Cold Sore Complex
afd3ddba-5649-4572-b71b-a57882c16db0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
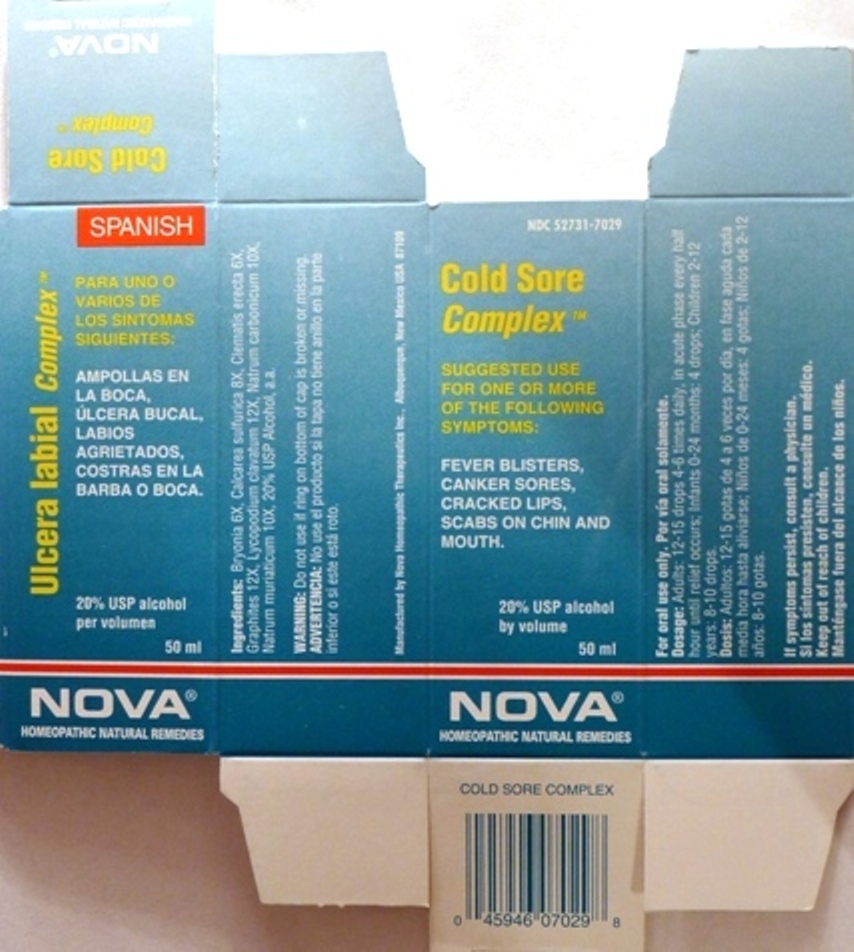
Suggested use for one or more of the following symptoms: Fever blisters, canker sores, cracked lips, scabs on chin and mouth.
Purpose
Suggested use for one or more of the following symptoms: Fever blisters, canker sores, cracked lips, scabs on chin and mouth.
Medication Information
Warnings and Precautions
If symptoms persist, consult a physician.
Do not use if ring on bottom of cap is broken or missing.
Purpose
Suggested use for one or more of the following symptoms: Fever blisters, canker sores, cracked lips, scabs on chin and mouth.
Dosage and Administration
Adults:
In Acute Phase:
12-15 drops until relief occurs
When Relief Occurs:
12-15 drops, 4-6 times per day
Children 2-12 years:
8-10 drops, 4-6 times per day
Infants 0-24 months:
4 drops, 4-6 times per day
Description
Suggested use for one or more of the following symptoms: Fever blisters, canker sores, cracked lips, scabs on chin and mouth.
Section 51945-4
Cold Sore Complex Product
Cold Sore Complex Bottle Label
Cold Sore Complex Box
Section 53413-1
Manufactured by Nova Homeopathic Therapeutics Inc., Albuquerque, New Mexico USA 87109
1-800-225-8094
Purpose:
Suggested use for one or more of the following symptoms:
Fever blisters, canker sores, cracked lips, scabs on chin and mouth.
Warnings:
Keep out of reach of children.
Usage and Dosage:
For oral use only.
Active Ingredients:
Bryonia 6X, Calcarea sulfurica 8X, Clematis erecta 6X, Graphites 12X, Lycopodium clavatum 12X, Natrum carbonicum 10X, Natrum muriaticum 10X
Inactive Ingredients:
Alcohol, 20% USP
Structured Label Content
Dosage and Administration (34068-7)
Adults:
In Acute Phase:
12-15 drops until relief occurs
When Relief Occurs:
12-15 drops, 4-6 times per day
Children 2-12 years:
8-10 drops, 4-6 times per day
Infants 0-24 months:
4 drops, 4-6 times per day
Warnings and Precautions (34071-1)
If symptoms persist, consult a physician.
Do not use if ring on bottom of cap is broken or missing.
Section 51945-4 (51945-4)
Cold Sore Complex Product
Cold Sore Complex Bottle Label
Cold Sore Complex Box
Section 53413-1 (53413-1)
Manufactured by Nova Homeopathic Therapeutics Inc., Albuquerque, New Mexico USA 87109
1-800-225-8094
Purpose:
Suggested use for one or more of the following symptoms:
Fever blisters, canker sores, cracked lips, scabs on chin and mouth.
Warnings:
Keep out of reach of children.
Usage and Dosage:
For oral use only.
Active Ingredients:
Bryonia 6X, Calcarea sulfurica 8X, Clematis erecta 6X, Graphites 12X, Lycopodium clavatum 12X, Natrum carbonicum 10X, Natrum muriaticum 10X
Inactive Ingredients:
Alcohol, 20% USP
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:09.756508 · Updated: 2026-03-14T23:14:19.537125