These Highlights Do Not Include All The Information Needed To Use Ultomiris Safely And Effectively. See Full Prescribing Information For Ultomiris.
a9a590d9-0217-43c7-908d-e62a71279791
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ULTOMIRIS, a complement inhibitor, increases the risk of serious infections caused by Neisseria meningitidis [see Warnings and Precautions (5.1) ] . Life-threatening and fatal meningococcal infections have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early . Complete or update vaccination for meningococcal bacteria (for serogroups A, C, W, Y, and B) at least 2 weeks prior to the first dose of ULTOMIRIS, unless the risks of delaying therapy with ULTOMIRIS outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against meningococcal bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by meningococcal bacteria. Patients receiving ULTOMIRIS are at increased risk for invasive disease caused by Neisseria meningitidis , even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious meningococcal infections and evaluate immediately if infection is suspected. Because of the risk of serious meningococcal infections, ULTOMIRIS is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called ULTOMIRIS and SOLIRIS REMS [see Warnings and Precautions (5.2) ].
Indications and Usage
ULTOMIRIS is a complement inhibitor indicated for: the treatment of adult and pediatric patients one month of age and older with paroxysmal nocturnal hemoglobinuria (PNH). ( 1.1 ) the treatment of adult and pediatric patients one month of age and older with atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy (TMA). ( 1.2 ) Limitations of Use: ULTOMIRIS is not indicated for the treatment of patients with Shiga toxin E. coli related hemolytic uremic syndrome (STEC-HUS). the treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody-positive. ( 1.3 ) the treatment of adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are anti-aquaporin-4 (AQP4) antibody-positive. ( 1.4 )
Dosage and Administration
See Full Prescribing Information for instructions on dosage, preparation, and administration. ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 ) Dilute ULTOMIRIS before use. ( 2.5 ) Only administer as an intravenous infusion through a 0.2 or 0.22 micron filter. ( 2.5 )
Warnings and Precautions
Other Infections: Use caution when administering ULTOMIRIS to patients with any other systemic infection. ( 5.3 ) Infusion-Related Reactions: Monitor during infusion, interrupt for reactions, and institute appropriate supportive measures. ( 5.6 )
Contraindications
ULTOMIRIS is contraindicated for initiation in patients with unresolved serious Neisseria meningitidis infection [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: Serious Meningococcal Infections [see Warnings and Precautions (5.1) ] Other Infections [see Warnings and Precautions (5.3) ] Infusion-Related Reactions [see Warnings and Precautions (5.6) ]
Drug Interactions
Plasma Exchange, Plasmapheresis, or Intravenous Immunoglobulins: concomitant use requires supplemental dose of ULTOMIRIS. ( 7.1 ) Neonatal Fc Receptor Blockers (FcRn): Closely monitor for reduced effectiveness of ULTOMIRIS. ( 7.2 )
Storage and Handling
Store ULTOMIRIS vials refrigerated at 2°C - 8°C (36°F - 46°F) in the original carton to protect from light. Do not freeze. Do not shake. Refer to Dosage and Administration (2) for information on the stability and storage of diluted solutions of ULTOMIRIS.
How Supplied
ULTOMIRIS (ravulizumab-cwvz) injection is translucent, clear to yellowish color solution supplied in one single-dose vial per carton as: 300 mg/3 mL (100 mg/mL) (NDC 25682-025-01) 1,100 mg/11 mL (100 mg/mL) (NDC 25682-028-01)
Medication Information
Warnings and Precautions
Other Infections: Use caution when administering ULTOMIRIS to patients with any other systemic infection. ( 5.3 ) Infusion-Related Reactions: Monitor during infusion, interrupt for reactions, and institute appropriate supportive measures. ( 5.6 )
Indications and Usage
ULTOMIRIS is a complement inhibitor indicated for: the treatment of adult and pediatric patients one month of age and older with paroxysmal nocturnal hemoglobinuria (PNH). ( 1.1 ) the treatment of adult and pediatric patients one month of age and older with atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy (TMA). ( 1.2 ) Limitations of Use: ULTOMIRIS is not indicated for the treatment of patients with Shiga toxin E. coli related hemolytic uremic syndrome (STEC-HUS). the treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody-positive. ( 1.3 ) the treatment of adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are anti-aquaporin-4 (AQP4) antibody-positive. ( 1.4 )
Dosage and Administration
See Full Prescribing Information for instructions on dosage, preparation, and administration. ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 ) Dilute ULTOMIRIS before use. ( 2.5 ) Only administer as an intravenous infusion through a 0.2 or 0.22 micron filter. ( 2.5 )
Contraindications
ULTOMIRIS is contraindicated for initiation in patients with unresolved serious Neisseria meningitidis infection [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: Serious Meningococcal Infections [see Warnings and Precautions (5.1) ] Other Infections [see Warnings and Precautions (5.3) ] Infusion-Related Reactions [see Warnings and Precautions (5.6) ]
Drug Interactions
Plasma Exchange, Plasmapheresis, or Intravenous Immunoglobulins: concomitant use requires supplemental dose of ULTOMIRIS. ( 7.1 ) Neonatal Fc Receptor Blockers (FcRn): Closely monitor for reduced effectiveness of ULTOMIRIS. ( 7.2 )
Storage and Handling
Store ULTOMIRIS vials refrigerated at 2°C - 8°C (36°F - 46°F) in the original carton to protect from light. Do not freeze. Do not shake. Refer to Dosage and Administration (2) for information on the stability and storage of diluted solutions of ULTOMIRIS.
How Supplied
ULTOMIRIS (ravulizumab-cwvz) injection is translucent, clear to yellowish color solution supplied in one single-dose vial per carton as: 300 mg/3 mL (100 mg/mL) (NDC 25682-025-01) 1,100 mg/11 mL (100 mg/mL) (NDC 25682-028-01)
Description
ULTOMIRIS, a complement inhibitor, increases the risk of serious infections caused by Neisseria meningitidis [see Warnings and Precautions (5.1) ] . Life-threatening and fatal meningococcal infections have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early . Complete or update vaccination for meningococcal bacteria (for serogroups A, C, W, Y, and B) at least 2 weeks prior to the first dose of ULTOMIRIS, unless the risks of delaying therapy with ULTOMIRIS outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against meningococcal bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by meningococcal bacteria. Patients receiving ULTOMIRIS are at increased risk for invasive disease caused by Neisseria meningitidis , even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious meningococcal infections and evaluate immediately if infection is suspected. Because of the risk of serious meningococcal infections, ULTOMIRIS is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called ULTOMIRIS and SOLIRIS REMS [see Warnings and Precautions (5.2) ].
Section 42229-5
Limitations of Use:
ULTOMIRIS is not indicated for the treatment of patients with Shiga toxin E. coli related hemolytic uremic syndrome (STEC-HUS).
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 09/2025 | ||
|
MEDICATION GUIDE ULTOMIRIS® (ul-toe-meer-is) (ravulizumab-cwvz) injection, for intravenous use |
|||
|
What is the most important information I should know about ULTOMIRIS? |
|||
|
|||
|
|
||
| Your healthcare provider will give you a Patient Safety Card about the risk of serious meningococcal infection. Carry it with you at all times during treatment and for 8 months after your last dose of ULTOMIRIS. Your risk of meningococcal infection may continue for several months after your last dose of ULTOMIRIS. It is important to show this card to any healthcare provider who treats you. This will help them diagnose and treat you quickly. | |||
| ULTOMIRIS is only available through a program called the ULTOMIRIS and SOLIRIS Risk Evaluation and Mitigation Strategy (REMS). Before you can receive ULTOMIRIS, your healthcare provider must: | |||
|
|||
| ULTOMIRIS may also increase the risk of other types of serious infections caused by encapsulated bacteria, including Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria gonorrhoeae . | |||
|
|||
| For more information about side effects, see "What are the possible side effects of ULTOMIRIS?" | |||
| What is ULTOMIRIS? | |||
ULTOMIRIS is a prescription medicine called a monoclonal antibody. ULTOMIRIS is used to treat:
|
|||
| It is not known if ULTOMIRIS is safe and effective in children younger than 1 month of age. It is not known if ULTOMIRIS is safe and effective for the treatment of gMG or NMOSD in children. |
|||
|
Who should not receive ULTOMIRIS?
Do not receive ULTOMIRIS if you have a serious meningococcal infection when you are starting ULTOMIRIS treatment. |
|||
| Before you receive ULTOMIRIS, tell your healthcare provider about all of your medical conditions, including if you: | |||
|
|||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. ULTOMIRIS and other medicines can affect each other causing side effects. Know the medicines you take and the vaccines you receive. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
| How should I receive ULTOMIRIS? | |||
|
|||
| Adults with PNH, aHUS, gMG, or NMOSD when administered intravenously (by vein) | |||
|
|||
| Children 1 month of age and older with PNH or aHUS when administered intravenously (by vein) | |||
| Your child will be given intravenous ULTOMIRIS infusion by a healthcare provider through a needle placed in a vein | |||
|
|||
If you are changing treatment from SOLIRIS to ULTOMIRIS, you should receive your starting dose of ULTOMIRIS at time of your next scheduled dose of SOLIRIS.
|
|||
|
|
||
|
|||
|
|
||
| If you miss an ULTOMIRIS infusion, call your healthcare provider right away. | |||
|
What are the possible side effects of ULTOMIRIS? |
|||
|
|||
|
|
||
| The most common side effects of ULTOMIRIS in people treated for PNH are: | |||
|
|
||
| The most common side effects of ULTOMIRIS in people treated for aHUS are: | |||
|
|
||
| The most common side effects of ULTOMIRIS in people with gMG are: | |||
|
|
||
| The most common side effects of ULTOMIRIS in people with NMOSD are: | |||
|
|
||
| Tell your healthcare provider about any side effect that bothers you or that does not go away. These are not all of the possible side effects of ULTOMIRIS. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of ULTOMIRIS.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about ULTOMIRIS that is written for health professionals. |
|||
| What are the ingredients in ULTOMIRIS? | |||
| Active ingredient: ravulizumab-cwvz. | |||
| Inactive ingredients: L-arginine, polysorbate 80 (vegetable origin), sodium phosphate dibasic, sodium phosphate monobasic, sucrose and Water for Injection. | |||
| Manufactured by Alexion Pharmaceuticals, Inc., 121 Seaport Boulevard, Boston, MA 02210 USA. U.S. License Number 1743 ULTOMIRIS is a registered trademark of Alexion Pharmaceuticals, Inc. © 2025 Alexion Pharmaceuticals, Inc. For more information, go to www.ULTOMIRIS.com or call: 1-888-765-4747. |
|||
Section 43683-2
11 Description
Ravulizumab-cwvz, a complement inhibitor, is a humanized monoclonal antibody (mAb) produced in Chinese hamster ovary (CHO) cells. Ravulizumab-cwvz consists of 2 identical 448 amino acid heavy chains and 2 identical 214 amino acid light chains and has a molecular weight of approximately 148 kDa. The constant regions of ravulizumab-cwvz include the human kappa light chain constant region, and the protein engineered "IgG2/4" heavy chain constant region.
The heavy chain CH1 domain, hinge region, and the first 5 amino acids of the CH2 domain match the human IgG2 amino acid sequence, residues 6 to 36 in the CH2 region (common to both human IgG2 and IgG4 amino acid sequences), while the remainder of the CH2 domain and the CH3 domain match the human IgG4 amino acid sequence. The heavy and light chain variable regions that form the human C5 binding site consist of human framework regions grafted to murine complementarity-determining regions.
ULTOMIRIS (ravulizumab-cwvz) injection is a sterile, translucent, clear to yellowish color, preservative-free solution for intravenous use. Each single-dose vial contains 300 mg or 1,100 mg ravulizumab-cwvz at a concentration of 100 mg/mL with a pH of 7.4. Each mL also contains L-arginine (4.33 mg), polysorbate 80 (0.5 mg) (vegetable origin), sodium phosphate dibasic (4.42 mg), sodium phosphate monobasic (4.57 mg), sucrose (50 mg), and Water for Injection, USP.
16.1 How Supplied
ULTOMIRIS (ravulizumab-cwvz) injection is translucent, clear to yellowish color solution supplied in one single-dose vial per carton as:
- 300 mg/3 mL (100 mg/mL) (NDC 25682-025-01)
- 1,100 mg/11 mL (100 mg/mL) (NDC 25682-028-01)
8.4 Pediatric Use
The safety and effectiveness of ULTOMIRIS for the treatment of PNH have been established in pediatric patients aged one month and older. Use of ULTOMIRIS for this indication is supported by evidence from adequate and well-controlled trials in adults with additional pharmacokinetic, efficacy and safety data in pediatric patients aged 9 to 17 years [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)]. The safety and efficacy for the treatment of pediatric and adult patients with PNH appear similar. Use of ULTOMIRIS in pediatric patients with PNH aged less than 9 years and body weight < 30 kg is based on extrapolation of pharmacokinetic / pharmacodynamic (PK/PD), and efficacy and safety data from aHUS and PNH clinical studies [see Clinical Pharmacology (12.3) and Clinical Studies (14)].
The safety and effectiveness of ULTOMIRIS for the treatment of aHUS have been established in pediatric patients aged one month and older. Use of ULTOMIRIS for this indication is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic, safety, and efficacy data in pediatric patients aged 10 months to < 17 years. The safety and efficacy of ULTOMIRIS for the treatment of aHUS appear similar in pediatric and adult patients [see Adverse Reactions (6.1), and Clinical Studies (14.2)].
The safety and effectiveness of ULTOMIRIS for the treatment of gMG or NMOSD in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ULTOMIRIS did not include sufficient numbers of subjects aged 65 and over (58 patients with PNH, 9 with aHUS, 54 with gMG, and 7 with NMOSD) to determine whether they respond differently from younger subjects.
Other reported clinical experience has not identified differences in responses between elderly and younger patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of ULTOMIRIS or other ravulizumab-cwvz products.
The immunogenicity of ravulizumab-cwvz has been evaluated using an enzyme-linked immunosorbent assay (ELISA) for the detection of binding anti-ravulizumab-cwvz antibodies. For patients whose sera tested positive in the screening immunoassay, an in vitro biological assay was performed to detect neutralizing antibodies.
In clinical studies with ULTOMIRIS, treatment-emergent antibodies to ravulizumab-cwvz were detected in 1 of 219 (0.5%) patients with PNH [see Clinical Studies (14.1)] and 1 of 71 (1.4%) patients with aHUS [see Clinical Studies (14.2)]. In these 2 patient populations, the observed ADA were non-neutralizing with no apparent impact on PK, safety, or efficacy. In the gMG study (N=86) and NMOSD study (N=58), no treatment-emergent antibodies to ravulizumab-cwvz were detected [see Clinical Studies (14.3 & 14.4)].
However, the assay used to measure anti-drug antibodies (ADA) is subject to interference by serum ravulizumab-cwvz, possibly resulting in an underestimation of the incidence of antibody formation. Due to the limitation of the assay conditions, the potential clinical impact of antibodies to ravulizumab-cwvz is not known.
4 Contraindications
ULTOMIRIS is contraindicated for initiation in patients with unresolved serious Neisseria meningitidis infection [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Serious Meningococcal Infections [see Warnings and Precautions (5.1)]
- Other Infections [see Warnings and Precautions (5.3)]
- Infusion-Related Reactions [see Warnings and Precautions (5.6)]
7 Drug Interactions
5.3 Other Infections
Serious infections with Neisseria species (other than Neisseria meningitidis), including disseminated gonococcal infections, have been reported.
ULTOMIRIS blocks terminal complement activation; therefore, patients may have increased susceptibility to infections, especially with encapsulated bacteria, such as infections caused by Neisseria meningitidis but also Streptococcus pneumoniae, Haemophilus influenzae, and to a lesser extent, Neisseria gonorrhoeae. Children treated with ULTOMIRIS may be at increased risk of developing serious infections due to Streptococcus pneumoniae and Haemophilus influenzae type b (Hib). Administer vaccinations for the prevention of Streptococcus pneumoniae and Haemophilus influenzae type b (Hib) infections according to ACIP recommendations. Patients receiving ULTOMIRIS are at increased risk for infections due to these organisms, even if they develop antibodies following vaccination.
12.2 Pharmacodynamics
Complete inhibition of serum free C5 (concentration of less than 0.5 mcg/mL) was observed by the end of the first ULTOMIRIS infusion and sustained throughout the entire 26-week treatment period in both adult and pediatric patients with PNH, in the majority (93%) of adult and pediatric patients with aHUS, in all adult patients with gMG, and in the majority (98.3%) of adult patients with NMOSD.
The extent and duration of the pharmacodynamic response in patients with PNH, aHUS, gMG, or NMOSD were exposure-dependent for ULTOMIRIS. Free C5 levels of < 0.5 mcg/mL were correlated with maximal intravascular hemolysis control and complete terminal complement inhibition in patients with PNH.
Complete terminal complement inhibition following initiation of ULTOMIRIS treatment led to normalization of serum LDH by week 4 in complement-inhibitor naïve patients with PNH, and maintained LDH normalization in patients previously treated with eculizumab with PNH [see Clinical Studies (14)].
12.3 Pharmacokinetics
Following ULTOMIRIS treatment, ravulizumab-cwvz pharmacokinetics increase proportionally over a dose range of 200 to 5400 mg. Ravulizumab-cwvz Cmax and Ctrough parameters are presented in Table 14, Table 15, Table 16, and Table 17.
| Pediatric Patients | Adult Patients | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| ALXN1210-PNH-304 | ALXN1210-PNH-301 | ALXN1210-PNH-302 | |||||||
| N | Complement Inhibitor-Naïve | N | Previously Treated with Eculizumab | N | Complement Inhibitor-Naïve | N | Previously Treated with Eculizumab | ||
| Abbreviations: LD = Loading Dose; MD = Maintenance Dose | |||||||||
| Cmax
(mcg/mL) |
LD | 4 | 733 (14.5) | 8 | 885 (19.3) | 125 | 771 (21.5) | 95 | 843 (24.1) |
| MD | 4 | 1490 (26.7) | 8 | 1705 (9.7) | 124 | 1,379 (20.0) | 95 | 1,386 (19.4) | |
| Ctrough
(mcg/mL) |
LD | 4 | 368 (14.7) | 8 | 452 (15.1) | 125 | 391 (35.0) | 96 | 405 (29.9) |
| MD | 4 | 495 (21.3) | 8 | 566 (12.2) | 124 | 473 (33.4) | 95 | 501 (28.6) |
| Pediatric Patients (ALXN1210-aHUS-312) |
Adult Patients (ALXN1210-aHUS-311) |
||||||
|---|---|---|---|---|---|---|---|
| N | < 20 kg MD Q4W |
N | ≥ 20 to < 40 kg MD Q8W |
N | ≥ 40 kg MD Q8W |
||
| Abbreviations: LD = Loading Dose; MD = Maintenance Dose; Q4W = Every 4 Weeks; Q8W = Every 8 Weeks | |||||||
| Cmax
(mcg/mL) |
LD | 8 | 656 (38.1) | 4 | 600 (17.3) | 52 | 754 (35.2) |
| MD | 7 | 1,467 (37.8) | 6 | 1,863 (15.3) | 46 | 1,458 (17.6) | |
| Ctrough
(mcg/mL) |
LD | 9 | 241 (52.1) | 5 | 186 (16.5) | 55 | 313 (33.9) |
| MD | 7 | 683 (46.1) | 6 | 549 (34.1) | 46 | 507 (42.5) |
| N | Adult Patients (ALXN1210-MG-306) | ||
|---|---|---|---|
| Abbreviations: LD = Loading Dose; MD=Maintenance Dose | |||
| Cmax
(mcg/mL) |
LD | 86 | 874 (21.1) |
| MD | 76 | 1548 (23.2) | |
| Ctrough
(mcg/mL) |
LD | 85 | 418 (27.6) |
| MD | 70 | 587 (29.6) |
| N | Adult Patients (ALXN1210-NMO-307) | ||
|---|---|---|---|
| Abbreviations: LD = Loading Dose; MD=Maintenance Dose | |||
| Cmax
(mcg/mL) |
LD | 58 | 935.3 (17.3) |
| MD | 56 | 1836.4 (19.4) | |
| Ctrough
(mcg/mL) |
LD | 58 | 459.1 (19.7) |
| MD | 54 | 796.9 (27.1) |
1 Indications and Usage
ULTOMIRIS is a complement inhibitor indicated for:
- the treatment of adult and pediatric patients one month of age and older with paroxysmal nocturnal hemoglobinuria (PNH). (1.1)
- the treatment of adult and pediatric patients one month of age and older with atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy (TMA). (1.2)
Limitations of Use:
ULTOMIRIS is not indicated for the treatment of patients with Shiga toxin E. coli related hemolytic uremic syndrome (STEC-HUS). - the treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody-positive. (1.3)
- the treatment of adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are anti-aquaporin-4 (AQP4) antibody-positive. (1.4)
12.1 Mechanism of Action
Ravulizumab-cwvz is a terminal complement inhibitor that specifically binds to the complement protein C5 with high affinity, thereby inhibiting its cleavage to C5a (the proinflammatory anaphylatoxin) and C5b (the initiating subunit of the membrane attack complex [MAC or C5b-9]) thus preventing MAC formation. ULTOMIRIS inhibits terminal complement-mediated intravascular hemolysis in patients with PNH and complement-mediated thrombotic microangiopathy (TMA) in patients with aHUS.
The precise mechanism by which ravulizumab-cwvz exerts its therapeutic effect in gMG patients is unknown, but is presumed to involve reduction of terminal complement complex C5b-9 deposition at the neuromuscular junction.
The precise mechanism by which ravulizumab-cwvz exerts its therapeutic effect in NMOSD is unknown, but is presumed to involve inhibition of aquaporin-4 antibody-induced terminal complement C5b-9 deposition.
16.2 Storage and Handling
Store ULTOMIRIS vials refrigerated at 2°C - 8°C (36°F - 46°F) in the original carton to protect from light. Do not freeze. Do not shake.
Refer to Dosage and Administration (2) for information on the stability and storage of diluted solutions of ULTOMIRIS.
5 Warnings and Precautions
2 Dosage and Administration
3 Dosage Forms and Strengths
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ULTOMIRIS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to ULTOMIRIS exposure.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.2 Ultomiris and Soliris Rems
ULTOMIRIS is available only through a restricted program under a REMS called ULTOMIRIS and SOLIRIS REMS, because of the risk of serious meningococcal infections [see Warnings and Precautions (5.1)].
Notable requirements of the ULTOMIRIS and SOLIRIS REMS include the following:
- Prescribers must enroll in the REMS.
- Prescribers must counsel patients about the risk of serious meningococcal infection.
- Prescribers must provide the patients with the REMS educational materials.
- Prescribers must assess patient vaccination status for meningococcal vaccines (against serogroups A, C, W, Y, and B) and vaccinate if needed according to current ACIP recommendations two weeks prior to the first dose of ULTOMIRIS.
- Prescribers must provide a prescription for antibacterial drug prophylaxis if treatment must be started urgently and the patient is not up to date with meningococcal vaccines according to current ACIP recommendations at least two weeks prior to the first dose of ULTOMIRIS.
- Healthcare settings and pharmacies that dispense ULTOMIRIS must be certified in the REMS and must verify prescribers are certified.
- Patients must receive counseling from the prescriber about the need to receive meningococcal vaccines per ACIP recommendations, the need to take antibiotics as directed by the prescriber, and the signs and symptoms of meningococcal infection.
- Patients must be instructed to carry the Patient Safety Card with them at all times during and for 8 months following treatment with ULTOMIRIS.
Further information is available at www.UltSolREMS.com or 1-888-765-4747.
5.6 Infusion Related Reactions
Administration of ULTOMIRIS may result in systemic infusion-related reactions, including anaphylaxis [see Adverse Reactions (6.2)] and hypersensitivity reactions. In clinical trials, infusion-related reactions occurred in approximately 1 to 7% of patients treated with ULTOMIRIS [see Adverse Reactions (6.1)]. These events included lower back pain, abdominal pain, muscle spasms, drop in blood pressure, elevation in blood pressure, rigors, limb discomfort, drug hypersensitivity (allergic reaction), and dysgeusia (bad taste). These reactions did not require discontinuation of ULTOMIRIS. If signs of cardiovascular instability or respiratory compromise occur, interrupt ULTOMIRIS infusion and institute appropriate supportive measures.
2.1 Important Dosage Information
ULTOMIRIS is intended to be administered only as an intravenous infusion in adult or pediatric patients one month of age and older.
1.3 Generalized Myasthenia Gravis
ULTOMIRIS is indicated for the treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody-positive.
17 Patient Counseling Information
Advise the patients and/or caregivers to read the FDA-approved patient labeling (Medication Guide).
7.2 Neonatal Fc Receptor Blockers
Concomitant use of ULTOMIRIS with neonatal Fc receptor (FcRn) blockers (e.g., efgartigimod) may lower systemic exposures and reduce effectiveness of ULTOMIRIS. Closely monitor for reduced effectiveness of ULTOMIRIS.
5.5 Thromboembolic Event Management
The effect of withdrawal of anticoagulant therapy during ULTOMIRIS treatment has not been established. Therefore, treatment with ULTOMIRIS should not alter anticoagulant management.
5.1 Serious Meningococcal Infections
ULTOMIRIS, a complement inhibitor, increases a patient's susceptibility to serious, life-threatening, or fatal infections caused by meningococcal bacteria (septicemia and/or meningitis) in any serogroup, including non-groupable strains. Life-threatening and fatal meningococcal infections have occurred in both vaccinated and unvaccinated patients treated with complement inhibitors. The initiation of ULTOMIRIS treatment is contraindicated in patients with unresolved serious Neisseria meningitidis infection.
Complete or update meningococcal vaccination (for serogroups A, C, W, Y and B) at least 2 weeks prior to administration of the first dose of ULTOMIRIS, according to current ACIP recommendations for patients receiving a complement inhibitor. Revaccinate patients in accordance with ACIP recommendations considering the duration of ULTOMIRIS therapy. Note that ACIP recommends an administration schedule in patients receiving complement inhibitors that differs from the administration schedule in the vaccine prescribing information. If urgent ULTOMIRIS therapy is indicated in a patient who is not up to date with meningococcal vaccines according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer meningococcal vaccines as soon as possible. Various durations and regimens of antibacterial drug prophylaxis have been considered, but the optimal durations and drug regimens for prophylaxis and their efficacy have not been studied in unvaccinated or vaccinated patients receiving complement inhibitors, including ULTOMIRIS. The benefits and risks of treatment with ULTOMIRIS, as well as the benefits and risks of antibacterial drug prophylaxis in unvaccinated or vaccinated patients, must be considered against the known risks for serious infections caused by Neisseria meningitidis.
Vaccination does not eliminate the risk of meningococcal infections, despite development of antibodies following vaccination.
Closely monitor patients for early signs and symptoms of meningococcal infection and evaluate patients immediately if infection is suspected. Inform patients of these signs and symptoms and instruct patients to seek immediate medical care if these signs and symptoms occur. Promptly treat known infections. Meningococcal infection may become rapidly life-threatening or fatal if not recognized and treated early. Consider interruption of ULTOMIRIS in patients who are undergoing treatment for serious meningococcal infection, depending on the risks of interrupting treatment in the disease being treated.
ULTOMIRIS is available only through a restricted program under a REMS [see Warnings and Precautions (5.2)].
1.2 Atypical Hemolytic Uremic Syndrome
ULTOMIRIS is indicated for the treatment of adult and pediatric patients one month of age and older with atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy (TMA).
1.1 Paroxysmal Nocturnal Hemoglobinuria
ULTOMIRIS is indicated for the treatment of adult and pediatric patients one month of age and older with paroxysmal nocturnal hemoglobinuria (PNH).
14.3 Generalized Myasthenia Gravis (gmg)
The efficacy of ULTOMIRIS for the treatment of gMG was demonstrated in a randomized, double-blind, placebo-controlled, multicenter study (ALXN1210-MG-306; NCT03920293). Patients were randomized 1:1 to either receive ULTOMIRIS (n=86) or placebo (n=89) for 26 weeks. ULTOMIRIS was administered intravenously according to the weight-based recommended dosage [see Dosage and Administration (2.3)].
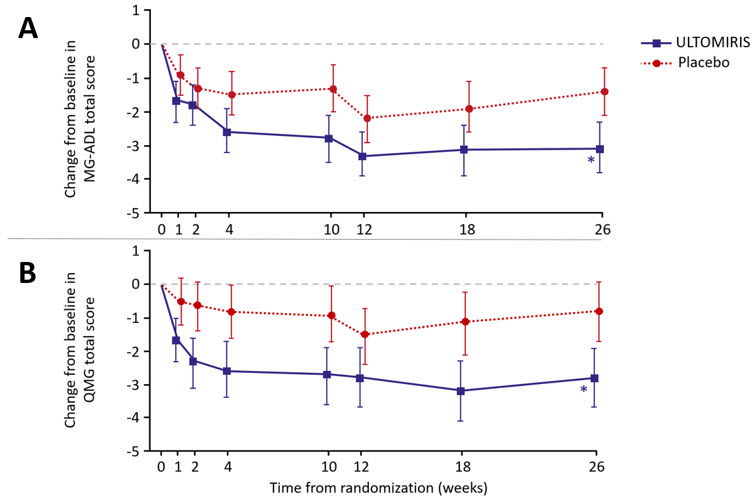
Patients with gMG with a positive serologic test for anti-AChR antibodies, Myasthenia Gravis Foundation of America (MGFA) clinical classification class II to IV, and Myasthenia Gravis-Activities of Daily Living (MG-ADL) total score ≥ 6 were enrolled. Baseline and disease characteristics were similar between treatment groups (including age at first dose [mean of 58 years for ULTOMIRIS versus 53 years for placebo], gender [51% female for ULTOMIRIS versus 51% female for placebo], race as White, Asian, and Black or African American [78%, 17%, and 2% for ULTOMIRIS versus 69%, 18%, and 5% for placebo, respectively], and duration of MG since diagnosis [mean of 10 years, ranging from 0.5 to 39.5 years, for ULTOMIRIS versus mean of 10.0 years, ranging from 0.5 to 36.1 years, for placebo].
Over 80% of patients were receiving acetylcholinesterase inhibitors, 70% were receiving corticosteroids, and 68% were receiving non-steroidal immunosuppressants (ISTs) at study entry. Patients on concomitant medications to treat gMG were permitted to continue on therapy throughout the course of the study.
The primary efficacy endpoint was a comparison of the change from baseline between treatment groups in the MG-ADL total score at Week 26. The MG-ADL is a categorical scale that assesses the impact on daily function of 8 signs or symptoms that are typically affected in gMG. Each item is assessed on a 4-point scale where a score of 0 represents normal function and a score of 3 represents loss of ability to perform that function. The total score ranges from 0 to 24, with the higher scores indicating more impairment.
The secondary endpoints, also assessed from baseline to Week 26, included the change in the Quantitative MG total score (QMG). The QMG is a 13-item categorical scale assessing muscle weakness. Each item is assessed on a 4-point scale where a score of 0 represents no weakness and a score of 3 represents severe weakness. A total score ranges from 0 to 39, where higher scores indicate more severe impairment.
Other secondary endpoints included the proportion of patients with improvements of at least 5 and 3 points in the QMG and MG-ADL total scores, respectively.
Treatment with ULTOMIRIS demonstrated a statistically significant change in the MG-ADL and QMG total scores from baseline at Week 26 as compared to placebo (Table 29).
| Efficacy Endpoints: Change from Baseline at Week 26 | Placebo (n = 89) LS Mean |
ULTOMIRIS (n = 86) LS Mean |
Treatment Effect (95% CI) |
p-value p-value calculated using mixed effect model for repeated measures
|
|---|---|---|---|---|
| Abbreviations: CI = confidence interval, LS = least squares; MG-ADL = Myasthenia Gravis Activities of Daily Living profile; QMG = Quantitative Myasthenia Gravis score for disease severity | ||||
| Primary Endpoint | ||||
| MG-ADL | -1.4 | -3.1 | -1.6 (-2.6, -0.7) | < 0.001 |
| Secondary Endpoint | ||||
| QMG | -0.8 | -2.8 | -2.0 (-3.2, -0.8) | < 0.001 |
The proportion of QMG responders with at least a 5-point improvement at week 26 was greater for ULTOMIRIS (30.0%) compared to placebo (11.3%) p = 0.005. The proportion of MG-ADL responders with at least a 3-point improvement at week 26 was also greater for ULTOMIRIS (56.7%) compared to placebo (34.1%). The proportion of clinical responders at higher response thresholds (≥ 4-, 5-, 6-, 7-, or 8-point improvement on MG-ADL, and ≥ 6-, 7-, 8-, 9-, or 10-point improvement on QMG) was consistently greater for ULTOMIRIS compared to placebo.
Figure 1: Change from Baseline in MG-ADL Total Score (A) and QMG Total Score (B) Through Week 26 of the Randomized Controlled Period of ALXN1210-MG-306 (Mean and 95% CI)
Note: *p<0.001 versus placebo
Warning: Serious Meningococcal Infections
ULTOMIRIS, a complement inhibitor, increases the risk of serious infections caused by Neisseria meningitidis [see Warnings and Precautions (5.1)]. Life-threatening and fatal meningococcal infections have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early.
- Complete or update vaccination for meningococcal bacteria (for serogroups A, C, W, Y, and B) at least 2 weeks prior to the first dose of ULTOMIRIS, unless the risks of delaying therapy with ULTOMIRIS outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against meningococcal bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by meningococcal bacteria.
- Patients receiving ULTOMIRIS are at increased risk for invasive disease caused by Neisseria meningitidis, even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious meningococcal infections and evaluate immediately if infection is suspected.
Because of the risk of serious meningococcal infections, ULTOMIRIS is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called ULTOMIRIS and SOLIRIS REMS [see Warnings and Precautions (5.2)].
1.4 Neuromyelitis Optica Spectrum Disorder
ULTOMIRIS is indicated for the treatment of adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are anti-aquaporin-4 (AQP4) antibody-positive.
Principal Display Panel 3 Ml Vial Carton
Rx only
NDC 25682-025-01
ULTOMIRIS®
(ravulizumab-cwvz)
Injection
300 mg/3 mL
(100 mg/mL)
For Intravenous Infusion
Dilute with 0.9% Sodium Chloride
Injection prior to use.
3 mL Single-dose vial
Discard Unused Portion
ATTENTION: Dispense the
enclosed Medication Guide to
each patient
Principal Display Panel 11 Ml Vial Carton
Rx only
NDC 25682-028-01
ULTOMIRIS®
(ravulizumab-cwvz)
Injection
1,100 mg/11mL
(100 mg/mL)
For Intravenous Infusion
Dilute with 0.9% Sodium Chloride
Injection prior to use.
11 mL Single-dose vial
Discard Unused Portion
ATTENTION: Dispense the
enclosed Medication Guide to
each patient.
Principal Display Panel 30 Ml Vial Carton
NDC 25682-022-01
ULTOMIRIS®
(ravulizumab-cwvz)
injection
300 mg/30 mL
(10 mg/mL)
For Intravenous Infusion
Dilute with 0.9% Sodium Chloride
Injection prior to use.
Rx only
30 mL Single-dose vial
Discard Unused Portion
ATTENTION: Dispense the enclosed
Medication Guide to each patient
14.1 Paroxysmal Nocturnal Hemoglobinuria (pnh)
The safety and efficacy of ULTOMIRIS in adult patients with PNH was assessed in 2 open-label, randomized, active-controlled, non-inferiority Phase 3 studies: PNH Study 301 and PNH Study 302. Study 301 enrolled patients with PNH who were complement inhibitor-naïve and had active hemolysis. Study 302 enrolled patients with PNH who were clinically stable after having been treated with eculizumab for at least the past 6 months. The safety and efficacy of ULTOMIRIS in pediatric patients with PNH was assessed in PNH Study 304, an open-label, Phase 3 study conducted in eculizumab-experienced and complement inhibitor treatment-naïve pediatric patients with PNH.
In Study 301 and Study 302, adults with PNH were dosed with ULTOMIRIS administered intravenously in accordance with the weight-based dosing described in Section 2.3 (4 infusions of ULTOMIRIS over 26 weeks) above. Eculizumab was administered on Days 1, 8, 15, and 22, followed by maintenance treatment with 900 mg of eculizumab on Day 29 and every 2 weeks (q2w) thereafter for a total of 26 weeks of treatment, according to the approved dosing regimen of eculizumab which was the standard-of-care for PNH at the time of the studies.
Patients were vaccinated against meningococcal infection prior to or at the time of initiating treatment with ULTOMIRIS or eculizumab, or received prophylactic treatment with appropriate antibiotics until 2 weeks after vaccination. Prophylactic treatment with appropriate antibiotics beyond 2 weeks after vaccination was at the discretion of the provider.
14.2 Atypical Hemolytic Uremic Syndrome (ahus)
The efficacy of ULTOMIRIS administered intravenously in patients with aHUS was assessed in 2 open-label, single-arm studies. Study ALXN1210-aHUS-311 enrolled adult patients who displayed signs of TMA. In order to qualify for enrollment, patients were required to have a platelet count ≤ 150 × 109/L, evidence of hemolysis such as an elevation in serum LDH, and serum creatinine above the upper limits of normal or required dialysis.
Study ALXN1210-aHUS-312 enrolled pediatric patients who displayed signs of TMA. In order to qualify for enrollment, patients were required to have a platelet count ≤ 150 × 109/L, evidence of hemolysis such as an elevation in serum LDH, and serum creatinine level ≥ 97.5% percentile at screening or required dialysis. In both studies, enrollment criteria excluded patients presenting with TMA due to a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) deficiency, Shiga toxin Escherichia coli related hemolytic uremic syndrome (STEC-HUS) and genetic defect in cobalamin C metabolism. Patients with confirmed diagnosis of STEC-HUS after enrollment were excluded from the efficacy evaluation.
14.4 Neuromyelitis Optica Spectrum Disorder (nmosd)
The efficacy and safety of ULTOMIRIS in adult patients with anti-AQP4 antibody positive NMOSD was assessed in an open-label multicenter study, Study ALXN1210-NMO-307 (NCT04291262). Patients participating in Study ALXN1210-NMO-307 received ULTOMIRIS intravenously in the Primary Treatment Period that ended when the last enrolled patient completed (or discontinued prior to) 50 weeks on study, representing a median study duration of 73.5 weeks (minimum 13.7, maximum 117.7). Efficacy assessments were based on a comparison of patients in Study ALXN1210-NMO-307 with an external placebo control group from another study (Study ECU-NMO-301, NCT01892345) composed of a comparable population of adult patients with anti-AQP4 antibody positive NMOSD.
Study ALXN1210-NMO-307 enrolled 58 adult patients with NMOSD who had a positive serologic test for anti-AQP4 antibodies, at least 1 relapse in the last 12 months prior to the Screening Period, and an Expanded Disability Status Scale (EDSS) score ≤ 7. In the external placebo control group, eligibility criteria were similar except patients were required to have at least 2 relapses in last 12 months or 3 relapses in the last 24 months with at least 1 relapse in the 12 months prior to screening. Prior treatment with immunosuppressant therapies (ISTs) was not required for enrollment. However, patients on selected ISTs (i.e., corticosteroids, azathioprine, mycophenolate mofetil, methotrexate, and tacrolimus) were permitted to continue on therapy, with a requirement for stable dosing until they reached Week 106 in the Study. Similar IST use was permitted in the external placebo control group.
ULTOMIRIS was administered intravenously according to the weight-based recommended dosage [see Dosage and Administration (2.3)].
The demographics were similar between the ULTOMIRIS treatment group from Study ALXN1210-NMO-307 and the placebo treatment group from Study ECU-NMO-301 (including age [median of 46.0 years for ULTOMIRIS versus 44.0 years for placebo] and sex [89.7% female for ULTOMIRIS versus 89.4% female for placebo]). The majority of patients were White or Asian. The median time from diagnosis to first dose was 0.9 years for ULTOMIRIS and 2.0 years for placebo. The median annualized relapse rate (ARR) in the last 24 months was 1.4 for ULTOMIRIS versus 1.9 for placebo, and the median number of historical relapses was 2 for ULTOMIRIS versus 4 for placebo. The median baseline EDSS score was 3.3 for ULTOMIRIS versus 4.0 for placebo. At baseline, 48% of patients in the ULTOMIRIS group received concomitant IST, including corticosteroids, versus 72% of subjects in the placebo group.
The primary endpoint of Study ALXN1210-NMO-307 was the time to first adjudicated on-trial relapse as determined by an independent adjudication committee. No adjudicated on-trial relapses were observed in ULTOMIRIS-treated patients during the Primary Treatment Period, representing a statistically significant difference between the ULTOMIRIS and placebo treatment arms in time to first adjudicated on-trial relapse (p < 0.0001). The hazard ratio (95% confidence interval [CI]) for ULTOMIRIS compared with placebo was 0.014 (0.000, 0.103), representing a 98.6% reduction in the risk of relapse (Figure 2). ULTOMIRIS-treated patients experienced similar improvement in time to first adjudicated on-trial relapse with or without concomitant treatment.
Figure 2: Kaplan-Meier Survival Estimates for Time to First Adjudicated On-Trial Relapse in Study ALXN1210-NMO-307 and Comparative Placebo Arm of Study ECU-NMO-301
Note: The placebo group data were collected as part of Study ECU-NMO-301. Patients who did not experience an adjudicated on-trial relapse were censored at the end of the study period. If a patient in the placebo group was followed longer than any of the patients in the Ultomiris group, then that patient was censored at the longest Ultomiris follow-up time.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal carcinogenicity studies of ravulizumab-cwvz have not been conducted.
Genotoxicity studies have not been conducted with ravulizumab-cwvz.
Effects of ravulizumab-cwvz upon fertility have not been studied in animals. Intravenous injections of male and female mice with a murine anti-C5 antibody at up to 0.8-2.2 times the equivalent of the clinical dose of ULTOMIRIS had no adverse effects on mating or fertility.
7.1 Plasma Exchange, Plasmapheresis, and Intravenous Immunoglobulins
Concomitant use of ULTOMIRIS with plasma exchange (PE), plasmapheresis (PP), or intravenous immunoglobulin (IVIg) treatment can reduce serum ravulizumab concentrations and requires a supplemental dose of ULTOMIRIS [see Dosage and Administration (2.5)].
2.2 Recommended Vaccination and Prophylaxis for Meningococcal Infection
Vaccinate patients against meningococcal infection (serogroups A, C, W, Y and B) according to current ACIP recommendations at least 2 weeks prior to initiation of ULTOMIRIS [see Warnings and Precautions (5.1)].
If urgent ULTOMIRIS therapy is indicated in a patient who is not up to date with meningococcal vaccines according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer these vaccines as soon as possible.
Healthcare providers who prescribe ULTOMIRIS must enroll in the ULTOMIRIS and SOLIRIS REMS [see Warnings and Precautions (5.2)].
2.3 Recommended Dosage for Intravenous Administration in Adult and Pediatric Patients With Pnh Or Ahus, and in Adult Patients With Gmg Or Nmosd
The recommended intravenous (IV) ULTOMIRIS loading and maintenance dosing in adult and pediatric patients, one month of age or older weighing 5 kg or greater, with PNH or aHUS, or in adult patients with gMG or NMOSD weighing 40 kg or greater, is based on the patient's body weight, as shown in Table 1, with maintenance doses administered every 4 or 8 weeks, starting 2 weeks after loading dose.
The IV dosing schedule is allowed to occasionally vary within 7 days of the scheduled infusion day (except for the first maintenance dose of ULTOMIRIS); but subsequent doses should be administered according to the original schedule.
Following a missed IV ULTOMIRIS dose, the patient should contact their health care provider immediately.
| Indications | Body Weight Range (kg) |
Loading Dose (mg) See Table 2 for ULTOMIRIS treatment initiation instruction and timing of loading dose and maintenance dose
|
Maintenance Dose (mg) and Dosing Interval | |
|---|---|---|---|---|
| PNH or aHUS | 5 to less than 10 | 600 | 300 | Every 4 weeks |
| 10 to less than 20 | 600 | 600 | ||
| 20 to less than 30 | 900 | 2,100 | Every 8 weeks |
|
| 30 to less than 40 | 1,200 | 2,700 | ||
| PNH, aHUS, gMG, or NMOSD | 40 to less than 60 | 2,400 | 3,000 | Every 8 weeks |
| 60 to less than 100 | 2,700 | 3,300 | ||
| 100 or greater | 3,000 | 3,600 |
Refer to Table 2 for treatment initiation instructions in patients who are complement inhibitor treatment-naïve or switching treatment from eculizumab.
| Population | Weight-based ULTOMIRIS Loading Dose | Time of First ULTOMIRIS Weight-based Maintenance Dose |
|---|---|---|
| Not currently on ULTOMIRIS or eculizumab treatment | At treatment start | 2 weeks after ULTOMIRIS loading dose |
| Currently treated with eculizumab | At time of next scheduled eculizumab dose | 2 weeks after ULTOMIRIS loading dose |
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use:
ULTOMIRIS is not indicated for the treatment of patients with Shiga toxin E. coli related hemolytic uremic syndrome (STEC-HUS).
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 09/2025 | ||
|
MEDICATION GUIDE ULTOMIRIS® (ul-toe-meer-is) (ravulizumab-cwvz) injection, for intravenous use |
|||
|
What is the most important information I should know about ULTOMIRIS? |
|||
|
|||
|
|
||
| Your healthcare provider will give you a Patient Safety Card about the risk of serious meningococcal infection. Carry it with you at all times during treatment and for 8 months after your last dose of ULTOMIRIS. Your risk of meningococcal infection may continue for several months after your last dose of ULTOMIRIS. It is important to show this card to any healthcare provider who treats you. This will help them diagnose and treat you quickly. | |||
| ULTOMIRIS is only available through a program called the ULTOMIRIS and SOLIRIS Risk Evaluation and Mitigation Strategy (REMS). Before you can receive ULTOMIRIS, your healthcare provider must: | |||
|
|||
| ULTOMIRIS may also increase the risk of other types of serious infections caused by encapsulated bacteria, including Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria gonorrhoeae . | |||
|
|||
| For more information about side effects, see "What are the possible side effects of ULTOMIRIS?" | |||
| What is ULTOMIRIS? | |||
ULTOMIRIS is a prescription medicine called a monoclonal antibody. ULTOMIRIS is used to treat:
|
|||
| It is not known if ULTOMIRIS is safe and effective in children younger than 1 month of age. It is not known if ULTOMIRIS is safe and effective for the treatment of gMG or NMOSD in children. |
|||
|
Who should not receive ULTOMIRIS?
Do not receive ULTOMIRIS if you have a serious meningococcal infection when you are starting ULTOMIRIS treatment. |
|||
| Before you receive ULTOMIRIS, tell your healthcare provider about all of your medical conditions, including if you: | |||
|
|||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. ULTOMIRIS and other medicines can affect each other causing side effects. Know the medicines you take and the vaccines you receive. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
| How should I receive ULTOMIRIS? | |||
|
|||
| Adults with PNH, aHUS, gMG, or NMOSD when administered intravenously (by vein) | |||
|
|||
| Children 1 month of age and older with PNH or aHUS when administered intravenously (by vein) | |||
| Your child will be given intravenous ULTOMIRIS infusion by a healthcare provider through a needle placed in a vein | |||
|
|||
If you are changing treatment from SOLIRIS to ULTOMIRIS, you should receive your starting dose of ULTOMIRIS at time of your next scheduled dose of SOLIRIS.
|
|||
|
|
||
|
|||
|
|
||
| If you miss an ULTOMIRIS infusion, call your healthcare provider right away. | |||
|
What are the possible side effects of ULTOMIRIS? |
|||
|
|||
|
|
||
| The most common side effects of ULTOMIRIS in people treated for PNH are: | |||
|
|
||
| The most common side effects of ULTOMIRIS in people treated for aHUS are: | |||
|
|
||
| The most common side effects of ULTOMIRIS in people with gMG are: | |||
|
|
||
| The most common side effects of ULTOMIRIS in people with NMOSD are: | |||
|
|
||
| Tell your healthcare provider about any side effect that bothers you or that does not go away. These are not all of the possible side effects of ULTOMIRIS. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of ULTOMIRIS.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about ULTOMIRIS that is written for health professionals. |
|||
| What are the ingredients in ULTOMIRIS? | |||
| Active ingredient: ravulizumab-cwvz. | |||
| Inactive ingredients: L-arginine, polysorbate 80 (vegetable origin), sodium phosphate dibasic, sodium phosphate monobasic, sucrose and Water for Injection. | |||
| Manufactured by Alexion Pharmaceuticals, Inc., 121 Seaport Boulevard, Boston, MA 02210 USA. U.S. License Number 1743 ULTOMIRIS is a registered trademark of Alexion Pharmaceuticals, Inc. © 2025 Alexion Pharmaceuticals, Inc. For more information, go to www.ULTOMIRIS.com or call: 1-888-765-4747. |
|||
Section 43683-2 (43683-2)
11 Description (11 DESCRIPTION)
Ravulizumab-cwvz, a complement inhibitor, is a humanized monoclonal antibody (mAb) produced in Chinese hamster ovary (CHO) cells. Ravulizumab-cwvz consists of 2 identical 448 amino acid heavy chains and 2 identical 214 amino acid light chains and has a molecular weight of approximately 148 kDa. The constant regions of ravulizumab-cwvz include the human kappa light chain constant region, and the protein engineered "IgG2/4" heavy chain constant region.
The heavy chain CH1 domain, hinge region, and the first 5 amino acids of the CH2 domain match the human IgG2 amino acid sequence, residues 6 to 36 in the CH2 region (common to both human IgG2 and IgG4 amino acid sequences), while the remainder of the CH2 domain and the CH3 domain match the human IgG4 amino acid sequence. The heavy and light chain variable regions that form the human C5 binding site consist of human framework regions grafted to murine complementarity-determining regions.
ULTOMIRIS (ravulizumab-cwvz) injection is a sterile, translucent, clear to yellowish color, preservative-free solution for intravenous use. Each single-dose vial contains 300 mg or 1,100 mg ravulizumab-cwvz at a concentration of 100 mg/mL with a pH of 7.4. Each mL also contains L-arginine (4.33 mg), polysorbate 80 (0.5 mg) (vegetable origin), sodium phosphate dibasic (4.42 mg), sodium phosphate monobasic (4.57 mg), sucrose (50 mg), and Water for Injection, USP.
16.1 How Supplied
ULTOMIRIS (ravulizumab-cwvz) injection is translucent, clear to yellowish color solution supplied in one single-dose vial per carton as:
- 300 mg/3 mL (100 mg/mL) (NDC 25682-025-01)
- 1,100 mg/11 mL (100 mg/mL) (NDC 25682-028-01)
8.4 Pediatric Use
The safety and effectiveness of ULTOMIRIS for the treatment of PNH have been established in pediatric patients aged one month and older. Use of ULTOMIRIS for this indication is supported by evidence from adequate and well-controlled trials in adults with additional pharmacokinetic, efficacy and safety data in pediatric patients aged 9 to 17 years [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)]. The safety and efficacy for the treatment of pediatric and adult patients with PNH appear similar. Use of ULTOMIRIS in pediatric patients with PNH aged less than 9 years and body weight < 30 kg is based on extrapolation of pharmacokinetic / pharmacodynamic (PK/PD), and efficacy and safety data from aHUS and PNH clinical studies [see Clinical Pharmacology (12.3) and Clinical Studies (14)].
The safety and effectiveness of ULTOMIRIS for the treatment of aHUS have been established in pediatric patients aged one month and older. Use of ULTOMIRIS for this indication is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic, safety, and efficacy data in pediatric patients aged 10 months to < 17 years. The safety and efficacy of ULTOMIRIS for the treatment of aHUS appear similar in pediatric and adult patients [see Adverse Reactions (6.1), and Clinical Studies (14.2)].
The safety and effectiveness of ULTOMIRIS for the treatment of gMG or NMOSD in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ULTOMIRIS did not include sufficient numbers of subjects aged 65 and over (58 patients with PNH, 9 with aHUS, 54 with gMG, and 7 with NMOSD) to determine whether they respond differently from younger subjects.
Other reported clinical experience has not identified differences in responses between elderly and younger patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of ULTOMIRIS or other ravulizumab-cwvz products.
The immunogenicity of ravulizumab-cwvz has been evaluated using an enzyme-linked immunosorbent assay (ELISA) for the detection of binding anti-ravulizumab-cwvz antibodies. For patients whose sera tested positive in the screening immunoassay, an in vitro biological assay was performed to detect neutralizing antibodies.
In clinical studies with ULTOMIRIS, treatment-emergent antibodies to ravulizumab-cwvz were detected in 1 of 219 (0.5%) patients with PNH [see Clinical Studies (14.1)] and 1 of 71 (1.4%) patients with aHUS [see Clinical Studies (14.2)]. In these 2 patient populations, the observed ADA were non-neutralizing with no apparent impact on PK, safety, or efficacy. In the gMG study (N=86) and NMOSD study (N=58), no treatment-emergent antibodies to ravulizumab-cwvz were detected [see Clinical Studies (14.3 & 14.4)].
However, the assay used to measure anti-drug antibodies (ADA) is subject to interference by serum ravulizumab-cwvz, possibly resulting in an underestimation of the incidence of antibody formation. Due to the limitation of the assay conditions, the potential clinical impact of antibodies to ravulizumab-cwvz is not known.
4 Contraindications (4 CONTRAINDICATIONS)
ULTOMIRIS is contraindicated for initiation in patients with unresolved serious Neisseria meningitidis infection [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Serious Meningococcal Infections [see Warnings and Precautions (5.1)]
- Other Infections [see Warnings and Precautions (5.3)]
- Infusion-Related Reactions [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.3 Other Infections
Serious infections with Neisseria species (other than Neisseria meningitidis), including disseminated gonococcal infections, have been reported.
ULTOMIRIS blocks terminal complement activation; therefore, patients may have increased susceptibility to infections, especially with encapsulated bacteria, such as infections caused by Neisseria meningitidis but also Streptococcus pneumoniae, Haemophilus influenzae, and to a lesser extent, Neisseria gonorrhoeae. Children treated with ULTOMIRIS may be at increased risk of developing serious infections due to Streptococcus pneumoniae and Haemophilus influenzae type b (Hib). Administer vaccinations for the prevention of Streptococcus pneumoniae and Haemophilus influenzae type b (Hib) infections according to ACIP recommendations. Patients receiving ULTOMIRIS are at increased risk for infections due to these organisms, even if they develop antibodies following vaccination.
12.2 Pharmacodynamics
Complete inhibition of serum free C5 (concentration of less than 0.5 mcg/mL) was observed by the end of the first ULTOMIRIS infusion and sustained throughout the entire 26-week treatment period in both adult and pediatric patients with PNH, in the majority (93%) of adult and pediatric patients with aHUS, in all adult patients with gMG, and in the majority (98.3%) of adult patients with NMOSD.
The extent and duration of the pharmacodynamic response in patients with PNH, aHUS, gMG, or NMOSD were exposure-dependent for ULTOMIRIS. Free C5 levels of < 0.5 mcg/mL were correlated with maximal intravascular hemolysis control and complete terminal complement inhibition in patients with PNH.
Complete terminal complement inhibition following initiation of ULTOMIRIS treatment led to normalization of serum LDH by week 4 in complement-inhibitor naïve patients with PNH, and maintained LDH normalization in patients previously treated with eculizumab with PNH [see Clinical Studies (14)].
12.3 Pharmacokinetics
Following ULTOMIRIS treatment, ravulizumab-cwvz pharmacokinetics increase proportionally over a dose range of 200 to 5400 mg. Ravulizumab-cwvz Cmax and Ctrough parameters are presented in Table 14, Table 15, Table 16, and Table 17.
| Pediatric Patients | Adult Patients | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| ALXN1210-PNH-304 | ALXN1210-PNH-301 | ALXN1210-PNH-302 | |||||||
| N | Complement Inhibitor-Naïve | N | Previously Treated with Eculizumab | N | Complement Inhibitor-Naïve | N | Previously Treated with Eculizumab | ||
| Abbreviations: LD = Loading Dose; MD = Maintenance Dose | |||||||||
| Cmax
(mcg/mL) |
LD | 4 | 733 (14.5) | 8 | 885 (19.3) | 125 | 771 (21.5) | 95 | 843 (24.1) |
| MD | 4 | 1490 (26.7) | 8 | 1705 (9.7) | 124 | 1,379 (20.0) | 95 | 1,386 (19.4) | |
| Ctrough
(mcg/mL) |
LD | 4 | 368 (14.7) | 8 | 452 (15.1) | 125 | 391 (35.0) | 96 | 405 (29.9) |
| MD | 4 | 495 (21.3) | 8 | 566 (12.2) | 124 | 473 (33.4) | 95 | 501 (28.6) |
| Pediatric Patients (ALXN1210-aHUS-312) |
Adult Patients (ALXN1210-aHUS-311) |
||||||
|---|---|---|---|---|---|---|---|
| N | < 20 kg MD Q4W |
N | ≥ 20 to < 40 kg MD Q8W |
N | ≥ 40 kg MD Q8W |
||
| Abbreviations: LD = Loading Dose; MD = Maintenance Dose; Q4W = Every 4 Weeks; Q8W = Every 8 Weeks | |||||||
| Cmax
(mcg/mL) |
LD | 8 | 656 (38.1) | 4 | 600 (17.3) | 52 | 754 (35.2) |
| MD | 7 | 1,467 (37.8) | 6 | 1,863 (15.3) | 46 | 1,458 (17.6) | |
| Ctrough
(mcg/mL) |
LD | 9 | 241 (52.1) | 5 | 186 (16.5) | 55 | 313 (33.9) |
| MD | 7 | 683 (46.1) | 6 | 549 (34.1) | 46 | 507 (42.5) |
| N | Adult Patients (ALXN1210-MG-306) | ||
|---|---|---|---|
| Abbreviations: LD = Loading Dose; MD=Maintenance Dose | |||
| Cmax
(mcg/mL) |
LD | 86 | 874 (21.1) |
| MD | 76 | 1548 (23.2) | |
| Ctrough
(mcg/mL) |
LD | 85 | 418 (27.6) |
| MD | 70 | 587 (29.6) |
| N | Adult Patients (ALXN1210-NMO-307) | ||
|---|---|---|---|
| Abbreviations: LD = Loading Dose; MD=Maintenance Dose | |||
| Cmax
(mcg/mL) |
LD | 58 | 935.3 (17.3) |
| MD | 56 | 1836.4 (19.4) | |
| Ctrough
(mcg/mL) |
LD | 58 | 459.1 (19.7) |
| MD | 54 | 796.9 (27.1) |
1 Indications and Usage (1 INDICATIONS AND USAGE)
ULTOMIRIS is a complement inhibitor indicated for:
- the treatment of adult and pediatric patients one month of age and older with paroxysmal nocturnal hemoglobinuria (PNH). (1.1)
- the treatment of adult and pediatric patients one month of age and older with atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy (TMA). (1.2)
Limitations of Use:
ULTOMIRIS is not indicated for the treatment of patients with Shiga toxin E. coli related hemolytic uremic syndrome (STEC-HUS). - the treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody-positive. (1.3)
- the treatment of adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are anti-aquaporin-4 (AQP4) antibody-positive. (1.4)
12.1 Mechanism of Action
Ravulizumab-cwvz is a terminal complement inhibitor that specifically binds to the complement protein C5 with high affinity, thereby inhibiting its cleavage to C5a (the proinflammatory anaphylatoxin) and C5b (the initiating subunit of the membrane attack complex [MAC or C5b-9]) thus preventing MAC formation. ULTOMIRIS inhibits terminal complement-mediated intravascular hemolysis in patients with PNH and complement-mediated thrombotic microangiopathy (TMA) in patients with aHUS.
The precise mechanism by which ravulizumab-cwvz exerts its therapeutic effect in gMG patients is unknown, but is presumed to involve reduction of terminal complement complex C5b-9 deposition at the neuromuscular junction.
The precise mechanism by which ravulizumab-cwvz exerts its therapeutic effect in NMOSD is unknown, but is presumed to involve inhibition of aquaporin-4 antibody-induced terminal complement C5b-9 deposition.
16.2 Storage and Handling
Store ULTOMIRIS vials refrigerated at 2°C - 8°C (36°F - 46°F) in the original carton to protect from light. Do not freeze. Do not shake.
Refer to Dosage and Administration (2) for information on the stability and storage of diluted solutions of ULTOMIRIS.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ULTOMIRIS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to ULTOMIRIS exposure.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.2 Ultomiris and Soliris Rems (5.2 ULTOMIRIS and SOLIRIS REMS)
ULTOMIRIS is available only through a restricted program under a REMS called ULTOMIRIS and SOLIRIS REMS, because of the risk of serious meningococcal infections [see Warnings and Precautions (5.1)].
Notable requirements of the ULTOMIRIS and SOLIRIS REMS include the following:
- Prescribers must enroll in the REMS.
- Prescribers must counsel patients about the risk of serious meningococcal infection.
- Prescribers must provide the patients with the REMS educational materials.
- Prescribers must assess patient vaccination status for meningococcal vaccines (against serogroups A, C, W, Y, and B) and vaccinate if needed according to current ACIP recommendations two weeks prior to the first dose of ULTOMIRIS.
- Prescribers must provide a prescription for antibacterial drug prophylaxis if treatment must be started urgently and the patient is not up to date with meningococcal vaccines according to current ACIP recommendations at least two weeks prior to the first dose of ULTOMIRIS.
- Healthcare settings and pharmacies that dispense ULTOMIRIS must be certified in the REMS and must verify prescribers are certified.
- Patients must receive counseling from the prescriber about the need to receive meningococcal vaccines per ACIP recommendations, the need to take antibiotics as directed by the prescriber, and the signs and symptoms of meningococcal infection.
- Patients must be instructed to carry the Patient Safety Card with them at all times during and for 8 months following treatment with ULTOMIRIS.
Further information is available at www.UltSolREMS.com or 1-888-765-4747.
5.6 Infusion Related Reactions (5.6 Infusion-Related Reactions)
Administration of ULTOMIRIS may result in systemic infusion-related reactions, including anaphylaxis [see Adverse Reactions (6.2)] and hypersensitivity reactions. In clinical trials, infusion-related reactions occurred in approximately 1 to 7% of patients treated with ULTOMIRIS [see Adverse Reactions (6.1)]. These events included lower back pain, abdominal pain, muscle spasms, drop in blood pressure, elevation in blood pressure, rigors, limb discomfort, drug hypersensitivity (allergic reaction), and dysgeusia (bad taste). These reactions did not require discontinuation of ULTOMIRIS. If signs of cardiovascular instability or respiratory compromise occur, interrupt ULTOMIRIS infusion and institute appropriate supportive measures.
2.1 Important Dosage Information
ULTOMIRIS is intended to be administered only as an intravenous infusion in adult or pediatric patients one month of age and older.
1.3 Generalized Myasthenia Gravis
ULTOMIRIS is indicated for the treatment of adult patients with generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody-positive.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patients and/or caregivers to read the FDA-approved patient labeling (Medication Guide).
7.2 Neonatal Fc Receptor Blockers
Concomitant use of ULTOMIRIS with neonatal Fc receptor (FcRn) blockers (e.g., efgartigimod) may lower systemic exposures and reduce effectiveness of ULTOMIRIS. Closely monitor for reduced effectiveness of ULTOMIRIS.
5.5 Thromboembolic Event Management
The effect of withdrawal of anticoagulant therapy during ULTOMIRIS treatment has not been established. Therefore, treatment with ULTOMIRIS should not alter anticoagulant management.
5.1 Serious Meningococcal Infections
ULTOMIRIS, a complement inhibitor, increases a patient's susceptibility to serious, life-threatening, or fatal infections caused by meningococcal bacteria (septicemia and/or meningitis) in any serogroup, including non-groupable strains. Life-threatening and fatal meningococcal infections have occurred in both vaccinated and unvaccinated patients treated with complement inhibitors. The initiation of ULTOMIRIS treatment is contraindicated in patients with unresolved serious Neisseria meningitidis infection.
Complete or update meningococcal vaccination (for serogroups A, C, W, Y and B) at least 2 weeks prior to administration of the first dose of ULTOMIRIS, according to current ACIP recommendations for patients receiving a complement inhibitor. Revaccinate patients in accordance with ACIP recommendations considering the duration of ULTOMIRIS therapy. Note that ACIP recommends an administration schedule in patients receiving complement inhibitors that differs from the administration schedule in the vaccine prescribing information. If urgent ULTOMIRIS therapy is indicated in a patient who is not up to date with meningococcal vaccines according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer meningococcal vaccines as soon as possible. Various durations and regimens of antibacterial drug prophylaxis have been considered, but the optimal durations and drug regimens for prophylaxis and their efficacy have not been studied in unvaccinated or vaccinated patients receiving complement inhibitors, including ULTOMIRIS. The benefits and risks of treatment with ULTOMIRIS, as well as the benefits and risks of antibacterial drug prophylaxis in unvaccinated or vaccinated patients, must be considered against the known risks for serious infections caused by Neisseria meningitidis.
Vaccination does not eliminate the risk of meningococcal infections, despite development of antibodies following vaccination.
Closely monitor patients for early signs and symptoms of meningococcal infection and evaluate patients immediately if infection is suspected. Inform patients of these signs and symptoms and instruct patients to seek immediate medical care if these signs and symptoms occur. Promptly treat known infections. Meningococcal infection may become rapidly life-threatening or fatal if not recognized and treated early. Consider interruption of ULTOMIRIS in patients who are undergoing treatment for serious meningococcal infection, depending on the risks of interrupting treatment in the disease being treated.
ULTOMIRIS is available only through a restricted program under a REMS [see Warnings and Precautions (5.2)].
1.2 Atypical Hemolytic Uremic Syndrome
ULTOMIRIS is indicated for the treatment of adult and pediatric patients one month of age and older with atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy (TMA).
1.1 Paroxysmal Nocturnal Hemoglobinuria
ULTOMIRIS is indicated for the treatment of adult and pediatric patients one month of age and older with paroxysmal nocturnal hemoglobinuria (PNH).
14.3 Generalized Myasthenia Gravis (gmg) (14.3 Generalized Myasthenia Gravis (gMG))
The efficacy of ULTOMIRIS for the treatment of gMG was demonstrated in a randomized, double-blind, placebo-controlled, multicenter study (ALXN1210-MG-306; NCT03920293). Patients were randomized 1:1 to either receive ULTOMIRIS (n=86) or placebo (n=89) for 26 weeks. ULTOMIRIS was administered intravenously according to the weight-based recommended dosage [see Dosage and Administration (2.3)].
Patients with gMG with a positive serologic test for anti-AChR antibodies, Myasthenia Gravis Foundation of America (MGFA) clinical classification class II to IV, and Myasthenia Gravis-Activities of Daily Living (MG-ADL) total score ≥ 6 were enrolled. Baseline and disease characteristics were similar between treatment groups (including age at first dose [mean of 58 years for ULTOMIRIS versus 53 years for placebo], gender [51% female for ULTOMIRIS versus 51% female for placebo], race as White, Asian, and Black or African American [78%, 17%, and 2% for ULTOMIRIS versus 69%, 18%, and 5% for placebo, respectively], and duration of MG since diagnosis [mean of 10 years, ranging from 0.5 to 39.5 years, for ULTOMIRIS versus mean of 10.0 years, ranging from 0.5 to 36.1 years, for placebo].
Over 80% of patients were receiving acetylcholinesterase inhibitors, 70% were receiving corticosteroids, and 68% were receiving non-steroidal immunosuppressants (ISTs) at study entry. Patients on concomitant medications to treat gMG were permitted to continue on therapy throughout the course of the study.
The primary efficacy endpoint was a comparison of the change from baseline between treatment groups in the MG-ADL total score at Week 26. The MG-ADL is a categorical scale that assesses the impact on daily function of 8 signs or symptoms that are typically affected in gMG. Each item is assessed on a 4-point scale where a score of 0 represents normal function and a score of 3 represents loss of ability to perform that function. The total score ranges from 0 to 24, with the higher scores indicating more impairment.
The secondary endpoints, also assessed from baseline to Week 26, included the change in the Quantitative MG total score (QMG). The QMG is a 13-item categorical scale assessing muscle weakness. Each item is assessed on a 4-point scale where a score of 0 represents no weakness and a score of 3 represents severe weakness. A total score ranges from 0 to 39, where higher scores indicate more severe impairment.
Other secondary endpoints included the proportion of patients with improvements of at least 5 and 3 points in the QMG and MG-ADL total scores, respectively.
Treatment with ULTOMIRIS demonstrated a statistically significant change in the MG-ADL and QMG total scores from baseline at Week 26 as compared to placebo (Table 29).
| Efficacy Endpoints: Change from Baseline at Week 26 | Placebo (n = 89) LS Mean |
ULTOMIRIS (n = 86) LS Mean |
Treatment Effect (95% CI) |
p-value p-value calculated using mixed effect model for repeated measures
|
|---|---|---|---|---|
| Abbreviations: CI = confidence interval, LS = least squares; MG-ADL = Myasthenia Gravis Activities of Daily Living profile; QMG = Quantitative Myasthenia Gravis score for disease severity | ||||
| Primary Endpoint | ||||
| MG-ADL | -1.4 | -3.1 | -1.6 (-2.6, -0.7) | < 0.001 |
| Secondary Endpoint | ||||
| QMG | -0.8 | -2.8 | -2.0 (-3.2, -0.8) | < 0.001 |
The proportion of QMG responders with at least a 5-point improvement at week 26 was greater for ULTOMIRIS (30.0%) compared to placebo (11.3%) p = 0.005. The proportion of MG-ADL responders with at least a 3-point improvement at week 26 was also greater for ULTOMIRIS (56.7%) compared to placebo (34.1%). The proportion of clinical responders at higher response thresholds (≥ 4-, 5-, 6-, 7-, or 8-point improvement on MG-ADL, and ≥ 6-, 7-, 8-, 9-, or 10-point improvement on QMG) was consistently greater for ULTOMIRIS compared to placebo.
Figure 1: Change from Baseline in MG-ADL Total Score (A) and QMG Total Score (B) Through Week 26 of the Randomized Controlled Period of ALXN1210-MG-306 (Mean and 95% CI)
Note: *p<0.001 versus placebo
Warning: Serious Meningococcal Infections (WARNING: SERIOUS MENINGOCOCCAL INFECTIONS)
ULTOMIRIS, a complement inhibitor, increases the risk of serious infections caused by Neisseria meningitidis [see Warnings and Precautions (5.1)]. Life-threatening and fatal meningococcal infections have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early.
- Complete or update vaccination for meningococcal bacteria (for serogroups A, C, W, Y, and B) at least 2 weeks prior to the first dose of ULTOMIRIS, unless the risks of delaying therapy with ULTOMIRIS outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccinations against meningococcal bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by meningococcal bacteria.
- Patients receiving ULTOMIRIS are at increased risk for invasive disease caused by Neisseria meningitidis, even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious meningococcal infections and evaluate immediately if infection is suspected.
Because of the risk of serious meningococcal infections, ULTOMIRIS is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called ULTOMIRIS and SOLIRIS REMS [see Warnings and Precautions (5.2)].
1.4 Neuromyelitis Optica Spectrum Disorder
ULTOMIRIS is indicated for the treatment of adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are anti-aquaporin-4 (AQP4) antibody-positive.
Principal Display Panel 3 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 3 mL Vial Carton)
Rx only
NDC 25682-025-01
ULTOMIRIS®
(ravulizumab-cwvz)
Injection
300 mg/3 mL
(100 mg/mL)
For Intravenous Infusion
Dilute with 0.9% Sodium Chloride
Injection prior to use.
3 mL Single-dose vial
Discard Unused Portion
ATTENTION: Dispense the
enclosed Medication Guide to
each patient
Principal Display Panel 11 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 11 mL Vial Carton)
Rx only
NDC 25682-028-01
ULTOMIRIS®
(ravulizumab-cwvz)
Injection
1,100 mg/11mL
(100 mg/mL)
For Intravenous Infusion
Dilute with 0.9% Sodium Chloride
Injection prior to use.
11 mL Single-dose vial
Discard Unused Portion
ATTENTION: Dispense the
enclosed Medication Guide to
each patient.
Principal Display Panel 30 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 30 mL Vial Carton)
NDC 25682-022-01
ULTOMIRIS®
(ravulizumab-cwvz)
injection
300 mg/30 mL
(10 mg/mL)
For Intravenous Infusion
Dilute with 0.9% Sodium Chloride
Injection prior to use.
Rx only
30 mL Single-dose vial
Discard Unused Portion
ATTENTION: Dispense the enclosed
Medication Guide to each patient
14.1 Paroxysmal Nocturnal Hemoglobinuria (pnh) (14.1 Paroxysmal Nocturnal Hemoglobinuria (PNH))
The safety and efficacy of ULTOMIRIS in adult patients with PNH was assessed in 2 open-label, randomized, active-controlled, non-inferiority Phase 3 studies: PNH Study 301 and PNH Study 302. Study 301 enrolled patients with PNH who were complement inhibitor-naïve and had active hemolysis. Study 302 enrolled patients with PNH who were clinically stable after having been treated with eculizumab for at least the past 6 months. The safety and efficacy of ULTOMIRIS in pediatric patients with PNH was assessed in PNH Study 304, an open-label, Phase 3 study conducted in eculizumab-experienced and complement inhibitor treatment-naïve pediatric patients with PNH.
In Study 301 and Study 302, adults with PNH were dosed with ULTOMIRIS administered intravenously in accordance with the weight-based dosing described in Section 2.3 (4 infusions of ULTOMIRIS over 26 weeks) above. Eculizumab was administered on Days 1, 8, 15, and 22, followed by maintenance treatment with 900 mg of eculizumab on Day 29 and every 2 weeks (q2w) thereafter for a total of 26 weeks of treatment, according to the approved dosing regimen of eculizumab which was the standard-of-care for PNH at the time of the studies.
Patients were vaccinated against meningococcal infection prior to or at the time of initiating treatment with ULTOMIRIS or eculizumab, or received prophylactic treatment with appropriate antibiotics until 2 weeks after vaccination. Prophylactic treatment with appropriate antibiotics beyond 2 weeks after vaccination was at the discretion of the provider.
14.2 Atypical Hemolytic Uremic Syndrome (ahus) (14.2 Atypical Hemolytic Uremic Syndrome (aHUS))
The efficacy of ULTOMIRIS administered intravenously in patients with aHUS was assessed in 2 open-label, single-arm studies. Study ALXN1210-aHUS-311 enrolled adult patients who displayed signs of TMA. In order to qualify for enrollment, patients were required to have a platelet count ≤ 150 × 109/L, evidence of hemolysis such as an elevation in serum LDH, and serum creatinine above the upper limits of normal or required dialysis.
Study ALXN1210-aHUS-312 enrolled pediatric patients who displayed signs of TMA. In order to qualify for enrollment, patients were required to have a platelet count ≤ 150 × 109/L, evidence of hemolysis such as an elevation in serum LDH, and serum creatinine level ≥ 97.5% percentile at screening or required dialysis. In both studies, enrollment criteria excluded patients presenting with TMA due to a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) deficiency, Shiga toxin Escherichia coli related hemolytic uremic syndrome (STEC-HUS) and genetic defect in cobalamin C metabolism. Patients with confirmed diagnosis of STEC-HUS after enrollment were excluded from the efficacy evaluation.
14.4 Neuromyelitis Optica Spectrum Disorder (nmosd) (14.4 Neuromyelitis Optica Spectrum Disorder (NMOSD))
The efficacy and safety of ULTOMIRIS in adult patients with anti-AQP4 antibody positive NMOSD was assessed in an open-label multicenter study, Study ALXN1210-NMO-307 (NCT04291262). Patients participating in Study ALXN1210-NMO-307 received ULTOMIRIS intravenously in the Primary Treatment Period that ended when the last enrolled patient completed (or discontinued prior to) 50 weeks on study, representing a median study duration of 73.5 weeks (minimum 13.7, maximum 117.7). Efficacy assessments were based on a comparison of patients in Study ALXN1210-NMO-307 with an external placebo control group from another study (Study ECU-NMO-301, NCT01892345) composed of a comparable population of adult patients with anti-AQP4 antibody positive NMOSD.
Study ALXN1210-NMO-307 enrolled 58 adult patients with NMOSD who had a positive serologic test for anti-AQP4 antibodies, at least 1 relapse in the last 12 months prior to the Screening Period, and an Expanded Disability Status Scale (EDSS) score ≤ 7. In the external placebo control group, eligibility criteria were similar except patients were required to have at least 2 relapses in last 12 months or 3 relapses in the last 24 months with at least 1 relapse in the 12 months prior to screening. Prior treatment with immunosuppressant therapies (ISTs) was not required for enrollment. However, patients on selected ISTs (i.e., corticosteroids, azathioprine, mycophenolate mofetil, methotrexate, and tacrolimus) were permitted to continue on therapy, with a requirement for stable dosing until they reached Week 106 in the Study. Similar IST use was permitted in the external placebo control group.
ULTOMIRIS was administered intravenously according to the weight-based recommended dosage [see Dosage and Administration (2.3)].
The demographics were similar between the ULTOMIRIS treatment group from Study ALXN1210-NMO-307 and the placebo treatment group from Study ECU-NMO-301 (including age [median of 46.0 years for ULTOMIRIS versus 44.0 years for placebo] and sex [89.7% female for ULTOMIRIS versus 89.4% female for placebo]). The majority of patients were White or Asian. The median time from diagnosis to first dose was 0.9 years for ULTOMIRIS and 2.0 years for placebo. The median annualized relapse rate (ARR) in the last 24 months was 1.4 for ULTOMIRIS versus 1.9 for placebo, and the median number of historical relapses was 2 for ULTOMIRIS versus 4 for placebo. The median baseline EDSS score was 3.3 for ULTOMIRIS versus 4.0 for placebo. At baseline, 48% of patients in the ULTOMIRIS group received concomitant IST, including corticosteroids, versus 72% of subjects in the placebo group.
The primary endpoint of Study ALXN1210-NMO-307 was the time to first adjudicated on-trial relapse as determined by an independent adjudication committee. No adjudicated on-trial relapses were observed in ULTOMIRIS-treated patients during the Primary Treatment Period, representing a statistically significant difference between the ULTOMIRIS and placebo treatment arms in time to first adjudicated on-trial relapse (p < 0.0001). The hazard ratio (95% confidence interval [CI]) for ULTOMIRIS compared with placebo was 0.014 (0.000, 0.103), representing a 98.6% reduction in the risk of relapse (Figure 2). ULTOMIRIS-treated patients experienced similar improvement in time to first adjudicated on-trial relapse with or without concomitant treatment.
Figure 2: Kaplan-Meier Survival Estimates for Time to First Adjudicated On-Trial Relapse in Study ALXN1210-NMO-307 and Comparative Placebo Arm of Study ECU-NMO-301
Note: The placebo group data were collected as part of Study ECU-NMO-301. Patients who did not experience an adjudicated on-trial relapse were censored at the end of the study period. If a patient in the placebo group was followed longer than any of the patients in the Ultomiris group, then that patient was censored at the longest Ultomiris follow-up time.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal carcinogenicity studies of ravulizumab-cwvz have not been conducted.
Genotoxicity studies have not been conducted with ravulizumab-cwvz.
Effects of ravulizumab-cwvz upon fertility have not been studied in animals. Intravenous injections of male and female mice with a murine anti-C5 antibody at up to 0.8-2.2 times the equivalent of the clinical dose of ULTOMIRIS had no adverse effects on mating or fertility.
7.1 Plasma Exchange, Plasmapheresis, and Intravenous Immunoglobulins
Concomitant use of ULTOMIRIS with plasma exchange (PE), plasmapheresis (PP), or intravenous immunoglobulin (IVIg) treatment can reduce serum ravulizumab concentrations and requires a supplemental dose of ULTOMIRIS [see Dosage and Administration (2.5)].
2.2 Recommended Vaccination and Prophylaxis for Meningococcal Infection
Vaccinate patients against meningococcal infection (serogroups A, C, W, Y and B) according to current ACIP recommendations at least 2 weeks prior to initiation of ULTOMIRIS [see Warnings and Precautions (5.1)].
If urgent ULTOMIRIS therapy is indicated in a patient who is not up to date with meningococcal vaccines according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer these vaccines as soon as possible.
Healthcare providers who prescribe ULTOMIRIS must enroll in the ULTOMIRIS and SOLIRIS REMS [see Warnings and Precautions (5.2)].
2.3 Recommended Dosage for Intravenous Administration in Adult and Pediatric Patients With Pnh Or Ahus, and in Adult Patients With Gmg Or Nmosd (2.3 Recommended Dosage for Intravenous Administration in Adult and Pediatric Patients with PNH or aHUS, and in Adult Patients with gMG or NMOSD)
The recommended intravenous (IV) ULTOMIRIS loading and maintenance dosing in adult and pediatric patients, one month of age or older weighing 5 kg or greater, with PNH or aHUS, or in adult patients with gMG or NMOSD weighing 40 kg or greater, is based on the patient's body weight, as shown in Table 1, with maintenance doses administered every 4 or 8 weeks, starting 2 weeks after loading dose.
The IV dosing schedule is allowed to occasionally vary within 7 days of the scheduled infusion day (except for the first maintenance dose of ULTOMIRIS); but subsequent doses should be administered according to the original schedule.
Following a missed IV ULTOMIRIS dose, the patient should contact their health care provider immediately.
| Indications | Body Weight Range (kg) |
Loading Dose (mg) See Table 2 for ULTOMIRIS treatment initiation instruction and timing of loading dose and maintenance dose
|
Maintenance Dose (mg) and Dosing Interval | |
|---|---|---|---|---|
| PNH or aHUS | 5 to less than 10 | 600 | 300 | Every 4 weeks |
| 10 to less than 20 | 600 | 600 | ||
| 20 to less than 30 | 900 | 2,100 | Every 8 weeks |
|
| 30 to less than 40 | 1,200 | 2,700 | ||
| PNH, aHUS, gMG, or NMOSD | 40 to less than 60 | 2,400 | 3,000 | Every 8 weeks |
| 60 to less than 100 | 2,700 | 3,300 | ||
| 100 or greater | 3,000 | 3,600 |
Refer to Table 2 for treatment initiation instructions in patients who are complement inhibitor treatment-naïve or switching treatment from eculizumab.
| Population | Weight-based ULTOMIRIS Loading Dose | Time of First ULTOMIRIS Weight-based Maintenance Dose |
|---|---|---|
| Not currently on ULTOMIRIS or eculizumab treatment | At treatment start | 2 weeks after ULTOMIRIS loading dose |
| Currently treated with eculizumab | At time of next scheduled eculizumab dose | 2 weeks after ULTOMIRIS loading dose |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:02.193169 · Updated: 2026-03-14T22:46:59.478961