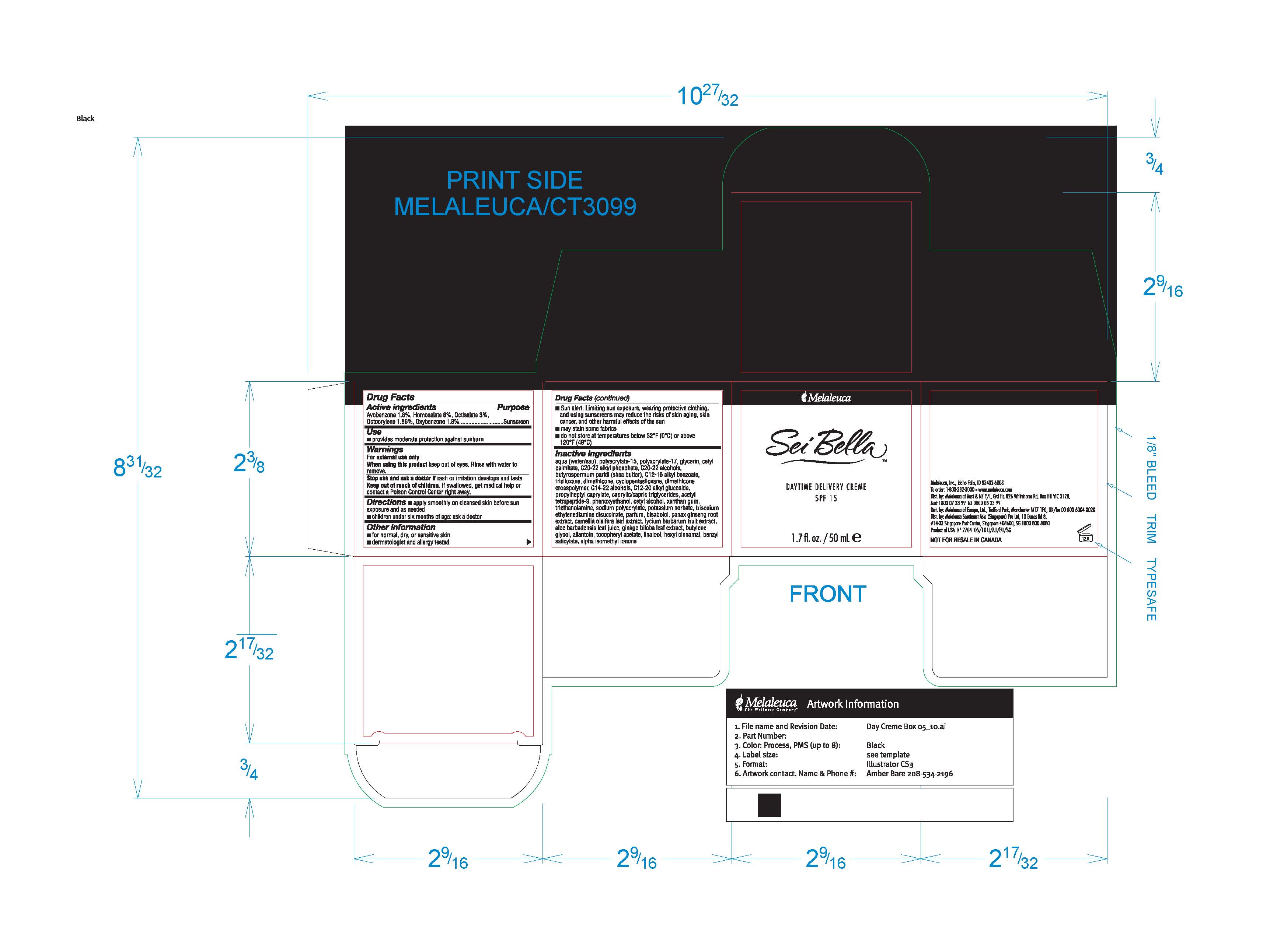
Sei Bella Daytime Delivery Creme Content Of Label
af2086a9-eae0-484e-aa9f-9697e2b05fb5
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients Avobenzone 1.8%, Homosalate 6%, Octisalate, 3%, Octocrylene 1.86%, Oxybenzone 1.8%
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Use
- provides moderate protection against sunburn
Dosage and Administration
Directions
- apply smoothly on cleansed skin before sun exposure and as needed
- children under six months of age: ask a doctor
Description
Active ingredients Avobenzone 1.8%, Homosalate 6%, Octisalate, 3%, Octocrylene 1.86%, Oxybenzone 1.8%
Section 34072-9
Other information
- Sun alert: Limiting sun exposure, wearing protective clothing, and using sunscreens may reduce the risks of skin aging, skin cancer, and other harmful effects of the sun
- may stain some fabrics
Section 34092-7
- dermatologist and allergy tested
Section 44425-7
Other information
- do not store at temperatures below 32°F (0°C) or above 120°F (49°C)
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if rash or irritation develops and lasts
Section 50567-7
When using this product keep out of eyes. Rinse with water to remove.
Section 51727-6
Inactive ingredients
aqua (water/eau), polyacrylate-15, polyacrylate-17, glycerin, cetyl palmitate, C20-22 alkyl phosphate, C20-22 alcohols, butyrospermum parkii (shea butter), C12-15 alkyl benzoate, trisiloxane, dimethicone, cyclopentasiloxane, dimethicone crosspolymer, C14-22 alcohols, C12-20 alkyl glucoside, propylheptyl caprylate, caprylic/capric triglycerides, acetyl tetrapeptide-9, phenoxyethanol, cetyl alcohol, xanthan gum, triethanolamine, sodium polyacrylate, potassium sorbate, trisodium ethylenediamine disuccinate, parfum, bisabolol, panax ginseng root extract, camellia oleifera leaf extract, lycium barbarum fruit extract, aloe barbadensis leaf juice, ginkgo biloba leaf extract, butylene glycol, allantoin, tocopheryl acetate, linalool, hexyl cinnamal, benzyl salicylate, alpha isomethyl ionone
Section 51945-4
Section 55105-1
Purpose
Sunscreen
Section 55106-9
Active ingredients
Avobenzone 1.8%, Homosalate 6%, Octisalate, 3%, Octocrylene 1.86%, Oxybenzone 1.8%
Structured Label Content
Indications and Usage (34067-9)
Use
- provides moderate protection against sunburn
Dosage and Administration (34068-7)
Directions
- apply smoothly on cleansed skin before sun exposure and as needed
- children under six months of age: ask a doctor
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 34072-9 (34072-9)
Other information
- Sun alert: Limiting sun exposure, wearing protective clothing, and using sunscreens may reduce the risks of skin aging, skin cancer, and other harmful effects of the sun
- may stain some fabrics
Section 34092-7 (34092-7)
- dermatologist and allergy tested
Section 44425-7 (44425-7)
Other information
- do not store at temperatures below 32°F (0°C) or above 120°F (49°C)
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if rash or irritation develops and lasts
Section 50567-7 (50567-7)
When using this product keep out of eyes. Rinse with water to remove.
Section 51727-6 (51727-6)
Inactive ingredients
aqua (water/eau), polyacrylate-15, polyacrylate-17, glycerin, cetyl palmitate, C20-22 alkyl phosphate, C20-22 alcohols, butyrospermum parkii (shea butter), C12-15 alkyl benzoate, trisiloxane, dimethicone, cyclopentasiloxane, dimethicone crosspolymer, C14-22 alcohols, C12-20 alkyl glucoside, propylheptyl caprylate, caprylic/capric triglycerides, acetyl tetrapeptide-9, phenoxyethanol, cetyl alcohol, xanthan gum, triethanolamine, sodium polyacrylate, potassium sorbate, trisodium ethylenediamine disuccinate, parfum, bisabolol, panax ginseng root extract, camellia oleifera leaf extract, lycium barbarum fruit extract, aloe barbadensis leaf juice, ginkgo biloba leaf extract, butylene glycol, allantoin, tocopheryl acetate, linalool, hexyl cinnamal, benzyl salicylate, alpha isomethyl ionone
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose
Sunscreen
Section 55106-9 (55106-9)
Active ingredients
Avobenzone 1.8%, Homosalate 6%, Octisalate, 3%, Octocrylene 1.86%, Oxybenzone 1.8%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:12.250532 · Updated: 2026-03-14T22:52:34.752454