Simvastatin Tablets
acaaab63-c9ff-44c9-8fde-9a4ea6454dd7
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Simvastatin is indicated: To reduce the risk of total mortality by reducing risk of coronary heart disease death, non-fatal myocardial infarction and stroke, and the need for coronary and non-coronary revascularization procedures in adults with established coronary heart disease, cerebrovascular disease, peripheral vascular disease, and/or diabetes, who are at high risk of coronary heart disease events. As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C): In adults with primary hyperlipidemia. In adults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH). As an adjunct to other LDL-C-lowering therapies to reduce LDL-C in adults with homozygous familial hypercholesterol emia (HoFH). As an adjunct to diet for the treatment of adults with: Primary dysbetalipoproteinemia. Hypertriglyceridemia.
Dosage and Administration
Important Dosage and Administration Information: ( 2.1 ) For dosing with the 5 mg strength, use another simvastatin product. Take simvastatin orally once daily in the evening. Maximum recommended dosage is simvastatin 40 mg once daily. An 80 mg daily dosage of simvastatin is restricted to patients who have been taking simvastatin 80 mg daily chronically (e.g., for 12 months or more) without evidence of muscle toxicity. For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving simvastatin 40 mg daily, prescribe alternative LDL-C lowering treatment. Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating, and adjust the dosage if necessary. Adults: Recommended dosage is 20 mg to 40 mg once daily. ( 2.2 ) Pediatric Patients Aged 10 Years and Older with HeFH: Recommended dosage is 10 mg to 40 mg once daily. ( 2.3 ) Patients with Severe Renal Impairment: Recommended starting dosage is simvastatin 5 mg once daily. ( 2.4 , 8.6 ) See full prescribing information for simvastatin dosage modifications due to drug interactions. ( 2.5 )
Contraindications
Simvastatin is contraindicated in the following conditions: Concomitant use of strong CYP3A4 inhibitors (select azole anti-fungals, macrolide antibiotics, anti-viral medications, and nefazodone) [see Drug Interactions (7.1) ] . Concomitant use of cyclosporine, danazol or gemfibrozil [see Drug Interactions (7.1) ] . Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3) ] . Hypersensitivity to simvastatin or any excipients in Simvastatin Tablets. Hypersensitivity reactions, including anaphylaxis, angioedema and Stevens-Johnson syndrome, have been reported [see Adverse Reactions (6.2) ] .
Warnings and Precautions
Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher simvastatin dosage. Chinese patients may be at higher risk for myopathy. Discontinue simvastatin if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue simvastatin in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing simvastatin dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever. ( 5.1 , 7.1 , 8.5 , 8.6 , 8.8 ) Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue simvastatin if IMNM is suspected. ( 5.2 ) Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzyme before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue simvastatin. ( 4 , 5.3 , 8.7 )
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1) ] Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2) ] Hepatic Dysfunction [see Warnings and Precautions (5.3) ] Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.4) ]
Drug Interactions
Concomitant use of simvastatin with the following drugs requires dosage modification of simvastatin [see Warnings and Precautions (5.1) and Drug Interactions (7.1) ] .
How Supplied
Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side. They are supplied as follows: NDC 69367-374-09 unit of use bottles of 90. NDC 69367-374-10 bottles of 1000. Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side. They are supplied as follows: NDC 69367-375-09 unit of use bottles of 90. NDC 69367-375-10 bottles of 1000. Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side. They are supplied as follows: NDC 69367-376-09 unit of use bottles of 90. NDC 69367-376-10 bottles of 1000. Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side. They are supplied as follows: NDC 69367-377-09 unit of use bottles of 90. NDC 69367-377-10 bottles of 1000.
Storage and Handling
Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side. They are supplied as follows: NDC 69367-374-09 unit of use bottles of 90. NDC 69367-374-10 bottles of 1000. Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side. They are supplied as follows: NDC 69367-375-09 unit of use bottles of 90. NDC 69367-375-10 bottles of 1000. Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side. They are supplied as follows: NDC 69367-376-09 unit of use bottles of 90. NDC 69367-376-10 bottles of 1000. Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side. They are supplied as follows: NDC 69367-377-09 unit of use bottles of 90. NDC 69367-377-10 bottles of 1000.
Description
Dosage and Administration ( 2.1 ) 03/2023
Medication Information
Warnings and Precautions
Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher simvastatin dosage. Chinese patients may be at higher risk for myopathy. Discontinue simvastatin if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue simvastatin in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing simvastatin dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever. ( 5.1 , 7.1 , 8.5 , 8.6 , 8.8 ) Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue simvastatin if IMNM is suspected. ( 5.2 ) Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzyme before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue simvastatin. ( 4 , 5.3 , 8.7 )
Indications and Usage
Simvastatin is indicated: To reduce the risk of total mortality by reducing risk of coronary heart disease death, non-fatal myocardial infarction and stroke, and the need for coronary and non-coronary revascularization procedures in adults with established coronary heart disease, cerebrovascular disease, peripheral vascular disease, and/or diabetes, who are at high risk of coronary heart disease events. As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C): In adults with primary hyperlipidemia. In adults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH). As an adjunct to other LDL-C-lowering therapies to reduce LDL-C in adults with homozygous familial hypercholesterol emia (HoFH). As an adjunct to diet for the treatment of adults with: Primary dysbetalipoproteinemia. Hypertriglyceridemia.
Dosage and Administration
Important Dosage and Administration Information: ( 2.1 ) For dosing with the 5 mg strength, use another simvastatin product. Take simvastatin orally once daily in the evening. Maximum recommended dosage is simvastatin 40 mg once daily. An 80 mg daily dosage of simvastatin is restricted to patients who have been taking simvastatin 80 mg daily chronically (e.g., for 12 months or more) without evidence of muscle toxicity. For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving simvastatin 40 mg daily, prescribe alternative LDL-C lowering treatment. Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating, and adjust the dosage if necessary. Adults: Recommended dosage is 20 mg to 40 mg once daily. ( 2.2 ) Pediatric Patients Aged 10 Years and Older with HeFH: Recommended dosage is 10 mg to 40 mg once daily. ( 2.3 ) Patients with Severe Renal Impairment: Recommended starting dosage is simvastatin 5 mg once daily. ( 2.4 , 8.6 ) See full prescribing information for simvastatin dosage modifications due to drug interactions. ( 2.5 )
Contraindications
Simvastatin is contraindicated in the following conditions: Concomitant use of strong CYP3A4 inhibitors (select azole anti-fungals, macrolide antibiotics, anti-viral medications, and nefazodone) [see Drug Interactions (7.1) ] . Concomitant use of cyclosporine, danazol or gemfibrozil [see Drug Interactions (7.1) ] . Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3) ] . Hypersensitivity to simvastatin or any excipients in Simvastatin Tablets. Hypersensitivity reactions, including anaphylaxis, angioedema and Stevens-Johnson syndrome, have been reported [see Adverse Reactions (6.2) ] .
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1) ] Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2) ] Hepatic Dysfunction [see Warnings and Precautions (5.3) ] Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.4) ]
Drug Interactions
Concomitant use of simvastatin with the following drugs requires dosage modification of simvastatin [see Warnings and Precautions (5.1) and Drug Interactions (7.1) ] .
Storage and Handling
Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side. They are supplied as follows: NDC 69367-374-09 unit of use bottles of 90. NDC 69367-374-10 bottles of 1000. Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side. They are supplied as follows: NDC 69367-375-09 unit of use bottles of 90. NDC 69367-375-10 bottles of 1000. Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side. They are supplied as follows: NDC 69367-376-09 unit of use bottles of 90. NDC 69367-376-10 bottles of 1000. Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side. They are supplied as follows: NDC 69367-377-09 unit of use bottles of 90. NDC 69367-377-10 bottles of 1000.
How Supplied
Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side. They are supplied as follows: NDC 69367-374-09 unit of use bottles of 90. NDC 69367-374-10 bottles of 1000. Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side. They are supplied as follows: NDC 69367-375-09 unit of use bottles of 90. NDC 69367-375-10 bottles of 1000. Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side. They are supplied as follows: NDC 69367-376-09 unit of use bottles of 90. NDC 69367-376-10 bottles of 1000. Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side. They are supplied as follows: NDC 69367-377-09 unit of use bottles of 90. NDC 69367-377-10 bottles of 1000.
Description
Dosage and Administration ( 2.1 ) 03/2023
Section 42229-5
Patients taking Lomitapide
Reduce the dosage of simvastatin by 50%. Do not exceed simvastatin 20 mg once daily (or 40 mg once daily for patients who have previously taken simvastatin 80 mg daily chronically while taking lomitapide) [see Dosage and Administration (2.1)].
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 01/2025 |
|
PATIENT INFORMATION
Simvastatin (sim" va stat' in) tablets, for oral use |
|
| Read this Patient Information before you start taking simvastatin tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is Simvastatin Tablets?
Simvastatin tablets are prescription medicine that contains the cholesterol lowering medicine, simvastatin Simvastatin tablets are used to lower:
The safety and effectiveness of simvastatin tablets have not been established in children younger than 10 years of age with heterozygous familial hypercholesterolemia (HeFH) or other types of hyperlipidemia (high levels of fat in the blood). |
|
Do not take Simvastatin tablets if you:
|
|
Before you take simvastatin tablets, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Talk to your healthcare provider before you start taking any new medicines. Tell your healthcare provider who prescribes simvastatin tablets if another healthcare provider increases the dose of another medicine you are taking. Simvastatin tablets may affect the way other medicines work, and other medicines may affect how simvastatin tablets work. Especially tell your healthcare provider if you take:
|
|
How should I take simvastatin tablets?
|
|
|
What are the possible side effects of simvastatin tablets? Simvastatin tablets may cause serious side effects including:
These are not all the possible side effects of simvastatin tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store simvastatin tablets?
|
|
|
General information about safe and effective use of simvastatin tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use simvastatin tablets for a condition for which it was not prescribed. Do not give simvastatin tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about simvastatin tablets that is written for health professionals. |
|
|
What are the ingredients in simvastatin tablets?
Active ingredient(s): simvastatin. Inactive ingredient(s): lactose monohydrate, pregelatinized starch, microcrystalline cellulose, ethanol, ascorbic acid, citric acid and magnesium stearate. Butylated hydroxyanisole is added as a preservative. |
|
| Manufacturer: Yiling Pharmaceutical Ltd No.36 Zhujiang Road, Shijiazhuang, 050035, China. Distributor: Westminster Pharmaceuticals, LLC 1321 Murfreesboro Pike, Ste 607, Nashville, TN 37217, USA |
Section 43683-2
| Dosage and Administration (2.1) | 03/2023 |
Section 44425-7
Storage
Store at 20-25°C (68-77°F) [see USP Controlled Room Temperature].
10 Overdosage
No specific antidotes for simvastatin are known. Contact Poison Control (1-800-222-1222) for latest recommendations.
11 Description
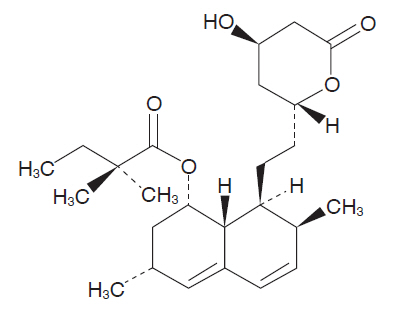
Simvastatin is a prodrug of 3-hydoroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor that is derived synthetically from a fermentation product of Aspergillus terreus.
Simvastatin is butanoic acid, 2,2-dimethyl-,1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-(tetra- hydro-4hydroxy-6-oxo-2H-pyran-2-yl)-ethyl]-1-naphthalenyl ester, [1S-[1α,3α,7β,8β(2S*,4S*),-8aβ]]. The empirical formula of simvastatin is C25H38O5 and its molecular weight is 418.57. Its structural formula is:
Simvastatin is a white to off-white, nonhygroscopic, crystalline powder that is practically insoluble in water, and freely soluble in chloroform, methanol and ethanol.
Simvastatin Tablets, USP for oral use contain 10 mg, 20 mg, 40 mg or 80 mg of simvastatin and the following inactive ingredients: anhydrous citric acid, ascorbic acid, butylated hydroxyanisole, ferric oxide red (iron oxide red), ferric oxide yellow (iron oxide yellow), ferrosoferric oxide (10 mg and 40 mg), hypromellose, lactose monohydrate, magnesium stearate, maltodextrin (10 mg and 80 mg), medium-chain triglycerides (10 mg and 80 mg), microcrystalline cellulose, polydextrose (10 mg and 80 mg), polyethylene glycol (macrogol/peg) (20 mg), pregelatinized starch (maize), (10 mg, 40 mg and 80 mg) and titanium dioxide.
FDA approved dissolution test specifications differ from USP.
8.4 Pediatric Use
The safety and effectiveness of simvastatin as an adjunct to diet to reduce LDL-C have been established in pediatric patients 10 years of age and older with HeFH. Use of simvastatin for this indication is based on a double-blind, placebo-controlled clinical study in 175 pediatric patients (99 boys and 76 girls at least 1 year post-menarche) 10 years of age and older with HeFH. In this limited controlled study, there was no significant effect on growth or sexual maturation in the boys or girls, or on menstrual cycle length in girls.
The safety and effectiveness of simvastatin have not been established in pediatric patients younger than 10 years of age with HeFH or in pediatric patients with other types of hyperlipidemia (other than HeFH).
8.5 Geriatric Use
Of the total number of simvastatin-treated patients in clinical studies 1,021 (23%) patients, 5,366 (52%) patients, and 363 (15%) patients were ≥65 years old, respectively. In Study HPS, 615 (6%) patients were ≥75 years old [see Clinical Studies (14)]. In a clinical study of patients treated with simvastatin 80 mg daily, patients ≥65 years of age had an increased risk of myopathy, including rhabdomyolysis, compared to patients <65 years of age.
A pharmacokinetic study with simvastatin use showed the mean plasma level of total inhibitors to be approximately 45% higher in geriatric patients between 70-78 years of age compared with patients between 18-30 years of age [see Clinical Pharmacology (12.3)].
Advanced age (≥65 years) is a risk factor for simvastatin-associated myopathy and rhabdomyolysis. Dose selection for an elderly patient should be cautious, recognizing the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy and the higher risk of myopathy. Monitor geriatric patients receiving simvastatin for the increased risk of myopathy [see Warnings and Precautions (5.1)].
4 Contraindications
Simvastatin is contraindicated in the following conditions:
- Concomitant use of strong CYP3A4 inhibitors (select azole anti-fungals, macrolide antibiotics, anti-viral medications, and nefazodone) [see Drug Interactions (7.1)].
- Concomitant use of cyclosporine, danazol or gemfibrozil [see Drug Interactions (7.1)].
- Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3)].
- Hypersensitivity to simvastatin or any excipients in Simvastatin Tablets. Hypersensitivity reactions, including anaphylaxis, angioedema and Stevens-Johnson syndrome, have been reported [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling:
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1)]
- Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2)]
- Hepatic Dysfunction [see Warnings and Precautions (5.3)]
- Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.4)]
7 Drug Interactions
- See full prescribing information for details regarding concomitant use of simvastatin with other drugs or grapefruit juice that increase the risk of myopathy and rhabdomyolysis. (2.5, 7.1)
- Coumarin Anticoagulants: Obtain INR before simvastatin initiation and monitor INR during simvastatin dosage initiation or adjustment. (7.2)
- Digoxin: During simvastatin initiation, monitor digoxin levels. (7.2)
8.6 Renal Impairment
Renal impairment is a risk factor for myopathy and rhabdomyolysis. Monitor all patients with renal impairment for development of myopathy. In patients with severe renal impairment (CLcr 15 – 29 mL/min), the recommended starting dosage is simvastatin 5 mg once daily [see Dosage and Administration (2.4), Warnings and Precautions (5.1)].
8.8 Chinese Patients
In a clinical study in which patients at high risk of CVD were treated with simvastatin 40 mg/day (median follow-up 3.9 years), the incidence of myopathy was approximately 0.05% for non-Chinese patients (n=7367) compared with 0.24% for Chinese patients (n=5468). In this study, the incidence of myopathy for Chinese patients on simvastatin 40 mg/day or ezetimibe/simvastatin 10/40 mg/day coadministered with extended-release niacin 2 g/day was 1.24%.
Chinese patients may be at higher risk for myopathy, monitor these patients appropriately. Coadministration of simvastatin with lipid-modifying doses of niacin-containing products (≥1 g/day niacin) is not recommended in Chinese patients [see Warnings and Precautions (5.1), Drug Interactions (7.1)].
12.2 Pharmacodynamics
Inhibition of HMG-CoA reductase by simvastatin acid accelerates the expression of LDL-receptors, followed by the uptake of LDL-C from blood to the liver, leading to a decrease in plasma LDL-C and total cholesterol. Sustained inhibition of cholesterol synthesis in the liver also decreases levels of very-lowdensity lipoproteins. The maximum LDL-C reduction of simvastatin is usually achieved by 4 weeks and is maintained after that.
12.3 Pharmacokinetics
Simvastatin is a lactone that is readily hydrolyzed in vivo to the corresponding β-hydroxyacid. Pharmacokinetics (PK) of simvastatin and its metabolites was originally characterized using inhibition of HMG-CoA reductase activity following base hydrolysis of plasma samples, as specific bioanalytical methods were not available. Inhibition of the enzyme activity (equivalent to the level of total inhibitors) represented the combination of activities in plasma following administration of simvastatin from both active (simvastatin acid and its metabolites) and latent forms (simvastatin and its metabolites) after conversion to the active forms in the presence of base.
8.7 Hepatic Impairment
Simvastatin is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4), Warnings and Precautions (5.3)].
1 Indications and Usage
Simvastatin is indicated:
- To reduce the risk of total mortality by reducing risk of coronary heart disease death, non-fatal myocardial infarction and stroke, and the need for coronary and non-coronary revascularization procedures in adults with established coronary heart disease, cerebrovascular disease, peripheral vascular disease, and/or diabetes, who are at high risk of coronary heart disease events.
- As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C):
- In adults with primary hyperlipidemia.
- In adults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH).
- As an adjunct to other LDL-C-lowering therapies to reduce LDL-C in adults with homozygous familial hypercholesterol emia (HoFH).
- As an adjunct to diet for the treatment of adults with:
- Primary dysbetalipoproteinemia.
- Hypertriglyceridemia.
5.3 Hepatic Dysfunction
Increases in serum transaminases have been reported with use of simvastatin [see Adverse Reactions (6.1)]. In most cases, these changes appeared soon after initiation, were transient, were not accompanied by symptoms, and resolved or improved on continued therapy or after a brief interruption in therapy. Persistent increases to more than 3×ULN in serum transaminases have occurred in approximately 1% of patients receiving simvastatin in clinical studies. Marked persistent increases of hepatic transaminases have also occurred with simvastatin. There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking statins, including simvastatin.
Patients who consume substantial quantities of alcohol and/or have a history of liver disease may be at increased risk for hepatic injury.
Consider liver enzyme testing before simvastatin initiation and when clinically indicated thereafter. Simvastatin is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)]. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue simvastatin.
12.1 Mechanism of Action
Simvastatin is a prodrug and is hydrolyzed to its active β-hydroxyacid form, simvastatin acid, after administration. Simvastatin acid and its metabolites are inhibitors of HMG-CoA reductase, the rate-limiting enzyme that converts HMG-CoA to mevalonate, a precursor of cholesterol.
5 Warnings and Precautions
- Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher simvastatin dosage. Chinese patients may be at higher risk for myopathy. Discontinue simvastatin if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue simvastatin in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing simvastatin dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever. (5.1, 7.1, 8.5, 8.6, 8.8)
- Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue simvastatin if IMNM is suspected. (5.2)
- Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzyme before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue simvastatin. (4, 5.3, 8.7)
2 Dosage and Administration
-
Important Dosage and Administration Information: (2.1)
- For dosing with the 5 mg strength, use another simvastatin product.
- Take simvastatin orally once daily in the evening.
- Maximum recommended dosage is simvastatin 40 mg once daily. An 80 mg daily dosage of simvastatin is restricted to patients who have been taking simvastatin 80 mg daily chronically (e.g., for 12 months or more) without evidence of muscle toxicity.
- For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving simvastatin 40 mg daily, prescribe alternative LDL-C lowering treatment.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating, and adjust the dosage if necessary.
- Adults: Recommended dosage is 20 mg to 40 mg once daily. (2.2)
- Pediatric Patients Aged 10 Years and Older with HeFH: Recommended dosage is 10 mg to 40 mg once daily. (2.3)
- Patients with Severe Renal Impairment: Recommended starting dosage is simvastatin 5 mg once daily. (2.4, 8.6)
- See full prescribing information for simvastatin dosage modifications due to drug interactions. (2.5)
3 Dosage Forms and Strengths
- Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side.
- Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side.
- Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side.
- Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of simvastatin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as whole: fever, chills, malaise, asthenia
Blood and Lymphatic System Disorders: anemia, thrombocytopenia, leukopenia, hemolytic anemia, positive ANA, ESR increase, eosinophilia
Gastrointestinal Disorders: pancreatitis, vomiting
Hepatic and Pancreatic Disorders: hepatitis/jaundice, fatal and non-fatal hepatic failure
Immune System Disorders: hypersensitivity syndrome including: anaphylaxis, angioedema, lupus erythematous-like syndrome, dermatomyositis, vasculitis
Musculoskeletal and Connective Tissue Disorders: muscle cramps, immune-mediated necrotizing myopathy, polymyalgia rheumatica, arthritis
Nervous System Disorders: dizziness, depression, paresthesia, peripheral neuropathy, Rare reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. Cognitive impairment was generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks).There have been rare reports of new-onset or exacerbation of myasthenia gravis, including ocular myasthenia, and reports of recurrence when the same or a different statin was administered.
Skin and Subcutaneous Tissue Disorders: pruritus, alopecia, a variety of skin changes (e.g., nodules, discoloration, dryness of skin/mucous membranes, changes to hair/nails), purpura, lichen planus, urticaria, photosensitivity, flushing, toxic epidermal necrolysis, erythema multiforme, including Stevens-Johnson syndrome
Respiratory and Thoracic: interstitial lung disease, dyspnea
Reproductive System Disorders: erectile dysfunction
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
In clinical studies, 2,423 adult patients were exposed to simvastatin with a median duration of follow-up of approximately 18 months. The most commonly reported adverse reactions (incidence ≥5%) in these simvastatin clinical studies were: upper respiratory infections (9%), headache (7%), abdominal pain (7%), constipation (7%), and nausea (5%). Overall, 1.4% of patients discontinued simvastatin due to adverse reactions. The most common adverse reactions that led to discontinuation were: gastrointestinal disorders (0.5%), myalgia (0.1%), and arthralgia (0.1%).
In a Cardiovascular Outcomes Study (the Scandinavian Simvastatin Survival Study [Study 4S]), adult patients (age range 35-71 years, 19% women, 100% Caucasians) were treated with 20-40 mg per day of simvastatin or placebo over a median of 5.4 years [see Clinical Studies (14)]; adverse reactions reported in ≥2% of patients and at a rate greater than placebo are shown in Table 1.
| %Placebo (N=2,223) |
%Simvastatin (N=2,221) |
|
|---|---|---|
| Bronchitis | 6.3 | 6.6 |
| Abdominal pain | 5.8 | 5.9 |
| Atrial fibrillation | 5.1 | 5.7 |
| Gastritis | 3.9 | 4.9 |
| Eczema | 3.0 | 4.5 |
| Vertigo | 4.2 | 4.5 |
| Diabetes mellitus | 3.6 | 4.2 |
| Insomnia | 3.8 | 4.0 |
| Myalgia | 3.2 | 3.7 |
| Urinary tract infection | 3.1 | 3.2 |
| Edema/swelling | 2.3 | 2.7 |
| Headache | 2.1 | 2.5 |
| Sinusitis | 1.8 | 2.3 |
| Constipation | 1.6 | 2.2 |
5.1 Myopathy and Rhabdomyolysis
Simvastatin may cause myopathy and rhabdomyolysis. Acute kidney injury secondary to myoglobinuria and rare fatalities have occurred as a result of rhabdomyolysis in patients treated with statins, including simvastatin.
In clinical studies of 24,747 simvastatin-treated patients with a median follow-up of 4 years, the incidence of myopathy, defined as unexplained muscle weakness, pain, or tenderness accompanied by creatinine kinase (CK) increases greater than ten times the upper limit of normal (10×ULN), were approximately 0.03%, 0.08%, and 0.61% in patients treated with simvastatin 20 mg, 40 mg, and 80 mg daily, respectively. In another clinical study of 12,064 simvastatin-treated patients (with a history of myocardial infarction) with a mean follow-up of 6.7 years, the incidences of myopathy in patients taking simvastatin 20 mg and 80 mg daily were approximately 0.02% and 0.9%, respectively. The incidences of rhabdomyolysis (defined as myopathy with a CK >40×ULN) in patients taking simvastatin 20 mg and 80 mg daily were approximately 0% and 0.4%, respectively [see Adverse Reactions (6.1)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side. They are supplied as follows:
NDC 69367-374-09 unit of use bottles of 90.
NDC 69367-374-10 bottles of 1000.
Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side. They are supplied as follows:
NDC 69367-375-09 unit of use bottles of 90.
NDC 69367-375-10 bottles of 1000.
Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side. They are supplied as follows:
NDC 69367-376-09 unit of use bottles of 90.
NDC 69367-376-10 bottles of 1000.
Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side. They are supplied as follows:
NDC 69367-377-09 unit of use bottles of 90.
NDC 69367-377-10 bottles of 1000.
7.2 Simvastatin Effects On Other Drugs
Table 3 presents Simvastatin's effect on other drugs and instructions for preventing or managing them.
| Coumarin Anticoagulants | |
|---|---|
| Clinical Impact: | Simvastatin may potentiate the effect of coumarin anticoagulants and increase the INR. The concomitant use of simvastatin (20 to 40 mg) and coumarin anticoagulants increased the INR from a baseline of 1.7 to 1.8 in healthy subjects and from 2.6 to 3.4 in patients with hyperlipidemia. There are postmarketing reports of clinically evident bleeding and/or increased INR in patients taking concomitant statins and warfarin. |
| Intervention: | In patients taking coumarin anticoagulants, obtain an INR before starting simvastatin and frequently enough after initiation, dose titration, or discontinuation to ensure that no significant alteration in INR occurs. Once the INR is stable, monitor INR at regularly recommended intervals. |
| Digoxin | |
| Clinical Impact: | Concomitant use of digoxin with simvastatin may result in elevated plasma digoxin concentrations [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor digoxin levels in patients taking digoxin when simvastatin is initiated. |
2.2 Recommended Dosage in Adult Patients
The recommended dosage range of simvastatin is 20 mg to 40 mg once daily.
5.2 Immune Mediated Necrotizing Myopathy
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use, including reports of recurrence when the same or a different statin was administered. IMNM is characterized by proximal muscle weakness and elevated serum creatine kinase that persist despite discontinuation of statin treatment; positive anti-HMG CoA reductase antibody; muscle biopsy showing necrotizing myopathy without significant inflammation; and improvement with immunosuppressive agents. Additional neuromuscular and serologic testing may be necessary. Treatment with immunosuppressive agents may be required. Discontinue simvastatin if IMNM is suspected.
2.5 Dosage Modifications Due to Drug Interactions
Concomitant use of simvastatin with the following drugs requires dosage modification of simvastatin [see Warnings and Precautions (5.1) and Drug Interactions (7.1)].
2.1 Important Dosage and Administration Information
- For dosing with the 5 mg strength, use another simvastatin product.
- Take simvastatin orally once daily in the evening.
- The maximum recommended dosage is simvastatin 40 mg once daily [see Dosage and Administration (2.2, 2.3)]. The simvastatin 80 mg daily dosage is restricted to patients who have been taking simvastatin 80 mg daily chronically (e.g., for 12 months or more) without evidence of muscle toxicity [see Warnings and Precautions (5.1)].
- If as dose is missed, take the missed dose as soon as possible. Do not double the next dose.
- For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving simvastatin 40 mg daily, prescribe alternative LDL-C-lowering treatment.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating simvastatin, and adjust the dosage if necessary.
Principal Display Panel 10 Mg Tablet Bottle Label
NDC 69367-374-10
Rx Only
Simvastatin
Tablets, USP
10 mg
1000 Tablets
Westminster
Pharmaceuticals
Principal Display Panel 20 Mg Tablet Bottle Label
NDC 69367-375-10
Rx Only
Simvastatin
Tablets, USP
20 mg
1000 Tablets
Westminster
Pharmaceuticals
Principal Display Panel 40 Mg Tablet Bottle Label
NDC 69367-376-10
Rx Only
Simvastatin
Tablets, USP
40 mg
1000 Tablets
Westminster
Pharmaceuticals
Principal Display Panel 80 Mg Tablet Bottle Label
NDC 69367-377-10
Rx Only
Simvastatin
Tablets, USP
80 mg
1000 Tablets
Westminster
Pharmaceuticals
5.4 Increases in Hba1c and Fasting Serum Glucose Levels
Increases in HbA1c and fasting serum glucose levels have been reported with statins, including simvastatin. Optimize lifestyle measures, including regular exercise, maintaining a healthy body weight, and making healthy food choices.
2.4 Recommended Dosage in Patients With Renal Impairment
For patients with severe renal impairment [creatinine clearance (CLcr) 15 – 29 mL/min], the recommended starting dosage of simvastatin is 5 mg once daily [see Warnings and Precautions (5.1) and Use in Specific Populations (8.6)]. Use another simvastatin product to initiate dosing in such patients [see Dosage and Administration (2.1)].
There are no dosage adjustment recommendations for patients with mild or moderate renal impairment.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 72-week carcinogenicity study, mice were administered daily doses of simvastatin of 25, 100, and 400 mg/kg body weight, which resulted in mean plasma drug levels approximately 1, 4, and 8 times higher than the mean human plasma drug level, respectively (as total inhibitory activity based on AUC) after an 80-mg oral dose. Liver carcinomas were significantly increased in high-dose females and mid- and high- dose males with a maximum incidence of 90% in males. The incidence of adenomas of the liver was significantly increased in mid- and high-dose females. Drug treatment also significantly increased the incidence of lung adenomas in mid- and high-dose males and females. Adenomas of the Harderian gland (a gland of the eye of rodents) were significantly higher in high-dose mice than in controls. No evidence of a tumorigenic effect was observed at 25 mg/kg/day.
In a separate 92-week carcinogenicity study in mice at doses up to 25 mg/kg/day, no evidence of a tumorigenic effect was observed (mean plasma drug levels were 1 times higher than humans given 80 mg simvastatin as measured by AUC).
In a two-year study in rats at 25 mg/kg/day, there was a statistically significant increase in the incidence of thyroid follicular adenomas in female rats exposed to approximately 11 times higher levels of simvastatin than in humans given 80 mg simvastatin (as measured by AUC).
A second two-year rat carcinogenicity study with doses of 50 and 100 mg/kg/day produced hepatocellular adenomas and carcinomas (in female rats at both doses and in males at 100 mg/kg/day). Thyroid follicular cell adenomas were increased in males and females at both doses; thyroid follicular cell carcinomas were increased in females at 100 mg/kg/day. The increased incidence of thyroid neoplasms appears to be consistent with findings from other statins. These treatment levels represented plasma drug levels (AUC) of approximately 7 and 15 times (males) and 22 and 25 times (females) the mean human plasma drug exposure after an 80 milligram daily dose.
No evidence of mutagenicity was observed in a microbial mutagenicity (Ames) test with or without rat or mouse liver metabolic activation. In addition, no evidence of damage to genetic material was noted in an in vitro alkaline elution assay using rat hepatocytes, a V-79 mammalian cell forward mutation study, an in vitro chromosome aberration study in CHO cells, or an in vivo chromosomal aberration assay in mouse bone marrow.
There was decreased fertility in male rats treated with simvastatin for 34 weeks at 25 mg/kg body weight (4 times the maximum human exposure level, based on AUC, in patients receiving 80 mg/day); however, this effect was not observed during a subsequent fertility study in which simvastatin was administered at this same dose level to male rats for 11 weeks (the entire cycle of spermatogenesis including epididymal maturation). No microscopic changes were observed in the testes of rats from either study. At 180 mg/kg/day, (which produces exposure levels 22 times higher than those in humans taking 80 mg/day based on surface area, mg/m2), seminiferous tubule degeneration (necrosis and loss of spermatogenic epithelium) was observed. In dogs, there was drug-related testicular atrophy, decreased spermatogenesis, spermatocytic degeneration and giant cell formation at 10 mg/kg/day, (approximately 2 times the human exposure, based on AUC, at 80 mg/day). The clinical significance of these findings is unclear.
2.3 Recommended Dosage in Pediatric Patients 10 Years of Age and Older With Hefh
The recommended dosage range of simvastatin is 10 mg to 40 mg daily.
7.1 Drug Interactions That Increase the Risk of Myopathy and Rhabdomyolysis With Simvastatin
Simvastatin is a substrate of CYP3A4 and of the transport protein OATP1B1. Simvastatin exposure can be significantly increased with concomitant administration of inhibitors of CYP3A4 and OATP1B1. Table 2 includes a list of drugs that increase the risk of myopathy and rhabdomyolysis when used concomitantly with simvastatin and instructions for preventing or managing them [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
| Strong CYP3A4 inhibitors | |
|---|---|
| Clinical Impact: | Simvastatin is a substrate of CYP3A4. Concomitant use of strong CYP3A4 inhibitors with simvastatin increases simvastatin exposure and increases the risk of myopathy and rhabdomyolysis, particularly with higher simvastatin dosages. |
| Intervention: | Concomitant use of strong CYP3A4 inhibitors with simvastatin is contraindicated [see Contraindications (4)]. If treatment with a CYP3A4 inhibitor is unavoidable, suspend simvastatin during the course of strong CYP3A4 inhibitor treatment. |
| Examples: | Select azole anti-fungals (e.g., itraconazole, ketoconazole, posaconazole, and voriconazole), select macrolide antibiotics (e.g., erythromycin and clarithromycin), select HIV protease inhibitors (e.g., nelfinavir, ritonavir, and darunavir/ritonavir), select HCV protease inhibitors (e.g., boceprevir and telaprevir), cobicistat-containing products, and nefazodone. |
| Cyclosporine, Danazol, or Gemfibrozil | |
| Clinical Impact: | The risk of myopathy and rhabdomyolysis is increased with concomitant use of cyclosporine, danazol, or gemfibrozil with simvastatin. Gemfibrozil may cause myopathy when given alone. |
| Intervention: | Concomitant use of cyclosporine, danazol, or gemfibrozil with simvastatin is contraindicated [see Contraindications (4)]. |
| Amiodarone, Dronedarone, Ranolazine, or Calcium Channel Blockers | |
| Clinical Impact: | The risk of myopathy and rhabdomyolysis is increased by concomitant use of amiodarone, dronedarone, ranolazine, or calcium channel blockers with simvastatin. |
| Intervention: | For patients taking verapamil, diltiazem, or dronedarone, do not exceed simvastatin 10 mg daily. For patients taking amiodarone, amlodipine, or ranolazine, do not exceed simvastatin 20 mg daily [see Dosage and Administration (2.5)]. |
| Lomitapide | |
| Clinical Impact: | Simvastatin exposure is approximately doubled with concomitant use of lomitapide and the risk of myopathy and rhabdomyolysis is increased. |
| Intervention: | Reduce the dose of simvastatin by 50% if initiating lomitapide. Do not exceed simvastatin 20 mg daily (or simvastatin 40 mg daily for patients who have previously taken simvastatin 80 mg daily chronically) while taking lomitapide [see Dosage and Administration (2.1,2.5)]. |
| Daptomycin | |
| Clinical Impact: | Cases of rhabdomyolysis have been reported with simvastatin administered with daptomycin. Both simvastatin and daptomycin can cause myopathy and rhabdomyolysis when given alone and the risk of myopathy and rhabdomyolysis may be increased by coadministration. |
| Intervention: | If treatment with daptomycin is required, consider temporarily suspending simvastatin during the course of daptomycin treatment. |
| Niacin | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been observed with concomitant use of lipid modifying dosages of niacin-containing products (≥1 gram/day niacin) with simvastatin. The risk of myopathy is greater in Chinese patients. In a clinical study (median follow-up 3.9 years) of patients at high risk of CVD and with well-controlled LDL-C levels on simvastatin 40 mg/day with or without ezetimibe 10 mg/day, there was no incremental benefit on cardiovascular outcomes with the addition of lipid-modifying doses of niacin |
| Intervention: | Concomitant use of simvastatin with lipid-modifying dosages of niacin is not recommended in Chinese patients [see Use in Specific Populations (8.8)]. For non-Chinese patients, consider if the benefit of using lipid-modifying doses of niacin concomitantly with simvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
| Fibrates (other than Gemfibrozil) | |
| Clinical Impact: | Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with simvastatin. |
| Intervention: | Consider if the benefit of using fibrates concomitantly with simvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
| Colchicine | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with simvastatin |
| Intervention: | Consider if the benefit of using colchicine concomitantly with simvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
| Grapefruit Juice | |
| Clinical Impact: | Grapefruit juice can raise the plasma levels of simvastatin and may increase the risk of myopathy and rhabdomyolysis. |
| Intervention: | Avoid grapefruit juice when taking simvastatin. |
Structured Label Content
Section 42229-5 (42229-5)
Patients taking Lomitapide
Reduce the dosage of simvastatin by 50%. Do not exceed simvastatin 20 mg once daily (or 40 mg once daily for patients who have previously taken simvastatin 80 mg daily chronically while taking lomitapide) [see Dosage and Administration (2.1)].
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 01/2025 |
|
PATIENT INFORMATION
Simvastatin (sim" va stat' in) tablets, for oral use |
|
| Read this Patient Information before you start taking simvastatin tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is Simvastatin Tablets?
Simvastatin tablets are prescription medicine that contains the cholesterol lowering medicine, simvastatin Simvastatin tablets are used to lower:
The safety and effectiveness of simvastatin tablets have not been established in children younger than 10 years of age with heterozygous familial hypercholesterolemia (HeFH) or other types of hyperlipidemia (high levels of fat in the blood). |
|
Do not take Simvastatin tablets if you:
|
|
Before you take simvastatin tablets, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Talk to your healthcare provider before you start taking any new medicines. Tell your healthcare provider who prescribes simvastatin tablets if another healthcare provider increases the dose of another medicine you are taking. Simvastatin tablets may affect the way other medicines work, and other medicines may affect how simvastatin tablets work. Especially tell your healthcare provider if you take:
|
|
How should I take simvastatin tablets?
|
|
|
What are the possible side effects of simvastatin tablets? Simvastatin tablets may cause serious side effects including:
These are not all the possible side effects of simvastatin tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store simvastatin tablets?
|
|
|
General information about safe and effective use of simvastatin tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use simvastatin tablets for a condition for which it was not prescribed. Do not give simvastatin tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about simvastatin tablets that is written for health professionals. |
|
|
What are the ingredients in simvastatin tablets?
Active ingredient(s): simvastatin. Inactive ingredient(s): lactose monohydrate, pregelatinized starch, microcrystalline cellulose, ethanol, ascorbic acid, citric acid and magnesium stearate. Butylated hydroxyanisole is added as a preservative. |
|
| Manufacturer: Yiling Pharmaceutical Ltd No.36 Zhujiang Road, Shijiazhuang, 050035, China. Distributor: Westminster Pharmaceuticals, LLC 1321 Murfreesboro Pike, Ste 607, Nashville, TN 37217, USA |
Section 43683-2 (43683-2)
| Dosage and Administration (2.1) | 03/2023 |
Section 44425-7 (44425-7)
Storage
Store at 20-25°C (68-77°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
No specific antidotes for simvastatin are known. Contact Poison Control (1-800-222-1222) for latest recommendations.
11 Description (11 DESCRIPTION)
Simvastatin is a prodrug of 3-hydoroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor that is derived synthetically from a fermentation product of Aspergillus terreus.
Simvastatin is butanoic acid, 2,2-dimethyl-,1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-(tetra- hydro-4hydroxy-6-oxo-2H-pyran-2-yl)-ethyl]-1-naphthalenyl ester, [1S-[1α,3α,7β,8β(2S*,4S*),-8aβ]]. The empirical formula of simvastatin is C25H38O5 and its molecular weight is 418.57. Its structural formula is:
Simvastatin is a white to off-white, nonhygroscopic, crystalline powder that is practically insoluble in water, and freely soluble in chloroform, methanol and ethanol.
Simvastatin Tablets, USP for oral use contain 10 mg, 20 mg, 40 mg or 80 mg of simvastatin and the following inactive ingredients: anhydrous citric acid, ascorbic acid, butylated hydroxyanisole, ferric oxide red (iron oxide red), ferric oxide yellow (iron oxide yellow), ferrosoferric oxide (10 mg and 40 mg), hypromellose, lactose monohydrate, magnesium stearate, maltodextrin (10 mg and 80 mg), medium-chain triglycerides (10 mg and 80 mg), microcrystalline cellulose, polydextrose (10 mg and 80 mg), polyethylene glycol (macrogol/peg) (20 mg), pregelatinized starch (maize), (10 mg, 40 mg and 80 mg) and titanium dioxide.
FDA approved dissolution test specifications differ from USP.
8.4 Pediatric Use
The safety and effectiveness of simvastatin as an adjunct to diet to reduce LDL-C have been established in pediatric patients 10 years of age and older with HeFH. Use of simvastatin for this indication is based on a double-blind, placebo-controlled clinical study in 175 pediatric patients (99 boys and 76 girls at least 1 year post-menarche) 10 years of age and older with HeFH. In this limited controlled study, there was no significant effect on growth or sexual maturation in the boys or girls, or on menstrual cycle length in girls.
The safety and effectiveness of simvastatin have not been established in pediatric patients younger than 10 years of age with HeFH or in pediatric patients with other types of hyperlipidemia (other than HeFH).
8.5 Geriatric Use
Of the total number of simvastatin-treated patients in clinical studies 1,021 (23%) patients, 5,366 (52%) patients, and 363 (15%) patients were ≥65 years old, respectively. In Study HPS, 615 (6%) patients were ≥75 years old [see Clinical Studies (14)]. In a clinical study of patients treated with simvastatin 80 mg daily, patients ≥65 years of age had an increased risk of myopathy, including rhabdomyolysis, compared to patients <65 years of age.
A pharmacokinetic study with simvastatin use showed the mean plasma level of total inhibitors to be approximately 45% higher in geriatric patients between 70-78 years of age compared with patients between 18-30 years of age [see Clinical Pharmacology (12.3)].
Advanced age (≥65 years) is a risk factor for simvastatin-associated myopathy and rhabdomyolysis. Dose selection for an elderly patient should be cautious, recognizing the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy and the higher risk of myopathy. Monitor geriatric patients receiving simvastatin for the increased risk of myopathy [see Warnings and Precautions (5.1)].
4 Contraindications (4 CONTRAINDICATIONS)
Simvastatin is contraindicated in the following conditions:
- Concomitant use of strong CYP3A4 inhibitors (select azole anti-fungals, macrolide antibiotics, anti-viral medications, and nefazodone) [see Drug Interactions (7.1)].
- Concomitant use of cyclosporine, danazol or gemfibrozil [see Drug Interactions (7.1)].
- Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3)].
- Hypersensitivity to simvastatin or any excipients in Simvastatin Tablets. Hypersensitivity reactions, including anaphylaxis, angioedema and Stevens-Johnson syndrome, have been reported [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described below and elsewhere in the labeling:
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1)]
- Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2)]
- Hepatic Dysfunction [see Warnings and Precautions (5.3)]
- Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- See full prescribing information for details regarding concomitant use of simvastatin with other drugs or grapefruit juice that increase the risk of myopathy and rhabdomyolysis. (2.5, 7.1)
- Coumarin Anticoagulants: Obtain INR before simvastatin initiation and monitor INR during simvastatin dosage initiation or adjustment. (7.2)
- Digoxin: During simvastatin initiation, monitor digoxin levels. (7.2)
8.6 Renal Impairment
Renal impairment is a risk factor for myopathy and rhabdomyolysis. Monitor all patients with renal impairment for development of myopathy. In patients with severe renal impairment (CLcr 15 – 29 mL/min), the recommended starting dosage is simvastatin 5 mg once daily [see Dosage and Administration (2.4), Warnings and Precautions (5.1)].
8.8 Chinese Patients
In a clinical study in which patients at high risk of CVD were treated with simvastatin 40 mg/day (median follow-up 3.9 years), the incidence of myopathy was approximately 0.05% for non-Chinese patients (n=7367) compared with 0.24% for Chinese patients (n=5468). In this study, the incidence of myopathy for Chinese patients on simvastatin 40 mg/day or ezetimibe/simvastatin 10/40 mg/day coadministered with extended-release niacin 2 g/day was 1.24%.
Chinese patients may be at higher risk for myopathy, monitor these patients appropriately. Coadministration of simvastatin with lipid-modifying doses of niacin-containing products (≥1 g/day niacin) is not recommended in Chinese patients [see Warnings and Precautions (5.1), Drug Interactions (7.1)].
12.2 Pharmacodynamics
Inhibition of HMG-CoA reductase by simvastatin acid accelerates the expression of LDL-receptors, followed by the uptake of LDL-C from blood to the liver, leading to a decrease in plasma LDL-C and total cholesterol. Sustained inhibition of cholesterol synthesis in the liver also decreases levels of very-lowdensity lipoproteins. The maximum LDL-C reduction of simvastatin is usually achieved by 4 weeks and is maintained after that.
12.3 Pharmacokinetics
Simvastatin is a lactone that is readily hydrolyzed in vivo to the corresponding β-hydroxyacid. Pharmacokinetics (PK) of simvastatin and its metabolites was originally characterized using inhibition of HMG-CoA reductase activity following base hydrolysis of plasma samples, as specific bioanalytical methods were not available. Inhibition of the enzyme activity (equivalent to the level of total inhibitors) represented the combination of activities in plasma following administration of simvastatin from both active (simvastatin acid and its metabolites) and latent forms (simvastatin and its metabolites) after conversion to the active forms in the presence of base.
8.7 Hepatic Impairment
Simvastatin is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4), Warnings and Precautions (5.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Simvastatin is indicated:
- To reduce the risk of total mortality by reducing risk of coronary heart disease death, non-fatal myocardial infarction and stroke, and the need for coronary and non-coronary revascularization procedures in adults with established coronary heart disease, cerebrovascular disease, peripheral vascular disease, and/or diabetes, who are at high risk of coronary heart disease events.
- As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C):
- In adults with primary hyperlipidemia.
- In adults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH).
- As an adjunct to other LDL-C-lowering therapies to reduce LDL-C in adults with homozygous familial hypercholesterol emia (HoFH).
- As an adjunct to diet for the treatment of adults with:
- Primary dysbetalipoproteinemia.
- Hypertriglyceridemia.
5.3 Hepatic Dysfunction
Increases in serum transaminases have been reported with use of simvastatin [see Adverse Reactions (6.1)]. In most cases, these changes appeared soon after initiation, were transient, were not accompanied by symptoms, and resolved or improved on continued therapy or after a brief interruption in therapy. Persistent increases to more than 3×ULN in serum transaminases have occurred in approximately 1% of patients receiving simvastatin in clinical studies. Marked persistent increases of hepatic transaminases have also occurred with simvastatin. There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking statins, including simvastatin.
Patients who consume substantial quantities of alcohol and/or have a history of liver disease may be at increased risk for hepatic injury.
Consider liver enzyme testing before simvastatin initiation and when clinically indicated thereafter. Simvastatin is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)]. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue simvastatin.
12.1 Mechanism of Action
Simvastatin is a prodrug and is hydrolyzed to its active β-hydroxyacid form, simvastatin acid, after administration. Simvastatin acid and its metabolites are inhibitors of HMG-CoA reductase, the rate-limiting enzyme that converts HMG-CoA to mevalonate, a precursor of cholesterol.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher simvastatin dosage. Chinese patients may be at higher risk for myopathy. Discontinue simvastatin if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue simvastatin in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing simvastatin dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever. (5.1, 7.1, 8.5, 8.6, 8.8)
- Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue simvastatin if IMNM is suspected. (5.2)
- Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzyme before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue simvastatin. (4, 5.3, 8.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
Important Dosage and Administration Information: (2.1)
- For dosing with the 5 mg strength, use another simvastatin product.
- Take simvastatin orally once daily in the evening.
- Maximum recommended dosage is simvastatin 40 mg once daily. An 80 mg daily dosage of simvastatin is restricted to patients who have been taking simvastatin 80 mg daily chronically (e.g., for 12 months or more) without evidence of muscle toxicity.
- For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving simvastatin 40 mg daily, prescribe alternative LDL-C lowering treatment.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating, and adjust the dosage if necessary.
- Adults: Recommended dosage is 20 mg to 40 mg once daily. (2.2)
- Pediatric Patients Aged 10 Years and Older with HeFH: Recommended dosage is 10 mg to 40 mg once daily. (2.3)
- Patients with Severe Renal Impairment: Recommended starting dosage is simvastatin 5 mg once daily. (2.4, 8.6)
- See full prescribing information for simvastatin dosage modifications due to drug interactions. (2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side.
- Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side.
- Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side.
- Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of simvastatin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as whole: fever, chills, malaise, asthenia
Blood and Lymphatic System Disorders: anemia, thrombocytopenia, leukopenia, hemolytic anemia, positive ANA, ESR increase, eosinophilia
Gastrointestinal Disorders: pancreatitis, vomiting
Hepatic and Pancreatic Disorders: hepatitis/jaundice, fatal and non-fatal hepatic failure
Immune System Disorders: hypersensitivity syndrome including: anaphylaxis, angioedema, lupus erythematous-like syndrome, dermatomyositis, vasculitis
Musculoskeletal and Connective Tissue Disorders: muscle cramps, immune-mediated necrotizing myopathy, polymyalgia rheumatica, arthritis
Nervous System Disorders: dizziness, depression, paresthesia, peripheral neuropathy, Rare reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. Cognitive impairment was generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks).There have been rare reports of new-onset or exacerbation of myasthenia gravis, including ocular myasthenia, and reports of recurrence when the same or a different statin was administered.
Skin and Subcutaneous Tissue Disorders: pruritus, alopecia, a variety of skin changes (e.g., nodules, discoloration, dryness of skin/mucous membranes, changes to hair/nails), purpura, lichen planus, urticaria, photosensitivity, flushing, toxic epidermal necrolysis, erythema multiforme, including Stevens-Johnson syndrome
Respiratory and Thoracic: interstitial lung disease, dyspnea
Reproductive System Disorders: erectile dysfunction
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
In clinical studies, 2,423 adult patients were exposed to simvastatin with a median duration of follow-up of approximately 18 months. The most commonly reported adverse reactions (incidence ≥5%) in these simvastatin clinical studies were: upper respiratory infections (9%), headache (7%), abdominal pain (7%), constipation (7%), and nausea (5%). Overall, 1.4% of patients discontinued simvastatin due to adverse reactions. The most common adverse reactions that led to discontinuation were: gastrointestinal disorders (0.5%), myalgia (0.1%), and arthralgia (0.1%).
In a Cardiovascular Outcomes Study (the Scandinavian Simvastatin Survival Study [Study 4S]), adult patients (age range 35-71 years, 19% women, 100% Caucasians) were treated with 20-40 mg per day of simvastatin or placebo over a median of 5.4 years [see Clinical Studies (14)]; adverse reactions reported in ≥2% of patients and at a rate greater than placebo are shown in Table 1.
| %Placebo (N=2,223) |
%Simvastatin (N=2,221) |
|
|---|---|---|
| Bronchitis | 6.3 | 6.6 |
| Abdominal pain | 5.8 | 5.9 |
| Atrial fibrillation | 5.1 | 5.7 |
| Gastritis | 3.9 | 4.9 |
| Eczema | 3.0 | 4.5 |
| Vertigo | 4.2 | 4.5 |
| Diabetes mellitus | 3.6 | 4.2 |
| Insomnia | 3.8 | 4.0 |
| Myalgia | 3.2 | 3.7 |
| Urinary tract infection | 3.1 | 3.2 |
| Edema/swelling | 2.3 | 2.7 |
| Headache | 2.1 | 2.5 |
| Sinusitis | 1.8 | 2.3 |
| Constipation | 1.6 | 2.2 |
5.1 Myopathy and Rhabdomyolysis
Simvastatin may cause myopathy and rhabdomyolysis. Acute kidney injury secondary to myoglobinuria and rare fatalities have occurred as a result of rhabdomyolysis in patients treated with statins, including simvastatin.
In clinical studies of 24,747 simvastatin-treated patients with a median follow-up of 4 years, the incidence of myopathy, defined as unexplained muscle weakness, pain, or tenderness accompanied by creatinine kinase (CK) increases greater than ten times the upper limit of normal (10×ULN), were approximately 0.03%, 0.08%, and 0.61% in patients treated with simvastatin 20 mg, 40 mg, and 80 mg daily, respectively. In another clinical study of 12,064 simvastatin-treated patients (with a history of myocardial infarction) with a mean follow-up of 6.7 years, the incidences of myopathy in patients taking simvastatin 20 mg and 80 mg daily were approximately 0.02% and 0.9%, respectively. The incidences of rhabdomyolysis (defined as myopathy with a CK >40×ULN) in patients taking simvastatin 20 mg and 80 mg daily were approximately 0% and 0.4%, respectively [see Adverse Reactions (6.1)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Simvastatin Tablets, USP 10 mg are yellow, oval, film-coated tablets, debossed with "Y22" on one side. They are supplied as follows:
NDC 69367-374-09 unit of use bottles of 90.
NDC 69367-374-10 bottles of 1000.
Simvastatin Tablets, USP 20 mg are peach, oval, film-coated tablets, debossed with "Y23" on one side. They are supplied as follows:
NDC 69367-375-09 unit of use bottles of 90.
NDC 69367-375-10 bottles of 1000.
Simvastatin Tablets, USP 40 mg are brown, oval, film-coated tablets, debossed with "Y24" on one side. They are supplied as follows:
NDC 69367-376-09 unit of use bottles of 90.
NDC 69367-376-10 bottles of 1000.
Simvastatin Tablets, USP 80 mg are pink, capsule-shaped, film-coated tablets, debossed with "Y25" on one side. They are supplied as follows:
NDC 69367-377-09 unit of use bottles of 90.
NDC 69367-377-10 bottles of 1000.
7.2 Simvastatin Effects On Other Drugs (7.2 Simvastatin Effects on Other Drugs)
Table 3 presents Simvastatin's effect on other drugs and instructions for preventing or managing them.
| Coumarin Anticoagulants | |
|---|---|
| Clinical Impact: | Simvastatin may potentiate the effect of coumarin anticoagulants and increase the INR. The concomitant use of simvastatin (20 to 40 mg) and coumarin anticoagulants increased the INR from a baseline of 1.7 to 1.8 in healthy subjects and from 2.6 to 3.4 in patients with hyperlipidemia. There are postmarketing reports of clinically evident bleeding and/or increased INR in patients taking concomitant statins and warfarin. |
| Intervention: | In patients taking coumarin anticoagulants, obtain an INR before starting simvastatin and frequently enough after initiation, dose titration, or discontinuation to ensure that no significant alteration in INR occurs. Once the INR is stable, monitor INR at regularly recommended intervals. |
| Digoxin | |
| Clinical Impact: | Concomitant use of digoxin with simvastatin may result in elevated plasma digoxin concentrations [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor digoxin levels in patients taking digoxin when simvastatin is initiated. |
2.2 Recommended Dosage in Adult Patients
The recommended dosage range of simvastatin is 20 mg to 40 mg once daily.
5.2 Immune Mediated Necrotizing Myopathy (5.2 Immune-Mediated Necrotizing Myopathy)
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use, including reports of recurrence when the same or a different statin was administered. IMNM is characterized by proximal muscle weakness and elevated serum creatine kinase that persist despite discontinuation of statin treatment; positive anti-HMG CoA reductase antibody; muscle biopsy showing necrotizing myopathy without significant inflammation; and improvement with immunosuppressive agents. Additional neuromuscular and serologic testing may be necessary. Treatment with immunosuppressive agents may be required. Discontinue simvastatin if IMNM is suspected.
2.5 Dosage Modifications Due to Drug Interactions
Concomitant use of simvastatin with the following drugs requires dosage modification of simvastatin [see Warnings and Precautions (5.1) and Drug Interactions (7.1)].
2.1 Important Dosage and Administration Information
- For dosing with the 5 mg strength, use another simvastatin product.
- Take simvastatin orally once daily in the evening.
- The maximum recommended dosage is simvastatin 40 mg once daily [see Dosage and Administration (2.2, 2.3)]. The simvastatin 80 mg daily dosage is restricted to patients who have been taking simvastatin 80 mg daily chronically (e.g., for 12 months or more) without evidence of muscle toxicity [see Warnings and Precautions (5.1)].
- If as dose is missed, take the missed dose as soon as possible. Do not double the next dose.
- For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving simvastatin 40 mg daily, prescribe alternative LDL-C-lowering treatment.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating simvastatin, and adjust the dosage if necessary.
Principal Display Panel 10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label)
NDC 69367-374-10
Rx Only
Simvastatin
Tablets, USP
10 mg
1000 Tablets
Westminster
Pharmaceuticals
Principal Display Panel 20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label)
NDC 69367-375-10
Rx Only
Simvastatin
Tablets, USP
20 mg
1000 Tablets
Westminster
Pharmaceuticals
Principal Display Panel 40 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 40 mg Tablet Bottle Label)
NDC 69367-376-10
Rx Only
Simvastatin
Tablets, USP
40 mg
1000 Tablets
Westminster
Pharmaceuticals
Principal Display Panel 80 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 80 mg Tablet Bottle Label)
NDC 69367-377-10
Rx Only
Simvastatin
Tablets, USP
80 mg
1000 Tablets
Westminster
Pharmaceuticals
5.4 Increases in Hba1c and Fasting Serum Glucose Levels (5.4 Increases in HbA1c and Fasting Serum Glucose Levels)
Increases in HbA1c and fasting serum glucose levels have been reported with statins, including simvastatin. Optimize lifestyle measures, including regular exercise, maintaining a healthy body weight, and making healthy food choices.
2.4 Recommended Dosage in Patients With Renal Impairment (2.4 Recommended Dosage in Patients with Renal Impairment)
For patients with severe renal impairment [creatinine clearance (CLcr) 15 – 29 mL/min], the recommended starting dosage of simvastatin is 5 mg once daily [see Warnings and Precautions (5.1) and Use in Specific Populations (8.6)]. Use another simvastatin product to initiate dosing in such patients [see Dosage and Administration (2.1)].
There are no dosage adjustment recommendations for patients with mild or moderate renal impairment.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 72-week carcinogenicity study, mice were administered daily doses of simvastatin of 25, 100, and 400 mg/kg body weight, which resulted in mean plasma drug levels approximately 1, 4, and 8 times higher than the mean human plasma drug level, respectively (as total inhibitory activity based on AUC) after an 80-mg oral dose. Liver carcinomas were significantly increased in high-dose females and mid- and high- dose males with a maximum incidence of 90% in males. The incidence of adenomas of the liver was significantly increased in mid- and high-dose females. Drug treatment also significantly increased the incidence of lung adenomas in mid- and high-dose males and females. Adenomas of the Harderian gland (a gland of the eye of rodents) were significantly higher in high-dose mice than in controls. No evidence of a tumorigenic effect was observed at 25 mg/kg/day.
In a separate 92-week carcinogenicity study in mice at doses up to 25 mg/kg/day, no evidence of a tumorigenic effect was observed (mean plasma drug levels were 1 times higher than humans given 80 mg simvastatin as measured by AUC).
In a two-year study in rats at 25 mg/kg/day, there was a statistically significant increase in the incidence of thyroid follicular adenomas in female rats exposed to approximately 11 times higher levels of simvastatin than in humans given 80 mg simvastatin (as measured by AUC).
A second two-year rat carcinogenicity study with doses of 50 and 100 mg/kg/day produced hepatocellular adenomas and carcinomas (in female rats at both doses and in males at 100 mg/kg/day). Thyroid follicular cell adenomas were increased in males and females at both doses; thyroid follicular cell carcinomas were increased in females at 100 mg/kg/day. The increased incidence of thyroid neoplasms appears to be consistent with findings from other statins. These treatment levels represented plasma drug levels (AUC) of approximately 7 and 15 times (males) and 22 and 25 times (females) the mean human plasma drug exposure after an 80 milligram daily dose.
No evidence of mutagenicity was observed in a microbial mutagenicity (Ames) test with or without rat or mouse liver metabolic activation. In addition, no evidence of damage to genetic material was noted in an in vitro alkaline elution assay using rat hepatocytes, a V-79 mammalian cell forward mutation study, an in vitro chromosome aberration study in CHO cells, or an in vivo chromosomal aberration assay in mouse bone marrow.
There was decreased fertility in male rats treated with simvastatin for 34 weeks at 25 mg/kg body weight (4 times the maximum human exposure level, based on AUC, in patients receiving 80 mg/day); however, this effect was not observed during a subsequent fertility study in which simvastatin was administered at this same dose level to male rats for 11 weeks (the entire cycle of spermatogenesis including epididymal maturation). No microscopic changes were observed in the testes of rats from either study. At 180 mg/kg/day, (which produces exposure levels 22 times higher than those in humans taking 80 mg/day based on surface area, mg/m2), seminiferous tubule degeneration (necrosis and loss of spermatogenic epithelium) was observed. In dogs, there was drug-related testicular atrophy, decreased spermatogenesis, spermatocytic degeneration and giant cell formation at 10 mg/kg/day, (approximately 2 times the human exposure, based on AUC, at 80 mg/day). The clinical significance of these findings is unclear.
2.3 Recommended Dosage in Pediatric Patients 10 Years of Age and Older With Hefh (2.3 Recommended Dosage in Pediatric Patients 10 Years of Age and Older with HeFH)
The recommended dosage range of simvastatin is 10 mg to 40 mg daily.
7.1 Drug Interactions That Increase the Risk of Myopathy and Rhabdomyolysis With Simvastatin (7.1 Drug Interactions that Increase the Risk of Myopathy and Rhabdomyolysis with Simvastatin)
Simvastatin is a substrate of CYP3A4 and of the transport protein OATP1B1. Simvastatin exposure can be significantly increased with concomitant administration of inhibitors of CYP3A4 and OATP1B1. Table 2 includes a list of drugs that increase the risk of myopathy and rhabdomyolysis when used concomitantly with simvastatin and instructions for preventing or managing them [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
| Strong CYP3A4 inhibitors | |
|---|---|
| Clinical Impact: | Simvastatin is a substrate of CYP3A4. Concomitant use of strong CYP3A4 inhibitors with simvastatin increases simvastatin exposure and increases the risk of myopathy and rhabdomyolysis, particularly with higher simvastatin dosages. |
| Intervention: | Concomitant use of strong CYP3A4 inhibitors with simvastatin is contraindicated [see Contraindications (4)]. If treatment with a CYP3A4 inhibitor is unavoidable, suspend simvastatin during the course of strong CYP3A4 inhibitor treatment. |
| Examples: | Select azole anti-fungals (e.g., itraconazole, ketoconazole, posaconazole, and voriconazole), select macrolide antibiotics (e.g., erythromycin and clarithromycin), select HIV protease inhibitors (e.g., nelfinavir, ritonavir, and darunavir/ritonavir), select HCV protease inhibitors (e.g., boceprevir and telaprevir), cobicistat-containing products, and nefazodone. |
| Cyclosporine, Danazol, or Gemfibrozil | |
| Clinical Impact: | The risk of myopathy and rhabdomyolysis is increased with concomitant use of cyclosporine, danazol, or gemfibrozil with simvastatin. Gemfibrozil may cause myopathy when given alone. |
| Intervention: | Concomitant use of cyclosporine, danazol, or gemfibrozil with simvastatin is contraindicated [see Contraindications (4)]. |
| Amiodarone, Dronedarone, Ranolazine, or Calcium Channel Blockers | |
| Clinical Impact: | The risk of myopathy and rhabdomyolysis is increased by concomitant use of amiodarone, dronedarone, ranolazine, or calcium channel blockers with simvastatin. |
| Intervention: | For patients taking verapamil, diltiazem, or dronedarone, do not exceed simvastatin 10 mg daily. For patients taking amiodarone, amlodipine, or ranolazine, do not exceed simvastatin 20 mg daily [see Dosage and Administration (2.5)]. |
| Lomitapide | |
| Clinical Impact: | Simvastatin exposure is approximately doubled with concomitant use of lomitapide and the risk of myopathy and rhabdomyolysis is increased. |
| Intervention: | Reduce the dose of simvastatin by 50% if initiating lomitapide. Do not exceed simvastatin 20 mg daily (or simvastatin 40 mg daily for patients who have previously taken simvastatin 80 mg daily chronically) while taking lomitapide [see Dosage and Administration (2.1,2.5)]. |
| Daptomycin | |
| Clinical Impact: | Cases of rhabdomyolysis have been reported with simvastatin administered with daptomycin. Both simvastatin and daptomycin can cause myopathy and rhabdomyolysis when given alone and the risk of myopathy and rhabdomyolysis may be increased by coadministration. |
| Intervention: | If treatment with daptomycin is required, consider temporarily suspending simvastatin during the course of daptomycin treatment. |
| Niacin | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been observed with concomitant use of lipid modifying dosages of niacin-containing products (≥1 gram/day niacin) with simvastatin. The risk of myopathy is greater in Chinese patients. In a clinical study (median follow-up 3.9 years) of patients at high risk of CVD and with well-controlled LDL-C levels on simvastatin 40 mg/day with or without ezetimibe 10 mg/day, there was no incremental benefit on cardiovascular outcomes with the addition of lipid-modifying doses of niacin |
| Intervention: | Concomitant use of simvastatin with lipid-modifying dosages of niacin is not recommended in Chinese patients [see Use in Specific Populations (8.8)]. For non-Chinese patients, consider if the benefit of using lipid-modifying doses of niacin concomitantly with simvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
| Fibrates (other than Gemfibrozil) | |
| Clinical Impact: | Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with simvastatin. |
| Intervention: | Consider if the benefit of using fibrates concomitantly with simvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
| Colchicine | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with simvastatin |
| Intervention: | Consider if the benefit of using colchicine concomitantly with simvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
| Grapefruit Juice | |
| Clinical Impact: | Grapefruit juice can raise the plasma levels of simvastatin and may increase the risk of myopathy and rhabdomyolysis. |
| Intervention: | Avoid grapefruit juice when taking simvastatin. |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:51.761110 · Updated: 2026-03-14T22:28:13.751851