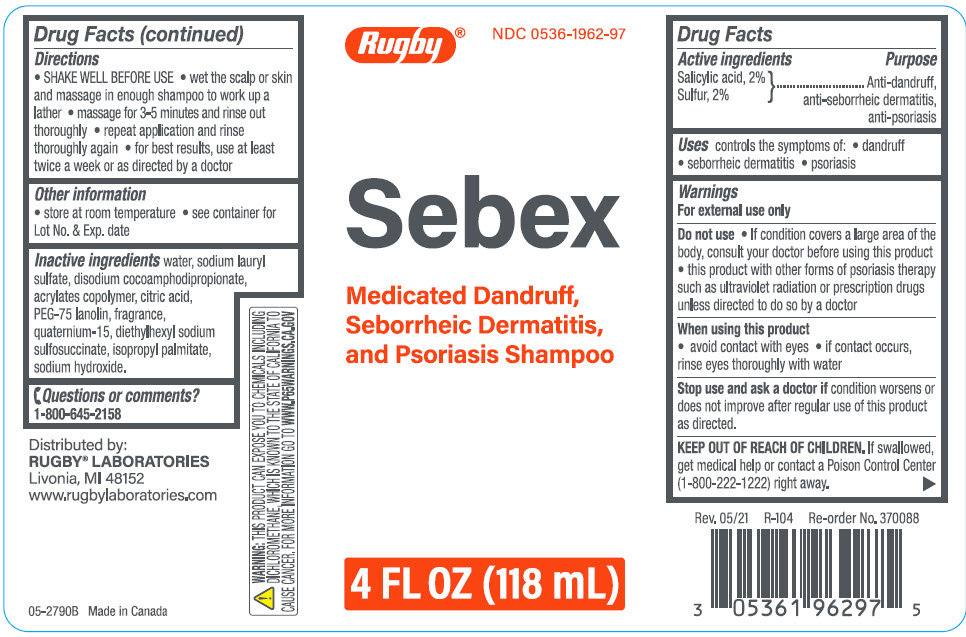
Rugby Sebex Medicated Dandruff, Seborrheic Dermatitis, And Psoriasis Shampoo
aec81d61-1d28-490f-9654-f66bd4a30ffa
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Description
Drug Facts
Uses
controls the symptoms of:
- dandruff
- seborrheic dermatitis
- psoriasis
Section 42229-5
Drug Facts
Section 50565-1
KEEP OUT OF REACH OF CHILDREN. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9
Stop use and ask a doctor if condition worsens or does not improve after regular use of this product as directed.
Section 55105-1
| Active ingredients | Purpose |
|---|---|
| Salicylic Acid, 2% }
Sulfur, 2% |
Anti-dandruff, anti-seborrheic dermatitis, anti-psoriasis |
Warnings
For external use only
Directions
- SHAKE WELL BEFORE USE
- wet the scalp or skin and massage in enough shampoo to work up a lather
- massage for 3-5 minutes and rinse out thoroughly
- repeat application and rinse thoroughly again
- for best results, use at least twice a week or as directed by a doctor
Do Not Use
- If condition covers a large area of the body, consult your doctor before using this product
- this product with other forms of psoriasis therapy such as ultraviolet radiation or prescription drugs unless directed to do so by a doctor
Other Information
- store at room temperature
- see container for Lot No. & Exp. date
Inactive Ingredients
water, sodium lauryl sulfate, disodium cocoamphodipropionate, acrylates copolymer, citric acid, PEG-75 lanolin, fragrance, quaternium-15, diethylhexyl sodium sulfosuccinate, isopropyl palmitate, sodium hydroxide.
Questions Or Comments?
1-800-645-2158
When Using This Product
- avoid contact with eyes
- If contact occurs, rinse eyes thoroughly with water
Principal Display Panel 118 Ml Bottle Label
Rugby®
NDC 0536-1962-97
Sebex
Medicated Dandruff,
Seborrheic Dermatitis,
and Psoriasis Shampoo
4 FL OZ (118 mL)
Structured Label Content
Uses
controls the symptoms of:
- dandruff
- seborrheic dermatitis
- psoriasis
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if condition worsens or does not improve after regular use of this product as directed.
Section 55105-1 (55105-1)
| Active ingredients | Purpose |
|---|---|
| Salicylic Acid, 2% }
Sulfur, 2% |
Anti-dandruff, anti-seborrheic dermatitis, anti-psoriasis |
Warnings
For external use only
Directions
- SHAKE WELL BEFORE USE
- wet the scalp or skin and massage in enough shampoo to work up a lather
- massage for 3-5 minutes and rinse out thoroughly
- repeat application and rinse thoroughly again
- for best results, use at least twice a week or as directed by a doctor
Do Not Use (Do not use)
- If condition covers a large area of the body, consult your doctor before using this product
- this product with other forms of psoriasis therapy such as ultraviolet radiation or prescription drugs unless directed to do so by a doctor
Other Information (Other information)
- store at room temperature
- see container for Lot No. & Exp. date
Inactive Ingredients (Inactive ingredients)
water, sodium lauryl sulfate, disodium cocoamphodipropionate, acrylates copolymer, citric acid, PEG-75 lanolin, fragrance, quaternium-15, diethylhexyl sodium sulfosuccinate, isopropyl palmitate, sodium hydroxide.
Questions Or Comments? (Questions or comments?)
1-800-645-2158
When Using This Product (When using this product)
- avoid contact with eyes
- If contact occurs, rinse eyes thoroughly with water
Principal Display Panel 118 Ml Bottle Label (PRINCIPAL DISPLAY PANEL - 118 mL Bottle Label)
Rugby®
NDC 0536-1962-97
Sebex
Medicated Dandruff,
Seborrheic Dermatitis,
and Psoriasis Shampoo
4 FL OZ (118 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:20.434600 · Updated: 2026-03-14T23:10:41.056891