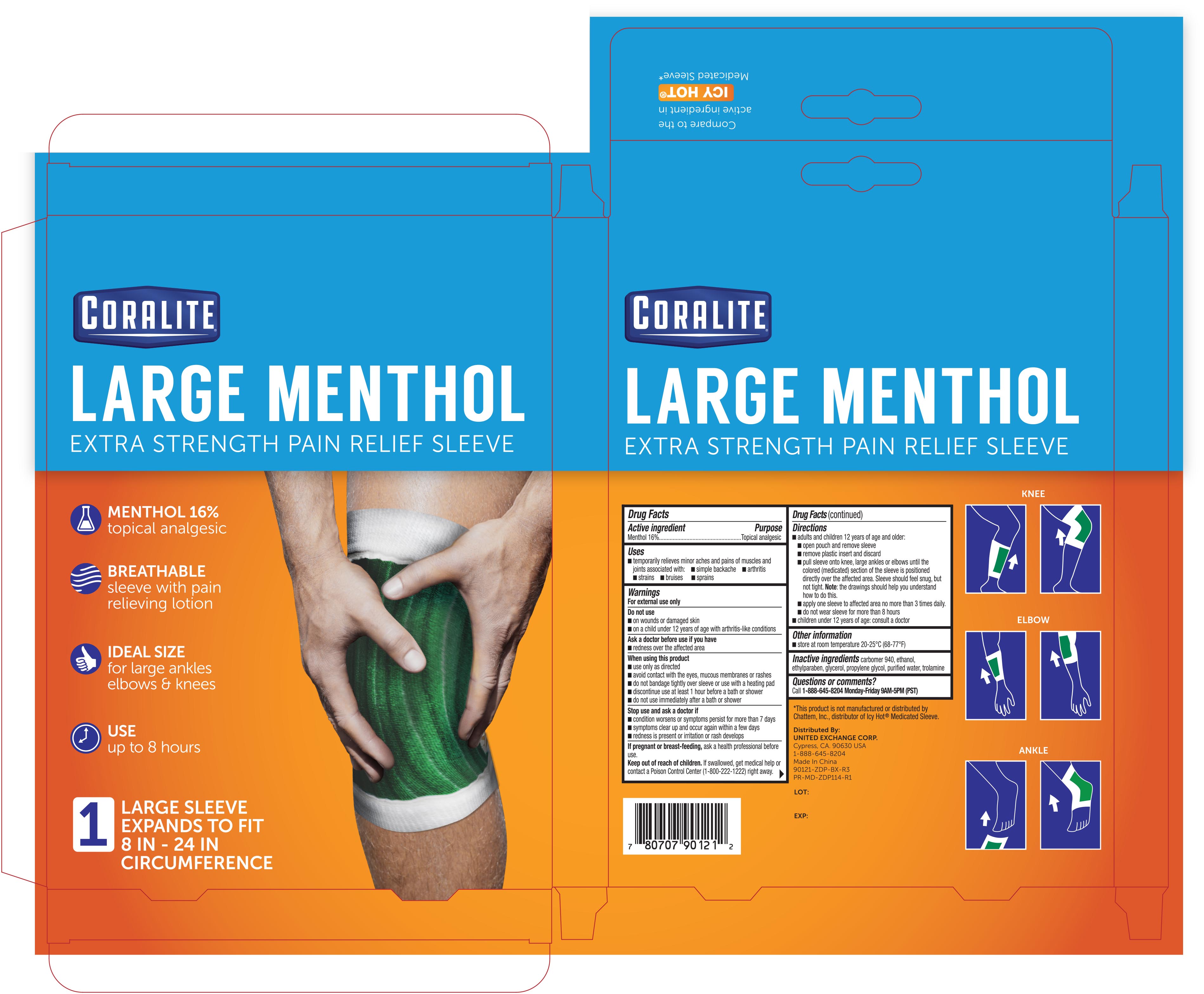
coralite large menthol pain relief sleeve 1ct 90121 zdp
aa5873bd-af64-41cc-8f16-7714c56bca96
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Purpose Menthol 16.0%..................................................Topical Analgesic Patch
Medication Information
Warnings and Precautions
Warnings
For external use only
Active Ingredient
Active ingredient Purpose
Menthol 16.0%..................................................Topical Analgesic Patch
Indications and Usage
Directions
- adults and children 12 years of age and older:
- open pouch and remove sleeve
- remove plastic insert and discard
- pull sleeve onto knee, large ankles or elbows until the colored (medicated) section of the sleeve is positioned directly over the affected area. Sleeve should feel snug, but not tight. Note: the drawings should help you understand how to do this.
- apply one sleeve to affected area no more than 3 times daily
- do not wear sleeve for more than 8 hours
- children under 12 years of age: consult a doctor
Dosage and Administration
Distributed by:
United Exchange Corp.
Cypress, CA 90630 USA
Made in China
Description
Active ingredient Purpose Menthol 16.0%..................................................Topical Analgesic Patch
Section 44425-7
Other information
- store at room temperature 20-25°C (68-77°F)
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away
Section 50566-9
Stop use and ask a doctor if
- condition worsens or symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
- redness is present or irritation or rash develops
Section 50567-7
When using this product
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- do not bandage tightly over sleeve or use with a heating pad
- discontinue use at least 1 hour before a bath or shower
- do not use immediately after a bath or shower
Section 50569-3
Ask a doctor before use if you have
- redness over the affected area
Section 50570-1
Do not use
- on wounds or damaged skin
- on a child under 12 years of age with arthritis-like-conditions
Section 51727-6
Inative ingredients
carbomer 940, ethanol, ethylparaben, glycerol, propylene glycol, purified water, trolamine
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use
Section 55105-1
Uses
- temporarily relieves minor aches and pains of muscles and joints associated with:
- simple backache
- arthritis
- strains
- bruises
- sprains
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only
Active Ingredient (55106-9)
Active ingredient Purpose
Menthol 16.0%..................................................Topical Analgesic Patch
Indications and Usage (34067-9)
Directions
- adults and children 12 years of age and older:
- open pouch and remove sleeve
- remove plastic insert and discard
- pull sleeve onto knee, large ankles or elbows until the colored (medicated) section of the sleeve is positioned directly over the affected area. Sleeve should feel snug, but not tight. Note: the drawings should help you understand how to do this.
- apply one sleeve to affected area no more than 3 times daily
- do not wear sleeve for more than 8 hours
- children under 12 years of age: consult a doctor
Dosage and Administration (34068-7)
Distributed by:
United Exchange Corp.
Cypress, CA 90630 USA
Made in China
Section 44425-7 (44425-7)
Other information
- store at room temperature 20-25°C (68-77°F)
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- condition worsens or symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
- redness is present or irritation or rash develops
Section 50567-7 (50567-7)
When using this product
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- do not bandage tightly over sleeve or use with a heating pad
- discontinue use at least 1 hour before a bath or shower
- do not use immediately after a bath or shower
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- redness over the affected area
Section 50570-1 (50570-1)
Do not use
- on wounds or damaged skin
- on a child under 12 years of age with arthritis-like-conditions
Section 51727-6 (51727-6)
Inative ingredients
carbomer 940, ethanol, ethylparaben, glycerol, propylene glycol, purified water, trolamine
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use
Section 55105-1 (55105-1)
Uses
- temporarily relieves minor aches and pains of muscles and joints associated with:
- simple backache
- arthritis
- strains
- bruises
- sprains
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:06.783679 · Updated: 2026-03-14T23:13:11.905913