressure and heart rate [see Warnings and Precautions (5.3)].
Psychiatric Adverse Reactions
Prior to initiating treatment with ADDERALL XR, adequately screen patients with comorbid depressive symptoms to determine if they are at risk for bipolar disorder. Such screening should include a detailed psychiatric history, including a family history of suicide, bipolar disorder, and/or depression. Additionally, ADDERALL XR therapy at usual doses may cause treatment-emergent psychotic or manic symptoms in patients without prior history of psychotic symptoms or mania [see Warnings and Precautions (5.4)].
Circulation Problems in Fingers and Toes [Peripheral Vasculopathy, including Raynaud’s Phenomenon]
Instruct patients beginning treatment with ADDERALL XR about the risk of peripheral vasculopathy, including Raynaud’s phenomenon, and in associated signs and symptoms: fingers or toes may feel numb, cool, painful, and/or may change color from pale, to blue, to red. Instruct patients to report to their physician any new numbness, pain, skin color change, or sensitivity to temperature in fingers or toes. Instruct patients to call their physician immediately with any signs of unexplained wounds appearing on fingers or toes while taking ADDERALL XR. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients [see Warnings and Precautions (5.7)].
Serotonin Syndrome
Caution patients about the risk of serotonin syndrome with concomitant use of ADDERALL XR and other serotonergic drugs including SSRIs, SNRIs, triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John’s Wort, and with drugs that impair metabolism of serotonin (in particular MAOIs, both those intended to treat psychiatric disorders and also others such as linezolid) [see Contraindications (4), Warnings and Precautions (5.8), Drug Interactions (7.1)]. Advise patients to contact their healthcare provider or report to the emergency room if they experience signs or symptoms of serotonin syndrome.
Concomitant Medications
Advise patients to notify their physicians if they are taking, or plan to take, any prescription or over-the-counter drugs because there is a potential for interactions [see Drug Interactions (7.1)].
Growth
Monitor growth in children during treatment with ADDERALL XR, and patients who are not growing or gaining weight as expected may need to have their treatment interrupted [see Warnings and Precautions (5.5)].
Motor and Verbal Tics, and Worsening of Tourette’s Syndrome
Advise patients that motor and verbal tics and worsening of Tourette’s syndrome may occur during treatment with ADDERALL XR. Instruct patients to notify their healthcare provider if emergence of new tics or worsening of tics or Tourette’s syndrome occurs [see Warnings and Precautions (5.9)].
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to ADDERALL XR during pregnancy [see Use in Specific Populations (8.1)].
Pregnancy
Advise patients to notify their healthcare provider if they become pregnant or intend to become pregnant during treatment with ADDERALL XR. Advise patients of the potential fetal effects from the use of ADDERALL XR during pregnancy [see Use in Specific Populations (8.1)].
Lactation
Advise women not to breastfeed if they are taking ADDERALL XR [see Use in Specific Populations (8.2)].
5.4 Psychiatric Adverse Reactions
Exacerbation of Pre-Existing Psychosis
Administration of stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with pre-existing psychotic disorder.
Induction of a Manic Episode in Patients with Bipolar Disease
CNS stimulants may induce a manic or mixed episode in patients. Prior to initiating ADDERALL XR treatment, screen patients for risk factors for developing a manic episode (e.g., comorbid or history of depressive symptoms or a family history of suicide, bipolar disorder, or depression).
New Psychotic or Manic Symptoms
CNS stimulants, at the recommended dosage, may cause psychotic or manic symptoms (e.g., hallucinations, delusional thinking, or mania) in patients without a prior history of psychotic illness or mania. In a pooled analysis of multiple short-term, placebo-controlled studies of CNS stimulants, psychotic or manic symptoms occurred in approximately 0.1% of CNS stimulant-treated patients compared to 0% of placebo-treated patients. If such symptoms occur, consider discontinuing ADDERALL XR.
16 How Supplied/storage and Handling
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg), bottles of 100, NDC 54092-381-01
ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg), bottles of 100, NDC 54092-383-01
ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg), bottles of 100, NDC 54092-385-01
ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg), bottles of 100, NDC 54092-387-01
ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg), bottles of 100, NDC 54092-389-01
ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg), bottles of 100, NDC 54092-391-01
7.2 Drug Laboratory Test Interactions
Amphetamines can cause a significant elevation in plasma corticosteroid levels. This increase is greatest in the evening. Amphetamines may interfere with urinary steroid determinations.
Warning: Abuse, Misuse, and Addiction
ADDERALL XR has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing ADDERALL XR, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout ADDERALL XR treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2)].
2.2 General Administration Information
Individualize the dosage according to the therapeutic needs and response of the patient. Administer ADDERALL XR at the lowest effective dosage.
Based on bioequivalence data, patients taking divided doses of immediate-release ADDERALL, (for example, twice daily), may be switched to ADDERALL XR at the same total daily dose taken once daily. Titrate at weekly intervals to appropriate efficacy and tolerability as indicated.
ADDERALL XR extended-release capsules may be taken whole, or the capsule may be opened and the entire contents sprinkled on applesauce. If the patient is using the sprinkle administration method, the sprinkled applesauce should be consumed immediately; it should not be stored. Patients should take the applesauce with sprinkled beads in its entirety without chewing. The dose of a single capsule should not be divided. The contents of the entire capsule should be taken, and patients should not take anything less than one capsule per day.
ADDERALL XR may be taken orally with or without food.
ADDERALL XR should be given upon awakening. Afternoon doses should be avoided because of the potential for insomnia.
13.2 Animal Toxicology And/or Pharmacology
Acute administration of high doses of amphetamine (d- or d, l-) has been shown to produce long-lasting neurotoxic effects, including irreversible nerve fiber damage in rodents. The significance of these findings to humans is unknown.
5.3 Increased Blood Pressure and Heart Rate
CNS stimulants may cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 bpm).
Monitor all ADDERALL XR-treated patients for hypertension and tachycardia.
1.1 Attention Deficit Hyperactivity Disorder
ADDERALL XR® is indicated for the treatment of attention deficit hyperactivity disorder (ADHD) in adults and pediatric patients 6 years and older.
Limitations of Use
The use of ADDERALL XR is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.5), Use in Specific Populations (8.4)].
2.6 Dosage in Patients With Renal Impairment
In adult patients with severe renal impairment (GFR 15 to <30 mL/min/1.73 m2), the recommended dose is 15 mg once daily in the morning. In pediatric patients (6 to 17 years of age) with severe renal impairment, the recommended dose is 5 mg once daily. The maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. ADDERALL XR is not recommended in patients with end stage renal disease (ESRD) (GFR <15 mL/min/1.73 m2) [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.7 Dosage Modification Due to Drug Interactions
Agents that alter urinary pH can impact excretion and alter blood levels of amphetamines. Acidifying agents (e.g., ascorbic acid) decrease blood levels; adjust ADDERALL XR dosage based on clinical response [see Drug Interactions (7)].
5.2 Risks to Patients With Serious Cardiac Disease
Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosage. Avoid ADDERALL XR use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmia, coronary artery disease, or other serious cardiac disease.
Principal Display Panel 5 Mg Capsule Bottle Label
NDC 54092-381-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
5 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 10 Mg Capsule Bottle Label
NDC 54092-383-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
10 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 15 Mg Capsule Bottle Label
NDC 54092-385-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
15 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 20 Mg Capsule Bottle Label
NDC 54092-387-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
20 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 25 Mg Capsule Bottle Label
NDC 54092-389-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
25 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 30 Mg Capsule Bottle Label
NDC 54092-391-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
30 mg
CII
100 Capsules
Rx only
Takeda
7.1 Clinically Important Interactions With Amphetamines
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact | Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure. |
| Intervention | Do not administer ADDERALL XR concomitantly or within 14 days after discontinuing MAOI [see Contraindications (4)]. |
| Serotonergic Drugs | |
| Clinical Impact | The concomitant use of ADDERALL XR and serotonergic drugs increases the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during ADDERALL XR initiation or dosage increase. If serotonin syndrome occurs, discontinue ADDERALL XR and the concomitant serotonergic drug(s) [see Warnings and Precautions (5.8)]. |
| CYP2D6 Inhibitors | |
| Clinical Impact | The concomitant use of ADDERALL XR and CYP2D6 inhibitors may increase the exposure of ADDERALL XR compared to the use of the drug alone and increase the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during ADDERALL XR initiation and after a dosage increase. If serotonin syndrome occurs, discontinue ADDERALL XR and the CYP2D6 inhibitor [see Warnings and Precautions (5.8), Overdosage (10)]. |
| Alkalinizing Agents | |
| Clinical Impact | Increase blood levels and potentiate the action of amphetamine. |
| Intervention | Coadministration of ADDERALL XR and gastrointestinal or urinary alkalinizing agents should be avoided. |
| Acidifying Agents | |
| Clinical Impact | Lower blood levels and efficacy of amphetamines. |
| Intervention | Increase dose based on clinical response. |
| Tricyclic Antidepressants | |
| Clinical Impact | May enhance the activity of tricyclic or sympathomimetic agents causing striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated. |
| Intervention | Monitor frequently and adjust or use alternative therapy based on clinical response. |
| Proton Pump Inhibitors | |
| Clinical Impact | Time to maximum concentration (Tmax) of amphetamine is decreased compared to when administered alone. |
| Intervention | Monitor patients for changes in clinical effect and adjust therapy based on clinical response. |
5.5 Long Term Suppression of Growth in Pediatric Patients
ADDERALL XR is not approved for use and is not recommended in pediatric patients below 6 years of age [see Use in Specific Populations (8.4)].
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients. Closely monitor growth (weight and height) in ADDERALL XR-treated pediatric patients treated with CNS stimulants.
In a controlled trial of ADDERALL XR in adolescents, mean weight change from baseline within the initial 4 weeks of therapy was -1.1 lbs. and -2.8 lbs., respectively, for patients receiving 10 mg and 20 mg ADDERALL XR. Higher doses were associated with greater weight loss within the initial 4 weeks of treatment. Chronic use of amphetamines can be expected to cause a similar suppression of growth [see Adverse Reactions (6.1)].
Pediatric patients who are not growing or gaining weight as expected may need to have their treatment interrupted.
2.3 Recommended Dosage in Pediatric Patients 6 to 12 Years
In pediatric patients 6 to 12 years of age with ADHD and are either starting treatment for the first time or switching from another medication, start with 10 mg once daily in the morning; daily dosage may be adjusted in increments of 5 mg or 10 mg at weekly intervals. When in the judgment of the clinician a lower initial dose is appropriate, patients may begin treatment with 5 mg once daily in the morning.
The maximum recommended dose for children 6 to 12 years of age is 30 mg/day; doses greater than 30 mg/day have not been studied in children. ADDERALL XR has not been studied in children under 6 years of age.
2.4 Recommended Dosage in Pediatric Patients 13 to 17 Years
The recommended starting dose for pediatric patients 13 to 17 years of age with ADHD and are either starting treatment for the first time or switching from another medication is 10 mg/day. The dose may be increased to 20 mg/day after one week if ADHD symptoms are not adequately controlled.
5.7 Peripheral Vasculopathy, Including Raynaud’s Phenomenon
CNS stimulants, including ADDERALL XR, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud’s phenomenon, were observed in postmarketing reports and at the therapeutic dosage of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during ADDERALL XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for ADDERALL XR-treated patients who develop signs or symptoms of peripheral vasculopathy.
5.9 Motor and Verbal Tics, and Worsening of Tourette’s Syndrome
CNS stimulants, including amphetamine, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette’s syndrome has also been reported [see Adverse Reactions (6.2)].
Before initiating ADDERALL XR, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor ADDERALL XR-treated patients for the emergence or worsening of tics or Tourette’s syndrome, and discontinue treatment if clinically appropriate.
6.2 Adverse Reactions Associated With the Use of Amphetamine, Adderall Xr, Or Adderall
The following adverse reactions have been identified during postapproval use of amphetamine, ADDERALL XR, or Adderall. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Allergic: Urticaria, rash, hypersensitivity reactions including angioedema and anaphylaxis. Serious skin rashes, including Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported.
Cardiovascular: Palpitations. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use.
Central Nervous System: Psychotic episodes at recommended doses, overstimulation, restlessness, irritability, euphoria, dyskinesia, dysphoria, depression, tremor, motor and verbal tics, aggression, anger, logorrhea, dermatillomania, paresthesia (including formication), and bruxism.
Endocrine: Impotence, changes in libido, frequent or prolonged erections.
Eye Disorders: Vision blurred, mydriasis.
Gastrointestinal: Unpleasant taste, constipation, intestinal ischemia, and other gastrointestinal disturbances.
Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis.
Skin: Alopecia.
Vascular Disorders: Raynaud’s phenomenon.
Structured Label Content
Section 42229-5 (42229-5)
Adverse Reactions Leading to Discontinuation of Treatment
In two placebo-controlled studies of up to 5 weeks duration among children with ADHD, 2.4% (10/425) of ADDERALL XR-treated patients discontinued due to adverse reactions (including three patients with loss of appetite, one of whom also reported insomnia) compared to 2.7% (7/259) receiving placebo.
The most frequent adverse reactions leading to discontinuation of ADDERALL XR in controlled and uncontrolled, multiple-dose clinical trials of children (N=595) were anorexia (loss of appetite) (2.9%), insomnia (1.5%), weight loss (1.2%), emotional lability (1%), and depression (0.7%). Over half of these patients were exposed to ADDERALL XR for 12 months or more.
In a separate placebo-controlled 4 week study in adolescents with ADHD, five patients (2.1%) discontinued treatment due to adverse events among ADDERALL XR-treated patients (N=233) compared to none who received placebo (N=54). The most frequent adverse event leading to discontinuation and considered to be drug-related (i.e., leading to discontinuation in at least 1% of ADDERALL XR-treated patients and at a rate at least twice that of placebo) was insomnia (1.3%, n=3).
In one placebo-controlled 4 week study among adults with ADHD with doses 20 to 60 mg, 23 patients (12.0%) discontinued treatment due to adverse events among ADDERALL XR-treated patients (N=191) compared to one patient (1.6%) who received placebo (N=64). The most frequent adverse events leading to discontinuation and considered to be drug-related (i.e., leading to discontinuation in at least 1% of ADDERALL XR-treated patients and at a rate at least twice that of placebo) were insomnia (5.2%, n=10), anxiety (2.1%, n=4), nervousness (1.6%, n=3), dry mouth (1.6%, n=3), anorexia (1.6%, n=3), tachycardia (1.6%, n=3), headache (1.6%, n=3), and asthenia (1.0%, n=2).
Section 42231-1 (42231-1)
| MEDICATION GUIDE ADDERALL XR® (ADD-ur-all X-R) (mixed salts of a single-entity amphetamine product) extended-release capsules, CII |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ADL364 Revised: 09/2025 | |
|
What is the most important information I should know about ADDERALL XR? ADDERALL XR may cause serious side effects, including: Abuse, misuse, and addiction. ADDERALL XR has a high chance for abuse and misuse and may lead to substance use problems, including addiction. Misuse and abuse of ADDERALL XR, other amphetamine containing medicines, and methylphenidate containing medicines, can lead to overdose and death. The risk of overdose and death is increased with higher doses of ADDERALL XR or when it is used in ways that are not approved, such as snorting or injection.
|
||
Tell your healthcare provider if you or your child have ever abused or been dependent on alcohol, prescription medicines, or street drugs.
|
||
| Your healthcare provider should check you or your child carefully for heart problems before starting treatment with ADDERALL XR. Tell your healthcare provider if you or your child have any heart problems, heart disease, or heart defects. | ||
Call your healthcare provider or go to the nearest hospital emergency room right away if you or your child have any signs of heart problems such as chest pain, shortness of breath, or fainting during treatment with ADDERALL XR.
|
||
| Tell your healthcare provider about any mental problems you or your child have or about a family history of suicide, bipolar illness, or depression. | ||
| Call your healthcare provider right away if you or your child have any new or worsening mental symptoms or problems during treatment with ADDERALL XR, especially hearing voices, seeing or believing things that are not real, or new manic symptoms. | ||
| What is ADDERALL XR? | ||
| ADDERALL XR is a central nervous system (CNS) stimulant prescription medicine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in adults and children 6 years of age and older. ADDERALL XR may help increase attention and decrease impulsiveness and hyperactivity in people with ADHD. | ||
| ADDERALL XR is not recommended for use in children under 6 years of age with ADHD. | ||
| ADDERALL XR is a federally controlled substance (CII) because it contains amphetamine that can be a target for people who abuse prescription medicines or street drugs. Keep ADDERALL XR in a safe place to protect it from theft. Never give your ADDERALL XR to anyone else because it may cause death or harm them. Selling or giving away ADDERALL XR may harm others and is against the law. | ||
Do not take ADDERALL XR if you or your child:
|
||
Before taking ADDERALL XR tell your healthcare provider about all of your or your child’s medical conditions, including if you or your child:
|
||
| Tell your healthcare provider about all of the medicines that you or your child take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | ||
| ADDERALL XR and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be changed during treatment with ADDERALL XR. | ||
| Your healthcare provider will decide if ADDERALL XR can be taken with other medicines. | ||
| Especially tell your healthcare provider if you or your child take: | ||
|
|
|
| Know the medicines that you or your child take. Keep a list of your or your child’s medicines with you to show your healthcare provider and pharmacist when you or your child get a new medicine. | ||
| Do not start any new medicine during treatment with ADDERALL XR without talking to your healthcare provider first. | ||
How should ADDERALL XR be taken?
|
||
|
What are the possible side effects of ADDERALL XR? ADDERALL XR may cause serious side effects, including:
|
||
|
||
|
|
|
|
||
| The most common side effects of ADDERALL XR in children ages 6 to 12 include: | ||
|
|
|
| The most common side effects of ADDERALL XR in adolescents ages 13 to 17 include: | ||
|
|
|
| The most common side effects of ADDERALL XR in adults include: | ||
|
|
|
| These are not all the possible side effects of ADDERALL XR. | ||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
How should I store ADDERALL XR?
|
||
|
General information about the safe and effective use of ADDERALL XR.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ADDERALL XR for a condition for which it was not prescribed. Do not give ADDERALL XR to other people, even if they have the same condition. It may harm them and it is against the law. |
||
| You can ask your healthcare provider or pharmacist for information about ADDERALL XR that is written for healthcare professionals. | ||
| What are the ingredients in ADDERALL XR? | ||
| Active ingredient: dextroamphetamine sulfate, amphetamine sulfate, dextroamphetamine saccharate, and amphetamine aspartate monohydrate | ||
| Inactive ingredients: gelatin capsules, hydroxypropyl methylcellulose, methacrylic acid copolymer, Opadry® beige, sugar spheres, talc, and triethyl citrate. Gelatin capsules contain edible inks, kosher gelatin, and titanium dioxide. The 5 mg, 10 mg, and 15 mg capsules also contain FD&C Blue #2. The 20 mg, 25 mg, and 30 mg capsules also contain red iron oxide and yellow iron oxide | ||
|
Distributed by: Takeda Pharmaceuticals America, Inc, Cambridge, MA 02142 |
||
| ADDERALL XR is a registered trademark of Takeda Pharmaceuticals U.S.A., Inc. OPADRY is a registered trademark of BPSI Holdings, LLC. © 2025 Takeda Pharmaceuticals U.S.A., Inc. All rights reserved. |
||
| For more information, you may also contact Takeda Pharmaceuticals (the maker of ADDERALL XR) at 1-877-825-3327 or visit the website at http://www.adderallxr.com. |
Section 44425-7 (44425-7)
Dispense in a tight, light-resistant container as defined in the USP.
Store at room temperature, 20 to 25ºC (68 to 77ºF). Excursions permitted to 15 to 30ºC (59 to 86ºF) [see USP Controlled Room Temperature].
9.2 Abuse
ADDERALL XR has a high potential for abuse and misuse which can lead to the development of a substance use disorder, including addiction [see Warnings and Precautions (5.1)]. ADDERALL XR can be diverted for non-medical use into illicit channels or distribution.
Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a healthcare provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of amphetamine may cause increased heart rate, respiratory rate, or blood pressure; sweating; dilated pupils; hyperactivity; restlessness; insomnia; decreased appetite; loss of coordination; tremors; flushed skin; vomiting; and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed with CNS stimulants abuse and/or misuse. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
5.6 Seizures
There is some clinical evidence that stimulants may lower the convulsive threshold in patients with prior history of seizures, in patients with prior EEG abnormalities in the absence of seizures, and very rarely, in patients without a history of seizures and no prior EEG evidence of seizures. In the presence of seizures, ADDERALL XR should be discontinued.
10 Overdosage (10 OVERDOSAGE)
Clinical Effects of Overdose
Overdose of CNS stimulants is characterized by the following sympathomimetic effects:
- Cardiovascular effects including tachyarrhythmias, and hypertension or hypotension. Vasospasm, myocardial infarction, or aortic dissection may precipitate sudden cardiac death. Takotsubo cardiomyopathy may develop.
- CNS effects including psychomotor agitation, confusion, and hallucinations. Serotonin syndrome, seizures, cerebral vascular accidents, and coma may occur.
- Life-threatening hyperthermia (temperatures greater than 104°F) and rhabdomyolysis may develop.
Overdose Management
Consider the possibility of multiple drug ingestion. The pharmacokinetic profile of ADDERALL XR should be considered when treating patients with overdose. D-amphetamine is not dialyzable. Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description (11 DESCRIPTION)
ADDERALL XR extended-release capsules contain mixed salts of a single-entity amphetamine, a CNS stimulant. ADDERALL XR contains equal amounts (by weight) of four salts: dextroamphetamine sulfate, amphetamine sulfate, dextroamphetamine saccharate and amphetamine (D, L)-aspartate monohydrate. This results in a 3.1:1 mixture of dextro- to levo-amphetamine base equivalent.
The 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, and 30 mg strength extended-release capsules are for oral administration. ADDERALL XR contains two types of drug-containing beads (immediate-release and delayed-release) which prolong the release of amphetamine compared to the Adderall (immediate-release) tablet formulation.
Each capsule contains:
| Capsule Strength | 5 mg | 10 mg | 15 mg | 20 mg | 25 mg | 30 mg |
|---|---|---|---|---|---|---|
| Dextroamphetamine Saccharate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine (D,L)-Aspartate Monohydrate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Dextroamphetamine Sulfate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine Sulfate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Total amphetamine base equivalence | 3.1 mg | 6.3 mg | 9.4 mg | 12.5 mg | 15.6 mg | 18.8 mg |
| d-amphetamine base equivalence | 2.4 mg | 4.7 mg | 7.1 mg | 9.5 mg | 11.9 mg | 14.2 mg |
| l-amphetamine base equivalence | 0.75 mg | 1.5 mg | 2.3 mg | 3.0 mg | 3.8 mg | 4.5 mg |
9.3 Dependence
Physical Dependence
ADDERALL XR may produce physical dependence. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
Withdrawal signs and symptoms after abrupt discontinuation or dose reduction following prolonged use of CNS stimulants including ADDERALL XR include dysphoric mood; depression; fatigue; vivid, unpleasant dreams; insomnia or hypersomnia; increased appetite; and psychomotor retardation or agitation.
Tolerance
ADDERALL XR may produce tolerance. Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose).
8.4 Pediatric Use
The safety and effectiveness of ADDERALL XR have not been established in pediatric patients less than 6 years of age.
The safety and effectiveness of ADDERALL XR have been established in pediatric patients with ADHD 6 years of age and older.
In studies evaluating extended-release amphetamine products, patients 4 to <6 years of age had higher systemic amphetamine exposures than those observed in older pediatric patients at the same dosage. Pediatric patients 4 to <6 years of age also had a higher incidence of adverse reactions, including weight loss.
8.5 Geriatric Use
ADDERALL XR has not been studied in the geriatric population.
4 Contraindications (4 CONTRAINDICATIONS)
ADDERALL XR administration is contraindicated in patients:
- known to be hypersensitive to amphetamine, or other components of ADDERALL XR. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions (6.2)].
- taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions (5.8), Drug Interactions (7.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Abuse, Misuse, and Addiction [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)]
- Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2)]
- Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4)]
- Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.5)]
- Seizures [see Warnings and Precautions (5.6)]
- Peripheral Vasculopathy, including Raynaud’s Phenomenon [see Warnings and Precautions (5.7)]
- Serotonin Syndrome [see Warnings and Precautions (5.8)]
- Motor and Verbal Tics, and Worsening of Tourette’s Syndrome [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.6 Renal Impairment
Due to reduced clearance of amphetamines in patients with severe renal impairment (GFR 15 to <30 mL/min/1.73 m2), the recommended dose should be reduced. ADDERALL XR is not recommended in patients with ESRD (GFR <15 mL/min/1.73 m2) [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
d-Amphetamine is not dialyzable.
12.2 Pharmacodynamics
Amphetamines block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
Pharmacokinetic studies of ADDERALL XR have been conducted in healthy adult and pediatric (children aged 6 to 12 yrs) subjects, adolescent (13 to 17 yrs), and children with ADHD. Both Adderall (immediate-release) tablets and ADDERALL XR extended-release capsules contain d-amphetamine and l-amphetamine salts in the ratio of 3:1. Following administration of Adderall (immediate-release), the peak plasma concentrations occurred in about 3 hours for both d-amphetamine and l-amphetamine.
The time to reach maximum plasma concentration (Tmax) for ADDERALL XR is about 7 hours, which is about 4 hours longer compared to Adderall (immediate-release). This is consistent with the extended-release nature of the product.
Figure 1: Mean d-amphetamine and l-amphetamine Plasma Concentrations Following Administration of ADDERALL XR 20 mg (8 am) and Adderall (immediate-release) 10 mg Twice Daily (8 am and 12 noon) in the Fed State.
A single dose of ADDERALL XR 20 mg extended-release capsules provided comparable plasma concentration profiles of both d-amphetamine and l-amphetamine to Adderall (immediate-release) 10 mg twice daily administered 4 hours apart.
The mean elimination half-life for d-amphetamine is 10 hours in adults; 11 hours in adolescents aged 13 to 17 years and weighing less than or equal to 75 kg/165 lbs; and 9 hours in children aged 6 to 12 years. For the l-amphetamine, the mean elimination half-life in adults is 13 hours; 13 to 14 hours in adolescents; and 11 hours in children aged 6 to 12 years. On a mg/kg body weight basis, children have a higher clearance than adolescents or adults (see Special Populations).
ADDERALL XR demonstrates linear pharmacokinetics over the dose range of 20 to 60 mg in adults and adolescents weighing greater than 75 kg/165 lbs, over the dose range of 10 to 40 mg in adolescents weighing less than or equal to 75 kg/165 lbs, and 5 to 30 mg in children aged 6 to 12 years. There is no unexpected accumulation at steady state in children.
Food does not affect the extent of absorption of d-amphetamine and l-amphetamine, but prolongs Tmax by 2.5 hours (from 5.2 hrs at fasted state to 7.7 hrs after a high-fat meal) for d-amphetamine and 2.7 hours (from 5.6 hrs at fasted state to 8.3 hrs after a high-fat meal) for l-amphetamine after administration of ADDERALL XR 30 mg. Opening the capsule and sprinkling the contents on applesauce results in comparable absorption to the intact capsule taken in the fasted state. Equal doses of ADDERALL XR strengths are bioequivalent.
5.8 Serotonin Syndrome
Serotonin syndrome, a potentially life-threatening reaction, may occur when amphetamines are used in combination with other drugs that affect the serotonergic neurotransmitter systems such as MAOIs, selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John’s Wort [see Drug Interactions (7.1)]. Amphetamines and amphetamine derivatives are known to be metabolized, to some degree, by cytochrome P450 2D6 (CYP2D6) and display minor inhibition of CYP2D6 metabolism [see Clinical Pharmacology (12.3)]. The potential for a pharmacokinetic interaction exists with the coadministration of CYP2D6 inhibitors which may increase the risk with increased exposure to ADDERALL XR. In these situations, consider an alternative nonserotonergic drug or an alternative drug that does not inhibit CYP2D6 [see Drug Interactions (7.1)]. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
Concomitant use of ADDERALL XR with MAOI drugs is contraindicated [see Contraindications (4)].
Discontinue treatment with ADDERALL XR and any concomitant serotonergic agents immediately if symptoms of serotonin syndrome occur, and initiate supportive symptomatic treatment. Concomitant use of ADDERALL XR with other serotonergic drugs or CYP2D6 inhibitors should be used only if the potential benefit justifies the potential risk. If clinically warranted, consider initiating ADDERALL XR with lower doses, monitoring patients for the emergence of serotonin syndrome during drug initiation or titration, and informing patients of the increased risk for serotonin syndrome.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ADDERALL XR, a CNS stimulant, is indicated for the treatment of attention deficit hyperactivity disorder (ADHD) in adults and pediatric patients 6 years and older. (1)
Limitations of Use
The use of ADDERALL XR is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage. (5.5, 8.4)
12.1 Mechanism of Action
Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
ADDERALL XR contains amphetamine, a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to treating patients with ADDERALL XR, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2)].
- the family history and clinically evaluate patients for motor or verbal tics or Tourette’s syndrome before initiating ADDERALL XR [see Warnings and Precautions (5.9)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. (5.2)
- Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse at appropriate intervals. (5.3)
- Psychiatric Adverse Reactions: Prior to initiating ADDERALL XR, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing ADDERALL XR. (5.4)
- Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. (5.5)
- Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. (5.6)
- Peripheral Vasculopathy, including Raynaud’s Phenomenon: Careful observation for digital changes is necessary during ADDERALL XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. (5.7)
- Serotonin Syndrome: Increased risk when coadministered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue ADDERALL XR and initiate supportive treatment. (4, 5.8, 10)
- Motor and Verbal Tics, and Worsening of Tourette’s Syndrome: Before initiating ADDERALL XR, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette’s syndrome. Discontinue treatment if clinically appropriate. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 years of age is 30 mg once daily. (2.2, 2.3, 2.4)
- Adults: 20 mg once daily in the morning. (2.5)
- Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. (2.6, 8.6)
- Adults with severe renal impairment: 15 mg once daily in the morning. (2.6, 8.6)
- Patients with end stage renal disease (ESRD): Not recommended. (2.6, 8.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg)
ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg)
ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg)
ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg)
ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg)
ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The premarketing development program for ADDERALL XR included exposures in a total of 1,315 participants in clinical trials (635 pediatric patients, 350 adolescent patients, 248 adult patients, and 82 healthy adult subjects). Of these, 635 patients (ages 6 to 12) were evaluated in two controlled clinical studies, one open-label clinical study, and two single-dose clinical pharmacology studies (N=40). Safety data on all patients are included in the discussion that follows. Adverse reactions were assessed by collecting adverse reactions, results of physical examinations, vital signs, weights, laboratory analyses, and ECGs.
Adverse reactions during exposure were obtained primarily by general inquiry and recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse reactions without first grouping similar types of reactions into a smaller number of standardized event categories. In the tables and listings that follow, COSTART terminology has been used to classify reported adverse reactions.
The stated frequencies of adverse reactions represent the proportion of individuals who experienced, at least once, a treatment-emergent adverse event of the type listed.
2.5 Recommended Dosage in Adults
In adults with ADHD who are either starting treatment for the first time or switching from another medication, the recommended dose is 20 mg/day.
5.1 Abuse, Misuse, and Addiction
ADDERALL XR has a high potential for abuse and misuse. The use of ADDERALL XR exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. ADDERALL XR can be diverted for non-medical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2)]. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing ADDERALL XR, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store ADDERALL XR in a safe place, preferably locked, and instruct patients to not give ADDERALL XR to anyone else. Throughout ADDERALL XR treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Abuse, Misuse, and Addiction
Educate patients and their families about the risks of abuse, misuse, and addiction of ADDERALL XR, which can lead to overdose and death, and proper disposal of any unused drug [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2), Overdosage (10)]. Advise patients to store ADDERALL XR in a safe place, preferably locked, and instruct patients to not give ADDERALL XR to anyone else.
Risks to Patients with Serious Cardiac Disease
Advise patients that there are potential risks to patients with serious cardiac disease, including sudden death, with ADDERALL XR use. Instruct patients to contact a healthcare provider immediately if they develop symptoms such as exertional chest pain, unexplained syncope, or other symptoms suggestive of cardiac disease [see Warnings and Precautions (5.2)].
Increased Blood Pressure and Heart Rate
Advise patients that ADDERALL XR can cause elevations in blood pressure and heart rate [see Warnings and Precautions (5.3)].
Psychiatric Adverse Reactions
Prior to initiating treatment with ADDERALL XR, adequately screen patients with comorbid depressive symptoms to determine if they are at risk for bipolar disorder. Such screening should include a detailed psychiatric history, including a family history of suicide, bipolar disorder, and/or depression. Additionally, ADDERALL XR therapy at usual doses may cause treatment-emergent psychotic or manic symptoms in patients without prior history of psychotic symptoms or mania [see Warnings and Precautions (5.4)].
Circulation Problems in Fingers and Toes [Peripheral Vasculopathy, including Raynaud’s Phenomenon]
Instruct patients beginning treatment with ADDERALL XR about the risk of peripheral vasculopathy, including Raynaud’s phenomenon, and in associated signs and symptoms: fingers or toes may feel numb, cool, painful, and/or may change color from pale, to blue, to red. Instruct patients to report to their physician any new numbness, pain, skin color change, or sensitivity to temperature in fingers or toes. Instruct patients to call their physician immediately with any signs of unexplained wounds appearing on fingers or toes while taking ADDERALL XR. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients [see Warnings and Precautions (5.7)].
Serotonin Syndrome
Caution patients about the risk of serotonin syndrome with concomitant use of ADDERALL XR and other serotonergic drugs including SSRIs, SNRIs, triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John’s Wort, and with drugs that impair metabolism of serotonin (in particular MAOIs, both those intended to treat psychiatric disorders and also others such as linezolid) [see Contraindications (4), Warnings and Precautions (5.8), Drug Interactions (7.1)]. Advise patients to contact their healthcare provider or report to the emergency room if they experience signs or symptoms of serotonin syndrome.
Concomitant Medications
Advise patients to notify their physicians if they are taking, or plan to take, any prescription or over-the-counter drugs because there is a potential for interactions [see Drug Interactions (7.1)].
Growth
Monitor growth in children during treatment with ADDERALL XR, and patients who are not growing or gaining weight as expected may need to have their treatment interrupted [see Warnings and Precautions (5.5)].
Motor and Verbal Tics, and Worsening of Tourette’s Syndrome
Advise patients that motor and verbal tics and worsening of Tourette’s syndrome may occur during treatment with ADDERALL XR. Instruct patients to notify their healthcare provider if emergence of new tics or worsening of tics or Tourette’s syndrome occurs [see Warnings and Precautions (5.9)].
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to ADDERALL XR during pregnancy [see Use in Specific Populations (8.1)].
Pregnancy
Advise patients to notify their healthcare provider if they become pregnant or intend to become pregnant during treatment with ADDERALL XR. Advise patients of the potential fetal effects from the use of ADDERALL XR during pregnancy [see Use in Specific Populations (8.1)].
Lactation
Advise women not to breastfeed if they are taking ADDERALL XR [see Use in Specific Populations (8.2)].
5.4 Psychiatric Adverse Reactions
Exacerbation of Pre-Existing Psychosis
Administration of stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with pre-existing psychotic disorder.
Induction of a Manic Episode in Patients with Bipolar Disease
CNS stimulants may induce a manic or mixed episode in patients. Prior to initiating ADDERALL XR treatment, screen patients for risk factors for developing a manic episode (e.g., comorbid or history of depressive symptoms or a family history of suicide, bipolar disorder, or depression).
New Psychotic or Manic Symptoms
CNS stimulants, at the recommended dosage, may cause psychotic or manic symptoms (e.g., hallucinations, delusional thinking, or mania) in patients without a prior history of psychotic illness or mania. In a pooled analysis of multiple short-term, placebo-controlled studies of CNS stimulants, psychotic or manic symptoms occurred in approximately 0.1% of CNS stimulant-treated patients compared to 0% of placebo-treated patients. If such symptoms occur, consider discontinuing ADDERALL XR.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ADDERALL XR 5 mg extended-release capsules: Clear/blue (imprinted ADDERALL XR 5 mg), bottles of 100, NDC 54092-381-01
ADDERALL XR 10 mg extended-release capsules: Blue/blue (imprinted ADDERALL XR 10 mg), bottles of 100, NDC 54092-383-01
ADDERALL XR 15 mg extended-release capsules: Blue/white (imprinted ADDERALL XR 15 mg), bottles of 100, NDC 54092-385-01
ADDERALL XR 20 mg extended-release capsules: Orange/orange (imprinted ADDERALL XR 20 mg), bottles of 100, NDC 54092-387-01
ADDERALL XR 25 mg extended-release capsules: Orange/white (imprinted ADDERALL XR 25 mg), bottles of 100, NDC 54092-389-01
ADDERALL XR 30 mg extended-release capsules: Natural/orange (imprinted ADDERALL XR 30 mg), bottles of 100, NDC 54092-391-01
7.2 Drug Laboratory Test Interactions (7.2 Drug-Laboratory Test Interactions)
Amphetamines can cause a significant elevation in plasma corticosteroid levels. This increase is greatest in the evening. Amphetamines may interfere with urinary steroid determinations.
Warning: Abuse, Misuse, and Addiction (WARNING: ABUSE, MISUSE, and ADDICTION)
ADDERALL XR has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including ADDERALL XR, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing ADDERALL XR, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout ADDERALL XR treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2)].
2.2 General Administration Information
Individualize the dosage according to the therapeutic needs and response of the patient. Administer ADDERALL XR at the lowest effective dosage.
Based on bioequivalence data, patients taking divided doses of immediate-release ADDERALL, (for example, twice daily), may be switched to ADDERALL XR at the same total daily dose taken once daily. Titrate at weekly intervals to appropriate efficacy and tolerability as indicated.
ADDERALL XR extended-release capsules may be taken whole, or the capsule may be opened and the entire contents sprinkled on applesauce. If the patient is using the sprinkle administration method, the sprinkled applesauce should be consumed immediately; it should not be stored. Patients should take the applesauce with sprinkled beads in its entirety without chewing. The dose of a single capsule should not be divided. The contents of the entire capsule should be taken, and patients should not take anything less than one capsule per day.
ADDERALL XR may be taken orally with or without food.
ADDERALL XR should be given upon awakening. Afternoon doses should be avoided because of the potential for insomnia.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Acute administration of high doses of amphetamine (d- or d, l-) has been shown to produce long-lasting neurotoxic effects, including irreversible nerve fiber damage in rodents. The significance of these findings to humans is unknown.
5.3 Increased Blood Pressure and Heart Rate
CNS stimulants may cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 bpm).
Monitor all ADDERALL XR-treated patients for hypertension and tachycardia.
1.1 Attention Deficit Hyperactivity Disorder
ADDERALL XR® is indicated for the treatment of attention deficit hyperactivity disorder (ADHD) in adults and pediatric patients 6 years and older.
Limitations of Use
The use of ADDERALL XR is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.5), Use in Specific Populations (8.4)].
2.6 Dosage in Patients With Renal Impairment (2.6 Dosage in Patients with Renal Impairment)
In adult patients with severe renal impairment (GFR 15 to <30 mL/min/1.73 m2), the recommended dose is 15 mg once daily in the morning. In pediatric patients (6 to 17 years of age) with severe renal impairment, the recommended dose is 5 mg once daily. The maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. ADDERALL XR is not recommended in patients with end stage renal disease (ESRD) (GFR <15 mL/min/1.73 m2) [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.7 Dosage Modification Due to Drug Interactions (2.7 Dosage Modification due to Drug Interactions)
Agents that alter urinary pH can impact excretion and alter blood levels of amphetamines. Acidifying agents (e.g., ascorbic acid) decrease blood levels; adjust ADDERALL XR dosage based on clinical response [see Drug Interactions (7)].
5.2 Risks to Patients With Serious Cardiac Disease (5.2 Risks to Patients with Serious Cardiac Disease)
Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosage. Avoid ADDERALL XR use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmia, coronary artery disease, or other serious cardiac disease.
Principal Display Panel 5 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg Capsule Bottle Label)
NDC 54092-381-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
5 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 10 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Label)
NDC 54092-383-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
10 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 15 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 15 mg Capsule Bottle Label)
NDC 54092-385-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
15 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 20 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Label)
NDC 54092-387-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
20 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 25 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Capsule Bottle Label)
NDC 54092-389-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
25 mg
CII
100 Capsules
Rx only
Takeda
Principal Display Panel 30 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Label)
NDC 54092-391-01
ADDERALL XR
®
(Mixed Salts of A Single-Entity
Amphetamine Product)
Extended-Release Capsules
30 mg
CII
100 Capsules
Rx only
Takeda
7.1 Clinically Important Interactions With Amphetamines (7.1 Clinically Important Interactions with Amphetamines)
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact | Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure. |
| Intervention | Do not administer ADDERALL XR concomitantly or within 14 days after discontinuing MAOI [see Contraindications (4)]. |
| Serotonergic Drugs | |
| Clinical Impact | The concomitant use of ADDERALL XR and serotonergic drugs increases the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during ADDERALL XR initiation or dosage increase. If serotonin syndrome occurs, discontinue ADDERALL XR and the concomitant serotonergic drug(s) [see Warnings and Precautions (5.8)]. |
| CYP2D6 Inhibitors | |
| Clinical Impact | The concomitant use of ADDERALL XR and CYP2D6 inhibitors may increase the exposure of ADDERALL XR compared to the use of the drug alone and increase the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during ADDERALL XR initiation and after a dosage increase. If serotonin syndrome occurs, discontinue ADDERALL XR and the CYP2D6 inhibitor [see Warnings and Precautions (5.8), Overdosage (10)]. |
| Alkalinizing Agents | |
| Clinical Impact | Increase blood levels and potentiate the action of amphetamine. |
| Intervention | Coadministration of ADDERALL XR and gastrointestinal or urinary alkalinizing agents should be avoided. |
| Acidifying Agents | |
| Clinical Impact | Lower blood levels and efficacy of amphetamines. |
| Intervention | Increase dose based on clinical response. |
| Tricyclic Antidepressants | |
| Clinical Impact | May enhance the activity of tricyclic or sympathomimetic agents causing striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated. |
| Intervention | Monitor frequently and adjust or use alternative therapy based on clinical response. |
| Proton Pump Inhibitors | |
| Clinical Impact | Time to maximum concentration (Tmax) of amphetamine is decreased compared to when administered alone. |
| Intervention | Monitor patients for changes in clinical effect and adjust therapy based on clinical response. |
5.5 Long Term Suppression of Growth in Pediatric Patients (5.5 Long-Term Suppression of Growth in Pediatric Patients)
ADDERALL XR is not approved for use and is not recommended in pediatric patients below 6 years of age [see Use in Specific Populations (8.4)].
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients. Closely monitor growth (weight and height) in ADDERALL XR-treated pediatric patients treated with CNS stimulants.
In a controlled trial of ADDERALL XR in adolescents, mean weight change from baseline within the initial 4 weeks of therapy was -1.1 lbs. and -2.8 lbs., respectively, for patients receiving 10 mg and 20 mg ADDERALL XR. Higher doses were associated with greater weight loss within the initial 4 weeks of treatment. Chronic use of amphetamines can be expected to cause a similar suppression of growth [see Adverse Reactions (6.1)].
Pediatric patients who are not growing or gaining weight as expected may need to have their treatment interrupted.
2.3 Recommended Dosage in Pediatric Patients 6 to 12 Years
In pediatric patients 6 to 12 years of age with ADHD and are either starting treatment for the first time or switching from another medication, start with 10 mg once daily in the morning; daily dosage may be adjusted in increments of 5 mg or 10 mg at weekly intervals. When in the judgment of the clinician a lower initial dose is appropriate, patients may begin treatment with 5 mg once daily in the morning.
The maximum recommended dose for children 6 to 12 years of age is 30 mg/day; doses greater than 30 mg/day have not been studied in children. ADDERALL XR has not been studied in children under 6 years of age.
2.4 Recommended Dosage in Pediatric Patients 13 to 17 Years
The recommended starting dose for pediatric patients 13 to 17 years of age with ADHD and are either starting treatment for the first time or switching from another medication is 10 mg/day. The dose may be increased to 20 mg/day after one week if ADHD symptoms are not adequately controlled.
5.7 Peripheral Vasculopathy, Including Raynaud’s Phenomenon (5.7 Peripheral Vasculopathy, including Raynaud’s Phenomenon)
CNS stimulants, including ADDERALL XR, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud’s phenomenon, were observed in postmarketing reports and at the therapeutic dosage of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during ADDERALL XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for ADDERALL XR-treated patients who develop signs or symptoms of peripheral vasculopathy.
5.9 Motor and Verbal Tics, and Worsening of Tourette’s Syndrome
CNS stimulants, including amphetamine, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette’s syndrome has also been reported [see Adverse Reactions (6.2)].
Before initiating ADDERALL XR, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor ADDERALL XR-treated patients for the emergence or worsening of tics or Tourette’s syndrome, and discontinue treatment if clinically appropriate.
6.2 Adverse Reactions Associated With the Use of Amphetamine, Adderall Xr, Or Adderall (6.2 Adverse Reactions Associated with the Use of Amphetamine, ADDERALL XR, or Adderall)
The following adverse reactions have been identified during postapproval use of amphetamine, ADDERALL XR, or Adderall. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Allergic: Urticaria, rash, hypersensitivity reactions including angioedema and anaphylaxis. Serious skin rashes, including Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported.
Cardiovascular: Palpitations. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use.
Central Nervous System: Psychotic episodes at recommended doses, overstimulation, restlessness, irritability, euphoria, dyskinesia, dysphoria, depression, tremor, motor and verbal tics, aggression, anger, logorrhea, dermatillomania, paresthesia (including formication), and bruxism.
Endocrine: Impotence, changes in libido, frequent or prolonged erections.
Eye Disorders: Vision blurred, mydriasis.
Gastrointestinal: Unpleasant taste, constipation, intestinal ischemia, and other gastrointestinal disturbances.
Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis.
Skin: Alopecia.
Vascular Disorders: Raynaud’s phenomenon.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:05.036915 · Updated: 2026-03-14T22:35:47.693311