Meclizine Hydrochloride Tablets, Usp 12.5 Mg
acdf636c-66f1-4a1f-87a8-786f51d75708
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Meclizine HCl, USP 12.5 mg
Purpose
Antiemetic
Medication Information
Warnings and Precautions
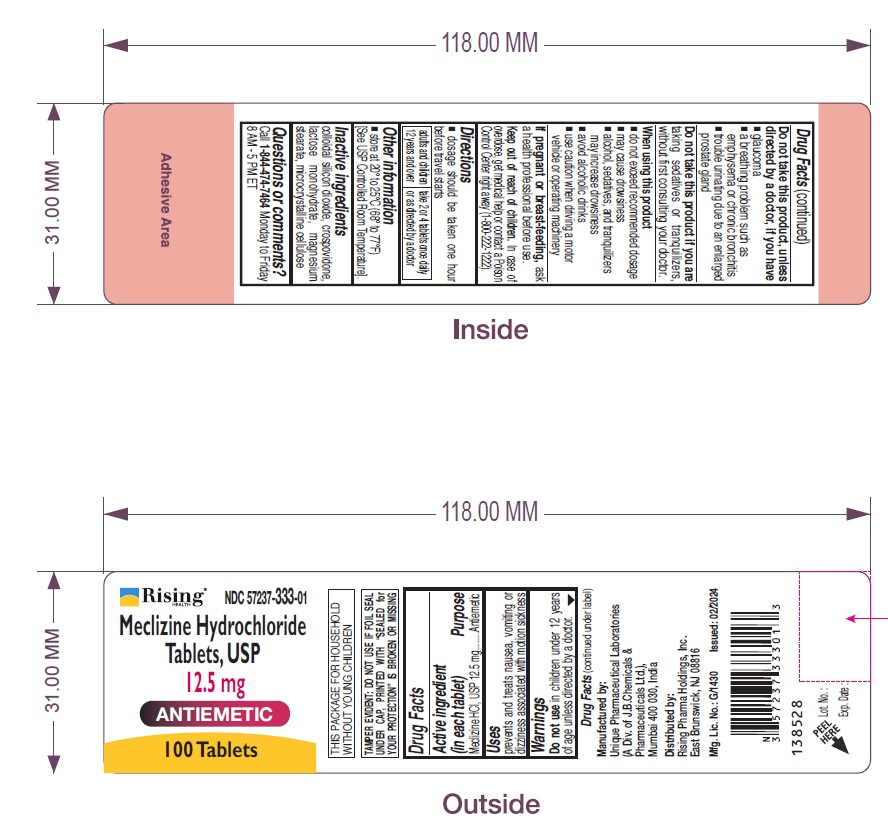
Do not take this product, unless directed by a doctor, if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Purpose
Antiemetic
Description
Meclizine HCl, USP 12.5 mg
Uses
prevents and treats nausea, vomiting or dizziness associated with motion sickness.
Section 42229-5
Manufactured by:
Unique Pharmaceutical Laboratories
(A Div. of J.B.Chemicals &
Pharmaceuticals Ltd.),
Mumbai 400 030, India
Mfg. Lic. No.: G/1430
Distributed by:
Rising Pharma Holdings, Inc.
East Brunswick, NJ 08816
Issued: 02/2024
138528
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Warnings
Do not use in children under 12 years of age unless directed by a doctor.
Directions
dosage should be taken one hour before travel starts
| adults and children 12 years and over | take 2 or 4 tablets once daily or as directed by a doctor |
Drug Facts
Meclizine HCl, USP 12.5 mg
Other Information
- store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Inactive Ingredients
colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate, microcrystalline cellulose
Questions Or Comments?
Call 1-844-474-7464 Monday to Friday 8 AM - 5 PM ET
When Using This Product
- do not exceed recommended dosage
- may cause drowsiness
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- use caution when driving a motor vehicle or operating machinery
Principal Display Panel 12.5 Mg Tablets Container Label
NDC 57237-333-01
Meclizine Hydrochloride Tablets, USP
12.5 mg
100 Tablets
Structured Label Content
Uses
prevents and treats nausea, vomiting or dizziness associated with motion sickness.
Warnings and Precautions (34071-1)
Do not take this product, unless directed by a doctor, if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Section 42229-5 (42229-5)
Manufactured by:
Unique Pharmaceutical Laboratories
(A Div. of J.B.Chemicals &
Pharmaceuticals Ltd.),
Mumbai 400 030, India
Mfg. Lic. No.: G/1430
Distributed by:
Rising Pharma Holdings, Inc.
East Brunswick, NJ 08816
Issued: 02/2024
138528
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Antiemetic
Warnings
Do not use in children under 12 years of age unless directed by a doctor.
Directions
dosage should be taken one hour before travel starts
| adults and children 12 years and over | take 2 or 4 tablets once daily or as directed by a doctor |
Drug Facts
Meclizine HCl, USP 12.5 mg
Other Information (Other information)
- store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Inactive Ingredients (Inactive ingredients)
colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate, microcrystalline cellulose
Questions Or Comments? (Questions or comments?)
Call 1-844-474-7464 Monday to Friday 8 AM - 5 PM ET
When Using This Product (When using this product)
- do not exceed recommended dosage
- may cause drowsiness
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- use caution when driving a motor vehicle or operating machinery
Principal Display Panel 12.5 Mg Tablets Container Label (PRINCIPAL DISPLAY PANEL - 12.5 mg tablets container label)
NDC 57237-333-01
Meclizine Hydrochloride Tablets, USP
12.5 mg
100 Tablets
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:26.687510 · Updated: 2026-03-14T23:05:11.503942