61010-2017 . Topical Antiseptic Wipe
aeecceb7-0753-4908-b1a2-d7e18ba350ec
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Drug Facts
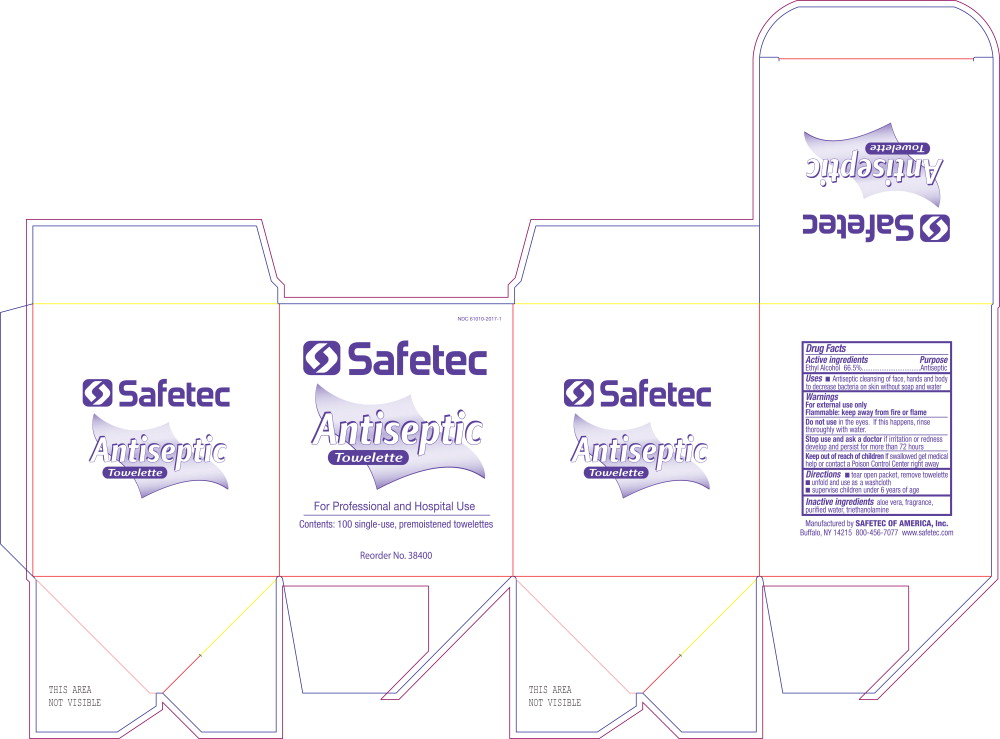
Uses
- Antiseptic cleansing of face, hands and body to decrease bacteria on skin without soap and water
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of childrenIf swallowed get medical help or contact a Poison Control Center right away
Section 50566-9
Stop use and ask a doctorif irritation or redness develop and persist for more than 72 hours
Section 50570-1
Do not usein the eyes. If this happens, rinse thoroughly with water.
Warnings
For external use only
Flammable: keep away from fire or flame
Directions
- tear open packet, remove towelette
- unfold and use as a washcloth
- supervise children under 6 years of age
Active Ingredients
Ethyl Alcohol 66.5%
Inactive Ingredients
aloe vera, fragrance, purified water, triethanolamine
Principal Display Panel Safetec Antiseptic Towelette Carton Label
NDC 61010-2017-1
Safetec
Antiseptic
Towelette
For Professional and Hospital Use
Contents: 100 single-use, premoistened towelettes
Reorder No. 38400
Principal Display Panel Safetec Antiseptic Towelette Packet Label
Safetec
Antiseptic
Towelette
For Professional and Hospital Use
Contents: 1 single-use, premoistened towelette
Manufactured by SAFETEC OF AMERICA, Inc.
Buffalo, NY 14215 800-456-7077 www.safetec.com
Structured Label Content
Uses
- Antiseptic cleansing of face, hands and body to decrease bacteria on skin without soap and water
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of childrenIf swallowed get medical help or contact a Poison Control Center right away
Section 50566-9 (50566-9)
Stop use and ask a doctorif irritation or redness develop and persist for more than 72 hours
Section 50570-1 (50570-1)
Do not usein the eyes. If this happens, rinse thoroughly with water.
Purpose
Antiseptic
Warnings
For external use only
Flammable: keep away from fire or flame
Directions
- tear open packet, remove towelette
- unfold and use as a washcloth
- supervise children under 6 years of age
Active Ingredients (Active ingredients)
Ethyl Alcohol 66.5%
Inactive Ingredients (Inactive ingredients)
aloe vera, fragrance, purified water, triethanolamine
Principal Display Panel Safetec Antiseptic Towelette Carton Label (Principal Display Panel - Safetec Antiseptic Towelette Carton Label)
NDC 61010-2017-1
Safetec
Antiseptic
Towelette
For Professional and Hospital Use
Contents: 100 single-use, premoistened towelettes
Reorder No. 38400
Principal Display Panel Safetec Antiseptic Towelette Packet Label (Principal Display Panel - Safetec Antiseptic Towelette Packet Label)
Safetec
Antiseptic
Towelette
For Professional and Hospital Use
Contents: 1 single-use, premoistened towelette
Manufactured by SAFETEC OF AMERICA, Inc.
Buffalo, NY 14215 800-456-7077 www.safetec.com
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:47.067522 · Updated: 2026-03-14T23:02:30.868416