Flavoxate Hcl Tablets
a9c44c4b-5b3c-45bf-9bc9-ce858e3d06c5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
PRESCRIBING INFORMATION
Indications and Usage
Flavoxate HCl tablets are indicated for symptomatic relief of dysuria, urgency, nocturia, suprapubic pain, frequency and incontinence as may occur in cystitis, prostatitis, urethritis, urethrocystitis/urethrotrigonitis. Flavoxate HCl tablets are not indicated for definitive treatment, but are compatible with drugs used for the treatment of urinary tract infections.
Dosage and Administration
Adults and children over 12 years of age: One or two 100 mg tablets 3 or 4 times a day. With improvement of symptoms, the dose may be reduced. This drug cannot be recommended for infants and children under 12 years of age because safety and efficacy have not been demonstrated in this age group.
Contraindications
Flavoxate HCl tablets are contraindicated in patients who have any of the following obstructive conditions: pyloric or duodenal obstruction, obstructive intestinal lesions or ileus, achalasia, gastrointestinal hemorrhage and obstructive uropathies of the lower urinary tract.
Adverse Reactions
The following adverse reactions have been observed, but there are not enough data to support an estimate of their frequency. Gastrointestinal: Nausea, vomiting, dry mouth. CNS: Vertigo, headache, mental confusion, especially in the elderly, drowsiness, nervousness. Hematologic: Leukopenia (one case which was reversible upon discontinuation of the drug). Cardiovascular: Tachycardia and palpitation. Allergic: Urticaria and other dermatoses, eosinophilia and hyperpyrexia. Ophthalmic: Increased ocular tension, blurred vision, disturbance in eye accommodation. Renal: Dysuria.
How Supplied
Flavoxate HCl Tablets, 100 mg, are supplied as round, white, film-coated tablets debossed "PAD" and "0115" on one side and plain on the other side, in bottles of 100. 100 mg 100's: NDC 0574- 0115 -01 Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature] Manufactured For Padagis ® Minneapolis, MN 55427 www.padagis.com Rev 05-23 7H700 RC PH3 Code 917A00
Medication Information
Indications and Usage
Flavoxate HCl tablets are indicated for symptomatic relief of dysuria, urgency, nocturia, suprapubic pain, frequency and incontinence as may occur in cystitis, prostatitis, urethritis, urethrocystitis/urethrotrigonitis. Flavoxate HCl tablets are not indicated for definitive treatment, but are compatible with drugs used for the treatment of urinary tract infections.
Dosage and Administration
Adults and children over 12 years of age:
One or two 100 mg tablets 3 or 4 times a day. With improvement of symptoms, the dose may be reduced. This drug cannot be recommended for infants and children under 12 years of age because safety and efficacy have not been demonstrated in this age group.
Contraindications
Flavoxate HCl tablets are contraindicated in patients who have any of the following obstructive conditions: pyloric or duodenal obstruction, obstructive intestinal lesions or ileus, achalasia, gastrointestinal hemorrhage and obstructive uropathies of the lower urinary tract.
Adverse Reactions
The following adverse reactions have been observed, but there are not enough data to support an estimate of their frequency.
Gastrointestinal: Nausea, vomiting, dry mouth.
CNS: Vertigo, headache, mental confusion, especially in the elderly, drowsiness, nervousness.
Hematologic: Leukopenia (one case which was reversible upon discontinuation of the drug).
Cardiovascular: Tachycardia and palpitation.
Allergic: Urticaria and other dermatoses, eosinophilia and hyperpyrexia.
Ophthalmic: Increased ocular tension, blurred vision, disturbance in eye accommodation.
Renal: Dysuria.
How Supplied
Flavoxate HCl Tablets, 100 mg, are supplied as round, white, film-coated tablets debossed "PAD" and "0115" on one side and plain on the other side, in bottles of 100.
100 mg 100's:
NDC 0574-0115-01
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]
Manufactured For
Padagis®
Minneapolis, MN 55427
www.padagis.com
Rev 05-23
7H700 RC PH3
Code 917A00
Description
Flavoxate HCl tablets contain flavoxate hydrochloride, a synthetic urinary tract spasmolytic.
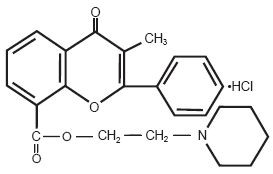
Chemically, flavoxate hydrochloride is 2-piperidinoethyl 3-methyl-4-oxo-2-phenyl-4H-1-benzopyran-8-carboxylate hydrochloride. The empirical formula of flavoxate hydrochloride is C24H25NO4•HCl. The molecular weight is 427.94. The structural formula appears below:
Flavoxate HCl is supplied in tablets for oral administration. Each round, white, film-coated Flavoxate HCl tablet is debossed "PAD" and "0115" on one side and plain on the other side and contains flavoxate hydrochloride, 100 mg. Inactive ingredients consist of colloidal silicon dioxide, ethyl acrylate, hypromellose, lactose monohydrate, magnesium stearate, methyl methacrylate, microcrystalline cellulose, nonoxynol 100 and sodium starch glycolate. Film coating is composed of hypromellose 2910 6cP and polyethylene glycol.
Section 42229-5
PRESCRIBING INFORMATION
Warnings
Flavoxate HCl should be given cautiously in patients with suspected glaucoma.
Overdosage
The oral LD50 for flavoxate HCl in rats is 4273 mg/kg. The oral LD50 for flavoxate HCl in mice is 1837 mg/kg.
It is not known whether flavoxate HCl is dialyzable.
Pregnancy:
Teratogenic Effects-Pregnancy Category B.
Reproduction studies have been performed in rats and rabbits at doses up to 34 times the human dose and revealed no evidence of impaired fertility or harm to the fetus due to flavoxate HCl. There are, however, no well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Pediatric Use:
Safety and effectiveness in children below the age of 12 years have not been established.
Nursing Mothers:
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when flavoxate HCl is administered to a nursing woman.
Clinical Pharmacology
Flavoxate hydrochloride counteracts smooth muscle spasm of the urinary tract and exerts its effect directly on the muscle.
In a single study of 11 normal male subjects, the time to onset of action was 55 minutes. The peak effect was observed at 112 minutes. 57% of the flavoxate HCl was excreted in the urine within 24 hours.
Information for Patients:
Patients should be informed that if drowsiness and blurred vision occur, they should not operate a motor vehicle or machinery or participate in activities where alertness is required.
Principal Display Panel 100 Mg Tablet Bottle
NDC 0574-0115-01
Rx Only
Flavoxate HCl Tablets
100 mg
100 Tablets
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Mutagenicity studies and long-term studies in animals to determine the carcinogenic potential of flavoxate HCl have not been performed.
Structured Label Content
Section 42229-5 (42229-5)
PRESCRIBING INFORMATION
Warnings (WARNINGS)
Flavoxate HCl should be given cautiously in patients with suspected glaucoma.
Overdosage (OVERDOSAGE)
The oral LD50 for flavoxate HCl in rats is 4273 mg/kg. The oral LD50 for flavoxate HCl in mice is 1837 mg/kg.
It is not known whether flavoxate HCl is dialyzable.
Pregnancy:
Teratogenic Effects-Pregnancy Category B.
Reproduction studies have been performed in rats and rabbits at doses up to 34 times the human dose and revealed no evidence of impaired fertility or harm to the fetus due to flavoxate HCl. There are, however, no well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Description (DESCRIPTION)
Flavoxate HCl tablets contain flavoxate hydrochloride, a synthetic urinary tract spasmolytic.
Chemically, flavoxate hydrochloride is 2-piperidinoethyl 3-methyl-4-oxo-2-phenyl-4H-1-benzopyran-8-carboxylate hydrochloride. The empirical formula of flavoxate hydrochloride is C24H25NO4•HCl. The molecular weight is 427.94. The structural formula appears below:
Flavoxate HCl is supplied in tablets for oral administration. Each round, white, film-coated Flavoxate HCl tablet is debossed "PAD" and "0115" on one side and plain on the other side and contains flavoxate hydrochloride, 100 mg. Inactive ingredients consist of colloidal silicon dioxide, ethyl acrylate, hypromellose, lactose monohydrate, magnesium stearate, methyl methacrylate, microcrystalline cellulose, nonoxynol 100 and sodium starch glycolate. Film coating is composed of hypromellose 2910 6cP and polyethylene glycol.
How Supplied (HOW SUPPLIED)
Flavoxate HCl Tablets, 100 mg, are supplied as round, white, film-coated tablets debossed "PAD" and "0115" on one side and plain on the other side, in bottles of 100.
100 mg 100's:
NDC 0574-0115-01
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]
Manufactured For
Padagis®
Minneapolis, MN 55427
www.padagis.com
Rev 05-23
7H700 RC PH3
Code 917A00
Pediatric Use:
Safety and effectiveness in children below the age of 12 years have not been established.
Nursing Mothers:
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when flavoxate HCl is administered to a nursing woman.
Adverse Reactions (ADVERSE REACTIONS)
The following adverse reactions have been observed, but there are not enough data to support an estimate of their frequency.
Gastrointestinal: Nausea, vomiting, dry mouth.
CNS: Vertigo, headache, mental confusion, especially in the elderly, drowsiness, nervousness.
Hematologic: Leukopenia (one case which was reversible upon discontinuation of the drug).
Cardiovascular: Tachycardia and palpitation.
Allergic: Urticaria and other dermatoses, eosinophilia and hyperpyrexia.
Ophthalmic: Increased ocular tension, blurred vision, disturbance in eye accommodation.
Renal: Dysuria.
Contraindications (CONTRAINDICATIONS)
Flavoxate HCl tablets are contraindicated in patients who have any of the following obstructive conditions: pyloric or duodenal obstruction, obstructive intestinal lesions or ileus, achalasia, gastrointestinal hemorrhage and obstructive uropathies of the lower urinary tract.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Flavoxate hydrochloride counteracts smooth muscle spasm of the urinary tract and exerts its effect directly on the muscle.
In a single study of 11 normal male subjects, the time to onset of action was 55 minutes. The peak effect was observed at 112 minutes. 57% of the flavoxate HCl was excreted in the urine within 24 hours.
Indications and Usage (INDICATIONS AND USAGE)
Flavoxate HCl tablets are indicated for symptomatic relief of dysuria, urgency, nocturia, suprapubic pain, frequency and incontinence as may occur in cystitis, prostatitis, urethritis, urethrocystitis/urethrotrigonitis. Flavoxate HCl tablets are not indicated for definitive treatment, but are compatible with drugs used for the treatment of urinary tract infections.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Adults and children over 12 years of age:
One or two 100 mg tablets 3 or 4 times a day. With improvement of symptoms, the dose may be reduced. This drug cannot be recommended for infants and children under 12 years of age because safety and efficacy have not been demonstrated in this age group.
Information for Patients:
Patients should be informed that if drowsiness and blurred vision occur, they should not operate a motor vehicle or machinery or participate in activities where alertness is required.
Principal Display Panel 100 Mg Tablet Bottle (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle)
NDC 0574-0115-01
Rx Only
Flavoxate HCl Tablets
100 mg
100 Tablets
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Mutagenicity studies and long-term studies in animals to determine the carcinogenic potential of flavoxate HCl have not been performed.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:11.931636 · Updated: 2026-03-14T22:49:40.462955