These Highlights Do Not Include All The Information Needed To Use Forteo Safely And Effectively. See Full Prescribing Information For Forteo.
aae667c5-381f-4f92-93df-2ed6158d07b0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
FORTEO is indicated: For the treatment of postmenopausal women with osteoporosis at high risk for fracture (defined herein as having a history of osteoporotic fracture or multiple risk factors for fracture) or who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, FORTEO reduces the risk of vertebral and nonvertebral fractures. To increase bone mass in men with primary or hypogonadal osteoporosis at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy. For the treatment of men and women with osteoporosis associated with sustained systemic glucocorticoid therapy (daily dosage equivalent to 5 mg or greater of prednisone) at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
Indications and Usage
FORTEO is indicated: For the treatment of postmenopausal women with osteoporosis at high risk for fracture (defined herein as having a history of osteoporotic fracture or multiple risk factors for fracture) or who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, FORTEO reduces the risk of vertebral and nonvertebral fractures. To increase bone mass in men with primary or hypogonadal osteoporosis at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy. For the treatment of men and women with osteoporosis associated with sustained systemic glucocorticoid therapy (daily dosage equivalent to 5 mg or greater of prednisone) at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
Dosage and Administration
Recommended dosage is 20 mcg subcutaneously once a day ( 2.1 ) Consider supplemental calcium and Vitamin D based on individual patient needs ( 2.1 ) Administer as a subcutaneous injection into the thigh or abdominal region ( 2.2 ) Administer initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur ( 2.2 ) Use of FORTEO for more than 2 years during a patient's lifetime should only be considered if a patient remains at or has returned to having a high risk for fracture ( 2.3 )
Warnings and Precautions
Osteosarcoma : Avoid use in patients with increased risk of osteosarcoma including patients with open epiphyses, metabolic bone diseases including Paget's disease, bone metastases or history of skeletal malignancies, prior external beam or implant radiation therapy involving the skeleton, and hereditary disorders predisposing to osteosarcoma. ( 5.1 ) Hypercalcemia and Cutaneous Calcification : Avoid in patients known to have an underlying hypercalcemic disorder. Discontinue in patients developing worsening of previously stable cutaneous calcification. ( 5.2 ) Risk of Urolithiasis : Consider the risk/benefit in patients with active or recent urolithiasis because of risk of exacerbation ( 5.3 ) Orthostatic Hypotension : Transient orthostatic hypotension may occur with initial doses of FORTEO ( 5.4 )
Contraindications
FORTEO is contraindicated in patients with hypersensitivity to teriparatide or to any of its excipients. Hypersensitivity reactions have included angioedema and anaphylaxis [see Adverse Reactions ( 6.3 )] .
Adverse Reactions
Most common adverse reactions (>10%) include: arthralgia, pain, and nausea ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
Digoxin: Transient hypercalcemia may predispose patients to digitalis toxicity ( 5.5 , 7.1 )
Storage and Handling
Store FORTEO under refrigeration at 2° to 8°C (36° to 46°F) at all times except when administering the product. Recap the delivery device (pen) when not in use to protect the cartridge from physical damage and light. When using FORTEO, minimize the time out of the refrigerator; deliver the dose immediately following removal from the refrigerator. Do not freeze. Do not use FORTEO if it has been frozen. Throw away the device 28 days after first use.
How Supplied
FORTEO (teriparatide injection) is a clear and colorless solution, available as single-patient-use prefilled delivery device (pen) in the following package size: 560 mcg/2.24 mL (250 mcg/mL) [intended to deliver 28 daily doses of 20 mcg] NDC 0002-9678-01 (MS8400).
Medication Information
Warnings and Precautions
Osteosarcoma : Avoid use in patients with increased risk of osteosarcoma including patients with open epiphyses, metabolic bone diseases including Paget's disease, bone metastases or history of skeletal malignancies, prior external beam or implant radiation therapy involving the skeleton, and hereditary disorders predisposing to osteosarcoma. ( 5.1 ) Hypercalcemia and Cutaneous Calcification : Avoid in patients known to have an underlying hypercalcemic disorder. Discontinue in patients developing worsening of previously stable cutaneous calcification. ( 5.2 ) Risk of Urolithiasis : Consider the risk/benefit in patients with active or recent urolithiasis because of risk of exacerbation ( 5.3 ) Orthostatic Hypotension : Transient orthostatic hypotension may occur with initial doses of FORTEO ( 5.4 )
Indications and Usage
FORTEO is indicated: For the treatment of postmenopausal women with osteoporosis at high risk for fracture (defined herein as having a history of osteoporotic fracture or multiple risk factors for fracture) or who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, FORTEO reduces the risk of vertebral and nonvertebral fractures. To increase bone mass in men with primary or hypogonadal osteoporosis at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy. For the treatment of men and women with osteoporosis associated with sustained systemic glucocorticoid therapy (daily dosage equivalent to 5 mg or greater of prednisone) at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
Dosage and Administration
Recommended dosage is 20 mcg subcutaneously once a day ( 2.1 ) Consider supplemental calcium and Vitamin D based on individual patient needs ( 2.1 ) Administer as a subcutaneous injection into the thigh or abdominal region ( 2.2 ) Administer initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur ( 2.2 ) Use of FORTEO for more than 2 years during a patient's lifetime should only be considered if a patient remains at or has returned to having a high risk for fracture ( 2.3 )
Contraindications
FORTEO is contraindicated in patients with hypersensitivity to teriparatide or to any of its excipients. Hypersensitivity reactions have included angioedema and anaphylaxis [see Adverse Reactions ( 6.3 )] .
Adverse Reactions
Most common adverse reactions (>10%) include: arthralgia, pain, and nausea ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
Digoxin: Transient hypercalcemia may predispose patients to digitalis toxicity ( 5.5 , 7.1 )
Storage and Handling
Store FORTEO under refrigeration at 2° to 8°C (36° to 46°F) at all times except when administering the product. Recap the delivery device (pen) when not in use to protect the cartridge from physical damage and light. When using FORTEO, minimize the time out of the refrigerator; deliver the dose immediately following removal from the refrigerator. Do not freeze. Do not use FORTEO if it has been frozen. Throw away the device 28 days after first use.
How Supplied
FORTEO (teriparatide injection) is a clear and colorless solution, available as single-patient-use prefilled delivery device (pen) in the following package size: 560 mcg/2.24 mL (250 mcg/mL) [intended to deliver 28 daily doses of 20 mcg] NDC 0002-9678-01 (MS8400).
Description
FORTEO is indicated: For the treatment of postmenopausal women with osteoporosis at high risk for fracture (defined herein as having a history of osteoporotic fracture or multiple risk factors for fracture) or who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, FORTEO reduces the risk of vertebral and nonvertebral fractures. To increase bone mass in men with primary or hypogonadal osteoporosis at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy. For the treatment of men and women with osteoporosis associated with sustained systemic glucocorticoid therapy (daily dosage equivalent to 5 mg or greater of prednisone) at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
Section 42229-5
Hypercalcemia
FORTEO has not been studied in patients with pre-existing hypercalcemia. FORTEO may cause hypercalcemia and may exacerbate hypercalcemia in patients with pre-existing hypercalcemia [see Adverse Reactions (6.1, 6.3)]. Avoid FORTEO in patients known to have an underlying hypercalcemic disorder, such as primary hyperparathyroidism.
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised: 11/2020 |
|
MEDICATION GUIDE
FORTEO ® (for-TAY-o) teriparatide injection for subcutaneous use |
|
| Read this Medication Guide before you start using FORTEO and each time you get a refill. There may be new information. Also, read the User Manual that comes with the FORTEO delivery device (pen) for information on how to use the device to inject your medicine the right way. This Medication Guide does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is the most important information I should know about FORTEO?
|
|
|
What is FORTEO?
FORTEO is a prescription medicine used to:
|
|
| It is not known if FORTEO is safe and effective in children. FORTEO should not be used in children and young adults whose bones are still growing. |
|
|
Who should not use FORTEO?
Do not use FORTEO if you:
|
|
| Symptoms of a serious allergic reaction of FORTEO may include swelling of the face, lips, tongue or throat that may cause difficulty in breathing or swallowing. Call your healthcare provider right away or get emergency medical help if you get any of these symptoms. | |
|
What should I tell my healthcare provider before using FORTEO?
Before you use FORTEO, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
How should I use FORTEO?
|
|
| If your healthcare provider recommends calcium and vitamin D supplements, you can take them at the same time you take FORTEO. | |
|
What are the possible side effects of FORTEO?
FORTEO may cause serious side effects including:
|
|
The most common side effects of FORTEO include:
|
|
| These are not all the possible side effects of FORTEO. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store FORTEO?
|
|
| Keep FORTEO and all medicines out of the reach of children. | |
|
General information about the safe and effective use of FORTEO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use FORTEO for a condition for which it was not prescribed. Do not give FORTEO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about FORTEO that is written for health professionals. |
|
|
What are the ingredients in FORTEO?
Active ingredient: teriparatide Inactive ingredients: glacial acetic acid, sodium acetate (anhydrous), mannitol, metacresol, and water for injection. In addition, hydrochloric acid solution 10% and/or sodium hydroxide solution 10% may have been added to adjust the product to pH 4. |
|
| For more information, go to www.FORTEO.com or call Lilly at 1-866-436-7836. Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA Copyright © 2002, 2020, Eli Lilly and Company. All rights reserved. FOR-0003-MG-20201116 |
Section 51945-4
PACKAGE LABEL – FORTEO 20 mcg per dose, 2.24 mL
Do NOT transfer contents to a syringe
ATTENTION PHARMACIST: Medication Guide and device User Manual for patient inside carton
NDC 0002-8400-01
MS8400
FORTEO®
teriparatide injection
20 mcg per dose (given once daily subcutaneously)
Each single-patient-use prefilled pen will deliver 28 subcutaneous doses.
560 mcg/ 2.24 mL (250 mcg/mL)
For Single-Patient-Use Only
REFRIGERATE / DO NOT FREEZE
For subcutaneous use / Rx only
Needles not included
Becton, Dickinson and Company pen needles are recommended for use with this device
www.forteo.com
Lilly
Section 59845-8
FORTEO® (for-TAY-o)
teriparatide injection
User Manual
Important: First read the Medication Guide that comes inside your FORTEO carton.
Before you use your new FORTEO delivery device, please read the entire front and back of this User Manual completely. Follow the directions carefully when using the FORTEO delivery device.
Do not share your delivery device or needles because infection or disease can be spread from one person to another.
The FORTEO delivery device contains 28 days of medicine. Throw away the FORTEO delivery device 28 days after first use, even if it is not completely empty. Do not inject more than one dose of FORTEO in the same day.
Do not transfer FORTEO to a syringe.
Wash your hands before every injection. Prepare the injection site as your healthcare provider instructed.
|
1
Pull off white cap |
Check the FORTEO delivery device label to make sure you have the right medicine and that it has not expired. Do not use if the FORTEO delivery device looks damaged, if the medicine in the cartridge is not clear and colorless, or if it has particles in it. |
|
2
Attach new needle |
||||
| Pull off paper tab. |
Push needle straight
onto medicine cartridge. |
Screw on needle clockwise until firmly attached. |
Pull off large needle cover and save it. |
|
3
Set dose |
|||
|
Pull out black injection button until it stops. If you cannot pull out the black injection button see Troubleshooting, Problem E, on back page. |
Check to make sure red stripe shows. |
Pull off small needle protector and throw away. |
|
4
Inject dose |
||
| Gently hold a fold of skin on your thigh or abdomen and insert needle straight into skin. |
Push in black injection button until it stops. Hold it in and count to 5 slowly. You must wait until the count of 5 to make sure you receive the correct dose. Then pull the needle from skin. |
| IMPORTANT | ||||
|
5
Confirm dose |
After completing the injection:
Once the needle is removed from the skin, take your thumb off the black injection button. Check to make sure the black injection button is all the way in. If the yellow shaft does not show, you have finished the injection steps the right way. |
You should NOT see any of the yellow shaft. If you do and have already injected the medicine, do not inject yourself a second time on the same day. Instead, you MUST reset the FORTEO delivery device (see Troubleshooting, Problem A, on back page). |
|
6
Remove needle |
||||
| Put large needle cover on needle. Do not try to put the needle cover back on with your hands. |
Unscrew the covered needle all the way by giving the large needle cover 3 to 5 counter-clockwise turns. |
Pull off needle and throw away in a puncture-resistant container. |
Push white cap back on. Right after use, place FORTEO delivery device in the refrigerator. |
For more information, or if you have any questions, turn to the back of this page.
|
|
FORTEO® (for-TAY-o) |
|
| Troubleshooting | ||
| Problem | Solution | |
|
A.
The yellow shaft is still showing |
|
To reset the FORTEO delivery device, follow the steps below.
|
| You can prevent this problem by always using a NEW needle for each injection, and by pushing the black injection button all the way in and slowly counting to five.
|
||
| B. How can I tell if my FORTEO delivery device works? |
|
The black injection button should be all the way in to show that the full dose of medicine has been injected from the FORTEO delivery device.
Use a new needle every time you inject to be sure your FORTEO delivery device will work properly. |
| C. I see an air bubble in my FORTEO delivery device. |
|
A small air bubble will not affect your dose and it will not harm you. You can continue to take your dose as usual. |
|
D. I cannot get the needle off. |
|
|
|
E. What should I do if I have difficulty pulling out the black injection button? |
|
Change to a new FORTEO delivery device to take your dose as instructed by your healthcare provider.
When the black injection button becomes hard to pull out, this means there is not enough medicine in your FORTEO delivery device for another dose. You may still see some medicine left in the cartridge. |
| Cleaning and Storage |
Cleaning Your FORTEO Delivery Device
Storing Your FORTEO Delivery Device
|
| Other Important Notes |
|
| Disposal of Pen Needles and Delivery Device | |
Disposal of Pen Needles and the FORTEO Delivery Device
|
|
| Dispose of the FORTEO delivery device 28 days after first use. | 1st use date ______ / ______ / ______ Throw away after ______ / ______ / ______ |
|
Contact Information |
|
If you have questions or need help with your FORTEO delivery device, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or your healthcare provider. For more information about FORTEO, go to www.FORTEO.com. Marketed by: Lilly USA, LLC Indianapolis, IN 46285, USA FORTEO is a registered trademark of Eli Lilly and Company. ® Registered trademarks owned by Eli Lilly and Company; used under license. Copyright © 2008, 2025, Eli Lilly and Company. All rights reserved. |
Literature revised Jun 03, 2025
FOR-0004-IFU-20250603
7.1 Digoxin
Sporadic case reports have suggested that hypercalcemia may predispose patients to digitalis toxicity. FORTEO may transiently increase serum calcium. Consider the potential onset of signs and symptoms of digitalis toxicity when FORTEO is used in patients receiving digoxin [see Warnings and Precaution (5.5) and Clinical Pharmacology (12.3)].
10 Overdosage
In postmarketing spontaneous reports, there have been cases of medication errors in which the entire contents (up to 800 mcg) (40 times the recommended dose) of the FORTEO prefilled delivery device (pen) have been administered as a single dose. Transient events reported have included nausea, weakness/lethargy and hypotension. No fatalities associated with overdose have been reported. Additional signs, symptoms, and complications of FORTEO overdosage may include a delayed hypercalcemic effect, vomiting, dizziness, and headache.
11 Description
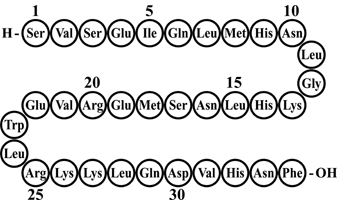
FORTEO (teriparatide injection) is a recombinant human parathyroid hormone analog (PTH 1-34). It has an identical sequence to the 34 N-terminal amino acids (the biologically active region) of the 84-amino acid human parathyroid hormone.
The molecular formula of teriparatide is C181H291N55O51S2 and molecular weight is 4117.8 daltons. Its amino acid sequence is shown below:
Teriparatide is manufactured using a strain of Escherichia coli modified by recombinant DNA technology.
FORTEO is supplied as a sterile, colorless, clear, isotonic solution in a glass cartridge which is pre-assembled into a single-patient-use delivery device (pen) for subcutaneous injection. Each delivery device (pen) is filled with volume to allow delivery of 2.24 mL. Each mL contains 250 mcg of teriparatide (as a free base), 0.41 mg of glacial acetic acid, 0.1 mg of sodium acetate (anhydrous), 45.4 mg of mannitol, 3 mg of Metacresol, and Water for Injection. In addition, hydrochloric acid solution 10% and/or sodium hydroxide solution 10% may have been added to adjust the pH to 4.
Each prefilled delivery device (pen) delivers 20 mcg of teriparatide per dose for up to 28 days. Each device contains additional volume to allow troubleshooting of the device 2 times.
5.1 Osteosarcoma
An increase in the incidence of osteosarcoma (a malignant bone tumor) was observed in male and female rats treated with teriparatide. Osteosarcoma has been reported in patients treated with FORTEO in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of FORTEO use [see Dosage and Administration (2.3), Adverse Reactions (6.3), and Nonclinical Toxicology (13.1)].
Avoid FORTEO use in patients with (these patients are at increased baseline risk of osteosarcoma):
- Open epiphyses (pediatric and young adult patients) (FORTEO is not approved in pediatric patients) [see Use in Specific Populations (8.4)].
- Metabolic bone diseases other than osteoporosis, including Paget's disease of the bone.
- Bone metastases or a history of skeletal malignancies.
- Prior external beam or implant radiation therapy involving the skeleton.
- Hereditary disorders predisposing to osteosarcoma.
16.1 How Supplied
FORTEO (teriparatide injection) is a clear and colorless solution, available as single-patient-use prefilled delivery device (pen) in the following package size:
- 560 mcg/2.24 mL (250 mcg/mL) [intended to deliver 28 daily doses of 20 mcg] NDC 0002-9678-01 (MS8400).
8.4 Pediatric Use
The safety and effectiveness of FORTEO have not been established in pediatric patients. Pediatric patients are at higher baseline risk of osteosarcoma because of open epiphyses [see Warnings and Precautions (5.1)].
8.5 Geriatric Use
Of the patients who received FORTEO in the osteoporosis trial of 1637 postmenopausal women, 75% were 65 years of age and older and 23% were 75 years of age and older. Of the patients who received FORTEO in the trial of 437 men with primary or hypogonadal osteoporosis, 39% were 65 years of age and over and 13% were 75 years of age and over. Of the 214 patients who received FORTEO in the glucocorticoid induced osteoporosis trial, 28% were 65 years of age and older and 9% were 75 years of age and older. No overall differences in safety or effectiveness of FORTEO have been observed between patients 65 years of age and older and younger adult patients.
6.2 Immunogenicity
As with all peptides, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors, including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies in the studies described below with the incidence of antibodies in other studies or to other teriparatide products may be misleading.
In the clinical trial of postmenopausal women with osteoporosis [see Clinical Studies (14.1)], antibodies that cross reacted with teriparatide were detected in 3% of women (15/541) who received FORTEO. Generally, antibodies were first detected following 12 months of treatment and diminished after withdrawal of therapy. There was no evidence of hypersensitivity reactions among these patients. Antibody formation did not appear to have effects on serum calcium, or on bone mineral density (BMD) response.
4 Contraindications
FORTEO is contraindicated in patients with hypersensitivity to teriparatide or to any of its excipients. Hypersensitivity reactions have included angioedema and anaphylaxis [see Adverse Reactions (6.3)].
6 Adverse Reactions
Most common adverse reactions (>10%) include: arthralgia, pain, and nausea (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions
8.7 Renal Impairment
In 5 patients with severe renal impairment (CrCl<30 mL/minute), the AUC and T1/2 of teriparatide were increased by 73% and 77%, respectively. Maximum serum concentration of teriparatide was not increased. It is unknown whether FORTEO alters the underlying metabolic bone disease seen in chronic renal impairment [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology
In single-dose rodent studies using subcutaneous injection of teriparatide, no mortality was seen in rats given doses of 1000 mcg/kg (540 times the human dose based on surface area, mcg/m2) or in mice given 10,000 mcg/kg (2700 times the human dose based on surface area, mcg/m2).
In a long-term study, skeletally mature ovariectomized female monkeys (N=30 per treatment group) were given either daily subcutaneous teriparatide injections of 5 mcg/kg or vehicle. Following the 18-month treatment period, the monkeys were removed from teriparatide treatment and were observed for an additional 3 years. The 5 mcg/kg dose resulted in systemic exposures that were approximately 6 times higher than the systemic exposure observed in humans following a subcutaneous dose of 20 mcg (based on AUC comparison). Bone tumors were not detected by radiographic or histologic evaluation in any monkey in the study.
2.1 Recommended Dosage
The recommended dosage is 20 mcg per dose given subcutaneously once a day. Instruct patients to take supplemental calcium and vitamin D if daily dietary intake is inadequate.
8.6 Hepatic Impairment
No studies have been performed in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage
FORTEO is indicated:
- For the treatment of postmenopausal women with osteoporosis at high risk for fracture (defined herein as having a history of osteoporotic fracture or multiple risk factors for fracture) or who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, FORTEO reduces the risk of vertebral and nonvertebral fractures.
- To increase bone mass in men with primary or hypogonadal osteoporosis at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
- For the treatment of men and women with osteoporosis associated with sustained systemic glucocorticoid therapy (daily dosage equivalent to 5 mg or greater of prednisone) at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
12.1 Mechanism of Action
Endogenous 84-amino acid parathyroid hormone (PTH) is the primary regulator of calcium and phosphate metabolism in bone and kidney. Physiological actions of PTH include regulation of bone metabolism, renal tubular reabsorption of calcium and phosphate, and intestinal calcium absorption. The biological actions of PTH and teriparatide are mediated through binding to specific high-affinity cell-surface receptors. Teriparatide and the 34 N-terminal amino acids of PTH bind to these receptors with the same affinity and have the same physiological actions on bone and kidney. Teriparatide is not expected to accumulate in bone or other tissues.
The skeletal effects of teriparatide depend upon the pattern of systemic exposure. Once-daily administration of teriparatide stimulates new bone formation on trabecular and cortical (periosteal and/or endosteal) bone surfaces by preferential stimulation of osteoblastic activity over osteoclastic activity. In monkey studies, teriparatide improved trabecular microarchitecture and increased bone mass and strength by stimulating new bone formation in both cancellous and cortical bone. In humans, the anabolic effects of teriparatide manifest as an increase in skeletal mass, an increase in markers of bone formation and resorption, and an increase in bone strength. By contrast, continuous excess of endogenous PTH, as occurs in hyperparathyroidism, may be detrimental to the skeleton because bone resorption may be stimulated more than bone formation.
5.3 Risk of Urolithiasis
In clinical trials, the frequency of urolithiasis was similar in patients treated with FORTEO and patients treated with placebo. However, FORTEO has not been studied in patients with active urolithiasis. If FORTEO-treated patients have pre-existing hypercalciuria or suspected/known active urolithiasis, consider measuring urinary calcium excretion. Consider the risks and benefits of use in patients with active or recent urolithiasis because of the potential to exacerbate this condition.
16.2 Storage and Handling
- Store FORTEO under refrigeration at 2° to 8°C (36° to 46°F) at all times except when administering the product.
- Recap the delivery device (pen) when not in use to protect the cartridge from physical damage and light.
- When using FORTEO, minimize the time out of the refrigerator; deliver the dose immediately following removal from the refrigerator.
- Do not freeze. Do not use FORTEO if it has been frozen.
- Throw away the device 28 days after first use.
5 Warnings and Precautions
- Osteosarcoma: Avoid use in patients with increased risk of osteosarcoma including patients with open epiphyses, metabolic bone diseases including Paget's disease, bone metastases or history of skeletal malignancies, prior external beam or implant radiation therapy involving the skeleton, and hereditary disorders predisposing to osteosarcoma. (5.1)
- Hypercalcemia and Cutaneous Calcification: Avoid in patients known to have an underlying hypercalcemic disorder. Discontinue in patients developing worsening of previously stable cutaneous calcification. (5.2)
- Risk of Urolithiasis: Consider the risk/benefit in patients with active or recent urolithiasis because of risk of exacerbation (5.3)
- Orthostatic Hypotension: Transient orthostatic hypotension may occur with initial doses of FORTEO (5.4)
2 Dosage and Administration
- Recommended dosage is 20 mcg subcutaneously once a day (2.1)
- Consider supplemental calcium and Vitamin D based on individual patient needs (2.1)
- Administer as a subcutaneous injection into the thigh or abdominal region (2.2)
- Administer initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur (2.2)
- Use of FORTEO for more than 2 years during a patient's lifetime should only be considered if a patient remains at or has returned to having a high risk for fracture (2.3)
5.4 Orthostatic Hypotension
FORTEO should be administered initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur. In short-term clinical pharmacology studies of FORTEO in healthy volunteers, transient episodes of symptomatic orthostatic hypotension were observed in 5% of volunteers. Typically, these events began within 4 hours of dosing and resolved (without treatment) within a few minutes to a few hours. When transient orthostatic hypotension occurred, it happened within the first several doses, it was relieved by placing the person in a reclining position, and it did not preclude continued treatment.
3 Dosage Forms and Strengths
Injection: 560 mcg/2.24 mL (250 mcg/mL) clear, colorless solution in a single-patient-use prefilled delivery device (pen) intended to deliver 28 daily doses of 20 mcg.
5.5 Risk of Digoxin Toxicity
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
2.2 Administration Instructions
- Administer FORTEO as a subcutaneous injection into the thigh or abdominal region. FORTEO is not approved for intravenous or intramuscular use.
- FORTEO should be administered initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur [see Warnings and Precautions (5.4)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration (FORTEO is a clear and colorless liquid). Do not use if solid particles appear or if the solution is cloudy or colored.
- Patients and/or caregivers who administer FORTEO should receive appropriate training and instruction on the proper use of the FORTEO prefilled delivery device (pen) from a qualified health professional.
- Discard the delivery device 28 days after first use.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide and the User Manual) before starting FORTEO and each time the prescription is renewed. Failure to follow the instructions may result in inaccurate dosing.
2.3 Recommended Treatment Duration
Use of FORTEO for more than 2 years during a patient's lifetime should only be considered if a patient remains at or has returned to having a high risk for fracture [see Warnings and Precautions (5.1)].
14.1 Treatment of Osteoporosis in Postmenopausal Women
The safety and efficacy of once-daily FORTEO, median exposure of 19 months, were examined in a double-blind, multicenter, placebo-controlled clinical study of 1637 postmenopausal women with osteoporosis. In this study 541 postmenopausal women were treated with 20 mcg FORTEO subcutaneously once daily.
All women received 1000 mg of calcium and at least 400 IU of vitamin D per day. Baseline and endpoint spinal radiographs were evaluated using the semiquantitative scoring. Ninety percent of the women in the study had 1 or more radiographically diagnosed vertebral fractures at baseline. The primary efficacy endpoint was the occurrence of new radiographically diagnosed vertebral fractures defined as changes in the height of previously undeformed vertebrae. Such fractures are not necessarily symptomatic.
14.3 Treatment of Men and Women With Glucocorticoid Induced Osteoporosis
The efficacy of FORTEO for treating glucocorticoid-induced osteoporosis was assessed in a randomized, double-blind, active-controlled trial of 428 patients (19% men, 81% women) aged 22 to 89 years (mean 57 years) treated with ≥5 mg/day prednisone or equivalent for a minimum of 3 months. The duration of the trial was 18 months. In the trial 214 patients were treated with FORTEO 20 mcg given subcutaneously once daily. In the FORTEO group, the baseline median glucocorticoid dose was 7.5 mg/day and the baseline median duration of glucocorticoid use was 1.5 years. The mean (SD) baseline lumbar spine BMD was 0.85 ± 0.13 g/cm2 and lumbar spine BMD T-score was –2.5 ± 1 (number of standard deviations below the mean BMD value for healthy adults). A total of 30% of patients had prevalent vertebral fracture(s) and 43% had prior non-vertebral fracture(s). The patients had chronic rheumatologic, respiratory or other diseases that required sustained glucocorticoid therapy. All patients received 1000 mg of calcium plus 800 IU of vitamin D supplementation per day.
Because of differences in mechanism of action (anabolic vs. anti-resorptive) and lack of clarity regarding differences in BMD as an adequate predictor of fracture efficacy, data on the active comparator are not presented.
14.2 Treatment to Increase Bone Mass in Men With Primary Or Hypogonadal Osteoporosis
The safety and efficacy of once-daily FORTEO, median exposure of 10 months, were examined in a double-blind, multicenter, placebo-controlled clinical study of 437 men with either primary (idiopathic) or hypogonadal osteoporosis. In this study, 151 men received 20 mcg of FORTEO given subcutaneously once daily. All men received 1000 mg of calcium and at least 400 IU of vitamin D per day. The primary efficacy endpoint was change in lumbar spine BMD.
FORTEO increased lumbar spine BMD in men with primary or hypogonadal osteoporosis. Statistically significant increases were seen at 3 months and continued throughout the treatment period. FORTEO was effective in increasing lumbar spine BMD regardless of age, baseline rate of bone turnover, and baseline BMD. The effects of FORTEO at additional skeletal sites are shown in Table 4.
FORTEO treatment for a median of 10 months increased lumbar spine BMD from baseline in 94% of men treated. Fifty-three percent of patients treated with FORTEO achieved at least a 5% increase in spine BMD, and 14% gained 10% or more.
|
a Intent-to-treat analysis, last observation carried forward. |
||
|
b p<0.001 compared with placebo. |
||
|
c p<0.05 compared with placebo. |
||
|
FORTEO N=151 |
Placebo N=147 |
|
| Lumbar spine BMD | 5.9b | 0.5 |
| Femoral neck BMD | 1.5c | 0.3 |
| Total hip BMD | 1.2 | 0.5 |
| Trochanter BMD | 1.3 | 1.1 |
| Intertrochanter BMD | 1.2 | 0.6 |
| Ward's triangle BMD | 2.8 | 1.1 |
| Total body BMD | 0.4 | -0.4 |
| Distal 1/3 radius BMD | -0.5 | -0.2 |
| Ultradistal radius BMD | -0.5 | -0.3 |
Structured Label Content
Section 42229-5 (42229-5)
Hypercalcemia
FORTEO has not been studied in patients with pre-existing hypercalcemia. FORTEO may cause hypercalcemia and may exacerbate hypercalcemia in patients with pre-existing hypercalcemia [see Adverse Reactions (6.1, 6.3)]. Avoid FORTEO in patients known to have an underlying hypercalcemic disorder, such as primary hyperparathyroidism.
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised: 11/2020 |
|
MEDICATION GUIDE
FORTEO ® (for-TAY-o) teriparatide injection for subcutaneous use |
|
| Read this Medication Guide before you start using FORTEO and each time you get a refill. There may be new information. Also, read the User Manual that comes with the FORTEO delivery device (pen) for information on how to use the device to inject your medicine the right way. This Medication Guide does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is the most important information I should know about FORTEO?
|
|
|
What is FORTEO?
FORTEO is a prescription medicine used to:
|
|
| It is not known if FORTEO is safe and effective in children. FORTEO should not be used in children and young adults whose bones are still growing. |
|
|
Who should not use FORTEO?
Do not use FORTEO if you:
|
|
| Symptoms of a serious allergic reaction of FORTEO may include swelling of the face, lips, tongue or throat that may cause difficulty in breathing or swallowing. Call your healthcare provider right away or get emergency medical help if you get any of these symptoms. | |
|
What should I tell my healthcare provider before using FORTEO?
Before you use FORTEO, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
How should I use FORTEO?
|
|
| If your healthcare provider recommends calcium and vitamin D supplements, you can take them at the same time you take FORTEO. | |
|
What are the possible side effects of FORTEO?
FORTEO may cause serious side effects including:
|
|
The most common side effects of FORTEO include:
|
|
| These are not all the possible side effects of FORTEO. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store FORTEO?
|
|
| Keep FORTEO and all medicines out of the reach of children. | |
|
General information about the safe and effective use of FORTEO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use FORTEO for a condition for which it was not prescribed. Do not give FORTEO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about FORTEO that is written for health professionals. |
|
|
What are the ingredients in FORTEO?
Active ingredient: teriparatide Inactive ingredients: glacial acetic acid, sodium acetate (anhydrous), mannitol, metacresol, and water for injection. In addition, hydrochloric acid solution 10% and/or sodium hydroxide solution 10% may have been added to adjust the product to pH 4. |
|
| For more information, go to www.FORTEO.com or call Lilly at 1-866-436-7836. Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA Copyright © 2002, 2020, Eli Lilly and Company. All rights reserved. FOR-0003-MG-20201116 |
Section 51945-4 (51945-4)
PACKAGE LABEL – FORTEO 20 mcg per dose, 2.24 mL
Do NOT transfer contents to a syringe
ATTENTION PHARMACIST: Medication Guide and device User Manual for patient inside carton
NDC 0002-8400-01
MS8400
FORTEO®
teriparatide injection
20 mcg per dose (given once daily subcutaneously)
Each single-patient-use prefilled pen will deliver 28 subcutaneous doses.
560 mcg/ 2.24 mL (250 mcg/mL)
For Single-Patient-Use Only
REFRIGERATE / DO NOT FREEZE
For subcutaneous use / Rx only
Needles not included
Becton, Dickinson and Company pen needles are recommended for use with this device
www.forteo.com
Lilly
Section 59845-8 (59845-8)
FORTEO® (for-TAY-o)
teriparatide injection
User Manual
Important: First read the Medication Guide that comes inside your FORTEO carton.
Before you use your new FORTEO delivery device, please read the entire front and back of this User Manual completely. Follow the directions carefully when using the FORTEO delivery device.
Do not share your delivery device or needles because infection or disease can be spread from one person to another.
The FORTEO delivery device contains 28 days of medicine. Throw away the FORTEO delivery device 28 days after first use, even if it is not completely empty. Do not inject more than one dose of FORTEO in the same day.
Do not transfer FORTEO to a syringe.
Wash your hands before every injection. Prepare the injection site as your healthcare provider instructed.
|
1
Pull off white cap |
Check the FORTEO delivery device label to make sure you have the right medicine and that it has not expired. Do not use if the FORTEO delivery device looks damaged, if the medicine in the cartridge is not clear and colorless, or if it has particles in it. |
|
2
Attach new needle |
||||
| Pull off paper tab. |
Push needle straight
onto medicine cartridge. |
Screw on needle clockwise until firmly attached. |
Pull off large needle cover and save it. |
|
3
Set dose |
|||
|
Pull out black injection button until it stops. If you cannot pull out the black injection button see Troubleshooting, Problem E, on back page. |
Check to make sure red stripe shows. |
Pull off small needle protector and throw away. |
|
4
Inject dose |
||
| Gently hold a fold of skin on your thigh or abdomen and insert needle straight into skin. |
Push in black injection button until it stops. Hold it in and count to 5 slowly. You must wait until the count of 5 to make sure you receive the correct dose. Then pull the needle from skin. |
| IMPORTANT | ||||
|
5
Confirm dose |
After completing the injection:
Once the needle is removed from the skin, take your thumb off the black injection button. Check to make sure the black injection button is all the way in. If the yellow shaft does not show, you have finished the injection steps the right way. |
You should NOT see any of the yellow shaft. If you do and have already injected the medicine, do not inject yourself a second time on the same day. Instead, you MUST reset the FORTEO delivery device (see Troubleshooting, Problem A, on back page). |
|
6
Remove needle |
||||
| Put large needle cover on needle. Do not try to put the needle cover back on with your hands. |
Unscrew the covered needle all the way by giving the large needle cover 3 to 5 counter-clockwise turns. |
Pull off needle and throw away in a puncture-resistant container. |
Push white cap back on. Right after use, place FORTEO delivery device in the refrigerator. |
For more information, or if you have any questions, turn to the back of this page.
|
|
FORTEO® (for-TAY-o) |
|
| Troubleshooting | ||
| Problem | Solution | |
|
A.
The yellow shaft is still showing |
|
To reset the FORTEO delivery device, follow the steps below.
|
| You can prevent this problem by always using a NEW needle for each injection, and by pushing the black injection button all the way in and slowly counting to five.
|
||
| B. How can I tell if my FORTEO delivery device works? |
|
The black injection button should be all the way in to show that the full dose of medicine has been injected from the FORTEO delivery device.
Use a new needle every time you inject to be sure your FORTEO delivery device will work properly. |
| C. I see an air bubble in my FORTEO delivery device. |
|
A small air bubble will not affect your dose and it will not harm you. You can continue to take your dose as usual. |
|
D. I cannot get the needle off. |
|
|
|
E. What should I do if I have difficulty pulling out the black injection button? |
|
Change to a new FORTEO delivery device to take your dose as instructed by your healthcare provider.
When the black injection button becomes hard to pull out, this means there is not enough medicine in your FORTEO delivery device for another dose. You may still see some medicine left in the cartridge. |
| Cleaning and Storage |
Cleaning Your FORTEO Delivery Device
Storing Your FORTEO Delivery Device
|
| Other Important Notes |
|
| Disposal of Pen Needles and Delivery Device | |
Disposal of Pen Needles and the FORTEO Delivery Device
|
|
| Dispose of the FORTEO delivery device 28 days after first use. | 1st use date ______ / ______ / ______ Throw away after ______ / ______ / ______ |
|
Contact Information |
|
If you have questions or need help with your FORTEO delivery device, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or your healthcare provider. For more information about FORTEO, go to www.FORTEO.com. Marketed by: Lilly USA, LLC Indianapolis, IN 46285, USA FORTEO is a registered trademark of Eli Lilly and Company. ® Registered trademarks owned by Eli Lilly and Company; used under license. Copyright © 2008, 2025, Eli Lilly and Company. All rights reserved. |
Literature revised Jun 03, 2025
FOR-0004-IFU-20250603
7.1 Digoxin
Sporadic case reports have suggested that hypercalcemia may predispose patients to digitalis toxicity. FORTEO may transiently increase serum calcium. Consider the potential onset of signs and symptoms of digitalis toxicity when FORTEO is used in patients receiving digoxin [see Warnings and Precaution (5.5) and Clinical Pharmacology (12.3)].
10 Overdosage (10 OVERDOSAGE)
In postmarketing spontaneous reports, there have been cases of medication errors in which the entire contents (up to 800 mcg) (40 times the recommended dose) of the FORTEO prefilled delivery device (pen) have been administered as a single dose. Transient events reported have included nausea, weakness/lethargy and hypotension. No fatalities associated with overdose have been reported. Additional signs, symptoms, and complications of FORTEO overdosage may include a delayed hypercalcemic effect, vomiting, dizziness, and headache.
11 Description (11 DESCRIPTION)
FORTEO (teriparatide injection) is a recombinant human parathyroid hormone analog (PTH 1-34). It has an identical sequence to the 34 N-terminal amino acids (the biologically active region) of the 84-amino acid human parathyroid hormone.
The molecular formula of teriparatide is C181H291N55O51S2 and molecular weight is 4117.8 daltons. Its amino acid sequence is shown below:
Teriparatide is manufactured using a strain of Escherichia coli modified by recombinant DNA technology.
FORTEO is supplied as a sterile, colorless, clear, isotonic solution in a glass cartridge which is pre-assembled into a single-patient-use delivery device (pen) for subcutaneous injection. Each delivery device (pen) is filled with volume to allow delivery of 2.24 mL. Each mL contains 250 mcg of teriparatide (as a free base), 0.41 mg of glacial acetic acid, 0.1 mg of sodium acetate (anhydrous), 45.4 mg of mannitol, 3 mg of Metacresol, and Water for Injection. In addition, hydrochloric acid solution 10% and/or sodium hydroxide solution 10% may have been added to adjust the pH to 4.
Each prefilled delivery device (pen) delivers 20 mcg of teriparatide per dose for up to 28 days. Each device contains additional volume to allow troubleshooting of the device 2 times.
5.1 Osteosarcoma
An increase in the incidence of osteosarcoma (a malignant bone tumor) was observed in male and female rats treated with teriparatide. Osteosarcoma has been reported in patients treated with FORTEO in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of FORTEO use [see Dosage and Administration (2.3), Adverse Reactions (6.3), and Nonclinical Toxicology (13.1)].
Avoid FORTEO use in patients with (these patients are at increased baseline risk of osteosarcoma):
- Open epiphyses (pediatric and young adult patients) (FORTEO is not approved in pediatric patients) [see Use in Specific Populations (8.4)].
- Metabolic bone diseases other than osteoporosis, including Paget's disease of the bone.
- Bone metastases or a history of skeletal malignancies.
- Prior external beam or implant radiation therapy involving the skeleton.
- Hereditary disorders predisposing to osteosarcoma.
16.1 How Supplied
FORTEO (teriparatide injection) is a clear and colorless solution, available as single-patient-use prefilled delivery device (pen) in the following package size:
- 560 mcg/2.24 mL (250 mcg/mL) [intended to deliver 28 daily doses of 20 mcg] NDC 0002-9678-01 (MS8400).
8.4 Pediatric Use
The safety and effectiveness of FORTEO have not been established in pediatric patients. Pediatric patients are at higher baseline risk of osteosarcoma because of open epiphyses [see Warnings and Precautions (5.1)].
8.5 Geriatric Use
Of the patients who received FORTEO in the osteoporosis trial of 1637 postmenopausal women, 75% were 65 years of age and older and 23% were 75 years of age and older. Of the patients who received FORTEO in the trial of 437 men with primary or hypogonadal osteoporosis, 39% were 65 years of age and over and 13% were 75 years of age and over. Of the 214 patients who received FORTEO in the glucocorticoid induced osteoporosis trial, 28% were 65 years of age and older and 9% were 75 years of age and older. No overall differences in safety or effectiveness of FORTEO have been observed between patients 65 years of age and older and younger adult patients.
6.2 Immunogenicity
As with all peptides, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors, including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies in the studies described below with the incidence of antibodies in other studies or to other teriparatide products may be misleading.
In the clinical trial of postmenopausal women with osteoporosis [see Clinical Studies (14.1)], antibodies that cross reacted with teriparatide were detected in 3% of women (15/541) who received FORTEO. Generally, antibodies were first detected following 12 months of treatment and diminished after withdrawal of therapy. There was no evidence of hypersensitivity reactions among these patients. Antibody formation did not appear to have effects on serum calcium, or on bone mineral density (BMD) response.
4 Contraindications (4 CONTRAINDICATIONS)
FORTEO is contraindicated in patients with hypersensitivity to teriparatide or to any of its excipients. Hypersensitivity reactions have included angioedema and anaphylaxis [see Adverse Reactions (6.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (>10%) include: arthralgia, pain, and nausea (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
In 5 patients with severe renal impairment (CrCl<30 mL/minute), the AUC and T1/2 of teriparatide were increased by 73% and 77%, respectively. Maximum serum concentration of teriparatide was not increased. It is unknown whether FORTEO alters the underlying metabolic bone disease seen in chronic renal impairment [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology
In single-dose rodent studies using subcutaneous injection of teriparatide, no mortality was seen in rats given doses of 1000 mcg/kg (540 times the human dose based on surface area, mcg/m2) or in mice given 10,000 mcg/kg (2700 times the human dose based on surface area, mcg/m2).
In a long-term study, skeletally mature ovariectomized female monkeys (N=30 per treatment group) were given either daily subcutaneous teriparatide injections of 5 mcg/kg or vehicle. Following the 18-month treatment period, the monkeys were removed from teriparatide treatment and were observed for an additional 3 years. The 5 mcg/kg dose resulted in systemic exposures that were approximately 6 times higher than the systemic exposure observed in humans following a subcutaneous dose of 20 mcg (based on AUC comparison). Bone tumors were not detected by radiographic or histologic evaluation in any monkey in the study.
2.1 Recommended Dosage
The recommended dosage is 20 mcg per dose given subcutaneously once a day. Instruct patients to take supplemental calcium and vitamin D if daily dietary intake is inadequate.
8.6 Hepatic Impairment
No studies have been performed in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
FORTEO is indicated:
- For the treatment of postmenopausal women with osteoporosis at high risk for fracture (defined herein as having a history of osteoporotic fracture or multiple risk factors for fracture) or who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, FORTEO reduces the risk of vertebral and nonvertebral fractures.
- To increase bone mass in men with primary or hypogonadal osteoporosis at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
- For the treatment of men and women with osteoporosis associated with sustained systemic glucocorticoid therapy (daily dosage equivalent to 5 mg or greater of prednisone) at high risk for fracture or who have failed or are intolerant to other available osteoporosis therapy.
12.1 Mechanism of Action
Endogenous 84-amino acid parathyroid hormone (PTH) is the primary regulator of calcium and phosphate metabolism in bone and kidney. Physiological actions of PTH include regulation of bone metabolism, renal tubular reabsorption of calcium and phosphate, and intestinal calcium absorption. The biological actions of PTH and teriparatide are mediated through binding to specific high-affinity cell-surface receptors. Teriparatide and the 34 N-terminal amino acids of PTH bind to these receptors with the same affinity and have the same physiological actions on bone and kidney. Teriparatide is not expected to accumulate in bone or other tissues.
The skeletal effects of teriparatide depend upon the pattern of systemic exposure. Once-daily administration of teriparatide stimulates new bone formation on trabecular and cortical (periosteal and/or endosteal) bone surfaces by preferential stimulation of osteoblastic activity over osteoclastic activity. In monkey studies, teriparatide improved trabecular microarchitecture and increased bone mass and strength by stimulating new bone formation in both cancellous and cortical bone. In humans, the anabolic effects of teriparatide manifest as an increase in skeletal mass, an increase in markers of bone formation and resorption, and an increase in bone strength. By contrast, continuous excess of endogenous PTH, as occurs in hyperparathyroidism, may be detrimental to the skeleton because bone resorption may be stimulated more than bone formation.
5.3 Risk of Urolithiasis
In clinical trials, the frequency of urolithiasis was similar in patients treated with FORTEO and patients treated with placebo. However, FORTEO has not been studied in patients with active urolithiasis. If FORTEO-treated patients have pre-existing hypercalciuria or suspected/known active urolithiasis, consider measuring urinary calcium excretion. Consider the risks and benefits of use in patients with active or recent urolithiasis because of the potential to exacerbate this condition.
16.2 Storage and Handling
- Store FORTEO under refrigeration at 2° to 8°C (36° to 46°F) at all times except when administering the product.
- Recap the delivery device (pen) when not in use to protect the cartridge from physical damage and light.
- When using FORTEO, minimize the time out of the refrigerator; deliver the dose immediately following removal from the refrigerator.
- Do not freeze. Do not use FORTEO if it has been frozen.
- Throw away the device 28 days after first use.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Osteosarcoma: Avoid use in patients with increased risk of osteosarcoma including patients with open epiphyses, metabolic bone diseases including Paget's disease, bone metastases or history of skeletal malignancies, prior external beam or implant radiation therapy involving the skeleton, and hereditary disorders predisposing to osteosarcoma. (5.1)
- Hypercalcemia and Cutaneous Calcification: Avoid in patients known to have an underlying hypercalcemic disorder. Discontinue in patients developing worsening of previously stable cutaneous calcification. (5.2)
- Risk of Urolithiasis: Consider the risk/benefit in patients with active or recent urolithiasis because of risk of exacerbation (5.3)
- Orthostatic Hypotension: Transient orthostatic hypotension may occur with initial doses of FORTEO (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dosage is 20 mcg subcutaneously once a day (2.1)
- Consider supplemental calcium and Vitamin D based on individual patient needs (2.1)
- Administer as a subcutaneous injection into the thigh or abdominal region (2.2)
- Administer initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur (2.2)
- Use of FORTEO for more than 2 years during a patient's lifetime should only be considered if a patient remains at or has returned to having a high risk for fracture (2.3)
5.4 Orthostatic Hypotension
FORTEO should be administered initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur. In short-term clinical pharmacology studies of FORTEO in healthy volunteers, transient episodes of symptomatic orthostatic hypotension were observed in 5% of volunteers. Typically, these events began within 4 hours of dosing and resolved (without treatment) within a few minutes to a few hours. When transient orthostatic hypotension occurred, it happened within the first several doses, it was relieved by placing the person in a reclining position, and it did not preclude continued treatment.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 560 mcg/2.24 mL (250 mcg/mL) clear, colorless solution in a single-patient-use prefilled delivery device (pen) intended to deliver 28 daily doses of 20 mcg.
5.5 Risk of Digoxin Toxicity
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
2.2 Administration Instructions
- Administer FORTEO as a subcutaneous injection into the thigh or abdominal region. FORTEO is not approved for intravenous or intramuscular use.
- FORTEO should be administered initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur [see Warnings and Precautions (5.4)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration (FORTEO is a clear and colorless liquid). Do not use if solid particles appear or if the solution is cloudy or colored.
- Patients and/or caregivers who administer FORTEO should receive appropriate training and instruction on the proper use of the FORTEO prefilled delivery device (pen) from a qualified health professional.
- Discard the delivery device 28 days after first use.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide and the User Manual) before starting FORTEO and each time the prescription is renewed. Failure to follow the instructions may result in inaccurate dosing.
2.3 Recommended Treatment Duration
Use of FORTEO for more than 2 years during a patient's lifetime should only be considered if a patient remains at or has returned to having a high risk for fracture [see Warnings and Precautions (5.1)].
14.1 Treatment of Osteoporosis in Postmenopausal Women
The safety and efficacy of once-daily FORTEO, median exposure of 19 months, were examined in a double-blind, multicenter, placebo-controlled clinical study of 1637 postmenopausal women with osteoporosis. In this study 541 postmenopausal women were treated with 20 mcg FORTEO subcutaneously once daily.
All women received 1000 mg of calcium and at least 400 IU of vitamin D per day. Baseline and endpoint spinal radiographs were evaluated using the semiquantitative scoring. Ninety percent of the women in the study had 1 or more radiographically diagnosed vertebral fractures at baseline. The primary efficacy endpoint was the occurrence of new radiographically diagnosed vertebral fractures defined as changes in the height of previously undeformed vertebrae. Such fractures are not necessarily symptomatic.
14.3 Treatment of Men and Women With Glucocorticoid Induced Osteoporosis (14.3 Treatment of Men and Women with Glucocorticoid-Induced Osteoporosis)
The efficacy of FORTEO for treating glucocorticoid-induced osteoporosis was assessed in a randomized, double-blind, active-controlled trial of 428 patients (19% men, 81% women) aged 22 to 89 years (mean 57 years) treated with ≥5 mg/day prednisone or equivalent for a minimum of 3 months. The duration of the trial was 18 months. In the trial 214 patients were treated with FORTEO 20 mcg given subcutaneously once daily. In the FORTEO group, the baseline median glucocorticoid dose was 7.5 mg/day and the baseline median duration of glucocorticoid use was 1.5 years. The mean (SD) baseline lumbar spine BMD was 0.85 ± 0.13 g/cm2 and lumbar spine BMD T-score was –2.5 ± 1 (number of standard deviations below the mean BMD value for healthy adults). A total of 30% of patients had prevalent vertebral fracture(s) and 43% had prior non-vertebral fracture(s). The patients had chronic rheumatologic, respiratory or other diseases that required sustained glucocorticoid therapy. All patients received 1000 mg of calcium plus 800 IU of vitamin D supplementation per day.
Because of differences in mechanism of action (anabolic vs. anti-resorptive) and lack of clarity regarding differences in BMD as an adequate predictor of fracture efficacy, data on the active comparator are not presented.
14.2 Treatment to Increase Bone Mass in Men With Primary Or Hypogonadal Osteoporosis (14.2 Treatment to Increase Bone Mass in Men with Primary or Hypogonadal Osteoporosis)
The safety and efficacy of once-daily FORTEO, median exposure of 10 months, were examined in a double-blind, multicenter, placebo-controlled clinical study of 437 men with either primary (idiopathic) or hypogonadal osteoporosis. In this study, 151 men received 20 mcg of FORTEO given subcutaneously once daily. All men received 1000 mg of calcium and at least 400 IU of vitamin D per day. The primary efficacy endpoint was change in lumbar spine BMD.
FORTEO increased lumbar spine BMD in men with primary or hypogonadal osteoporosis. Statistically significant increases were seen at 3 months and continued throughout the treatment period. FORTEO was effective in increasing lumbar spine BMD regardless of age, baseline rate of bone turnover, and baseline BMD. The effects of FORTEO at additional skeletal sites are shown in Table 4.
FORTEO treatment for a median of 10 months increased lumbar spine BMD from baseline in 94% of men treated. Fifty-three percent of patients treated with FORTEO achieved at least a 5% increase in spine BMD, and 14% gained 10% or more.
|
a Intent-to-treat analysis, last observation carried forward. |
||
|
b p<0.001 compared with placebo. |
||
|
c p<0.05 compared with placebo. |
||
|
FORTEO N=151 |
Placebo N=147 |
|
| Lumbar spine BMD | 5.9b | 0.5 |
| Femoral neck BMD | 1.5c | 0.3 |
| Total hip BMD | 1.2 | 0.5 |
| Trochanter BMD | 1.3 | 1.1 |
| Intertrochanter BMD | 1.2 | 0.6 |
| Ward's triangle BMD | 2.8 | 1.1 |
| Total body BMD | 0.4 | -0.4 |
| Distal 1/3 radius BMD | -0.5 | -0.2 |
| Ultradistal radius BMD | -0.5 | -0.3 |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:35.755713 · Updated: 2026-03-14T22:30:18.174146