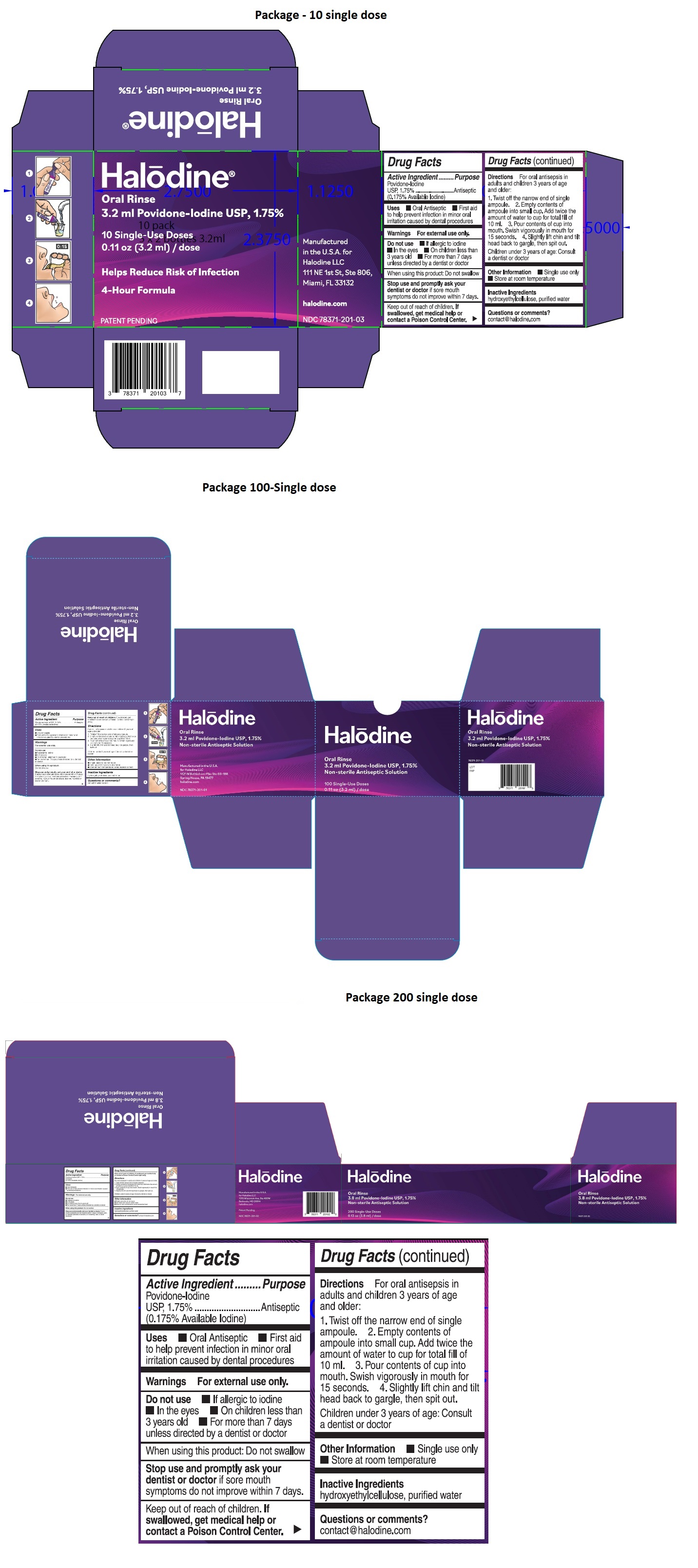
Halodine Oral Rinse
a9ee4c33-4a5b-3a1f-e053-2995a90aa515
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Povidone-Iodine USP, 1.75% (0.175% Available Iodine)
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Povidone-Iodine USP, 1.75% (0.175% Available Iodine)
Uses
- Oral Antiseptic
- First aid to help prevent infection in minor oral irritation caused by dental procedures
Section 42229-5
Helps Reduce Risk of Infection
4-Hour Formula
Non-Sterile Antiseptic Solution
PATENT PENDING
Manufactured in the U.S.A. for Halodine LLC
111 NE 1st St, Ste 806, Miami, FL 33132
halodine.com
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only.
Do not use • If allergic to iodine • In the eyes • On children less than 3 years old • For more than 7 days unless directed by a dentist or doctor
When using this product: Do not swallow
Stop use and promptly ask your dentist or doctor if sore mouth symptoms do not improve within 7 days, if irritation, pain, or redness persists or worsens, or if swelling, rash, or fever develops.
Packaging
Directions
For oral antisepsis in adults and children 3 years of age and older:
- Twist off the narrow end of single ampoule.
- Empty contents of ampoule into small cup. Add twice the amount of water to cup for total fill of 10 ml.
- Pour contents of cup into mouth. Swish vigorously in mouth for 15 seconds.
- Slightly lift chin and tilt head back to gargle, then spit out.
Children under 3 years of age: Consult a dentist or doctor
Drug Facts
Active Ingredient
Povidone-Iodine USP, 1.75%
(0.175% Available Iodine)
Other Information
- Single use only; do not reuse
- Not made with natural rubber latex
- Store at room temperature; avoid excessive heat
Inactive Ingredients
hydroxyethylcellulose, purified water
Questions Or Comments?
Structured Label Content
Uses
- Oral Antiseptic
- First aid to help prevent infection in minor oral irritation caused by dental procedures
Section 42229-5 (42229-5)
Helps Reduce Risk of Infection
4-Hour Formula
Non-Sterile Antiseptic Solution
PATENT PENDING
Manufactured in the U.S.A. for Halodine LLC
111 NE 1st St, Ste 806, Miami, FL 33132
halodine.com
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Antiseptic
Warnings
For external use only.
Do not use • If allergic to iodine • In the eyes • On children less than 3 years old • For more than 7 days unless directed by a dentist or doctor
When using this product: Do not swallow
Stop use and promptly ask your dentist or doctor if sore mouth symptoms do not improve within 7 days, if irritation, pain, or redness persists or worsens, or if swelling, rash, or fever develops.
Packaging
Directions
For oral antisepsis in adults and children 3 years of age and older:
- Twist off the narrow end of single ampoule.
- Empty contents of ampoule into small cup. Add twice the amount of water to cup for total fill of 10 ml.
- Pour contents of cup into mouth. Swish vigorously in mouth for 15 seconds.
- Slightly lift chin and tilt head back to gargle, then spit out.
Children under 3 years of age: Consult a dentist or doctor
Drug Facts
Active Ingredient
Povidone-Iodine USP, 1.75%
(0.175% Available Iodine)
Other Information
- Single use only; do not reuse
- Not made with natural rubber latex
- Store at room temperature; avoid excessive heat
Inactive Ingredients
hydroxyethylcellulose, purified water
Questions Or Comments? (Questions or comments?)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:03.242902 · Updated: 2026-03-14T22:58:52.562418