These Highlights Do Not Include All The Information Needed To Use Ondansetron Oral Solution Safely And Effectively. See Full Prescribing Information For Ondansetron Oral Solution
a95ca4f4-c4de-4822-9a08-209d10bf5136
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ondansetron is indicated for the prevention of nausea and vomiting associated with: highly emetogenic cancer chemotherapy, including cisplatin greater than or equal to 50 mg/m 2 . initial and repeat courses of moderately emetogenic cancer chemotherapy. radiotherapy in patients receiving either total body irradiation, single high-dose fraction to the abdomen, or daily fractions to the abdomen. Ondansetron is also indicated for the prevention of postoperative nausea and/or vomiting.
Indications and Usage
Ondansetron is indicated for the prevention of nausea and vomiting associated with: highly emetogenic cancer chemotherapy, including cisplatin greater than or equal to 50 mg/m 2 . initial and repeat courses of moderately emetogenic cancer chemotherapy. radiotherapy in patients receiving either total body irradiation, single high-dose fraction to the abdomen, or daily fractions to the abdomen. Ondansetron is also indicated for the prevention of postoperative nausea and/or vomiting.
Dosage and Administration
See full prescribing information for the recommended dosage in adults and pediatrics. ( 2 ) Patients with severe hepatic impairment: do not exceed a total daily dose of 8 mg. ( 2.2 , 8.6 )
Warnings and Precautions
Hypersensitivity reactions including anaphylaxis and bronchospasm: Discontinue ondansetron if suspected. Monitor and treat promptly per standard of care until signs and symptoms resolve. ( 5.1 ) QT interval prolongation and Torsade de Pointes : Avoid in patients with congenital long QT syndrome; monitor with electrocardiograms (ECGs) if concomitant electrolyte abnormalities, cardiac failure or arrhythmias, or use of other QT prolonging drugs. ( 5.2 ) Serotonin syndrome: Reported with 5-HT 3 receptor antagonists alone but particularly with concomitant use of serotonergic drugs. If such symptoms occur, discontinue ondansetron and initiate supportive treatment. If concomitant use of ondansetron with other serotonergic drugs is clinically warranted, patients should be made aware of a potential increased risk for serotonin syndrome. ( 5.3 ) Masking of progressive ileus and/or gastric distention following abdominal surgery or chemotherapy-induced nausea and vomiting : Monitor for decreased bowel activity, particularly in patients with risk factors for gastrointestinal obstruction. ( 5.4 )
Contraindications
Ondansetron is contraindicated in patients: known to have hypersensitivity (e.g., anaphylaxis) to ondansetron or any of the components of the formulation [see Adverse Reactions (6.2) ] . receiving concomitant apomorphine due to the risk of profound hypotension and loss of consciousness.
Adverse Reactions
The most common adverse reactions in adults for the: prevention of chemotherapy-induced (greater than or equal to 5%) are: headache, malaise/fatigue, constipation, diarrhea. ( 6.1 ) prevention of radiation-induced nausea and vomiting (greater than or equal to 2%) are: headache, constipation, and diarrhea. ( 6.1 ) prevention of postoperative nausea and vomiting (greater than or equal to 9%) are: headache and hypoxia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
Ondansetron Oral Solution USP, 4 mg/5mL, a clear, colorless to light yellow liquid with a characteristic strawberry odor, contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron per 5 mL in bottles of 50 mL with child-resistant closures (NDC 51672-4091-3).
How Supplied
Ondansetron Oral Solution USP, 4 mg/5mL, a clear, colorless to light yellow liquid with a characteristic strawberry odor, contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron per 5 mL in bottles of 50 mL with child-resistant closures (NDC 51672-4091-3).
Medication Information
Warnings and Precautions
Hypersensitivity reactions including anaphylaxis and bronchospasm: Discontinue ondansetron if suspected. Monitor and treat promptly per standard of care until signs and symptoms resolve. ( 5.1 ) QT interval prolongation and Torsade de Pointes : Avoid in patients with congenital long QT syndrome; monitor with electrocardiograms (ECGs) if concomitant electrolyte abnormalities, cardiac failure or arrhythmias, or use of other QT prolonging drugs. ( 5.2 ) Serotonin syndrome: Reported with 5-HT 3 receptor antagonists alone but particularly with concomitant use of serotonergic drugs. If such symptoms occur, discontinue ondansetron and initiate supportive treatment. If concomitant use of ondansetron with other serotonergic drugs is clinically warranted, patients should be made aware of a potential increased risk for serotonin syndrome. ( 5.3 ) Masking of progressive ileus and/or gastric distention following abdominal surgery or chemotherapy-induced nausea and vomiting : Monitor for decreased bowel activity, particularly in patients with risk factors for gastrointestinal obstruction. ( 5.4 )
Indications and Usage
Ondansetron is indicated for the prevention of nausea and vomiting associated with: highly emetogenic cancer chemotherapy, including cisplatin greater than or equal to 50 mg/m 2 . initial and repeat courses of moderately emetogenic cancer chemotherapy. radiotherapy in patients receiving either total body irradiation, single high-dose fraction to the abdomen, or daily fractions to the abdomen. Ondansetron is also indicated for the prevention of postoperative nausea and/or vomiting.
Dosage and Administration
See full prescribing information for the recommended dosage in adults and pediatrics. ( 2 ) Patients with severe hepatic impairment: do not exceed a total daily dose of 8 mg. ( 2.2 , 8.6 )
Contraindications
Ondansetron is contraindicated in patients: known to have hypersensitivity (e.g., anaphylaxis) to ondansetron or any of the components of the formulation [see Adverse Reactions (6.2) ] . receiving concomitant apomorphine due to the risk of profound hypotension and loss of consciousness.
Adverse Reactions
The most common adverse reactions in adults for the: prevention of chemotherapy-induced (greater than or equal to 5%) are: headache, malaise/fatigue, constipation, diarrhea. ( 6.1 ) prevention of radiation-induced nausea and vomiting (greater than or equal to 2%) are: headache, constipation, and diarrhea. ( 6.1 ) prevention of postoperative nausea and vomiting (greater than or equal to 9%) are: headache and hypoxia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
Ondansetron Oral Solution USP, 4 mg/5mL, a clear, colorless to light yellow liquid with a characteristic strawberry odor, contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron per 5 mL in bottles of 50 mL with child-resistant closures (NDC 51672-4091-3).
How Supplied
Ondansetron Oral Solution USP, 4 mg/5mL, a clear, colorless to light yellow liquid with a characteristic strawberry odor, contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron per 5 mL in bottles of 50 mL with child-resistant closures (NDC 51672-4091-3).
Description
Ondansetron is indicated for the prevention of nausea and vomiting associated with: highly emetogenic cancer chemotherapy, including cisplatin greater than or equal to 50 mg/m 2 . initial and repeat courses of moderately emetogenic cancer chemotherapy. radiotherapy in patients receiving either total body irradiation, single high-dose fraction to the abdomen, or daily fractions to the abdomen. Ondansetron is also indicated for the prevention of postoperative nausea and/or vomiting.
Section 42229-5
Prevention of Chemotherapy-induced Nausea and Vomiting
The most common adverse reactions reported in greater than or equal to 4% of 300 adults receiving a single 24-mg dose of ondansetron orally in 2 trials for the prevention of nausea and vomiting associated with highly emetogenic chemotherapy (cisplatin greater than or equal to 50 mg/m2) were: headache (11%) and diarrhea (4%).
The most common adverse reactions reported in 4 trials in adults for the prevention of nausea and vomiting associated with moderately emetogenic chemotherapy (primarily cyclophosphamide-based regimens) are shown in Table 3.
| Adverse Reaction | Ondansetron 8 mg Twice Daily (n = 242) |
Placebo (n = 262) |
|---|---|---|
| Headache | 58 (24%) | 34 (13%) |
| Malaise/fatigue | 32 (13%) | 6 (2%) |
| Constipation | 22 (9%) | 1 (<1%) |
| Diarrhea | 15 (6%) | 10 (4%) |
Section 44425-7
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from light.
Store bottles upright in cartons.
2.1 Dosage
The recommended dosage regimens for adult and pediatric patients are described in Table 1 and Table 2, respectively.
Corresponding doses of ondansetron tablets and ondansetron oral solution may be used interchangeably.
| Indication | Dosage Regimen |
|---|---|
| Highly Emetogenic Cancer Chemotherapy | A single 24-mg dose administered 30 minutes before the start of single-day highly emetogenic chemotherapy, including cisplatin greater than or equal to 50 mg/m2 |
| Moderately Emetogenic Cancer Chemotherapy | 8 mg administered 30 minutes before the start of chemotherapy, with a subsequent 8-mg dose 8 hours after the first dose. Then administer 8 mg twice a day (every 12 hours) for 1 to 2 days after completion of chemotherapy. |
| Radiotherapy |
For total body irradiation: 8 mg administered 1 to 2 hours before each fraction of radiotherapy each day. For single high-dose fraction radiotherapy to the abdomen: 8 mg administered 1 to 2 hours before radiotherapy, with subsequent 8-mg doses every 8 hours after the first dose for 1 to 2 days after completion of radiotherapy. For daily fractionated radiotherapy to the abdomen: 8 mg administered 1 to 2 hours before radiotherapy, with subsequent 8-mg doses every 8 hours after the first dose for each day radiotherapy is given. |
| Postoperative | 16 mg administered 1 hour before induction of anesthesia. |
| Indication | Dosage Regimen |
|---|---|
| Moderately Emetogenic Cancer Chemotherapy |
12 to 17 years of age: 8 mg administered 30 minutes before the start of chemotherapy, with a subsequent 8-mg dose 8 hours after the first dose. Then administer 8 mg twice a day (every 12 hours) for 1 to 2 days after completion of chemotherapy. 4 to 11 years of age: 4 mg administered 30 minutes before the start of chemotherapy, with a subsequent 4-mg dose 4 and 8 hours after the first dose. Then administer 4 mg three times a day for 1 to 2 days after completion of chemotherapy. |
7.3 Tramadol
Although no pharmacokinetic drug interaction between ondansetron and tramadol has been observed, data from 2 small trials indicate that when used together, ondansetron may increase patient-controlled administration of tramadol. Monitor patients to ensure adequate pain control when ondansetron is administered with tramadol.
10 Overdosage
There is no specific antidote for ondansetron overdose. Patients should be managed with appropriate supportive therapy.
In addition to the adverse reactions listed above, the following adverse reactions have been described in the setting of ondansetron overdose: "Sudden blindness" (amaurosis) of 2 to 3 minutes' duration plus severe constipation occurred in one patient that was administered 72 mg of ondansetron intravenously as a single dose. Hypotension (and faintness) occurred in a patient that took 48 mg of ondansetron tablets. Following infusion of 32 mg over only a 4-minute period, a vasovagal episode with transient second-degree heart block was observed. In all instances, the adverse reactions resolved completely.
Pediatric cases consistent with serotonin syndrome have been reported after inadvertent oral overdoses of ondansetron (exceeding estimated ingestion of 5 mg per kg) in young children. Reported symptoms included somnolence, agitation, tachycardia, tachypnea, hypertension, flushing, mydriasis, diaphoresis, myoclonic movements, horizontal nystagmus, hyperreflexia, and seizure. Patients required supportive care, including intubation in some cases, with complete recovery without sequelae within 1 to 2 days.
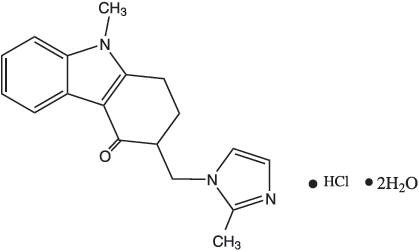
11 Description
The active ingredient in ondansetron oral solution, USP is ondansetron hydrochloride as the dihydrate, the racemic form of ondansetron and a selective blocking agent of the serotonin 5-HT3 receptor type. Chemically it is (±) 1, 2, 3, 9-tetrahydro-9-methyl-3-[(2-methyl-1H-imidazol-1-yl)methyl]-4H-carbazol-4-one, monohydrochloride, dihydrate. It has the following structural formula:
The empirical formula is C18H19N3O∙HCl∙2H2O, representing a molecular weight of 365.9.
Ondansetron hydrochloride dihydrate is a white to off-white powder that is soluble in water and normal saline. Each 5 mL of ondansetron oral solution contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron. Ondansetron oral solution contains the inactive ingredients citric acid anhydrous, purified water, sodium benzoate, sodium citrate dihydrate, strawberry flavor, and sucralose.
7.4 Chemotherapy
Carmustine, etoposide, and cisplatin do not affect the pharmacokinetics of ondansetron.
In a crossover trial in 76 pediatric patients, intravenous ondansetron did not increase systemic concentrations of high-dose methotrexate.
8.4 Pediatric Use
The safety and effectiveness of orally administered ondansetron have been established in pediatric patients 4 years and older for the prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy. Use of ondansetron in these age-groups is supported by evidence from adequate and well-controlled studies of ondansetron in adults with additional data from 3 open-label, uncontrolled, non-US trials in 182 pediatric patients aged 4 to 18 years with cancer who were given a variety of cisplatin or noncisplatin regimens [see Dosage and Administration (2.2), Clinical Studies (14.1)].
Additional information on the use of ondansetron in pediatric patients may be found in ondansetron injection prescribing information.
The safety and effectiveness of orally administered ondansetron have not been established in pediatric patients for:
- prevention of nausea and vomiting associated with highly emetogenic cancer chemotherapy.
- prevention of nausea and vomiting associated with radiotherapy.
- prevention of postoperative nausea and/or vomiting.
8.5 Geriatric Use
Of the total number of subjects enrolled in cancer chemotherapy-induced and postoperative nausea and vomiting in U.S.- and foreign-controlled clinical trials, for which there were subgroup analyses, 938 (19%) were aged 65 years and older.
No overall differences in safety or effectiveness were observed between subjects 65 years of age and older and younger subjects. A reduction in clearance and increase in elimination half-life were seen in patients older than 75 years compared with younger subjects [see Clinical Pharmacology (12.3)]. There were an insufficient number of patients older than 75 years of age and older in the clinical trials to permit safety or efficacy conclusions in this age-group. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. No dosage adjustment is needed in elderly patients.
4 Contraindications
Ondansetron is contraindicated in patients:
- known to have hypersensitivity (e.g., anaphylaxis) to ondansetron or any of the components of the formulation [see Adverse Reactions (6.2)].
- receiving concomitant apomorphine due to the risk of profound hypotension and loss of consciousness.
5.2 Qt Prolongation
Electrocardiogram (ECG) changes including QT interval prolongation have been seen in patients receiving ondansetron. In addition, postmarketing cases of Torsade de Pointes have been reported in patients using ondansetron. Avoid ondansetron in patients with congenital long QT syndrome. ECG monitoring is recommended in patients with electrolyte abnormalities (e.g., hypokalemia or hypomagnesemia), congestive heart failure, bradyarrhythmias, or patients taking other medicinal products that lead to QT prolongation [see Clinical Pharmacology (12.2)].
6 Adverse Reactions
The most common adverse reactions in adults for the:
- prevention of chemotherapy-induced (greater than or equal to 5%) are: headache, malaise/fatigue, constipation, diarrhea. (6.1)
- prevention of radiation-induced nausea and vomiting (greater than or equal to 2%) are: headache, constipation, and diarrhea. (6.1)
- prevention of postoperative nausea and vomiting (greater than or equal to 9%) are: headache and hypoxia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
8.7 Renal Impairment
No dosage adjustment is recommended for patients with any degree of renal impairment (mild, moderate, or severe). There is no experience beyond first-day administration of ondansetron [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In healthy subjects, single intravenous doses of 0.15 mg/kg of ondansetron had no effect on esophageal motility, gastric motility, lower esophageal sphincter pressure, or small intestinal transit time. Multiday administration of ondansetron has been shown to slow colonic transit in healthy subjects. Ondansetron has no effect on plasma prolactin concentrations.
5.3 Serotonin Syndrome
The development of serotonin syndrome has been reported with 5-HT3 receptor antagonists alone. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors, mirtazapine, fentanyl, lithium, tramadol, and intravenous methylene blue). Some of the reported cases were fatal. Serotonin syndrome occurring with overdose of ondansetron alone has also been reported. The majority of reports of serotonin syndrome related to 5-HT3 receptor antagonist use occurred in a post-anesthesia care unit or an infusion center.
Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, with or without gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome, especially with concomitant use of ondansetron and other serotonergic drugs. If symptoms of serotonin syndrome occur, discontinue ondansetron and initiate supportive treatment. Patients should be informed of the increased risk of serotonin syndrome, especially if ondansetron is used concomitantly with other serotonergic drugs [see Drug Interactions (7.1), Overdosage (10)].
7.1 Serotonergic Drugs
Serotonin syndrome (including altered mental status, autonomic instability, and neuromuscular symptoms) has been described following the concomitant use of 5-HT3 receptor antagonists and other serotonergic drugs, including selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs). Monitor for the emergence of serotonin syndrome. If symptoms occur, discontinue ondansetron and initiate supportive treatment [see Warnings and Precautions (5.3)].
8.6 Hepatic Impairment
No dosage adjustment is needed in patients with mild or moderate hepatic impairment.
In patients with severe hepatic impairment, clearance is reduced and the apparent volume of distribution is increased, resulting in a significant increase in the half-life of ondansetron. Therefore, do not exceed a total daily dose of 8 mg in patients with severe hepatic impairment (Child-Pugh score of 10 or greater) [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
1 Indications and Usage
Ondansetron is indicated for the prevention of nausea and vomiting associated with:
- highly emetogenic cancer chemotherapy, including cisplatin greater than or equal to 50 mg/m2.
- initial and repeat courses of moderately emetogenic cancer chemotherapy.
- radiotherapy in patients receiving either total body irradiation, single high-dose fraction to the abdomen, or daily fractions to the abdomen.
Ondansetron is also indicated for the prevention of postoperative nausea and/or vomiting.
12.1 Mechanism of Action
Ondansetron is a selective 5-HT3 receptor antagonist. While its mechanism of action has not been fully characterized, ondansetron is not a dopamine-receptor antagonist. Serotonin receptors of the 5-HT3 type are present both peripherally on vagal nerve terminals and centrally in the chemoreceptor trigger zone of the area postrema. It is not certain whether ondansetron's antiemetic action is mediated centrally, peripherally, or in both sites. However, cytotoxic chemotherapy appears to be associated with release of serotonin from the enterochromaffin cells of the small intestine. In humans, urinary 5-hydroxyindoleacetic acid (5-HIAA) excretion increases after cisplatin administration in parallel with the onset of emesis. The released serotonin may stimulate the vagal afferents through the 5-HT3 receptors and initiate the vomiting reflex.
5 Warnings and Precautions
- Hypersensitivity reactions including anaphylaxis and bronchospasm: Discontinue ondansetron if suspected. Monitor and treat promptly per standard of care until signs and symptoms resolve. (5.1)
- QT interval prolongation and Torsade de Pointes: Avoid in patients with congenital long QT syndrome; monitor with electrocardiograms (ECGs) if concomitant electrolyte abnormalities, cardiac failure or arrhythmias, or use of other QT prolonging drugs. (5.2)
- Serotonin syndrome: Reported with 5-HT3 receptor antagonists alone but particularly with concomitant use of serotonergic drugs. If such symptoms occur, discontinue ondansetron and initiate supportive treatment. If concomitant use of ondansetron with other serotonergic drugs is clinically warranted, patients should be made aware of a potential increased risk for serotonin syndrome. (5.3)
- Masking of progressive ileus and/or gastric distention following abdominal surgery or chemotherapy-induced nausea and vomiting: Monitor for decreased bowel activity, particularly in patients with risk factors for gastrointestinal obstruction. (5.4)
2 Dosage and Administration
9 Drug Abuse and Dependence
Animal studies have shown that ondansetron is not discriminated as a benzodiazepine nor does it substitute for benzodiazepines in direct addiction studies.
3 Dosage Forms and Strengths
Ondansetron oral solution USP, 4 mg/5 mL, is a clear, colorless to light yellow liquid with a characteristic strawberry odor available in a 50-mL bottle.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ondansetron. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
7.5 Alfentanil and Atracurium
Ondansetron does not alter the respiratory depressant effects produced by alfentanil or the degree of neuromuscular blockade produced by atracurium. Interactions with general or local anesthetics have not been studied.
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis and bronchospasm, have been reported in patients who have exhibited hypersensitivity to other selective 5-HT3 receptor antagonists. If hypersensitivity reactions occur, discontinue use of ondansetron; treat promptly per standard of care and monitor until signs and symptoms resolve [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following adverse reactions have been reported in clinical trials of patients treated with ondansetron, the active ingredient of ondansetron oral solution. A causal relationship to therapy with ondansetron was unclear in many cases.
2.2 Dosage in Hepatic Impairment
In patients with severe hepatic impairment (Child-Pugh score of 10 or greater), do not exceed a total daily dose of 8 mg [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling
Ondansetron Oral Solution USP, 4 mg/5mL, a clear, colorless to light yellow liquid with a characteristic strawberry odor, contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron per 5 mL in bottles of 50 mL with child-resistant closures (NDC 51672-4091-3).
14.3 Postoperative Nausea and Vomiting
In two placebo-controlled, double-blind trials (one conducted in the US and the other outside the US) in 865 females undergoing inpatient surgical procedures, ondansetron 16 mg as a single dose or placebo was administered one hour before the induction of general balanced anesthesia (barbiturate, opioid, nitrous oxide, neuromuscular blockade, and supplemental isoflurane or enflurane), ondansetron tablets was significantly more effective than placebo in preventing postoperative nausea and vomiting.
No trials have been performed in males.
7.2 Drugs Affecting Cytochrome P 450 Enzymes
Ondansetron does not itself appear to induce or inhibit the cytochrome P-450 drug-metabolizing enzyme system of the liver [see Clinical Pharmacology (12.3)]. Because ondansetron is metabolized by hepatic cytochrome P-450 drug-metabolizing enzymes (CYP3A4, CYP2D6, CYP1A2), inducers or inhibitors of these enzymes may change the clearance and, hence, the half-life of ondansetron. In patients treated with potent inducers of CYP3A4 (i.e., phenytoin, carbamazepine, and rifampin), the clearance of ondansetron was significantly increased and ondansetron blood concentrations were decreased. However, on the basis of available data, no dosage adjustment for ondansetron is recommended for patients on these drugs [see Clinical Pharmacology (12.3)].
Principal Display Panel 50 Ml Bottle Carton
50 mL
NDC 51672-4091-3
Ondansetron
Oral Solution
USP, 4 mg/5 mL
Rx only
TARO
5.4 Masking of Progressive Ileus and Gastric Distension
The use of ondansetron in patients following abdominal surgery or in patients with chemotherapy-induced nausea and vomiting may mask a progressive ileus and/or gastric distension. Monitor for decreased bowel activity, particularly in patients with risk factors for gastrointestinal obstruction.
Ondansetron is not a drug that stimulates gastric or intestinal peristalsis. It should not be used instead of nasogastric suction.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenic effects were not seen in 2-year studies in rats and mice with oral ondansetron doses up to 10 mg/kg per day and 30 mg/kg per day, respectively (approximately 4 and 6 times the maximum recommended human oral dose of 24 mg per day, based on body surface area).
Ondansetron was not mutagenic in standard tests for mutagenicity.
Oral administration of ondansetron up to 15 mg/kg per day (approximately 6 times the maximum recommended human oral dose of 24 mg per day, based on body surface area) did not affect fertility or general reproductive performance of male and female rats.
Structured Label Content
Section 42229-5 (42229-5)
Prevention of Chemotherapy-induced Nausea and Vomiting
The most common adverse reactions reported in greater than or equal to 4% of 300 adults receiving a single 24-mg dose of ondansetron orally in 2 trials for the prevention of nausea and vomiting associated with highly emetogenic chemotherapy (cisplatin greater than or equal to 50 mg/m2) were: headache (11%) and diarrhea (4%).
The most common adverse reactions reported in 4 trials in adults for the prevention of nausea and vomiting associated with moderately emetogenic chemotherapy (primarily cyclophosphamide-based regimens) are shown in Table 3.
| Adverse Reaction | Ondansetron 8 mg Twice Daily (n = 242) |
Placebo (n = 262) |
|---|---|---|
| Headache | 58 (24%) | 34 (13%) |
| Malaise/fatigue | 32 (13%) | 6 (2%) |
| Constipation | 22 (9%) | 1 (<1%) |
| Diarrhea | 15 (6%) | 10 (4%) |
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from light.
Store bottles upright in cartons.
2.1 Dosage
The recommended dosage regimens for adult and pediatric patients are described in Table 1 and Table 2, respectively.
Corresponding doses of ondansetron tablets and ondansetron oral solution may be used interchangeably.
| Indication | Dosage Regimen |
|---|---|
| Highly Emetogenic Cancer Chemotherapy | A single 24-mg dose administered 30 minutes before the start of single-day highly emetogenic chemotherapy, including cisplatin greater than or equal to 50 mg/m2 |
| Moderately Emetogenic Cancer Chemotherapy | 8 mg administered 30 minutes before the start of chemotherapy, with a subsequent 8-mg dose 8 hours after the first dose. Then administer 8 mg twice a day (every 12 hours) for 1 to 2 days after completion of chemotherapy. |
| Radiotherapy |
For total body irradiation: 8 mg administered 1 to 2 hours before each fraction of radiotherapy each day. For single high-dose fraction radiotherapy to the abdomen: 8 mg administered 1 to 2 hours before radiotherapy, with subsequent 8-mg doses every 8 hours after the first dose for 1 to 2 days after completion of radiotherapy. For daily fractionated radiotherapy to the abdomen: 8 mg administered 1 to 2 hours before radiotherapy, with subsequent 8-mg doses every 8 hours after the first dose for each day radiotherapy is given. |
| Postoperative | 16 mg administered 1 hour before induction of anesthesia. |
| Indication | Dosage Regimen |
|---|---|
| Moderately Emetogenic Cancer Chemotherapy |
12 to 17 years of age: 8 mg administered 30 minutes before the start of chemotherapy, with a subsequent 8-mg dose 8 hours after the first dose. Then administer 8 mg twice a day (every 12 hours) for 1 to 2 days after completion of chemotherapy. 4 to 11 years of age: 4 mg administered 30 minutes before the start of chemotherapy, with a subsequent 4-mg dose 4 and 8 hours after the first dose. Then administer 4 mg three times a day for 1 to 2 days after completion of chemotherapy. |
7.3 Tramadol
Although no pharmacokinetic drug interaction between ondansetron and tramadol has been observed, data from 2 small trials indicate that when used together, ondansetron may increase patient-controlled administration of tramadol. Monitor patients to ensure adequate pain control when ondansetron is administered with tramadol.
10 Overdosage (10 OVERDOSAGE)
There is no specific antidote for ondansetron overdose. Patients should be managed with appropriate supportive therapy.
In addition to the adverse reactions listed above, the following adverse reactions have been described in the setting of ondansetron overdose: "Sudden blindness" (amaurosis) of 2 to 3 minutes' duration plus severe constipation occurred in one patient that was administered 72 mg of ondansetron intravenously as a single dose. Hypotension (and faintness) occurred in a patient that took 48 mg of ondansetron tablets. Following infusion of 32 mg over only a 4-minute period, a vasovagal episode with transient second-degree heart block was observed. In all instances, the adverse reactions resolved completely.
Pediatric cases consistent with serotonin syndrome have been reported after inadvertent oral overdoses of ondansetron (exceeding estimated ingestion of 5 mg per kg) in young children. Reported symptoms included somnolence, agitation, tachycardia, tachypnea, hypertension, flushing, mydriasis, diaphoresis, myoclonic movements, horizontal nystagmus, hyperreflexia, and seizure. Patients required supportive care, including intubation in some cases, with complete recovery without sequelae within 1 to 2 days.
11 Description (11 DESCRIPTION)
The active ingredient in ondansetron oral solution, USP is ondansetron hydrochloride as the dihydrate, the racemic form of ondansetron and a selective blocking agent of the serotonin 5-HT3 receptor type. Chemically it is (±) 1, 2, 3, 9-tetrahydro-9-methyl-3-[(2-methyl-1H-imidazol-1-yl)methyl]-4H-carbazol-4-one, monohydrochloride, dihydrate. It has the following structural formula:
The empirical formula is C18H19N3O∙HCl∙2H2O, representing a molecular weight of 365.9.
Ondansetron hydrochloride dihydrate is a white to off-white powder that is soluble in water and normal saline. Each 5 mL of ondansetron oral solution contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron. Ondansetron oral solution contains the inactive ingredients citric acid anhydrous, purified water, sodium benzoate, sodium citrate dihydrate, strawberry flavor, and sucralose.
7.4 Chemotherapy
Carmustine, etoposide, and cisplatin do not affect the pharmacokinetics of ondansetron.
In a crossover trial in 76 pediatric patients, intravenous ondansetron did not increase systemic concentrations of high-dose methotrexate.
8.4 Pediatric Use
The safety and effectiveness of orally administered ondansetron have been established in pediatric patients 4 years and older for the prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy. Use of ondansetron in these age-groups is supported by evidence from adequate and well-controlled studies of ondansetron in adults with additional data from 3 open-label, uncontrolled, non-US trials in 182 pediatric patients aged 4 to 18 years with cancer who were given a variety of cisplatin or noncisplatin regimens [see Dosage and Administration (2.2), Clinical Studies (14.1)].
Additional information on the use of ondansetron in pediatric patients may be found in ondansetron injection prescribing information.
The safety and effectiveness of orally administered ondansetron have not been established in pediatric patients for:
- prevention of nausea and vomiting associated with highly emetogenic cancer chemotherapy.
- prevention of nausea and vomiting associated with radiotherapy.
- prevention of postoperative nausea and/or vomiting.
8.5 Geriatric Use
Of the total number of subjects enrolled in cancer chemotherapy-induced and postoperative nausea and vomiting in U.S.- and foreign-controlled clinical trials, for which there were subgroup analyses, 938 (19%) were aged 65 years and older.
No overall differences in safety or effectiveness were observed between subjects 65 years of age and older and younger subjects. A reduction in clearance and increase in elimination half-life were seen in patients older than 75 years compared with younger subjects [see Clinical Pharmacology (12.3)]. There were an insufficient number of patients older than 75 years of age and older in the clinical trials to permit safety or efficacy conclusions in this age-group. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. No dosage adjustment is needed in elderly patients.
4 Contraindications (4 CONTRAINDICATIONS)
Ondansetron is contraindicated in patients:
- known to have hypersensitivity (e.g., anaphylaxis) to ondansetron or any of the components of the formulation [see Adverse Reactions (6.2)].
- receiving concomitant apomorphine due to the risk of profound hypotension and loss of consciousness.
5.2 Qt Prolongation (5.2 QT Prolongation)
Electrocardiogram (ECG) changes including QT interval prolongation have been seen in patients receiving ondansetron. In addition, postmarketing cases of Torsade de Pointes have been reported in patients using ondansetron. Avoid ondansetron in patients with congenital long QT syndrome. ECG monitoring is recommended in patients with electrolyte abnormalities (e.g., hypokalemia or hypomagnesemia), congestive heart failure, bradyarrhythmias, or patients taking other medicinal products that lead to QT prolongation [see Clinical Pharmacology (12.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions in adults for the:
- prevention of chemotherapy-induced (greater than or equal to 5%) are: headache, malaise/fatigue, constipation, diarrhea. (6.1)
- prevention of radiation-induced nausea and vomiting (greater than or equal to 2%) are: headache, constipation, and diarrhea. (6.1)
- prevention of postoperative nausea and vomiting (greater than or equal to 9%) are: headache and hypoxia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
8.7 Renal Impairment
No dosage adjustment is recommended for patients with any degree of renal impairment (mild, moderate, or severe). There is no experience beyond first-day administration of ondansetron [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In healthy subjects, single intravenous doses of 0.15 mg/kg of ondansetron had no effect on esophageal motility, gastric motility, lower esophageal sphincter pressure, or small intestinal transit time. Multiday administration of ondansetron has been shown to slow colonic transit in healthy subjects. Ondansetron has no effect on plasma prolactin concentrations.
5.3 Serotonin Syndrome
The development of serotonin syndrome has been reported with 5-HT3 receptor antagonists alone. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors, mirtazapine, fentanyl, lithium, tramadol, and intravenous methylene blue). Some of the reported cases were fatal. Serotonin syndrome occurring with overdose of ondansetron alone has also been reported. The majority of reports of serotonin syndrome related to 5-HT3 receptor antagonist use occurred in a post-anesthesia care unit or an infusion center.
Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, with or without gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome, especially with concomitant use of ondansetron and other serotonergic drugs. If symptoms of serotonin syndrome occur, discontinue ondansetron and initiate supportive treatment. Patients should be informed of the increased risk of serotonin syndrome, especially if ondansetron is used concomitantly with other serotonergic drugs [see Drug Interactions (7.1), Overdosage (10)].
7.1 Serotonergic Drugs
Serotonin syndrome (including altered mental status, autonomic instability, and neuromuscular symptoms) has been described following the concomitant use of 5-HT3 receptor antagonists and other serotonergic drugs, including selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs). Monitor for the emergence of serotonin syndrome. If symptoms occur, discontinue ondansetron and initiate supportive treatment [see Warnings and Precautions (5.3)].
8.6 Hepatic Impairment
No dosage adjustment is needed in patients with mild or moderate hepatic impairment.
In patients with severe hepatic impairment, clearance is reduced and the apparent volume of distribution is increased, resulting in a significant increase in the half-life of ondansetron. Therefore, do not exceed a total daily dose of 8 mg in patients with severe hepatic impairment (Child-Pugh score of 10 or greater) [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Ondansetron is indicated for the prevention of nausea and vomiting associated with:
- highly emetogenic cancer chemotherapy, including cisplatin greater than or equal to 50 mg/m2.
- initial and repeat courses of moderately emetogenic cancer chemotherapy.
- radiotherapy in patients receiving either total body irradiation, single high-dose fraction to the abdomen, or daily fractions to the abdomen.
Ondansetron is also indicated for the prevention of postoperative nausea and/or vomiting.
12.1 Mechanism of Action
Ondansetron is a selective 5-HT3 receptor antagonist. While its mechanism of action has not been fully characterized, ondansetron is not a dopamine-receptor antagonist. Serotonin receptors of the 5-HT3 type are present both peripherally on vagal nerve terminals and centrally in the chemoreceptor trigger zone of the area postrema. It is not certain whether ondansetron's antiemetic action is mediated centrally, peripherally, or in both sites. However, cytotoxic chemotherapy appears to be associated with release of serotonin from the enterochromaffin cells of the small intestine. In humans, urinary 5-hydroxyindoleacetic acid (5-HIAA) excretion increases after cisplatin administration in parallel with the onset of emesis. The released serotonin may stimulate the vagal afferents through the 5-HT3 receptors and initiate the vomiting reflex.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity reactions including anaphylaxis and bronchospasm: Discontinue ondansetron if suspected. Monitor and treat promptly per standard of care until signs and symptoms resolve. (5.1)
- QT interval prolongation and Torsade de Pointes: Avoid in patients with congenital long QT syndrome; monitor with electrocardiograms (ECGs) if concomitant electrolyte abnormalities, cardiac failure or arrhythmias, or use of other QT prolonging drugs. (5.2)
- Serotonin syndrome: Reported with 5-HT3 receptor antagonists alone but particularly with concomitant use of serotonergic drugs. If such symptoms occur, discontinue ondansetron and initiate supportive treatment. If concomitant use of ondansetron with other serotonergic drugs is clinically warranted, patients should be made aware of a potential increased risk for serotonin syndrome. (5.3)
- Masking of progressive ileus and/or gastric distention following abdominal surgery or chemotherapy-induced nausea and vomiting: Monitor for decreased bowel activity, particularly in patients with risk factors for gastrointestinal obstruction. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
9 Drug Abuse and Dependence (9 DRUG ABUSE AND DEPENDENCE)
Animal studies have shown that ondansetron is not discriminated as a benzodiazepine nor does it substitute for benzodiazepines in direct addiction studies.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Ondansetron oral solution USP, 4 mg/5 mL, is a clear, colorless to light yellow liquid with a characteristic strawberry odor available in a 50-mL bottle.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ondansetron. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
7.5 Alfentanil and Atracurium
Ondansetron does not alter the respiratory depressant effects produced by alfentanil or the degree of neuromuscular blockade produced by atracurium. Interactions with general or local anesthetics have not been studied.
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis and bronchospasm, have been reported in patients who have exhibited hypersensitivity to other selective 5-HT3 receptor antagonists. If hypersensitivity reactions occur, discontinue use of ondansetron; treat promptly per standard of care and monitor until signs and symptoms resolve [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following adverse reactions have been reported in clinical trials of patients treated with ondansetron, the active ingredient of ondansetron oral solution. A causal relationship to therapy with ondansetron was unclear in many cases.
2.2 Dosage in Hepatic Impairment
In patients with severe hepatic impairment (Child-Pugh score of 10 or greater), do not exceed a total daily dose of 8 mg [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Ondansetron Oral Solution USP, 4 mg/5mL, a clear, colorless to light yellow liquid with a characteristic strawberry odor, contains 5 mg of ondansetron hydrochloride dihydrate equivalent to 4 mg of ondansetron per 5 mL in bottles of 50 mL with child-resistant closures (NDC 51672-4091-3).
14.3 Postoperative Nausea and Vomiting
In two placebo-controlled, double-blind trials (one conducted in the US and the other outside the US) in 865 females undergoing inpatient surgical procedures, ondansetron 16 mg as a single dose or placebo was administered one hour before the induction of general balanced anesthesia (barbiturate, opioid, nitrous oxide, neuromuscular blockade, and supplemental isoflurane or enflurane), ondansetron tablets was significantly more effective than placebo in preventing postoperative nausea and vomiting.
No trials have been performed in males.
7.2 Drugs Affecting Cytochrome P 450 Enzymes (7.2 Drugs Affecting Cytochrome P-450 Enzymes)
Ondansetron does not itself appear to induce or inhibit the cytochrome P-450 drug-metabolizing enzyme system of the liver [see Clinical Pharmacology (12.3)]. Because ondansetron is metabolized by hepatic cytochrome P-450 drug-metabolizing enzymes (CYP3A4, CYP2D6, CYP1A2), inducers or inhibitors of these enzymes may change the clearance and, hence, the half-life of ondansetron. In patients treated with potent inducers of CYP3A4 (i.e., phenytoin, carbamazepine, and rifampin), the clearance of ondansetron was significantly increased and ondansetron blood concentrations were decreased. However, on the basis of available data, no dosage adjustment for ondansetron is recommended for patients on these drugs [see Clinical Pharmacology (12.3)].
Principal Display Panel 50 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 50 mL Bottle Carton)
50 mL
NDC 51672-4091-3
Ondansetron
Oral Solution
USP, 4 mg/5 mL
Rx only
TARO
5.4 Masking of Progressive Ileus and Gastric Distension
The use of ondansetron in patients following abdominal surgery or in patients with chemotherapy-induced nausea and vomiting may mask a progressive ileus and/or gastric distension. Monitor for decreased bowel activity, particularly in patients with risk factors for gastrointestinal obstruction.
Ondansetron is not a drug that stimulates gastric or intestinal peristalsis. It should not be used instead of nasogastric suction.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenic effects were not seen in 2-year studies in rats and mice with oral ondansetron doses up to 10 mg/kg per day and 30 mg/kg per day, respectively (approximately 4 and 6 times the maximum recommended human oral dose of 24 mg per day, based on body surface area).
Ondansetron was not mutagenic in standard tests for mutagenicity.
Oral administration of ondansetron up to 15 mg/kg per day (approximately 6 times the maximum recommended human oral dose of 24 mg per day, based on body surface area) did not affect fertility or general reproductive performance of male and female rats.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:44.070289 · Updated: 2026-03-14T21:41:24.246163