Humco Sodium Bicarbonate, Usp
aeaa34f6-216d-49f3-8393-c7bda96a3e32
34390-5
HUMAN OTC DRUG LABEL
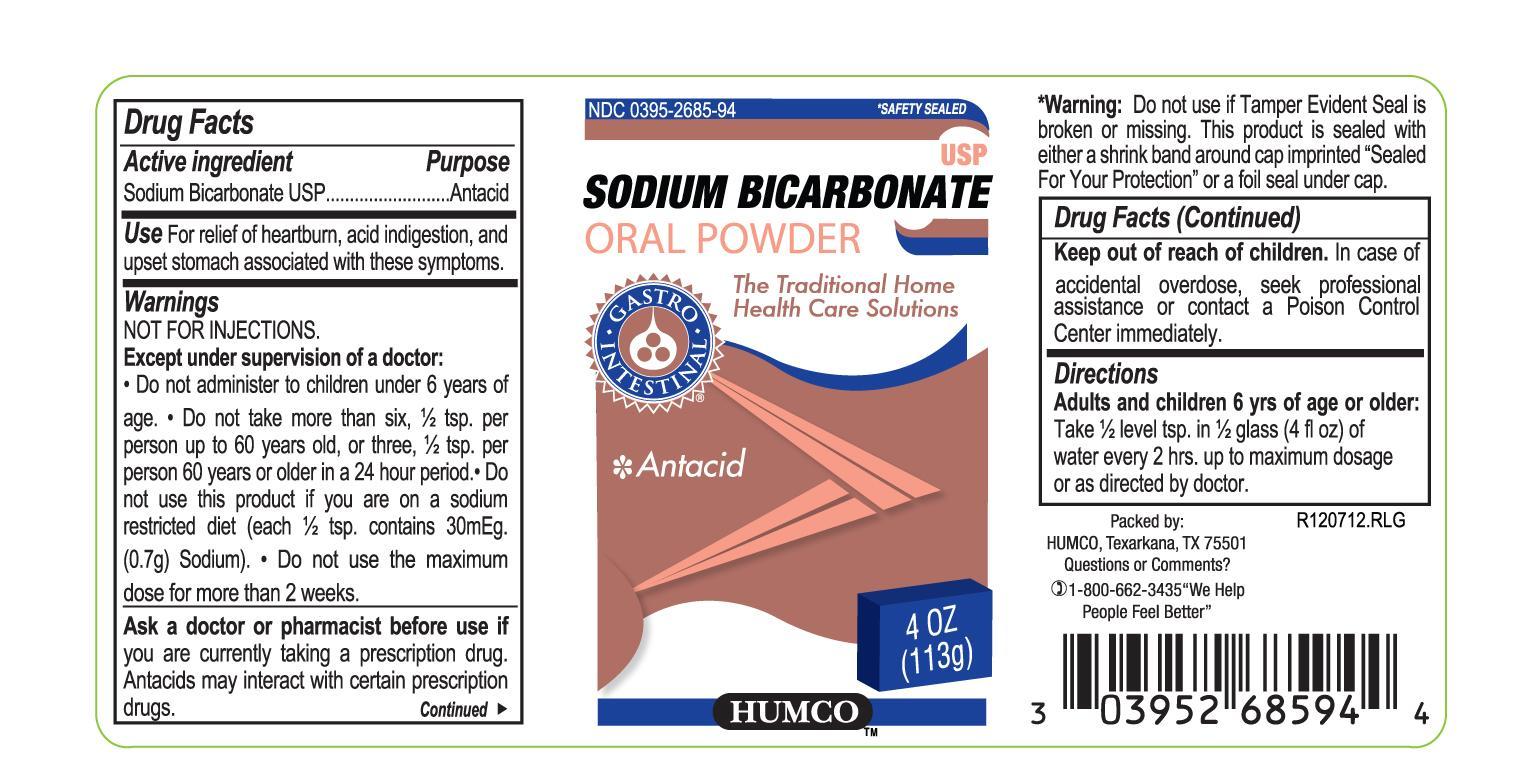
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antacid
Medication Information
Purpose
Antacid
Description
Drug Facts
Use
For relief of heartburn, acid indigestion, and upset stomach associated with these symptoms.
Section 42229-5
Drug Facts
Section 43683-2
FOR ORAL USE ONLY
Except under supervision of a doctor:
Do not administer to children under 12 years of age.
Do not take more than six, 1/2 tsp. per person up to 60 years old, or three 1/2 tsp. per person 60 years or older in a 24 hour period.
STOMACH WARNING: TO AVOID SERIOUS INJURY, DO NOT TAKE UNTIL POWDER IS COMPLETELY DISSOLVED. IT IS VERY IMPORTANT NOT TO TAKE THIS PRODUCT WHEN OVERLY FULL FROM FOOD OR DRINK.
Section 50568-5
Ask a doctor or pharmacist before use if you are currently taking a prescription drug. Antacids may interact with certain prescription drugs.
Ask a doctor before use if you are on a sodium restricted diet.
Stop use and consult a doctor if you have used the maximum dose for two weeks, or severe stomach pain occurs after using this product.
Warnings
FOR ORAL USE ONLY
Except under supervision of a doctor:
Do not administer to children under 12 years of age.
Do not take more than six, 1/2 tsp. per person up to 60 years old, or three 1/2 tsp. per person 60 years or older in a 24 hour period.
STOMACH WARNING: TO AVOID SERIOUS INJURY, DO NOT TAKE UNTIL POWDER IS COMPLETELY DISSOLVED. IT IS VERY IMPORTANT NOT TO TAKE THIS PRODUCT WHEN OVERLY FULL FROM FOOD OR DRINK.
Directions
Adult and children 12 yrs of age and older:
Dissolve 1/2 level teaspoon in glass (4 fl oz) of water every 2 hours up to maximum dosafe or as directed by doctor. Dissolve completely in water before drinking. Do not exceed recommended dose. SEE WARNINGS.
2017 Label Update
2025 Label Update
Active Ingredient
Soduim Bicarbonate, USP
Inactive Ingredients
none
Principal Display Pannel
Label 16 oz
Keep Out of Reach of Children.
In case of accidental overdose, seek professional assistance or contact a Poison Control Center immediately.
Packae Principal Display Pannel
Label 4 oz
Structured Label Content
Use
For relief of heartburn, acid indigestion, and upset stomach associated with these symptoms.
Section 42229-5 (42229-5)
Drug Facts
Section 43683-2 (43683-2)
FOR ORAL USE ONLY
Except under supervision of a doctor:
Do not administer to children under 12 years of age.
Do not take more than six, 1/2 tsp. per person up to 60 years old, or three 1/2 tsp. per person 60 years or older in a 24 hour period.
STOMACH WARNING: TO AVOID SERIOUS INJURY, DO NOT TAKE UNTIL POWDER IS COMPLETELY DISSOLVED. IT IS VERY IMPORTANT NOT TO TAKE THIS PRODUCT WHEN OVERLY FULL FROM FOOD OR DRINK.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are currently taking a prescription drug. Antacids may interact with certain prescription drugs.
Ask a doctor before use if you are on a sodium restricted diet.
Stop use and consult a doctor if you have used the maximum dose for two weeks, or severe stomach pain occurs after using this product.
Purpose
Antacid
Warnings
FOR ORAL USE ONLY
Except under supervision of a doctor:
Do not administer to children under 12 years of age.
Do not take more than six, 1/2 tsp. per person up to 60 years old, or three 1/2 tsp. per person 60 years or older in a 24 hour period.
STOMACH WARNING: TO AVOID SERIOUS INJURY, DO NOT TAKE UNTIL POWDER IS COMPLETELY DISSOLVED. IT IS VERY IMPORTANT NOT TO TAKE THIS PRODUCT WHEN OVERLY FULL FROM FOOD OR DRINK.
Directions
Adult and children 12 yrs of age and older:
Dissolve 1/2 level teaspoon in glass (4 fl oz) of water every 2 hours up to maximum dosafe or as directed by doctor. Dissolve completely in water before drinking. Do not exceed recommended dose. SEE WARNINGS.
2017 Label Update
2025 Label Update
Active Ingredient
Soduim Bicarbonate, USP
Inactive Ingredients (Inactive ingredients)
none
Principal Display Pannel
Label 16 oz
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of accidental overdose, seek professional assistance or contact a Poison Control Center immediately.
Packae Principal Display Pannel
Label 4 oz
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:46.711719 · Updated: 2026-03-14T23:11:33.591824