79805-011
ad49532e-4a4a-5ed9-e053-2a95a90ac053
34390-5
HUMAN OTC DRUG LABEL
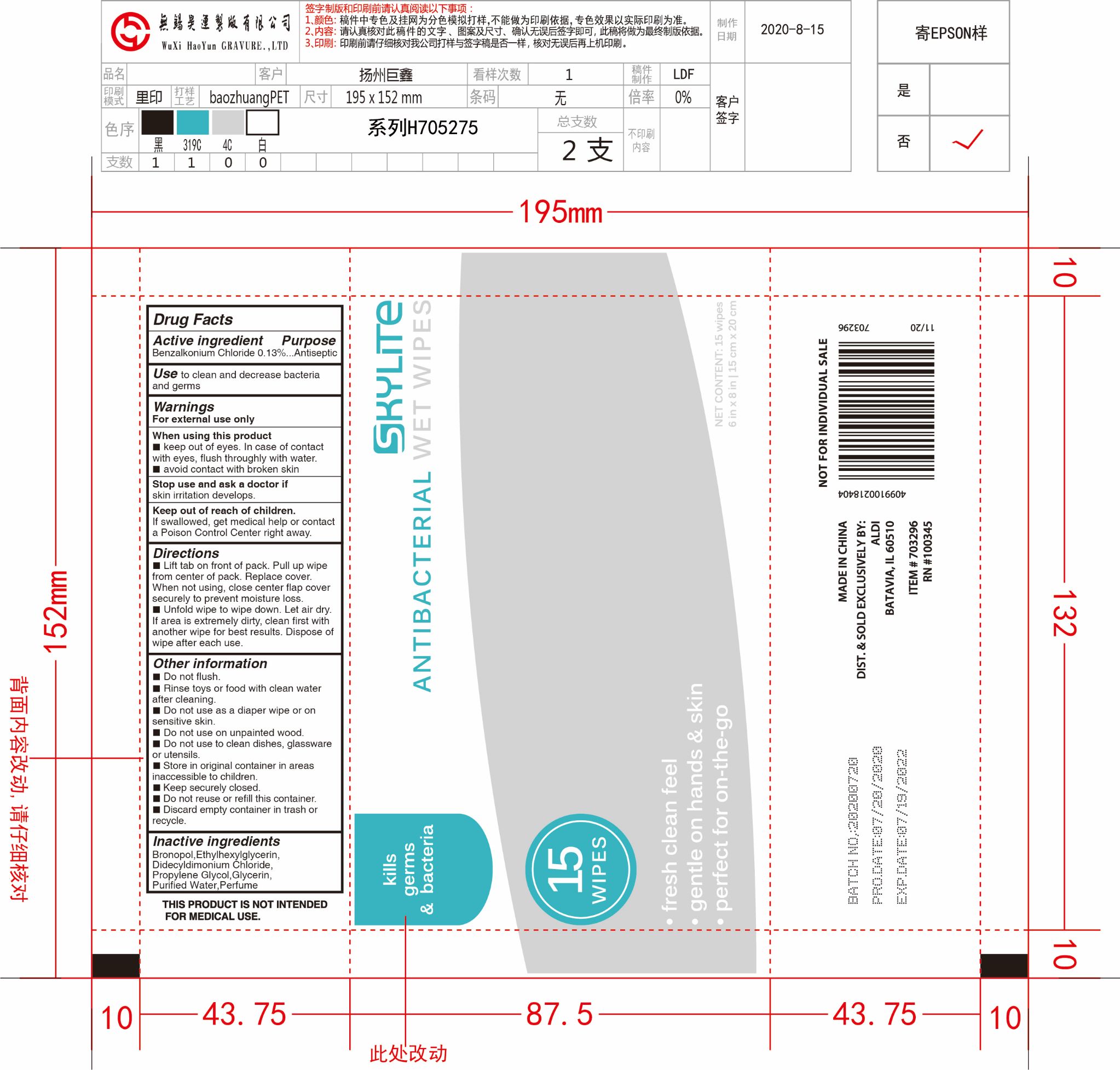
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzalkonium Chloride 0.13%
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Benzalkonium Chloride 0.13%
Use
Use to clean and decrease bacteria and germs.
Section 50565-1
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if skin irritation develops.
Section 50567-7
When using this product
- Keep out of eyes. In case of contact with eyes, flush throughly with water.
- Avoid contact with broken skin.
Warnings
- For external use only
Directions
- Lift tab on front of pack. Pull up wipe from center of pack. Replace cover. When not using, close center flap cover securely to prevert moisture loss.
- Unfold wipe to wipe down. Let air dry. If area is extremely dirty, clean first with another wipe for best results. Dispose of wipe after each use.
Other Information
- Do not flush.
- Rinse toys or food with clean water after cleaning.
- Do not use as a diaper wipe or on sensitive skin.
- Do not use on unpainted wood.
- Do not use to clean dishes, glassware or utensils.
- Store in original container in areas inaccessible to children.
- Keep securely closed.
- Do not reuse or refill this container.
- Discard empty container in trash or recycle.
Active Ingredient(s)
Benzalkonium Chloride 0.13%
Inactive Ingredients
Bronopol, Ethylhexylglycerin, Didecyldimonium Chloride, Propylene Glycol, Glycerin, Purified Water, Perfume.
Package Label Principal Display Panel
15pcs NDC: 79805-011-01
Structured Label Content
Use
Use to clean and decrease bacteria and germs.
Section 50565-1 (50565-1)
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if skin irritation develops.
Section 50567-7 (50567-7)
When using this product
- Keep out of eyes. In case of contact with eyes, flush throughly with water.
- Avoid contact with broken skin.
Purpose
Antiseptic
Warnings
- For external use only
Directions
- Lift tab on front of pack. Pull up wipe from center of pack. Replace cover. When not using, close center flap cover securely to prevert moisture loss.
- Unfold wipe to wipe down. Let air dry. If area is extremely dirty, clean first with another wipe for best results. Dispose of wipe after each use.
Other Information (Other information)
- Do not flush.
- Rinse toys or food with clean water after cleaning.
- Do not use as a diaper wipe or on sensitive skin.
- Do not use on unpainted wood.
- Do not use to clean dishes, glassware or utensils.
- Store in original container in areas inaccessible to children.
- Keep securely closed.
- Do not reuse or refill this container.
- Discard empty container in trash or recycle.
Active Ingredient(s)
Benzalkonium Chloride 0.13%
Inactive Ingredients (Inactive ingredients)
Bronopol, Ethylhexylglycerin, Didecyldimonium Chloride, Propylene Glycol, Glycerin, Purified Water, Perfume.
Package Label Principal Display Panel (Package Label - Principal Display Panel)
15pcs NDC: 79805-011-01
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:26.250750 · Updated: 2026-03-14T23:00:52.351984