a921ee49-2c16-9a79-e053-2a95a90aaa03
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
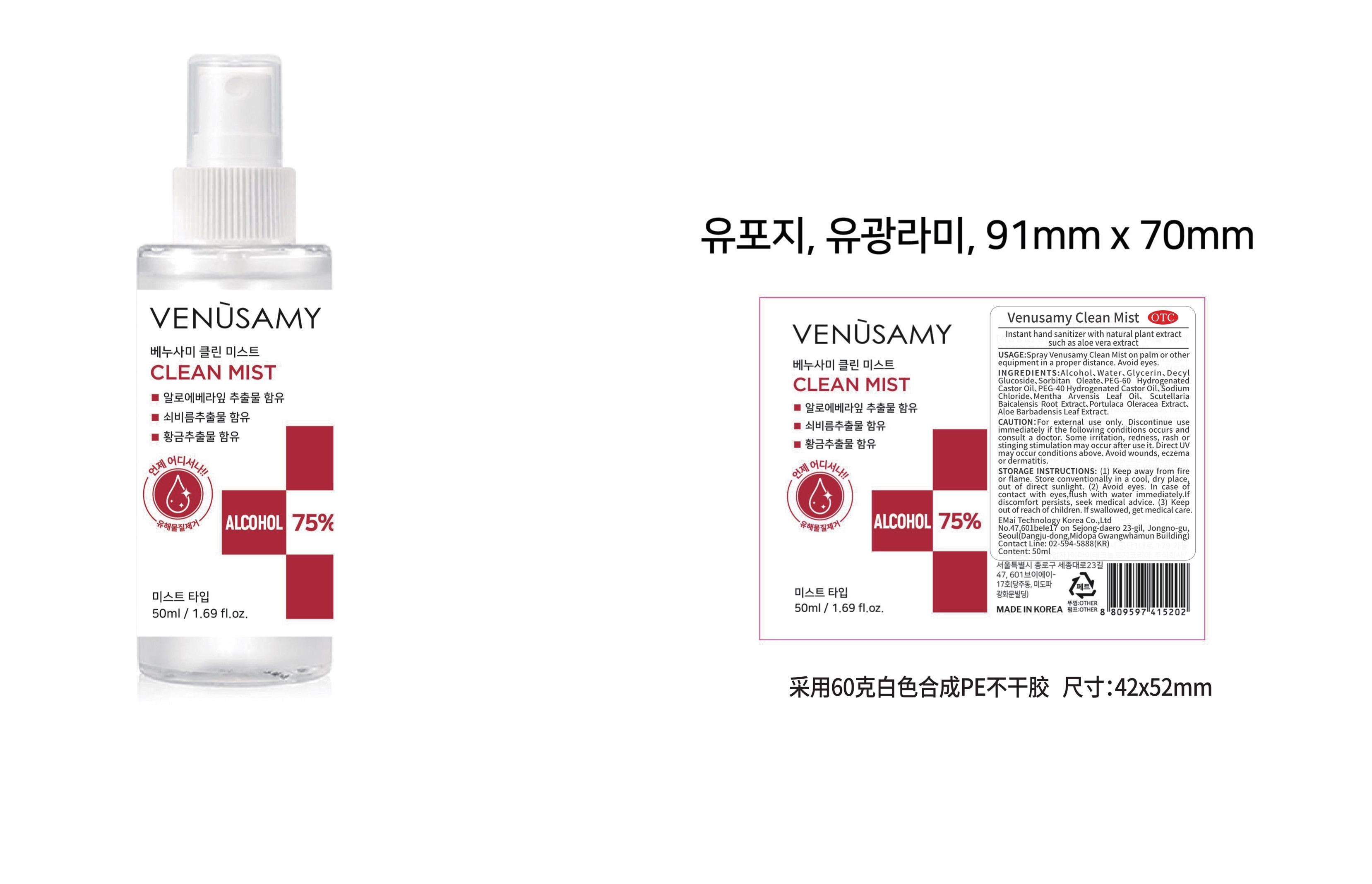
Store conventionally in a cool, dry place,out of direct sunlight.
Medication Information
Warnings and Precautions
For external use only. Discontinue use immediately if the following conditions occurs and consult a doctor. Some irritation, redness, rash or stinging stimulation may occur after use it. Direct UV may occur conditions above. Avoid wounds, eczema or dermatitis.
Indications and Usage
Spray Venusamy Clean Mist on palm or other equipment in a proper distance. Avoid eyes.
Dosage and Administration
Store conventionally in a cool, dry place,out of direct sunlight.
Description
Store conventionally in a cool, dry place,out of direct sunlight.
Section 50565-1
keep out of reach of children
Section 51727-6
Water
Glycerin
Decyl Glucoside
Sorbitan Oleate
PEG-60 Hydrogenated Castor Oil
PEG-40 Hydrogenated Castor Oil
Sodium Chloride
Mentha Arvensis Leaf Oil
Scutellaria Baicalensis Root Extract
Portulaca Oleracea Extract
Aloe Barbadensis Leaf Extract
Section 51945-4
Section 55105-1
Disinfection
Sterilization
Section 55106-9
Alcohol
Structured Label Content
Indications and Usage (34067-9)
Spray Venusamy Clean Mist on palm or other equipment in a proper distance. Avoid eyes.
Dosage and Administration (34068-7)
Store conventionally in a cool, dry place,out of direct sunlight.
Warnings and Precautions (34071-1)
For external use only. Discontinue use immediately if the following conditions occurs and consult a doctor. Some irritation, redness, rash or stinging stimulation may occur after use it. Direct UV may occur conditions above. Avoid wounds, eczema or dermatitis.
Section 50565-1 (50565-1)
keep out of reach of children
Section 51727-6 (51727-6)
Water
Glycerin
Decyl Glucoside
Sorbitan Oleate
PEG-60 Hydrogenated Castor Oil
PEG-40 Hydrogenated Castor Oil
Sodium Chloride
Mentha Arvensis Leaf Oil
Scutellaria Baicalensis Root Extract
Portulaca Oleracea Extract
Aloe Barbadensis Leaf Extract
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Disinfection
Sterilization
Section 55106-9 (55106-9)
Alcohol
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:04.394017 · Updated: 2026-03-14T22:57:25.927839