ac41d70d-0046-783e-e053-2995a90a9a58
34390-5
HUMAN OTC DRUG LABEL
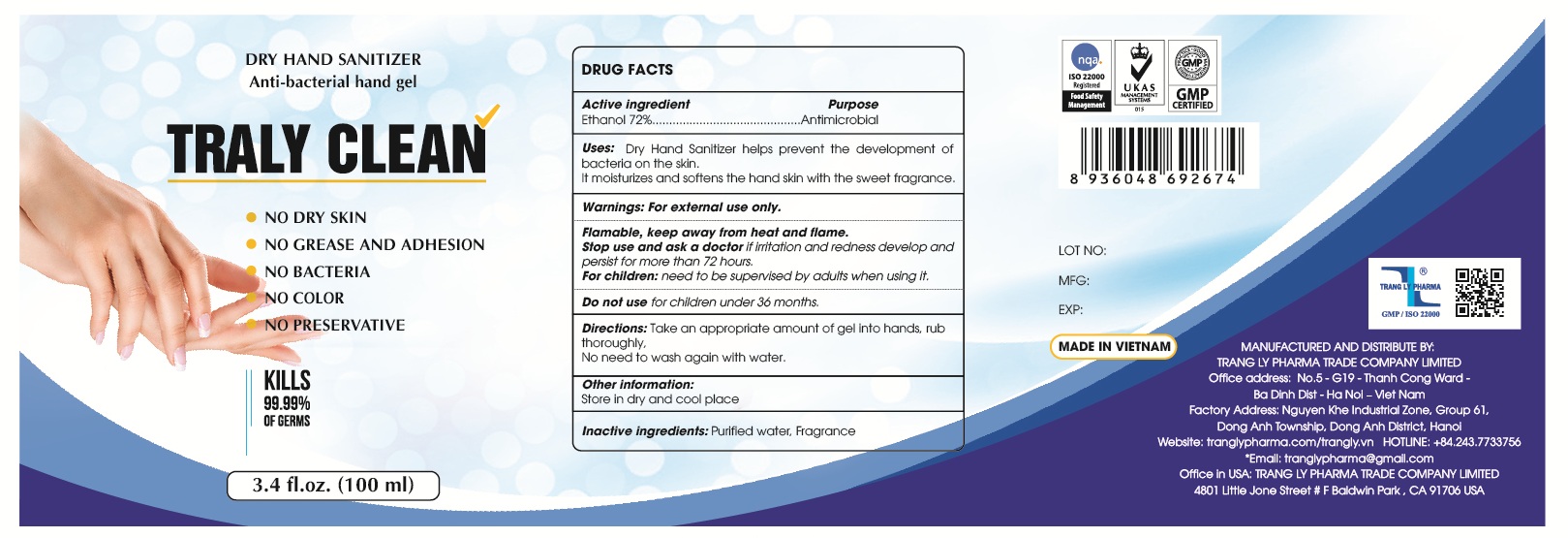
Drug Facts
Composition & Product
Identifiers & Packaging
Description
This is a hand sanitizer manufactured according to the Temporary Policy for Preparation of Certain Alcohol-Based Hand Sanitizer Products During the Public Health Emergency (CoViD-19); Guidance for Industry . The hand sanitizer is manufactured using only the following United States Pharmacopoeia (USP) grade ingredients in the preparation of the product (percentage in final product formulation) consistent with World Health Organization (WHO) recommendations: Alcohol (ethanol) (USP or Food Chemical Codex (FCC) grade) (80%, volume/volume (v/v)) in an aqueous solution denatured according to Alcohol and Tobacco Tax and Trade Bureau regulations in 27 CFR part 20. Glycerol (1.45% v/v). Hydrogen peroxide (0.125% v/v). Sterile distilled water or boiled cold water. The firm does not add other active or inactive ingredients. Different or additional ingredients may impact the quality and potency of the product.
Purpose
Antimicrobial
Medication Information
Purpose
Antimicrobial
Description
This is a hand sanitizer manufactured according to the Temporary Policy for Preparation of Certain Alcohol-Based Hand Sanitizer Products During the Public Health Emergency (CoViD-19); Guidance for Industry . The hand sanitizer is manufactured using only the following United States Pharmacopoeia (USP) grade ingredients in the preparation of the product (percentage in final product formulation) consistent with World Health Organization (WHO) recommendations: Alcohol (ethanol) (USP or Food Chemical Codex (FCC) grade) (80%, volume/volume (v/v)) in an aqueous solution denatured according to Alcohol and Tobacco Tax and Trade Bureau regulations in 27 CFR part 20. Glycerol (1.45% v/v). Hydrogen peroxide (0.125% v/v). Sterile distilled water or boiled cold water. The firm does not add other active or inactive ingredients. Different or additional ingredients may impact the quality and potency of the product.
Use
Dry Hand Sanitizer helps prevent the development of bacteria on the skin.
It moisturizes and softens the hand skin with the sweet fragrance.
Section 42229-5
This is a hand sanitizer manufactured according to the Temporary Policy for Preparation of Certain Alcohol-Based Hand Sanitizer Products During the Public Health Emergency (CoViD-19); Guidance for Industry.
The hand sanitizer is manufactured using only the following United States Pharmacopoeia (USP) grade ingredients in the preparation of the product (percentage in final product formulation) consistent with World Health Organization (WHO) recommendations:
- Alcohol (ethanol) (USP or Food Chemical Codex (FCC) grade) (80%, volume/volume (v/v)) in an aqueous solution denatured according to Alcohol and Tobacco Tax and Trade Bureau regulations in 27 CFR part 20.
- Glycerol (1.45% v/v).
- Hydrogen peroxide (0.125% v/v).
- Sterile distilled water or boiled cold water.
The firm does not add other active or inactive ingredients. Different or additional ingredients may impact the quality and potency of the product.
Section 43683-2
Update Uses: from template to label description.
Section 50565-1
Keep our of reach of children
Section 50566-9
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Section 50567-7
Stop use and ask a doctor if irritation and redness develop and persist for more than 72 hours.
For children: need to be supervised by adults when using it.
Warnings
For external use only. Flammable. Keep away from heat or flame
Directions
- Place enough product on hands to cover all surfaces. Rub hands together until dry.
- Supervise children under 6 years of age when using this product to avoid swallowing.
Do Not Use
- for children under 36 months.
Other Information
- Store in dry and cool place
Active Ingredient(s)
Alcohol 80% v/v. Purpose: Antiseptic
Inactive Ingredients
glycerin, hydrogen peroxide, purified water USP
Package Label Principal Display Panel
100 mL NDC: 90098-100-01
250 mL NDC: 90098-100-02
500 mL NDC: 90098-100-03
1000 mL NDC: 90098-100-04
2000 mL NDC: 90098-100-05
10000 mL NDC: 90098-100-06
20000 mL NDC: 90098-100-07
59 mL NDC: 90098-100-08
1 Gallon NDC: 90098-100-09
Structured Label Content
Use
Dry Hand Sanitizer helps prevent the development of bacteria on the skin.
It moisturizes and softens the hand skin with the sweet fragrance.
Section 42229-5 (42229-5)
This is a hand sanitizer manufactured according to the Temporary Policy for Preparation of Certain Alcohol-Based Hand Sanitizer Products During the Public Health Emergency (CoViD-19); Guidance for Industry.
The hand sanitizer is manufactured using only the following United States Pharmacopoeia (USP) grade ingredients in the preparation of the product (percentage in final product formulation) consistent with World Health Organization (WHO) recommendations:
- Alcohol (ethanol) (USP or Food Chemical Codex (FCC) grade) (80%, volume/volume (v/v)) in an aqueous solution denatured according to Alcohol and Tobacco Tax and Trade Bureau regulations in 27 CFR part 20.
- Glycerol (1.45% v/v).
- Hydrogen peroxide (0.125% v/v).
- Sterile distilled water or boiled cold water.
The firm does not add other active or inactive ingredients. Different or additional ingredients may impact the quality and potency of the product.
Section 43683-2 (43683-2)
Update Uses: from template to label description.
Section 50565-1 (50565-1)
Keep our of reach of children
Section 50566-9 (50566-9)
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Section 50567-7 (50567-7)
Stop use and ask a doctor if irritation and redness develop and persist for more than 72 hours.
For children: need to be supervised by adults when using it.
Purpose
Antimicrobial
Warnings
For external use only. Flammable. Keep away from heat or flame
Directions
- Place enough product on hands to cover all surfaces. Rub hands together until dry.
- Supervise children under 6 years of age when using this product to avoid swallowing.
Do Not Use (Do not use)
- for children under 36 months.
Other Information (Other information)
- Store in dry and cool place
Active Ingredient(s)
Alcohol 80% v/v. Purpose: Antiseptic
Inactive Ingredients (Inactive ingredients)
glycerin, hydrogen peroxide, purified water USP
Package Label Principal Display Panel (Package Label - Principal Display Panel)
100 mL NDC: 90098-100-01
250 mL NDC: 90098-100-02
500 mL NDC: 90098-100-03
1000 mL NDC: 90098-100-04
2000 mL NDC: 90098-100-05
10000 mL NDC: 90098-100-06
20000 mL NDC: 90098-100-07
59 mL NDC: 90098-100-08
1 Gallon NDC: 90098-100-09
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:53.880893 · Updated: 2026-03-14T22:58:42.480630