Asepso Wipes Bkc0.1
ada5d5e3-a125-0b0b-e053-2a95a90a3625
34390-5
HUMAN OTC DRUG LABEL
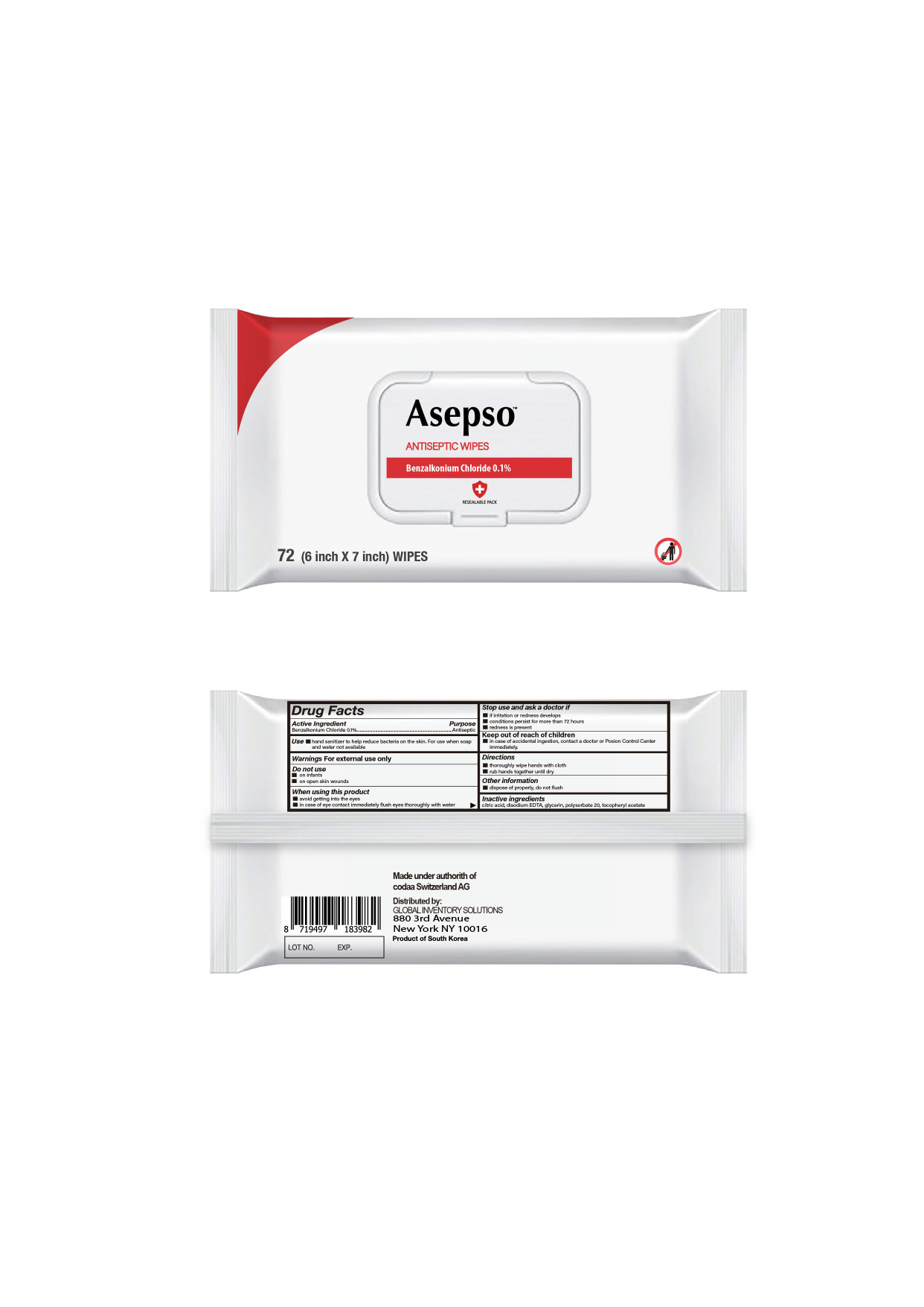
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzalkonium chloride 0.1%
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Benzalkonium chloride 0.1%
Use
■ hand sanitizer to help reduce bacteria on the skin. For use when soap and water not available
Warnings
For external use only
Directions
■ thoroughly wipe hands with cloth
■ rub hands together until dry
Do Not Use
■ on infants
■ on open skin wounds
Package Label
Active Ingredient
Benzalkonium chloride 0.1%
Other Information
■ dispose of properly, do not flush
Inactive Ingredients
citric acid,disodium EDTA, glycerin, polysorbate 20, tocopheryl acetate
When Using This Product
■ avoid getting into the eyes
■ in case of eye contact immediately flush eyes thoroughly with water
Stop Use and Ask A Doctor If
■ irritation or redness develops
■ conditions persist for more than 72 hours
■ redness is present
Keep Out of Reach of Children
■ in case of accidental ingestion, contact a doctor or Poison Control Center immediately.
Structured Label Content
Use
■ hand sanitizer to help reduce bacteria on the skin. For use when soap and water not available
Purpose
Antiseptic
Warnings
For external use only
Directions
■ thoroughly wipe hands with cloth
■ rub hands together until dry
Do Not Use (Do not use)
■ on infants
■ on open skin wounds
Package Label
Active Ingredient (Active ingredient)
Benzalkonium chloride 0.1%
Other Information (Other information)
■ dispose of properly, do not flush
Inactive Ingredients (Inactive ingredients)
citric acid,disodium EDTA, glycerin, polysorbate 20, tocopheryl acetate
When Using This Product (When using this product)
■ avoid getting into the eyes
■ in case of eye contact immediately flush eyes thoroughly with water
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
■ irritation or redness develops
■ conditions persist for more than 72 hours
■ redness is present
Keep Out of Reach of Children (Keep out of reach of children)
■ in case of accidental ingestion, contact a doctor or Poison Control Center immediately.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:04.075560 · Updated: 2026-03-14T23:00:21.684898