adac0ead-3ca7-ca22-e053-2995a90aacbb
34390-5
HUMAN OTC DRUG LABEL
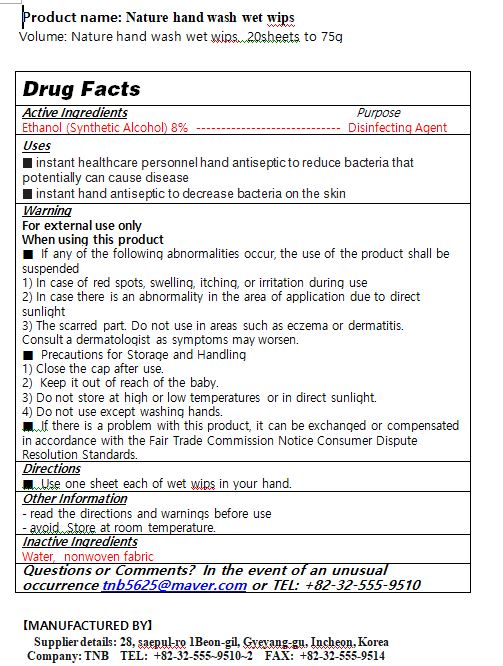
Drug Facts
Composition & Product
Identifiers & Packaging
Description
alcohol
Medication Information
Warnings and Precautions
For external use only
When using this product
■ If any of the following abnormalities occur, the use of the product shall be suspended
1) In case of red spots, swelling, itching, or irritation during use
2) In case there is an abnormality in the area of application due to direct sunlight
3) The scarred part. Do not use in areas such as eczema or dermatitis.
Consult a dermatologist as symptoms may worsen.
■ Precautions for Storage and Handling
1) Close the cap after use.
2) Keep it out of reach of the baby.
3) Do not store at high or low temperatures or in direct sunlight.
4) Do not use except washing hands.
■ If there is a problem with this product, it can be exchanged or compensated in accordance with the Fair Trade Commission Notice Consumer Dispute Resolution Standards.
Indications and Usage
■ Use one sheet each of wet wips in your hand.
Dosage and Administration
for external use only
Description
alcohol
Section 50565-1
keep out of reach of the children
Section 51727-6
Dihydrogen Oxid
Alcohol
Glycerin
Dipropyene Glycol
Sodium Hyaluronate
Butylene Glycol
Centella asiatica Extract
Camellia sinensis Leaf Extract
Aloe barbadensis Leaf Extract
Portulaca Oleracea Exract
Peppermint
Disodium EDTA
Chlorphenesin
Phenoxyethanol
Section 51945-4
Section 55105-1
■ instant healthcare personnel hand antiseptic to reduce bacteria that potentially can cause disease
■ instant hand antiseptic to decrease bacteria on the skin
Section 55106-9
alcohol
Structured Label Content
Indications and Usage (34067-9)
■ Use one sheet each of wet wips in your hand.
Dosage and Administration (34068-7)
for external use only
Warnings and Precautions (34071-1)
For external use only
When using this product
■ If any of the following abnormalities occur, the use of the product shall be suspended
1) In case of red spots, swelling, itching, or irritation during use
2) In case there is an abnormality in the area of application due to direct sunlight
3) The scarred part. Do not use in areas such as eczema or dermatitis.
Consult a dermatologist as symptoms may worsen.
■ Precautions for Storage and Handling
1) Close the cap after use.
2) Keep it out of reach of the baby.
3) Do not store at high or low temperatures or in direct sunlight.
4) Do not use except washing hands.
■ If there is a problem with this product, it can be exchanged or compensated in accordance with the Fair Trade Commission Notice Consumer Dispute Resolution Standards.
Section 50565-1 (50565-1)
keep out of reach of the children
Section 51727-6 (51727-6)
Dihydrogen Oxid
Alcohol
Glycerin
Dipropyene Glycol
Sodium Hyaluronate
Butylene Glycol
Centella asiatica Extract
Camellia sinensis Leaf Extract
Aloe barbadensis Leaf Extract
Portulaca Oleracea Exract
Peppermint
Disodium EDTA
Chlorphenesin
Phenoxyethanol
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
■ instant healthcare personnel hand antiseptic to reduce bacteria that potentially can cause disease
■ instant hand antiseptic to decrease bacteria on the skin
Section 55106-9 (55106-9)
alcohol
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:33.536201 · Updated: 2026-03-14T22:58:08.346905