Gentak®
aebfe247-cdf7-4acb-97b3-6d4341d7c815
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sterile Rx only
Indications and Usage
Gentamicin sulfate ophthalmic ointment is indicated in the topical treatment of ocular bacterial infections including conjunctivitis, keratitis, keratoconjunctivitis, corneal ulcers, blepharitis, blepharonconjunctivitis: acute meibomianitis, and dacryocystitis, caused by susceptible strains of the following microorganisms: Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Pseudomonas aeruginosa, and Serratia marcescens.
Dosage and Administration
Apply a small amount (approximately 1/2 inch ribbon) of ointment to the affected eye(s) two or three times a day.
Contraindications
Gentamicin sulfate ophthalmic ointment is con-traindicated in patients with known hypersensitivity to any of the components.
Adverse Reactions
Bacterial and fungal corneal ulcers have developed during treatment with gentamicin ophthalmic preparations. The most frequently reported adverse reactions are ocular burning and irritation upon drug instillation, non-specific conjunctivitis, conjunctival epithelial defects and conjunctival hyperemia. Other adverse reactions which have occurred rarely are allergic reactions, thrombocytopenic purpura and hallucinations.
How Supplied
GENTAK ® Gentamicin sulfate ophthalmic ointment USP, 0.3% is supplied in 3.5 g tube, box of one. (NDC 63187-873-35)
Medication Information
Indications and Usage
Gentamicin sulfate ophthalmic ointment is indicated in the topical treatment of ocular bacterial infections including conjunctivitis, keratitis, keratoconjunctivitis, corneal ulcers, blepharitis, blepharonconjunctivitis: acute meibomianitis, and dacryocystitis, caused by susceptible strains of the following microorganisms:
-
Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Pseudomonas aeruginosa, and Serratia marcescens.
Dosage and Administration
Apply a small amount (approximately 1/2 inch ribbon) of ointment to the affected eye(s) two or three times a day.
Contraindications
Gentamicin sulfate ophthalmic ointment is con-traindicated in patients with known hypersensitivity to any of the components.
Adverse Reactions
Bacterial and fungal corneal ulcers have developed during treatment with gentamicin ophthalmic preparations.
The most frequently reported adverse reactions are ocular burning and irritation upon drug instillation, non-specific conjunctivitis, conjunctival epithelial defects and conjunctival hyperemia.
Other adverse reactions which have occurred rarely are allergic reactions, thrombocytopenic purpura and hallucinations.
How Supplied
GENTAK® Gentamicin sulfate ophthalmic ointment USP, 0.3% is supplied in 3.5 g tube, box of one.
(NDC 63187-873-35)
Description
GENTAK® Gentamicin sulfate is a water soluble antibiotic of the aminoglycoside group.
Gentamicin sulfate ophthalmic ointment is a sterile ointment for ophthalmic use. Each gram contains gentamicin sulfate equivalent to 3 mg gentamicin in a base of white petrolatum and mineral oil, with methylparaben and propylparaben as preservatives.
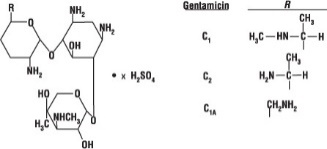
Gentamicin is obtained from cultures of Micromonospora purpurea. It is a mixture of the sulfate salts of gentamicin C1 C2, and C1A. All three components appear to have similar antimicrobial activities. Gentamicin sulfate occurs as a white to buff powder and is soluble in water and insoluble in alcohol. The structural formula is as follows:
Section 34072-9
General: Prolonged use of topical antibiotics may give rise to overgrowth of nonsusceptible organisms including fungi. Bacterial resistance to gentamicin may also develop. If purulent discharge, inflammation or pain becomes aggravated, the patient should discontinue use of the medication and consult a physician.
If irritation or hypersensitivity to any component of the drug develops, the patient should discontinue use of this preparation and appropriate therapy should be instituted.
Ophthalmic ointments may retard corneal healing.
Section 34076-0
Information for patients: To avoid contamination, do not touch tip of container to the eye, eyelid or any surface.
Section 34081-0
Pediatric Use: Safety and effectiveness in neonates have not been established.
Section 34083-6
Carcinogenesis, Mutagenesis, Impairment of Fertility: There are no published carcinogenicity or impairment of fertility studies on gentamicin. Aminoglycoside antibiotics have been found to be non-mutagenic.
Section 42228-7
Pregnancy: Pregnancy Category C. Gentamicin has been shown to depress body weights, kidney weights and median glomerular counts in newborn rats when administered systemically to pregnant rats in daily doses approximately 500 times the maximum recommended ophthalmic human dose. There are no adequate and well-controlled studies in pregnant women. Gentamicin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 42229-5
Sterile
Rx only
Section 44425-7
STORAGE: Store at 2° to 30°C (36° to 86°F).
Akorn
Manufactured by: Akorn, Inc.
Lake Forest, IL 60045
Relabeled by: Proficient Rx LP.
Thousand Oaks, CA 91320
GKO00N Rev. 09/08
Section 49489-8
Microbiology: Gentamicin sulfate is active in vitro against many strains of the following microorganisms:
-
Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Pseudomonas aeruginosa, and Serratia marcescens.
Section 51945-4
Principal Display Panel Text for Container Label:
NDC 63187-873-35 Akorn Logo
Gentak®
brand of Gentamicin Sulfate
Ophthalmic Ointment USP, 0.3%
(equivalent to 3 mg gentamicin per gram)
Net Wt 3.5 g (1/8 oz) Sterile
Rx only
Warnings
NOT FOR INJECTION INTO THE EYE.
Gentamicin sulfate ophthalmic ointment is not for injection. It should never be injected subconjunctivally, nor should it be directly introduced into the anterior chamber of the eye.
Structured Label Content
Section 34072-9 (34072-9)
General: Prolonged use of topical antibiotics may give rise to overgrowth of nonsusceptible organisms including fungi. Bacterial resistance to gentamicin may also develop. If purulent discharge, inflammation or pain becomes aggravated, the patient should discontinue use of the medication and consult a physician.
If irritation or hypersensitivity to any component of the drug develops, the patient should discontinue use of this preparation and appropriate therapy should be instituted.
Ophthalmic ointments may retard corneal healing.
Section 34076-0 (34076-0)
Information for patients: To avoid contamination, do not touch tip of container to the eye, eyelid or any surface.
Section 34081-0 (34081-0)
Pediatric Use: Safety and effectiveness in neonates have not been established.
Section 34083-6 (34083-6)
Carcinogenesis, Mutagenesis, Impairment of Fertility: There are no published carcinogenicity or impairment of fertility studies on gentamicin. Aminoglycoside antibiotics have been found to be non-mutagenic.
Section 42228-7 (42228-7)
Pregnancy: Pregnancy Category C. Gentamicin has been shown to depress body weights, kidney weights and median glomerular counts in newborn rats when administered systemically to pregnant rats in daily doses approximately 500 times the maximum recommended ophthalmic human dose. There are no adequate and well-controlled studies in pregnant women. Gentamicin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 42229-5 (42229-5)
Sterile
Rx only
Section 44425-7 (44425-7)
STORAGE: Store at 2° to 30°C (36° to 86°F).
Akorn
Manufactured by: Akorn, Inc.
Lake Forest, IL 60045
Relabeled by: Proficient Rx LP.
Thousand Oaks, CA 91320
GKO00N Rev. 09/08
Section 49489-8 (49489-8)
Microbiology: Gentamicin sulfate is active in vitro against many strains of the following microorganisms:
-
Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Pseudomonas aeruginosa, and Serratia marcescens.
Section 51945-4 (51945-4)
Principal Display Panel Text for Container Label:
NDC 63187-873-35 Akorn Logo
Gentak®
brand of Gentamicin Sulfate
Ophthalmic Ointment USP, 0.3%
(equivalent to 3 mg gentamicin per gram)
Net Wt 3.5 g (1/8 oz) Sterile
Rx only
Warnings (WARNINGS)
NOT FOR INJECTION INTO THE EYE.
Gentamicin sulfate ophthalmic ointment is not for injection. It should never be injected subconjunctivally, nor should it be directly introduced into the anterior chamber of the eye.
Description (DESCRIPTION)
GENTAK® Gentamicin sulfate is a water soluble antibiotic of the aminoglycoside group.
Gentamicin sulfate ophthalmic ointment is a sterile ointment for ophthalmic use. Each gram contains gentamicin sulfate equivalent to 3 mg gentamicin in a base of white petrolatum and mineral oil, with methylparaben and propylparaben as preservatives.
Gentamicin is obtained from cultures of Micromonospora purpurea. It is a mixture of the sulfate salts of gentamicin C1 C2, and C1A. All three components appear to have similar antimicrobial activities. Gentamicin sulfate occurs as a white to buff powder and is soluble in water and insoluble in alcohol. The structural formula is as follows:
How Supplied (HOW SUPPLIED)
GENTAK® Gentamicin sulfate ophthalmic ointment USP, 0.3% is supplied in 3.5 g tube, box of one.
(NDC 63187-873-35)
Adverse Reactions (ADVERSE REACTIONS)
Bacterial and fungal corneal ulcers have developed during treatment with gentamicin ophthalmic preparations.
The most frequently reported adverse reactions are ocular burning and irritation upon drug instillation, non-specific conjunctivitis, conjunctival epithelial defects and conjunctival hyperemia.
Other adverse reactions which have occurred rarely are allergic reactions, thrombocytopenic purpura and hallucinations.
Contraindications (CONTRAINDICATIONS)
Gentamicin sulfate ophthalmic ointment is con-traindicated in patients with known hypersensitivity to any of the components.
Indications and Usage (INDICATIONS AND USAGE)
Gentamicin sulfate ophthalmic ointment is indicated in the topical treatment of ocular bacterial infections including conjunctivitis, keratitis, keratoconjunctivitis, corneal ulcers, blepharitis, blepharonconjunctivitis: acute meibomianitis, and dacryocystitis, caused by susceptible strains of the following microorganisms:
-
Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Pseudomonas aeruginosa, and Serratia marcescens.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Apply a small amount (approximately 1/2 inch ribbon) of ointment to the affected eye(s) two or three times a day.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:57.196848 · Updated: 2026-03-14T21:41:56.665367