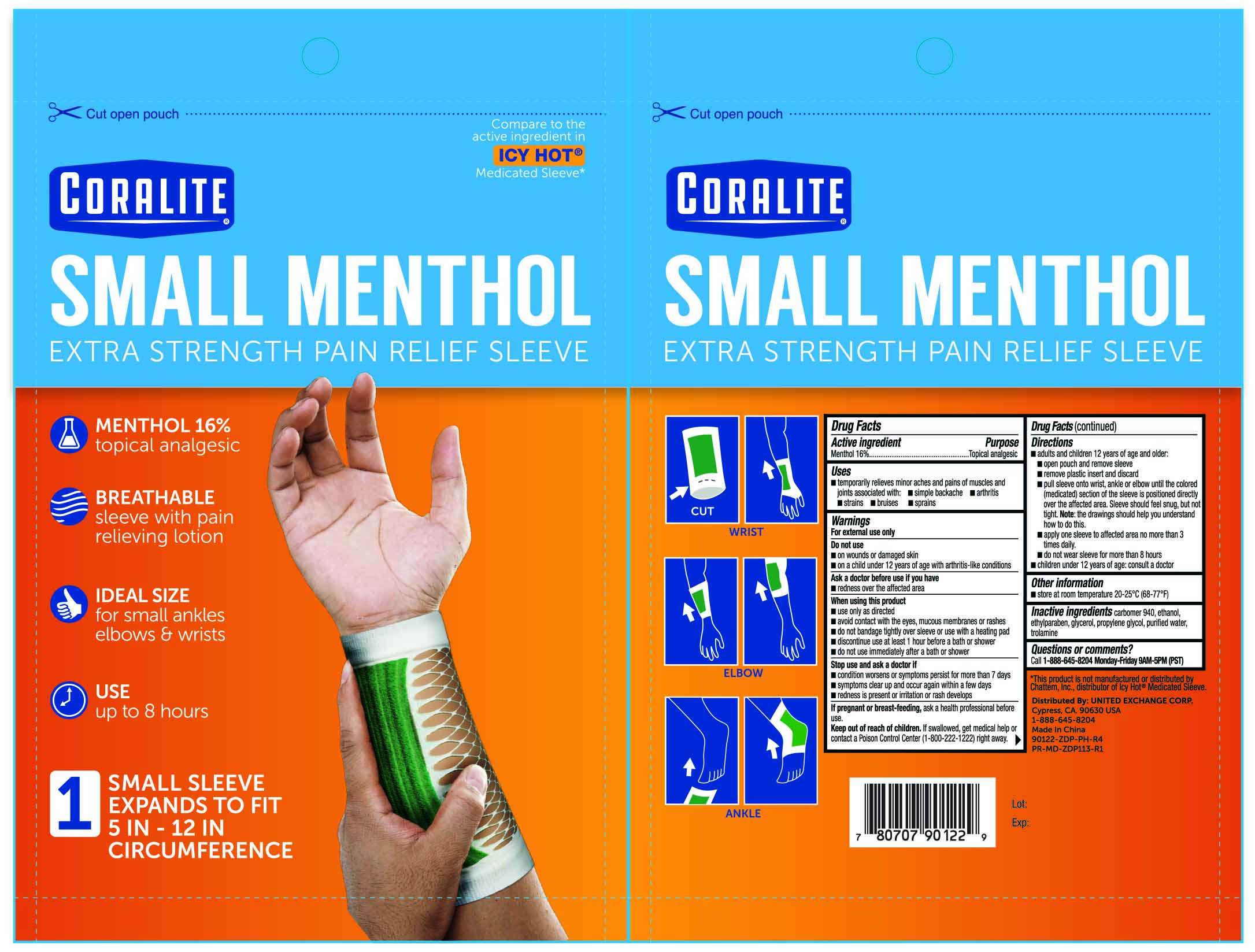
coralite small menthol pain relief sleeve 1ct 90122 zdp
ae314478-b8ab-4e90-bd0a-7a882d5c1b2e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient Purpose Menthol 16.0%.......................................Topical Analgesic Patch
Medication Information
Warnings and Precautions
Warnings
For external use only
Active Ingredient
Active Ingredient Purpose
Menthol 16.0%.......................................Topical Analgesic Patch
Indications and Usage
Directions
- Adults and children 12 years of age and older:
- open pouch and remove sleeve
- remove plastic insert and discard
- pull sleeve onto wrist, ankle or elbow until the colored (medicated) section of the sleeve is positioned directly over the affected area. Sleeve should feel snug, but not tight. Note: the drawings should help you understand how to do this.
- apply one sleeve to affected area no more than 3 times daily
- do not wear sleeve for more than 8 hours
- children under 12 years of age: consult a doctor
Dosage and Administration
Distributed by:
United Exchange Corp.
Cypress, CA 90630 USA
Made in China
Description
Active Ingredient Purpose Menthol 16.0%.......................................Topical Analgesic Patch
Section 44425-7
Other information
- store at room temperature 20-25°C (68-77°F)
Section 50565-1
keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Section 50566-9
Stop use and ask a doctor if
- condition worsens, or if symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
- redness is present or irritation or rash develops
Section 50567-7
When using this product
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- do not bandage tightly over sleeve or use with a heating pad
- discontinue use at least 1 hour before a bath or shower
- do not use immediately after a bath or shower
Section 50569-3
Ask a doctor before use if you have
- redness over the affected area
Section 50570-1
Do not use
- on wounds or damaged skin
- on a child under 12 years of age with arthritis-like conditions
Section 51727-6
Inactive ingredients
carbomber 940, ethanol, ethylparaben, glycerol, propylene glycol, purified water, trolamine
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use
Section 55105-1
Uses
- temporarily relieves minor aches and pains of muscles and joints associated with
- simple backache
- arthritis
- strains
- bruises
- sprains
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only
Active Ingredient (55106-9)
Active Ingredient Purpose
Menthol 16.0%.......................................Topical Analgesic Patch
Indications and Usage (34067-9)
Directions
- Adults and children 12 years of age and older:
- open pouch and remove sleeve
- remove plastic insert and discard
- pull sleeve onto wrist, ankle or elbow until the colored (medicated) section of the sleeve is positioned directly over the affected area. Sleeve should feel snug, but not tight. Note: the drawings should help you understand how to do this.
- apply one sleeve to affected area no more than 3 times daily
- do not wear sleeve for more than 8 hours
- children under 12 years of age: consult a doctor
Dosage and Administration (34068-7)
Distributed by:
United Exchange Corp.
Cypress, CA 90630 USA
Made in China
Section 44425-7 (44425-7)
Other information
- store at room temperature 20-25°C (68-77°F)
Section 50565-1 (50565-1)
keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- condition worsens, or if symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
- redness is present or irritation or rash develops
Section 50567-7 (50567-7)
When using this product
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- do not bandage tightly over sleeve or use with a heating pad
- discontinue use at least 1 hour before a bath or shower
- do not use immediately after a bath or shower
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- redness over the affected area
Section 50570-1 (50570-1)
Do not use
- on wounds or damaged skin
- on a child under 12 years of age with arthritis-like conditions
Section 51727-6 (51727-6)
Inactive ingredients
carbomber 940, ethanol, ethylparaben, glycerol, propylene glycol, purified water, trolamine
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use
Section 55105-1 (55105-1)
Uses
- temporarily relieves minor aches and pains of muscles and joints associated with
- simple backache
- arthritis
- strains
- bruises
- sprains
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:18.519963 · Updated: 2026-03-14T23:13:11.905913