Drug Facts
ae9d1672-9ade-4139-9126-5289b87fe574
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
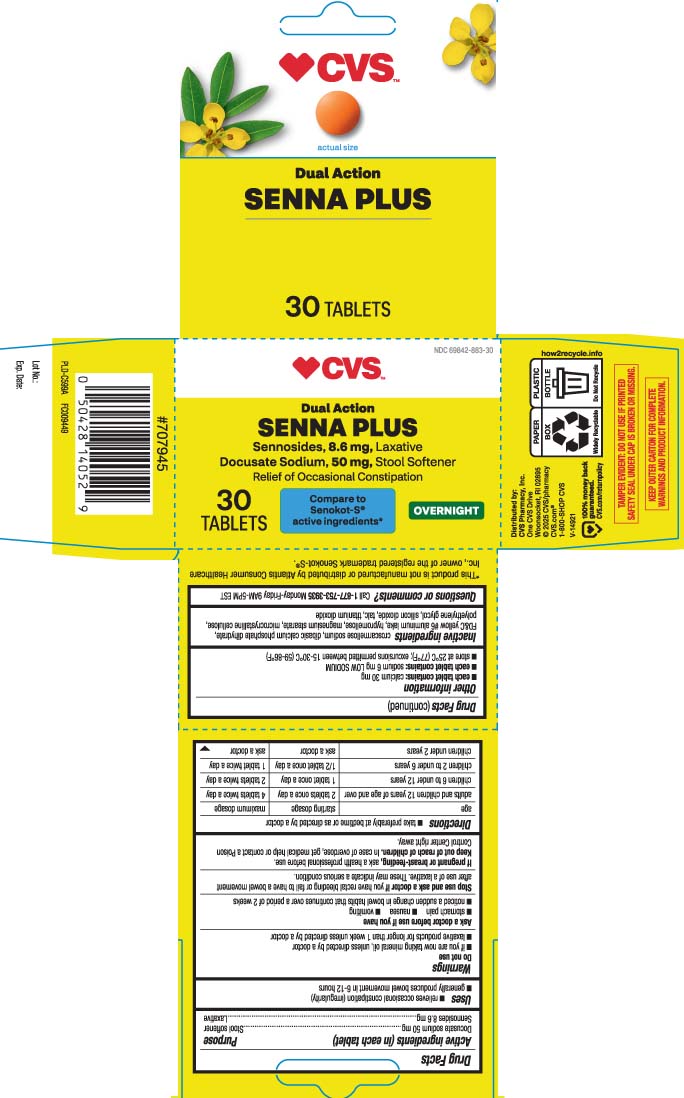
Docusate sodium 50 mg Sennosides 8.6 mg
Purpose
Stool softener Laxative
Medication Information
Purpose
Stool softener
Laxative
Description
Docusate sodium 50 mg Sennosides 8.6 mg
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 6-12 hours
Directions
- take preferably at bedtime or as directed by a doctor
| age | starting dosage | maximum dosage |
| adults and children 12 years of age and over | 2 tablets once a day | 4 tablets twice a day |
| children 6 to under 12 years | 1 tablet once a day | 2 tablets twice a day |
| children 2 to under 6 years | 1/2 tablet once a day | 1 tablet twice a day |
| children under 2 years | ask a doctor | ask a doctor |
Do Not Use
- if you are now taking mineral oil, unless directed by a doctor
- laxative products for longer than 1 week unless directed by a doctor
Product Labeling
Other Information
- each tablet contains: calcium 30 mg
- each tablet contains: sodium 6 mg VERY LOW SODIUM
- store at 25ºC (77ºF); excursions permitted between 15-30ºC (59-86ºF)
Inactive Ingredients
croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, silicon dioxide, talc, titanium dioxide
Questions Or Comments?
Call 1-877-753-3935 Monday-Friday 9AM-5PM EST
Principal Display Panel
Compare to Senokot-S® active ingredients*
Dual action
SENNA PLUS
Sennosides, 8.6 mg, Laxative
Docusate Sodium, 50 mg, Stool Softener
Relief of Occasional Constipation
OVERNIGHT
TABLETS
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
*This product is not manufactured or distributed by Atlantis Consumer Healthcare Inc., owner of the registered trademark Senokot-S®.
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
Stop Use and Ask A Doctor If
you have rectal bleeding or fail to have a bowel movement after use of a laxative. These may indicate a serious condition.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredients (in Each Tablet)
Docusate sodium 50 mg
Sennosides 8.6 mg
Ask A Doctor Before Use If You Have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that continues over a periodof 2 weeks
Structured Label Content
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 6-12 hours
Purpose
Stool softener
Laxative
Directions
- take preferably at bedtime or as directed by a doctor
| age | starting dosage | maximum dosage |
| adults and children 12 years of age and over | 2 tablets once a day | 4 tablets twice a day |
| children 6 to under 12 years | 1 tablet once a day | 2 tablets twice a day |
| children 2 to under 6 years | 1/2 tablet once a day | 1 tablet twice a day |
| children under 2 years | ask a doctor | ask a doctor |
Do Not Use (Do not use)
- if you are now taking mineral oil, unless directed by a doctor
- laxative products for longer than 1 week unless directed by a doctor
Product Labeling
Other Information (Other information)
- each tablet contains: calcium 30 mg
- each tablet contains: sodium 6 mg VERY LOW SODIUM
- store at 25ºC (77ºF); excursions permitted between 15-30ºC (59-86ºF)
Inactive Ingredients (Inactive ingredients)
croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, silicon dioxide, talc, titanium dioxide
Questions Or Comments? (Questions or comments?)
Call 1-877-753-3935 Monday-Friday 9AM-5PM EST
Principal Display Panel
Compare to Senokot-S® active ingredients*
Dual action
SENNA PLUS
Sennosides, 8.6 mg, Laxative
Docusate Sodium, 50 mg, Stool Softener
Relief of Occasional Constipation
OVERNIGHT
TABLETS
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
*This product is not manufactured or distributed by Atlantis Consumer Healthcare Inc., owner of the registered trademark Senokot-S®.
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
you have rectal bleeding or fail to have a bowel movement after use of a laxative. These may indicate a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredients (in Each Tablet) (Active ingredients (in each tablet))
Docusate sodium 50 mg
Sennosides 8.6 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that continues over a periodof 2 weeks
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:46.710383 · Updated: 2026-03-14T23:11:33.591824