These Highlights Do Not Include All The Information Needed To Use Voydeya Safely And Effectively. See Full Prescribing Information For Voydeya.
ab8d1cbd-a28b-4d49-82ab-5c7460719ac7
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
VOYDEYA, a complement inhibitor, increases the risk of serious infections, especially those caused by encapsulated bacteria, such as Neisseria meningitidis , Streptococcus pneumoniae , and Haemophilus influenzae type B [ see Warnings and Precautions (5.1) ]. Life-threatening and fatal infections with encapsulated bacteria have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early. Complete or update vaccination for encapsulated bacteria specifically, Neisseria meningitidis and Streptococcus pneumoniae at least 2 weeks prior to the first dose of VOYDEYA, unless the risks of delaying therapy with VOYDEYA outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against encapsulated bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by encapsulated bacteria. Patients receiving VOYDEYA are at increased risk for invasive disease caused by encapsulated bacteria, even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious infections and evaluate immediately if infection is suspected. Because of the risk of serious infections caused by encapsulated bacteria, VOYDEYA is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the VOYDEYA REMS [see Warnings and Precautions (5.2) ] .
Indications and Usage
VOYDEYA is indicated as add-on therapy to ravulizumab or eculizumab for the treatment of extravascular hemolysis (EVH) in adults with paroxysmal nocturnal hemoglobinuria (PNH).
Dosage and Administration
Start 150 mg three times a day orally, with or without food. Depending on clinical response, can increase to 200 mg three times a day. See Full Prescribing Information for instructions on dosage and administration ( 2.1 , 2.2 ).
Warnings and Precautions
Hepatic Enzyme Increases: Assess liver enzymes before treatment initiation and periodically during treatment. Consider treatment interruption or discontinuation if elevations are clinically significant or if the patient becomes symptomatic ( 5.3 ). Hyperlipidemia: Monitor serum lipids periodically during treatment and initiate cholesterol-lowering medication if indicated ( 5.4 ).
Contraindications
VOYDEYA is contraindicated for initiation in patients with unresolved serious infection caused by encapsulated bacteria, including Neisseria meningitidis , Streptococcus pneumoniae , or Haemophilus influenzae type B [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: Serious Infections Caused by Encapsulated Bacteria [see Warnings and Precautions (5.1) ] Hepatic Enzyme Increases [see Warnings and Precautions (5.3) ] Hyperlipidemia [see Warnings and Precautions (5.4) ]
Drug Interactions
BCRP substrates: Monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. For rosuvastatin, the dose should not exceed 10 mg once daily ( 7.1 ). P-gp substrates: Dose adjustment might be necessary for P-gp substrates where minimal concentration changes may lead to serious adverse reactions ( 7.2 ).
Storage and Handling
VOYDEYA (danicopan) tablets are available in the doses and packages listed in Table 4. Table 4 VOYDEYA Tablet Presentations Dose Tablet Strength Film-Coated Tablet Markings Tablet Color/Shape Pack Size NDC Code Each carton contains two high density polyethylene bottles with desiccant and child resistant seal, with 180 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets One bottle with 90 × 50 mg per tablet (25682-040-90) One bottle with 90 × 100 mg per tablet (25682-043-90) 25682-046-92 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Two bottles with 90 tablets per bottle: 90 × 100 mg per tablet (25682-043-90) 25682-043-92 Each carton contains four 7-day blister cards with 168 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 21 × 50 mg per tablet and 21 × 100 mg per tablet (25682-049-42) 25682-049-04 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 42 × 100 mg per tablet (25682-043-42) 25682-043-04
How Supplied
VOYDEYA (danicopan) tablets are available in the doses and packages listed in Table 4. Table 4 VOYDEYA Tablet Presentations Dose Tablet Strength Film-Coated Tablet Markings Tablet Color/Shape Pack Size NDC Code Each carton contains two high density polyethylene bottles with desiccant and child resistant seal, with 180 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets One bottle with 90 × 50 mg per tablet (25682-040-90) One bottle with 90 × 100 mg per tablet (25682-043-90) 25682-046-92 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Two bottles with 90 tablets per bottle: 90 × 100 mg per tablet (25682-043-90) 25682-043-92 Each carton contains four 7-day blister cards with 168 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 21 × 50 mg per tablet and 21 × 100 mg per tablet (25682-049-42) 25682-049-04 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 42 × 100 mg per tablet (25682-043-42) 25682-043-04
Medication Information
Warnings and Precautions
Hepatic Enzyme Increases: Assess liver enzymes before treatment initiation and periodically during treatment. Consider treatment interruption or discontinuation if elevations are clinically significant or if the patient becomes symptomatic ( 5.3 ). Hyperlipidemia: Monitor serum lipids periodically during treatment and initiate cholesterol-lowering medication if indicated ( 5.4 ).
Indications and Usage
VOYDEYA is indicated as add-on therapy to ravulizumab or eculizumab for the treatment of extravascular hemolysis (EVH) in adults with paroxysmal nocturnal hemoglobinuria (PNH).
Dosage and Administration
Start 150 mg three times a day orally, with or without food. Depending on clinical response, can increase to 200 mg three times a day. See Full Prescribing Information for instructions on dosage and administration ( 2.1 , 2.2 ).
Contraindications
VOYDEYA is contraindicated for initiation in patients with unresolved serious infection caused by encapsulated bacteria, including Neisseria meningitidis , Streptococcus pneumoniae , or Haemophilus influenzae type B [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: Serious Infections Caused by Encapsulated Bacteria [see Warnings and Precautions (5.1) ] Hepatic Enzyme Increases [see Warnings and Precautions (5.3) ] Hyperlipidemia [see Warnings and Precautions (5.4) ]
Drug Interactions
BCRP substrates: Monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. For rosuvastatin, the dose should not exceed 10 mg once daily ( 7.1 ). P-gp substrates: Dose adjustment might be necessary for P-gp substrates where minimal concentration changes may lead to serious adverse reactions ( 7.2 ).
Storage and Handling
VOYDEYA (danicopan) tablets are available in the doses and packages listed in Table 4. Table 4 VOYDEYA Tablet Presentations Dose Tablet Strength Film-Coated Tablet Markings Tablet Color/Shape Pack Size NDC Code Each carton contains two high density polyethylene bottles with desiccant and child resistant seal, with 180 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets One bottle with 90 × 50 mg per tablet (25682-040-90) One bottle with 90 × 100 mg per tablet (25682-043-90) 25682-046-92 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Two bottles with 90 tablets per bottle: 90 × 100 mg per tablet (25682-043-90) 25682-043-92 Each carton contains four 7-day blister cards with 168 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 21 × 50 mg per tablet and 21 × 100 mg per tablet (25682-049-42) 25682-049-04 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 42 × 100 mg per tablet (25682-043-42) 25682-043-04
How Supplied
VOYDEYA (danicopan) tablets are available in the doses and packages listed in Table 4. Table 4 VOYDEYA Tablet Presentations Dose Tablet Strength Film-Coated Tablet Markings Tablet Color/Shape Pack Size NDC Code Each carton contains two high density polyethylene bottles with desiccant and child resistant seal, with 180 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets One bottle with 90 × 50 mg per tablet (25682-040-90) One bottle with 90 × 100 mg per tablet (25682-043-90) 25682-046-92 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Two bottles with 90 tablets per bottle: 90 × 100 mg per tablet (25682-043-90) 25682-043-92 Each carton contains four 7-day blister cards with 168 tablets per carton: 150 mg 50 mg Debossed on one side with "DCN 50" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 21 × 50 mg per tablet and 21 × 100 mg per tablet (25682-049-42) 25682-049-04 100 mg Debossed on one side with "DCN 100" 200 mg 100 mg Debossed on one side with "DCN 100" White to off-white, round film-coated tablets Four blister cards with 42 tablets per card: 42 × 100 mg per tablet (25682-043-42) 25682-043-04
Description
VOYDEYA, a complement inhibitor, increases the risk of serious infections, especially those caused by encapsulated bacteria, such as Neisseria meningitidis , Streptococcus pneumoniae , and Haemophilus influenzae type B [ see Warnings and Precautions (5.1) ]. Life-threatening and fatal infections with encapsulated bacteria have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early. Complete or update vaccination for encapsulated bacteria specifically, Neisseria meningitidis and Streptococcus pneumoniae at least 2 weeks prior to the first dose of VOYDEYA, unless the risks of delaying therapy with VOYDEYA outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against encapsulated bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by encapsulated bacteria. Patients receiving VOYDEYA are at increased risk for invasive disease caused by encapsulated bacteria, even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious infections and evaluate immediately if infection is suspected. Because of the risk of serious infections caused by encapsulated bacteria, VOYDEYA is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the VOYDEYA REMS [see Warnings and Precautions (5.2) ] .
Section 42229-5
Limitations of Use
VOYDEYA has not been shown to be effective as monotherapy and should only be prescribed as an add-on to ravulizumab or eculizumab.
Section 42231-1
| MEDICATION GUIDE VOYDEYA™(voi-day-uh) (danicopan) tablets, for oral use |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Issued: 03/2024 | |
| What is the most important information I should know about VOYDEYA? | ||
VOYDEYA is a medicine that affects your immune system. VOYDEYA may lower the ability of your immune system to fight infections.
|
||
|
|
|
Your healthcare provider will give you a Patient Safety Card about the risk of serious infections. Carry it with you at all times during treatment and for 1 week after your last VOYDEYA dose. Your risk of serious infections may continue for a few days after your last dose of VOYDEYA. If you get any of the symptoms listed on this card you should get medical help right away. It is important to show this card to any healthcare provider who treats you. This will help them diagnose and treat you quickly.
|
||
| For more information about side effects, see " What are the possible side effects of VOYDEYA? " | ||
| What is VOYDEYA? | ||
| VOYDEYA is a prescription medicine used along with ravulizumab or eculizumab to treat breakdown of red blood cells that takes place outside of blood vessels (extravascular hemolysis), in adults with paroxysmal nocturnal hemoglobinuria (PNH). It is not known if VOYDEYA is safe and effective in children. |
||
| Who should not take VOYDEYA? | ||
| Do not take VOYDEYA if you have a serious infection caused by encapsulated bacteria, including Neisseria meningitidis, Streptococcus pneumoniae, or Haemophilus influenzae type B when you are starting VOYDEYA treatment. | ||
Before taking VOYDEYA, tell your healthcare provider about all of your medical conditions, including if you:
|
||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. VOYDEYA may affect the way other medicines work. | ||
| Know the medicines you take and the vaccines you receive. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. | ||
How should I take VOYDEYA?
|
||
| What are the possible side effects of VOYDEYA? | ||
VOYDEYA may cause side effects, including:
|
||
| The most common side effect of VOYDEYA is headache. | ||
| Tell your healthcare provider about any side effect that bothers you or that does not go away. | ||
| These are not all of the possible side effects of VOYDEYA. | ||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
How should I store VOYDEYA?
|
||
| General information about the safe and effective use of VOYDEYA. | ||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VOYDEYA for a condition for which it was not prescribed. Do not give VOYDEYA to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VOYDEYA that is written for health professionals. | ||
| What are the ingredients in VOYDEYA? | ||
| Active ingredient: danicopan | ||
| Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. | ||
| The film coatings contain the following inactive ingredients: polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. | ||
| Manufactured for: Alexion Pharmaceuticals, Inc., 121 Seaport Boulevard, Boston, MA 02210 USA VOYDEYA is a trademark of Alexion Pharmaceuticals, Inc. © 2024 Alexion Pharmaceuticals, Inc For more information, go to www.VOYDEYA.com or call 1-888-765-4747. |
Section 44425-7
Store and dispense in the original container at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP controlled room temperature].
10 Overdosage
Serum ALT elevations occurred after treatment cessation without a taper in 2 healthy subjects who received danicopan 500 mg and 800 mg twice a day. These abnormal ALT findings were transient, with no evidence of hepatic function abnormality and resolved spontaneously. In case of overdose, elevations in liver enzymes may occur. General supportive measures are recommended. It is not known if VOYDEYA can be removed by dialysis.
11 Description
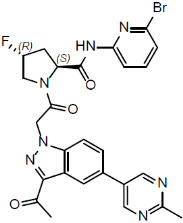
Danicopan is a small molecule complement Factor D inhibitor. Its chemical name is (2S,4R)-1-{[3-acetyl-5-(2-methylpyrimidin-5-yl)-1H-indazol-1-yl] acetyl}-N-(6-bromopyridin-2-yl)-4-fluoropyrrolidine-2-carboxamide. Its molecular formula is C26H23BrFN7O3 and its molecular weight is 580.4. Danicopan has the following structural formula:
Danicopan is a white/off-white to pale yellow powder. In aqueous solutions, danicopan is considered slightly soluble at pH 1.2 and insoluble from pH 4 to pH 7.
Danicopan tablets are available as white to off-white, round, film-coated, immediate release tablets in strengths of 50 mg and 100 mg, intended for oral administration. Each tablet contains the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. The tablet coating components are polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
5.2 Voydeya Rems
VOYDEYA is available only through a restricted program under a REMS called VOYDEYA REMS, because of the risk of serious infections caused by encapsulated bacteria [see Warnings and Precautions (5.1)].
Notable requirements of the VOYDEYA REMS include the following:
- Prescribers must enroll in the REMS.
- Prescribers must counsel patients about the risk of serious infections caused by encapsulated bacteria.
- Prescribers must provide patients with the REMS educational materials.
- Prescribers must assess patient vaccination status for vaccines against encapsulated bacteria and vaccinate if needed according to current ACIP recommendations two weeks prior to the first dose of VOYDEYA.
- Prescribers must provide a prescription for antibacterial drug prophylaxis if treatment must be started urgently, and the patient is not up to date with vaccines against encapsulated bacteria according to current ACIP recommendations at least two weeks prior to the first dose of VOYDEYA.
- Pharmacies that dispense VOYDEYA must be certified in the VOYDEYA REMS and must verify prescribers are certified.
- Patients must receive counseling from the prescriber about the need to receive vaccinations against encapsulated bacteria per ACIP recommendations, the need to take antibiotics as directed by the prescriber, and the early signs and symptoms of serious infections.
- Patients must be instructed to carry the Patient Safety Card with them at all times during treatment and for 1 week following the last dose of VOYDEYA.
Further information is available by telephone: 1-888-765-4747 or online at www.VoydeyaREMS.com.
8.4 Pediatric Use
Safety and effectiveness of VOYDEYA for the treatment of PNH in pediatric patients have not been established.
8.5 Geriatric Use
There were 22 patients 65 years of age and older in the clinical studies for PNH [see Clinical Studies (14)]. Of the total number of VOYDEYA-treated patients in these studies, 16 (28.1%) were 65 years of age and older, and 7 (12.3%) were 75 years of age and older. Clinical studies of VOYDEYA did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects.
5.5 Hyperlipidemia
VOYDEYA increases total cholesterol and LDL-cholesterol.
Of the 50 VOYDEYA-treated patients who had a normal total cholesterol level at baseline in Study ALXN2040-PNH-301, 30% developed Grade 1 hypercholesterolemia. Of the 6 VOYDEYA treated patients who had Grade 1 hypercholesterolemia at baseline in Study ALXN2040-PNH-301, 1 patient experienced increased total cholesterol that worsened to Grade 2. Of the 54 VOYDEYA-treated patients who had LDL-cholesterol ≤130 mg/dL at baseline in Study ALXN2040-PNH-301, 13% developed LDL-cholesterol >130-160 mg/dL and 9% developed LDL-cholesterol >160-190 mg/dL.
Some patients required cholesterol-lowering medications.
Monitor serum lipid parameters periodically during treatment with VOYDEYA and initiate cholesterol lowering medication, if indicated.
4 Contraindications
VOYDEYA is contraindicated for initiation in patients with unresolved serious infection caused by encapsulated bacteria, including Neisseria meningitidis, Streptococcus pneumoniae, or Haemophilus influenzae type B [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Serious Infections Caused by Encapsulated Bacteria [see Warnings and Precautions (5.1)]
- Hepatic Enzyme Increases [see Warnings and Precautions (5.3)]
- Hyperlipidemia [see Warnings and Precautions (5.4)]
7 Drug Interactions
- BCRP substrates: Monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. For rosuvastatin, the dose should not exceed 10 mg once daily (7.1).
- P-gp substrates: Dose adjustment might be necessary for P-gp substrates where minimal concentration changes may lead to serious adverse reactions (7.2).
7.1 Bcrp Substrates
Danicopan is a Breast Cancer Resistance Protein (BCRP) inhibitor. Concomitant use of VOYDEYA with a BCRP substrate increases the plasma concentrations of the BCRP substrate [see Clinical Pharmacology (12.3)], which may increase the risk for adverse reactions associated with the BCRP substrate. If used together, monitor patients more frequently for adverse reactions associated with the BCRP substrate, and consider dose reduction of the BCRP substrate according to its prescribing information.
7.2 P Gp Substrates
Danicopan is an inhibitor of P-glycoprotein (P-gp). Concomitant administration of VOYDEYA with a P-gp substrate may increase the plasma concentration of the P-gp substrate. Dose adjustment might be necessary for P-gp substrates where minimal concentration changes may lead to serious adverse reactions [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Danicopan inhibits the AP of the complement system, as demonstrated by the decrease in ex vivo serum AP activity and in vivo plasma Bb concentration. Danicopan also reduces complement C3 fragment deposition on circulating red blood cells (RBCs) in PNH patients.
In patients with PNH undergoing treatment with ravulizumab or eculizumab, co-administration of VOYDEYA from 150 mg three times a day to 200 mg three times a day inhibited AP activity by >90%. Additionally, plasma Bb levels decreased by about 50% and the fraction of circulating PNH RBCs with measured C3 fragment deposition decreased by over 50%.
12.3 Pharmacokinetics
At the recommended dosages of 150 or 200 mg three times a day, the median systemic exposure of danicopan at steady state has a maximum plasma concentration (Cmax,ss) of 535 or 665 ng/mL, respectively, and has an area under the plasma drug concentration time curve (AUC24,ss) of 8180 or 10200 ng × h/mL, respectively.
Danicopan exposures at steady state generally increase in a dose-proportional manner from 150 mg three times a day to 200 mg three times a day. Danicopan systemic exposure reaches steady state in approximately 2 days. An approximately 2-fold accumulation of danicopan exposure is expected at steady state following thrice daily dosing compared to a single dose.
8.6 Hepatic Impairment
No dose adjustment is required in patients with mild to moderate hepatic impairment (Child-Pugh Class A and B). Studies have not been conducted in patients with severe hepatic impairment, therefore, avoid use of VOYDEYA in this patient population [see Warnings and Precautions (5.3)].
1 Indications and Usage
VOYDEYA is indicated as add-on therapy to ravulizumab or eculizumab for the treatment of extravascular hemolysis (EVH) in adults with paroxysmal nocturnal hemoglobinuria (PNH).
12.1 Mechanism of Action
Danicopan binds reversibly to complement Factor D and selectively inhibits the alternative complement pathway. Danicopan prevents the cleavage of complement Factor B into the Ba and Bb fragments which are required for the formation of the alternative pathway (AP) complement component C3 convertase (C3bBb), the generation of downstream effectors including C3 fragment opsonization, and the amplification of the terminal pathway.
In PNH, intravascular hemolysis (IVH) is mediated by the terminal membrane attack complex (MAC), while extravascular hemolysis (EVH) is facilitated by C3 fragment opsonization. Danicopan acts proximally in the alternative pathway of the complement cascade to control preferentially C3 fragment-mediated EVH, while co-administered ravulizumab or eculizumab is anticipated to maintain control over MAC-mediated IVH.
5 Warnings and Precautions
- Hepatic Enzyme Increases: Assess liver enzymes before treatment initiation and periodically during treatment. Consider treatment interruption or discontinuation if elevations are clinically significant or if the patient becomes symptomatic (5.3).
- Hyperlipidemia: Monitor serum lipids periodically during treatment and initiate cholesterol-lowering medication if indicated (5.4).
2 Dosage and Administration
3 Dosage Forms and Strengths
Tablets: 50 mg and 100 mg (3)
5.3 Hepatic Enzyme Increases
Hepatic enzyme elevations have been observed in patients treated with VOYDEYA [see Adverse Reactions (6.1)]. Fourteen percent of patients receiving VOYDEYA in Study ALXN2040-PNH-301 had elevations in serum alanine aminotransferase (ALT). ALT elevations > 3 × the upper limit of normal (ULN) and ≤ 5 × ULN occurred in 9% of VOYDEYA-treated patients, and ALT elevations > 5 × ULN and ≤ 10 × ULN occurred in 5% of VOYDEYA-treated patients.
Assess liver enzyme test results prior to the initiation of VOYDEYA and periodically during treatment. Consider treatment interruption or discontinuation if elevations are clinically significant or if the patient becomes symptomatic. VOYDEYA has not been studied in patients with severe hepatic impairment [see Use in Specific Populations (8.7)].
8 Use in Specific Populations
Hepatic Impairment: Avoid use in patients with severe hepatic impairment (Child-Pugh C) (8.6).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of VOYDEYA was evaluated in 86 adults with PNH in Study ALXN2040-PNH-301 [see Clinical Studies (14)]. Study ALXN2040-PNH-301 enrolled adults with PNH with clinically significant EVH who had been treated with a stable dose of ravulizumab or eculizumab for at least the previous 6 months. Patients were randomly assigned 2:1 to receive double-blind VOYDEYA 150 mg (n=57) or placebo (n=29) orally three times a day in addition to ravulizumab or eculizumab for 12 weeks. Patients received either US-approved or non-US-approved ravulizumab or eculizumab in the trial. Among patients who were randomized to receive VOYDEYA, 84% were exposed for at least 12 weeks.
Serious adverse reactions were reported in 5% of patients who received VOYDEYA and included pancreatitis, cholecystitis, and blood bilirubin increased. No specific serious adverse reaction was reported in more than 1 patient treated with VOYDEYA.
Permanent discontinuation of VOYDEYA due to an adverse reaction occurred in 5% of patients and included 1 patient with blood bilirubin increase and pancreatitis, 1 patient with hepatic enzyme increased, and 1 patient with ALT increased and aspartate aminotransferase increased. Dosage reduction due to an adverse reaction occurred in 1 patient and the adverse reaction was COVID-19.
Among the 57 patients treated with VOYDEYA in Study ALXN2040-PNH-301, the most common adverse reaction (≥10%) was headache.
Table 1 describes adverse reactions reported in ≥5% of patients treated with VOYDEYA and greater than placebo in the randomized, controlled period of Study ALXN2040-PNH-301.
| Adverse Reactions Common Toxicity Criteria Adverse Events (CTCAE)
|
VOYDEYA (with ravulizumab or eculizumab) N = 57 |
Placebo (ravulizumab or eculizumab only) N = 29 |
|---|---|---|
| n (%) | n (%) | |
| Headache | 6 (11) | 3 (10) |
| Vomiting Represents a composite of multiple, related adverse reactions
|
4 (7) | 0 (0) |
| Pyrexia | 4 (7) | 0 (0) |
| Alanine aminotransferase increased | 3 (5) | 1 (3) |
| Hypertension | 3 (5) | 1 (3) |
| Pain in extremity | 3 (5) | 0 (0) |
Clinically relevant adverse reactions in <5% of patients include increased serum triglycerides.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
VOYDEYA (danicopan) tablets are available in the doses and packages listed in Table 4.
| Dose | Tablet Strength | Film-Coated Tablet Markings | Tablet Color/Shape | Pack Size | NDC Code |
|---|---|---|---|---|---|
| Each carton contains two high density polyethylene bottles with desiccant and child resistant seal, with 180 tablets per carton: | |||||
| 150 mg | 50 mg | Debossed on one side with "DCN 50" | White to off-white, round film-coated tablets | One bottle with 90 × 50 mg per tablet (25682-040-90) One bottle with 90 × 100 mg per tablet (25682-043-90) |
25682-046-92 |
| 100 mg | Debossed on one side with "DCN 100" | ||||
| 200 mg | 100 mg | Debossed on one side with "DCN 100" | White to off-white, round film-coated tablets | Two bottles with 90 tablets per bottle: 90 × 100 mg per tablet (25682-043-90) |
25682-043-92 |
| Each carton contains four 7-day blister cards with 168 tablets per carton: | |||||
| 150 mg | 50 mg | Debossed on one side with "DCN 50" | White to off-white, round film-coated tablets | Four blister cards with 42 tablets per card: 21 × 50 mg per tablet and 21 × 100 mg per tablet (25682-049-42) |
25682-049-04 |
| 100 mg | Debossed on one side with "DCN 100" | ||||
| 200 mg | 100 mg | Debossed on one side with "DCN 100" | White to off-white, round film-coated tablets | Four blister cards with 42 tablets per card: 42 × 100 mg per tablet (25682-043-42) |
25682-043-04 |
13.2 Animal Toxicology And/or Pharmacology
Ocular phototoxicity was observed in pigmented rats at systemic exposures 15-times and 28-times the exposure at the MRHD (based on AUC and Cmax, respectively). As danicopan is expected to accumulate in the eye, a risk of developing ocular phototoxicity cannot be excluded in patients on long-term danicopan therapy who are exposed to unprotected ultraviolet radiation for extended periods of time. The clinical significance of these findings is unknown.
Principal Display Panel Kit Carton 046
NDC 25682-046-92
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
50 mg
and
100 mg
Take one 100 mg tablet and one 50 mg tablet
three times a day
(150 mg dose three times a day)
180 tablets
90 x 50 mg tablets per bottle
90 x 100 mg tablets per bottle
Principal Display Panel Kit Carton 049
NDC 25682-049-04
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
50 mg per tablet
and
100 mg per tablet
Take one 100 mg tablet and one 50 mg tablet
three times a day
(150 mg dose three times a day)
Four 7-day blister cards with 42 tablets per card (168 tablets)
21 x 50 mg per tablet
21 x 100 mg per tablet
5.1 Serious Infections Caused By Encapsulated Bacteria
VOYDEYA, a complement inhibitor, increases a patient's susceptibility to serious, life-threatening, or fatal infections caused by encapsulated bacteria including Neisseria meningitidis (caused by any serogroup, including non-groupable strains), Streptococcus pneumoniae, and Haemophilus influenzae type B. Life-threatening and fatal infections with encapsulated bacteria have occurred in both vaccinated and unvaccinated patients treated with complement inhibitors. The initiation of VOYDEYA treatment is contraindicated in patients with unresolved serious infections caused by encapsulated bacteria.
Complete or update vaccination against encapsulated bacteria, specifically Neisseria meningitidis and Streptococcus pneumoniae at least 2 weeks prior to administration of the first dose of VOYDEYA, according to the current ACIP recommendations for patients receiving a complement inhibitor. Revaccinate patients in accordance with ACIP recommendations considering the duration of therapy with VOYDEYA. Note that ACIP recommends an administration schedule in patients receiving complement inhibitors that differs from the administration schedule in the vaccine prescribing information. If urgent VOYDEYA therapy is indicated in a patient who is not up to date with vaccines against encapsulated bacteria according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer these vaccines as soon as possible. Various durations and regimens of antibacterial drug prophylaxis have been considered, but the optimal durations and drug regimens for prophylaxis and their efficacy have not been studied in unvaccinated or vaccinated patients receiving complement inhibitors, including VOYDEYA. The benefits and risks of treatment with VOYDEYA, as well as the benefits and risks of antibacterial drug prophylaxis in unvaccinated or vaccinated patients, must be considered against the known risks for serious infections caused by encapsulated bacteria.
Vaccination does not eliminate the risk of serious encapsulated bacterial infections, despite development of antibodies following vaccination. Closely monitor patients for early signs and symptoms of serious infection and evaluate patients immediately if an infection is suspected. Inform patients of these signs and symptoms and instruct patients to seek immediate medical care if these signs and symptoms occur. Promptly treat known infections. Serious infection may become rapidly life-threatening or fatal if not recognized and treated early. Consider interruption of VOYDEYA in patients who are undergoing treatment for serious infections.
VOYDEYA is available only through a restricted program under a REMS [see Warnings and Precautions (5.2)].
Warning: Serious Infections Caused By Encapsulated Bacteria
VOYDEYA, a complement inhibitor, increases the risk of serious infections, especially those caused by encapsulated bacteria, such as Neisseria meningitidis, Streptococcus pneumoniae, and Haemophilus influenzae type B [see Warnings and Precautions (5.1) ]. Life-threatening and fatal infections with encapsulated bacteria have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early.
- Complete or update vaccination for encapsulated bacteria specifically, Neisseria meningitidis and Streptococcus pneumoniae at least 2 weeks prior to the first dose of VOYDEYA, unless the risks of delaying therapy with VOYDEYA outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against encapsulated bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by encapsulated bacteria.
- Patients receiving VOYDEYA are at increased risk for invasive disease caused by encapsulated bacteria, even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious infections and evaluate immediately if infection is suspected.
Because of the risk of serious infections caused by encapsulated bacteria, VOYDEYA is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the VOYDEYA REMS [see Warnings and Precautions (5.2)].
Principal Display Panel 100 Mg Tablet Bottle Carton 043 92
NDC 25682-043-92
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
100 mg
180 tablets
90 x 100 mg tablets per bottle
5.4 Monitoring of Pnh Manifestations After Voydeya Discontinuation
After discontinuing treatment with VOYDEYA, closely monitor patients for at least 2 weeks after the last dose for signs and symptoms of hemolysis. If discontinuation of VOYDEYA is necessary, continue background treatment with ravulizumab or eculizumab or consider alternative therapy if necessary. The signs and symptoms of hemolysis may include a sudden decrease in hemoglobin or fatigue.
If hemolysis occurs after discontinuation of VOYDEYA, consider restarting treatment with VOYDEYA if appropriate.
Principal Display Panel 100 Mg Tablet Blister Pack Carton 043 04
NDC 25682-043-04
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
100 mg per tablet
Take two 100 mg tablets three times a day
(200 mg dose three times a day)
Four 7-day blister cards with 42 tablets per card (168 tablets)
2.1 Recommended Vaccination and Prophylaxis for Encapsulated Bacterial Infections
Vaccinate patients against encapsulated bacteria, including Neisseria meningitidis (serogroups A, C, W, Y, and B) and Streptococcus pneumoniae according to current ACIP recommendations at least 2 weeks prior to initiation of VOYDEYA.
If urgent VOYDEYA therapy is indicated in a patient who is not up to date with vaccines for Neisseria meningitidis and Streptococcus pneumoniae according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer these vaccines as soon as possible [see Warnings and Precautions (5.1)].
Healthcare professionals who prescribe VOYDEYA must enroll in the VOYDEYA REMS [see Warnings and Precautions (5.2)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
VOYDEYA has not been shown to be effective as monotherapy and should only be prescribed as an add-on to ravulizumab or eculizumab.
Section 42231-1 (42231-1)
| MEDICATION GUIDE VOYDEYA™(voi-day-uh) (danicopan) tablets, for oral use |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Issued: 03/2024 | |
| What is the most important information I should know about VOYDEYA? | ||
VOYDEYA is a medicine that affects your immune system. VOYDEYA may lower the ability of your immune system to fight infections.
|
||
|
|
|
Your healthcare provider will give you a Patient Safety Card about the risk of serious infections. Carry it with you at all times during treatment and for 1 week after your last VOYDEYA dose. Your risk of serious infections may continue for a few days after your last dose of VOYDEYA. If you get any of the symptoms listed on this card you should get medical help right away. It is important to show this card to any healthcare provider who treats you. This will help them diagnose and treat you quickly.
|
||
| For more information about side effects, see " What are the possible side effects of VOYDEYA? " | ||
| What is VOYDEYA? | ||
| VOYDEYA is a prescription medicine used along with ravulizumab or eculizumab to treat breakdown of red blood cells that takes place outside of blood vessels (extravascular hemolysis), in adults with paroxysmal nocturnal hemoglobinuria (PNH). It is not known if VOYDEYA is safe and effective in children. |
||
| Who should not take VOYDEYA? | ||
| Do not take VOYDEYA if you have a serious infection caused by encapsulated bacteria, including Neisseria meningitidis, Streptococcus pneumoniae, or Haemophilus influenzae type B when you are starting VOYDEYA treatment. | ||
Before taking VOYDEYA, tell your healthcare provider about all of your medical conditions, including if you:
|
||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. VOYDEYA may affect the way other medicines work. | ||
| Know the medicines you take and the vaccines you receive. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. | ||
How should I take VOYDEYA?
|
||
| What are the possible side effects of VOYDEYA? | ||
VOYDEYA may cause side effects, including:
|
||
| The most common side effect of VOYDEYA is headache. | ||
| Tell your healthcare provider about any side effect that bothers you or that does not go away. | ||
| These are not all of the possible side effects of VOYDEYA. | ||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
How should I store VOYDEYA?
|
||
| General information about the safe and effective use of VOYDEYA. | ||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VOYDEYA for a condition for which it was not prescribed. Do not give VOYDEYA to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VOYDEYA that is written for health professionals. | ||
| What are the ingredients in VOYDEYA? | ||
| Active ingredient: danicopan | ||
| Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. | ||
| The film coatings contain the following inactive ingredients: polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. | ||
| Manufactured for: Alexion Pharmaceuticals, Inc., 121 Seaport Boulevard, Boston, MA 02210 USA VOYDEYA is a trademark of Alexion Pharmaceuticals, Inc. © 2024 Alexion Pharmaceuticals, Inc For more information, go to www.VOYDEYA.com or call 1-888-765-4747. |
Section 44425-7 (44425-7)
Store and dispense in the original container at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP controlled room temperature].
10 Overdosage (10 OVERDOSAGE)
Serum ALT elevations occurred after treatment cessation without a taper in 2 healthy subjects who received danicopan 500 mg and 800 mg twice a day. These abnormal ALT findings were transient, with no evidence of hepatic function abnormality and resolved spontaneously. In case of overdose, elevations in liver enzymes may occur. General supportive measures are recommended. It is not known if VOYDEYA can be removed by dialysis.
11 Description (11 DESCRIPTION)
Danicopan is a small molecule complement Factor D inhibitor. Its chemical name is (2S,4R)-1-{[3-acetyl-5-(2-methylpyrimidin-5-yl)-1H-indazol-1-yl] acetyl}-N-(6-bromopyridin-2-yl)-4-fluoropyrrolidine-2-carboxamide. Its molecular formula is C26H23BrFN7O3 and its molecular weight is 580.4. Danicopan has the following structural formula:
Danicopan is a white/off-white to pale yellow powder. In aqueous solutions, danicopan is considered slightly soluble at pH 1.2 and insoluble from pH 4 to pH 7.
Danicopan tablets are available as white to off-white, round, film-coated, immediate release tablets in strengths of 50 mg and 100 mg, intended for oral administration. Each tablet contains the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. The tablet coating components are polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
5.2 Voydeya Rems (5.2 VOYDEYA REMS)
VOYDEYA is available only through a restricted program under a REMS called VOYDEYA REMS, because of the risk of serious infections caused by encapsulated bacteria [see Warnings and Precautions (5.1)].
Notable requirements of the VOYDEYA REMS include the following:
- Prescribers must enroll in the REMS.
- Prescribers must counsel patients about the risk of serious infections caused by encapsulated bacteria.
- Prescribers must provide patients with the REMS educational materials.
- Prescribers must assess patient vaccination status for vaccines against encapsulated bacteria and vaccinate if needed according to current ACIP recommendations two weeks prior to the first dose of VOYDEYA.
- Prescribers must provide a prescription for antibacterial drug prophylaxis if treatment must be started urgently, and the patient is not up to date with vaccines against encapsulated bacteria according to current ACIP recommendations at least two weeks prior to the first dose of VOYDEYA.
- Pharmacies that dispense VOYDEYA must be certified in the VOYDEYA REMS and must verify prescribers are certified.
- Patients must receive counseling from the prescriber about the need to receive vaccinations against encapsulated bacteria per ACIP recommendations, the need to take antibiotics as directed by the prescriber, and the early signs and symptoms of serious infections.
- Patients must be instructed to carry the Patient Safety Card with them at all times during treatment and for 1 week following the last dose of VOYDEYA.
Further information is available by telephone: 1-888-765-4747 or online at www.VoydeyaREMS.com.
8.4 Pediatric Use
Safety and effectiveness of VOYDEYA for the treatment of PNH in pediatric patients have not been established.
8.5 Geriatric Use
There were 22 patients 65 years of age and older in the clinical studies for PNH [see Clinical Studies (14)]. Of the total number of VOYDEYA-treated patients in these studies, 16 (28.1%) were 65 years of age and older, and 7 (12.3%) were 75 years of age and older. Clinical studies of VOYDEYA did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects.
5.5 Hyperlipidemia
VOYDEYA increases total cholesterol and LDL-cholesterol.
Of the 50 VOYDEYA-treated patients who had a normal total cholesterol level at baseline in Study ALXN2040-PNH-301, 30% developed Grade 1 hypercholesterolemia. Of the 6 VOYDEYA treated patients who had Grade 1 hypercholesterolemia at baseline in Study ALXN2040-PNH-301, 1 patient experienced increased total cholesterol that worsened to Grade 2. Of the 54 VOYDEYA-treated patients who had LDL-cholesterol ≤130 mg/dL at baseline in Study ALXN2040-PNH-301, 13% developed LDL-cholesterol >130-160 mg/dL and 9% developed LDL-cholesterol >160-190 mg/dL.
Some patients required cholesterol-lowering medications.
Monitor serum lipid parameters periodically during treatment with VOYDEYA and initiate cholesterol lowering medication, if indicated.
4 Contraindications (4 CONTRAINDICATIONS)
VOYDEYA is contraindicated for initiation in patients with unresolved serious infection caused by encapsulated bacteria, including Neisseria meningitidis, Streptococcus pneumoniae, or Haemophilus influenzae type B [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Serious Infections Caused by Encapsulated Bacteria [see Warnings and Precautions (5.1)]
- Hepatic Enzyme Increases [see Warnings and Precautions (5.3)]
- Hyperlipidemia [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- BCRP substrates: Monitor patients more frequently for adverse reactions and consider dose reduction of the BCRP substrate drug. For rosuvastatin, the dose should not exceed 10 mg once daily (7.1).
- P-gp substrates: Dose adjustment might be necessary for P-gp substrates where minimal concentration changes may lead to serious adverse reactions (7.2).
7.1 Bcrp Substrates (7.1 BCRP Substrates)
Danicopan is a Breast Cancer Resistance Protein (BCRP) inhibitor. Concomitant use of VOYDEYA with a BCRP substrate increases the plasma concentrations of the BCRP substrate [see Clinical Pharmacology (12.3)], which may increase the risk for adverse reactions associated with the BCRP substrate. If used together, monitor patients more frequently for adverse reactions associated with the BCRP substrate, and consider dose reduction of the BCRP substrate according to its prescribing information.
7.2 P Gp Substrates (7.2 P-gp Substrates)
Danicopan is an inhibitor of P-glycoprotein (P-gp). Concomitant administration of VOYDEYA with a P-gp substrate may increase the plasma concentration of the P-gp substrate. Dose adjustment might be necessary for P-gp substrates where minimal concentration changes may lead to serious adverse reactions [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Danicopan inhibits the AP of the complement system, as demonstrated by the decrease in ex vivo serum AP activity and in vivo plasma Bb concentration. Danicopan also reduces complement C3 fragment deposition on circulating red blood cells (RBCs) in PNH patients.
In patients with PNH undergoing treatment with ravulizumab or eculizumab, co-administration of VOYDEYA from 150 mg three times a day to 200 mg three times a day inhibited AP activity by >90%. Additionally, plasma Bb levels decreased by about 50% and the fraction of circulating PNH RBCs with measured C3 fragment deposition decreased by over 50%.
12.3 Pharmacokinetics
At the recommended dosages of 150 or 200 mg three times a day, the median systemic exposure of danicopan at steady state has a maximum plasma concentration (Cmax,ss) of 535 or 665 ng/mL, respectively, and has an area under the plasma drug concentration time curve (AUC24,ss) of 8180 or 10200 ng × h/mL, respectively.
Danicopan exposures at steady state generally increase in a dose-proportional manner from 150 mg three times a day to 200 mg three times a day. Danicopan systemic exposure reaches steady state in approximately 2 days. An approximately 2-fold accumulation of danicopan exposure is expected at steady state following thrice daily dosing compared to a single dose.
8.6 Hepatic Impairment
No dose adjustment is required in patients with mild to moderate hepatic impairment (Child-Pugh Class A and B). Studies have not been conducted in patients with severe hepatic impairment, therefore, avoid use of VOYDEYA in this patient population [see Warnings and Precautions (5.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
VOYDEYA is indicated as add-on therapy to ravulizumab or eculizumab for the treatment of extravascular hemolysis (EVH) in adults with paroxysmal nocturnal hemoglobinuria (PNH).
12.1 Mechanism of Action
Danicopan binds reversibly to complement Factor D and selectively inhibits the alternative complement pathway. Danicopan prevents the cleavage of complement Factor B into the Ba and Bb fragments which are required for the formation of the alternative pathway (AP) complement component C3 convertase (C3bBb), the generation of downstream effectors including C3 fragment opsonization, and the amplification of the terminal pathway.
In PNH, intravascular hemolysis (IVH) is mediated by the terminal membrane attack complex (MAC), while extravascular hemolysis (EVH) is facilitated by C3 fragment opsonization. Danicopan acts proximally in the alternative pathway of the complement cascade to control preferentially C3 fragment-mediated EVH, while co-administered ravulizumab or eculizumab is anticipated to maintain control over MAC-mediated IVH.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatic Enzyme Increases: Assess liver enzymes before treatment initiation and periodically during treatment. Consider treatment interruption or discontinuation if elevations are clinically significant or if the patient becomes symptomatic (5.3).
- Hyperlipidemia: Monitor serum lipids periodically during treatment and initiate cholesterol-lowering medication if indicated (5.4).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 50 mg and 100 mg (3)
5.3 Hepatic Enzyme Increases
Hepatic enzyme elevations have been observed in patients treated with VOYDEYA [see Adverse Reactions (6.1)]. Fourteen percent of patients receiving VOYDEYA in Study ALXN2040-PNH-301 had elevations in serum alanine aminotransferase (ALT). ALT elevations > 3 × the upper limit of normal (ULN) and ≤ 5 × ULN occurred in 9% of VOYDEYA-treated patients, and ALT elevations > 5 × ULN and ≤ 10 × ULN occurred in 5% of VOYDEYA-treated patients.
Assess liver enzyme test results prior to the initiation of VOYDEYA and periodically during treatment. Consider treatment interruption or discontinuation if elevations are clinically significant or if the patient becomes symptomatic. VOYDEYA has not been studied in patients with severe hepatic impairment [see Use in Specific Populations (8.7)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Hepatic Impairment: Avoid use in patients with severe hepatic impairment (Child-Pugh C) (8.6).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of VOYDEYA was evaluated in 86 adults with PNH in Study ALXN2040-PNH-301 [see Clinical Studies (14)]. Study ALXN2040-PNH-301 enrolled adults with PNH with clinically significant EVH who had been treated with a stable dose of ravulizumab or eculizumab for at least the previous 6 months. Patients were randomly assigned 2:1 to receive double-blind VOYDEYA 150 mg (n=57) or placebo (n=29) orally three times a day in addition to ravulizumab or eculizumab for 12 weeks. Patients received either US-approved or non-US-approved ravulizumab or eculizumab in the trial. Among patients who were randomized to receive VOYDEYA, 84% were exposed for at least 12 weeks.
Serious adverse reactions were reported in 5% of patients who received VOYDEYA and included pancreatitis, cholecystitis, and blood bilirubin increased. No specific serious adverse reaction was reported in more than 1 patient treated with VOYDEYA.
Permanent discontinuation of VOYDEYA due to an adverse reaction occurred in 5% of patients and included 1 patient with blood bilirubin increase and pancreatitis, 1 patient with hepatic enzyme increased, and 1 patient with ALT increased and aspartate aminotransferase increased. Dosage reduction due to an adverse reaction occurred in 1 patient and the adverse reaction was COVID-19.
Among the 57 patients treated with VOYDEYA in Study ALXN2040-PNH-301, the most common adverse reaction (≥10%) was headache.
Table 1 describes adverse reactions reported in ≥5% of patients treated with VOYDEYA and greater than placebo in the randomized, controlled period of Study ALXN2040-PNH-301.
| Adverse Reactions Common Toxicity Criteria Adverse Events (CTCAE)
|
VOYDEYA (with ravulizumab or eculizumab) N = 57 |
Placebo (ravulizumab or eculizumab only) N = 29 |
|---|---|---|
| n (%) | n (%) | |
| Headache | 6 (11) | 3 (10) |
| Vomiting Represents a composite of multiple, related adverse reactions
|
4 (7) | 0 (0) |
| Pyrexia | 4 (7) | 0 (0) |
| Alanine aminotransferase increased | 3 (5) | 1 (3) |
| Hypertension | 3 (5) | 1 (3) |
| Pain in extremity | 3 (5) | 0 (0) |
Clinically relevant adverse reactions in <5% of patients include increased serum triglycerides.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
VOYDEYA (danicopan) tablets are available in the doses and packages listed in Table 4.
| Dose | Tablet Strength | Film-Coated Tablet Markings | Tablet Color/Shape | Pack Size | NDC Code |
|---|---|---|---|---|---|
| Each carton contains two high density polyethylene bottles with desiccant and child resistant seal, with 180 tablets per carton: | |||||
| 150 mg | 50 mg | Debossed on one side with "DCN 50" | White to off-white, round film-coated tablets | One bottle with 90 × 50 mg per tablet (25682-040-90) One bottle with 90 × 100 mg per tablet (25682-043-90) |
25682-046-92 |
| 100 mg | Debossed on one side with "DCN 100" | ||||
| 200 mg | 100 mg | Debossed on one side with "DCN 100" | White to off-white, round film-coated tablets | Two bottles with 90 tablets per bottle: 90 × 100 mg per tablet (25682-043-90) |
25682-043-92 |
| Each carton contains four 7-day blister cards with 168 tablets per carton: | |||||
| 150 mg | 50 mg | Debossed on one side with "DCN 50" | White to off-white, round film-coated tablets | Four blister cards with 42 tablets per card: 21 × 50 mg per tablet and 21 × 100 mg per tablet (25682-049-42) |
25682-049-04 |
| 100 mg | Debossed on one side with "DCN 100" | ||||
| 200 mg | 100 mg | Debossed on one side with "DCN 100" | White to off-white, round film-coated tablets | Four blister cards with 42 tablets per card: 42 × 100 mg per tablet (25682-043-42) |
25682-043-04 |
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Ocular phototoxicity was observed in pigmented rats at systemic exposures 15-times and 28-times the exposure at the MRHD (based on AUC and Cmax, respectively). As danicopan is expected to accumulate in the eye, a risk of developing ocular phototoxicity cannot be excluded in patients on long-term danicopan therapy who are exposed to unprotected ultraviolet radiation for extended periods of time. The clinical significance of these findings is unknown.
Principal Display Panel Kit Carton 046 (PRINCIPAL DISPLAY PANEL - Kit Carton - 046)
NDC 25682-046-92
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
50 mg
and
100 mg
Take one 100 mg tablet and one 50 mg tablet
three times a day
(150 mg dose three times a day)
180 tablets
90 x 50 mg tablets per bottle
90 x 100 mg tablets per bottle
Principal Display Panel Kit Carton 049 (PRINCIPAL DISPLAY PANEL - Kit Carton - 049)
NDC 25682-049-04
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
50 mg per tablet
and
100 mg per tablet
Take one 100 mg tablet and one 50 mg tablet
three times a day
(150 mg dose three times a day)
Four 7-day blister cards with 42 tablets per card (168 tablets)
21 x 50 mg per tablet
21 x 100 mg per tablet
5.1 Serious Infections Caused By Encapsulated Bacteria (5.1 Serious Infections Caused by Encapsulated Bacteria)
VOYDEYA, a complement inhibitor, increases a patient's susceptibility to serious, life-threatening, or fatal infections caused by encapsulated bacteria including Neisseria meningitidis (caused by any serogroup, including non-groupable strains), Streptococcus pneumoniae, and Haemophilus influenzae type B. Life-threatening and fatal infections with encapsulated bacteria have occurred in both vaccinated and unvaccinated patients treated with complement inhibitors. The initiation of VOYDEYA treatment is contraindicated in patients with unresolved serious infections caused by encapsulated bacteria.
Complete or update vaccination against encapsulated bacteria, specifically Neisseria meningitidis and Streptococcus pneumoniae at least 2 weeks prior to administration of the first dose of VOYDEYA, according to the current ACIP recommendations for patients receiving a complement inhibitor. Revaccinate patients in accordance with ACIP recommendations considering the duration of therapy with VOYDEYA. Note that ACIP recommends an administration schedule in patients receiving complement inhibitors that differs from the administration schedule in the vaccine prescribing information. If urgent VOYDEYA therapy is indicated in a patient who is not up to date with vaccines against encapsulated bacteria according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer these vaccines as soon as possible. Various durations and regimens of antibacterial drug prophylaxis have been considered, but the optimal durations and drug regimens for prophylaxis and their efficacy have not been studied in unvaccinated or vaccinated patients receiving complement inhibitors, including VOYDEYA. The benefits and risks of treatment with VOYDEYA, as well as the benefits and risks of antibacterial drug prophylaxis in unvaccinated or vaccinated patients, must be considered against the known risks for serious infections caused by encapsulated bacteria.
Vaccination does not eliminate the risk of serious encapsulated bacterial infections, despite development of antibodies following vaccination. Closely monitor patients for early signs and symptoms of serious infection and evaluate patients immediately if an infection is suspected. Inform patients of these signs and symptoms and instruct patients to seek immediate medical care if these signs and symptoms occur. Promptly treat known infections. Serious infection may become rapidly life-threatening or fatal if not recognized and treated early. Consider interruption of VOYDEYA in patients who are undergoing treatment for serious infections.
VOYDEYA is available only through a restricted program under a REMS [see Warnings and Precautions (5.2)].
Warning: Serious Infections Caused By Encapsulated Bacteria (WARNING: SERIOUS INFECTIONS CAUSED BY ENCAPSULATED BACTERIA)
VOYDEYA, a complement inhibitor, increases the risk of serious infections, especially those caused by encapsulated bacteria, such as Neisseria meningitidis, Streptococcus pneumoniae, and Haemophilus influenzae type B [see Warnings and Precautions (5.1) ]. Life-threatening and fatal infections with encapsulated bacteria have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early.
- Complete or update vaccination for encapsulated bacteria specifically, Neisseria meningitidis and Streptococcus pneumoniae at least 2 weeks prior to the first dose of VOYDEYA, unless the risks of delaying therapy with VOYDEYA outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against encapsulated bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by encapsulated bacteria.
- Patients receiving VOYDEYA are at increased risk for invasive disease caused by encapsulated bacteria, even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious infections and evaluate immediately if infection is suspected.
Because of the risk of serious infections caused by encapsulated bacteria, VOYDEYA is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the VOYDEYA REMS [see Warnings and Precautions (5.2)].
Principal Display Panel 100 Mg Tablet Bottle Carton 043 92 (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Carton - 043-92)
NDC 25682-043-92
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
100 mg
180 tablets
90 x 100 mg tablets per bottle
5.4 Monitoring of Pnh Manifestations After Voydeya Discontinuation (5.4 Monitoring of PNH Manifestations After VOYDEYA Discontinuation)
After discontinuing treatment with VOYDEYA, closely monitor patients for at least 2 weeks after the last dose for signs and symptoms of hemolysis. If discontinuation of VOYDEYA is necessary, continue background treatment with ravulizumab or eculizumab or consider alternative therapy if necessary. The signs and symptoms of hemolysis may include a sudden decrease in hemoglobin or fatigue.
If hemolysis occurs after discontinuation of VOYDEYA, consider restarting treatment with VOYDEYA if appropriate.
Principal Display Panel 100 Mg Tablet Blister Pack Carton 043 04 (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Blister Pack Carton - 043-04)
NDC 25682-043-04
Rx only
Voydeya™
(danicopan) tablets
Dispense with enclosed Medication Guide.
100 mg per tablet
Take two 100 mg tablets three times a day
(200 mg dose three times a day)
Four 7-day blister cards with 42 tablets per card (168 tablets)
2.1 Recommended Vaccination and Prophylaxis for Encapsulated Bacterial Infections
Vaccinate patients against encapsulated bacteria, including Neisseria meningitidis (serogroups A, C, W, Y, and B) and Streptococcus pneumoniae according to current ACIP recommendations at least 2 weeks prior to initiation of VOYDEYA.
If urgent VOYDEYA therapy is indicated in a patient who is not up to date with vaccines for Neisseria meningitidis and Streptococcus pneumoniae according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer these vaccines as soon as possible [see Warnings and Precautions (5.1)].
Healthcare professionals who prescribe VOYDEYA must enroll in the VOYDEYA REMS [see Warnings and Precautions (5.2)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:06.439243 · Updated: 2026-03-14T22:18:42.758225