drug facts
ad9fde8b-10cd-4778-917d-1ed6b67e5ab1
34390-5
HUMAN OTC DRUG LABEL
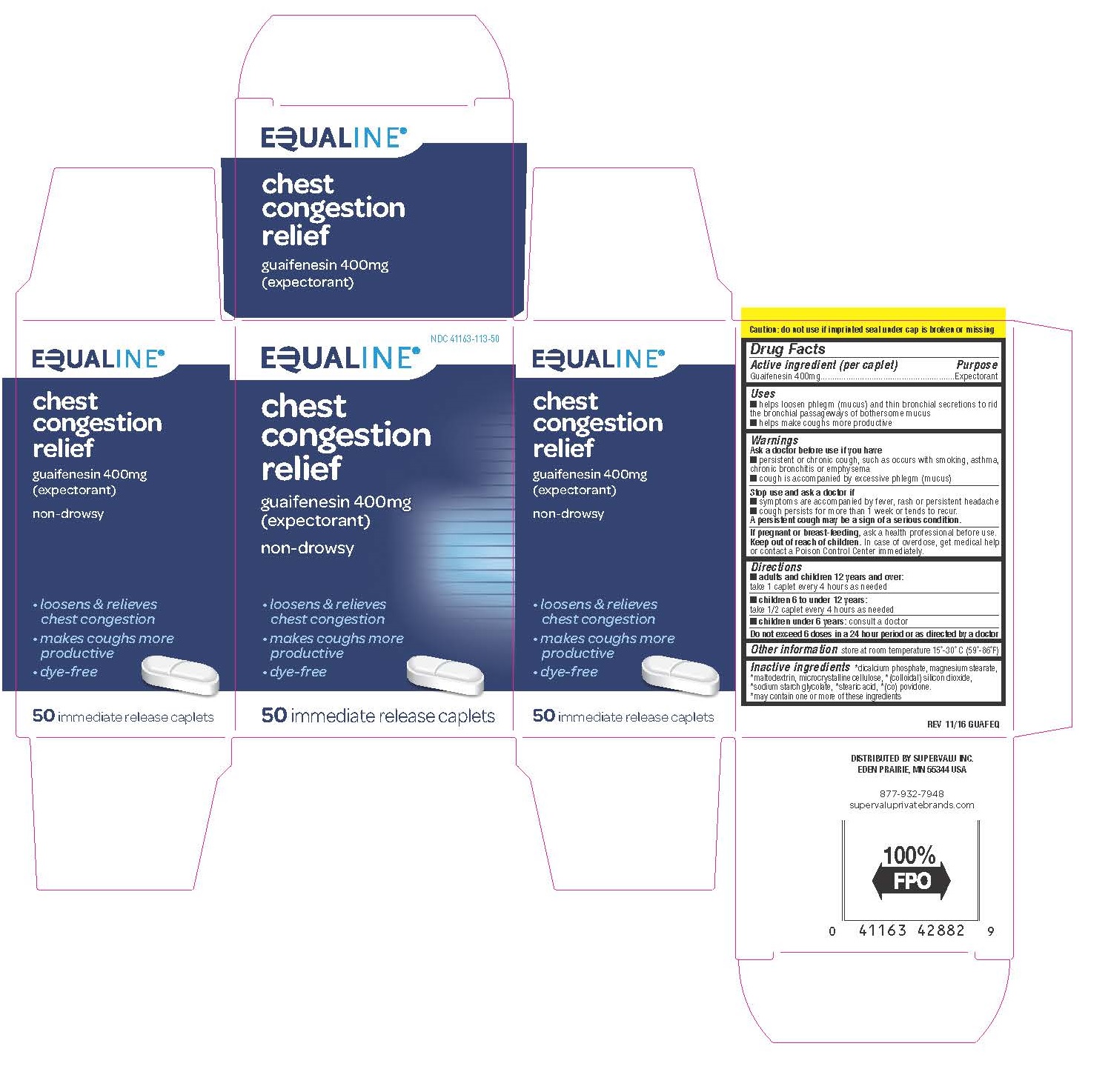
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Expectorant
Description
Active ingredient (per tablet) Guaifenesin 400mg
Medication Information
Warnings
Ask doctor before use if you have
- persistent or chronic cough, such as occurs with smoking, asthma, bronchitis or emphysema
- cough is accompanied by excessive phlegm (mucus)
Active Ingredient
Active ingredient (per tablet)
Guaifenesin 400mg
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus
- helps make coughs more productive
Purpose
Expectorant
Directions
- Adults and children 12 years of age and over: take 1 tablet every 4 hours as needed
- Children 6 to 10 under 12 years of age: take 1/2 tablet every 4 hours as needed
- Children under 6 years of age: consult a doctor
Other Information
store at 15°-30°C (59°-86°F)
Inactive Ingredients
magnesium stearate, microcrystalline cellulose. May also contain (colloidal) silicon dioxide, (co) povidone, dicalcium phosphate, maltodextrin, sodium starch glycolate, stearic acid.
Description
Active ingredient (per tablet) Guaifenesin 400mg
Section 50565-1
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control
Center immediately.
Section 50566-9
Stop use and ask doctor if
- Symptoms are accompanied by fever, rash or persistent headache
- cough persists for more than 1 week or tends to recur
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Structured Label Content
Warnings
Ask doctor before use if you have
- persistent or chronic cough, such as occurs with smoking, asthma, bronchitis or emphysema
- cough is accompanied by excessive phlegm (mucus)
Active Ingredient (55106-9)
Active ingredient (per tablet)
Guaifenesin 400mg
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus
- helps make coughs more productive
Purpose
Expectorant
Directions
- Adults and children 12 years of age and over: take 1 tablet every 4 hours as needed
- Children 6 to 10 under 12 years of age: take 1/2 tablet every 4 hours as needed
- Children under 6 years of age: consult a doctor
Other Information
store at 15°-30°C (59°-86°F)
Inactive Ingredients (Inactive ingredients)
magnesium stearate, microcrystalline cellulose. May also contain (colloidal) silicon dioxide, (co) povidone, dicalcium phosphate, maltodextrin, sodium starch glycolate, stearic acid.
Section 50565-1 (50565-1)
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control
Center immediately.
Section 50566-9 (50566-9)
Stop use and ask doctor if
- Symptoms are accompanied by fever, rash or persistent headache
- cough persists for more than 1 week or tends to recur
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:02.336017 · Updated: 2026-03-14T23:06:18.731306