Giltuss Honey Dm
a8c08532-1169-6603-e053-2a95a90a3484
34390-5
HUMAN OTC DRUG LABEL
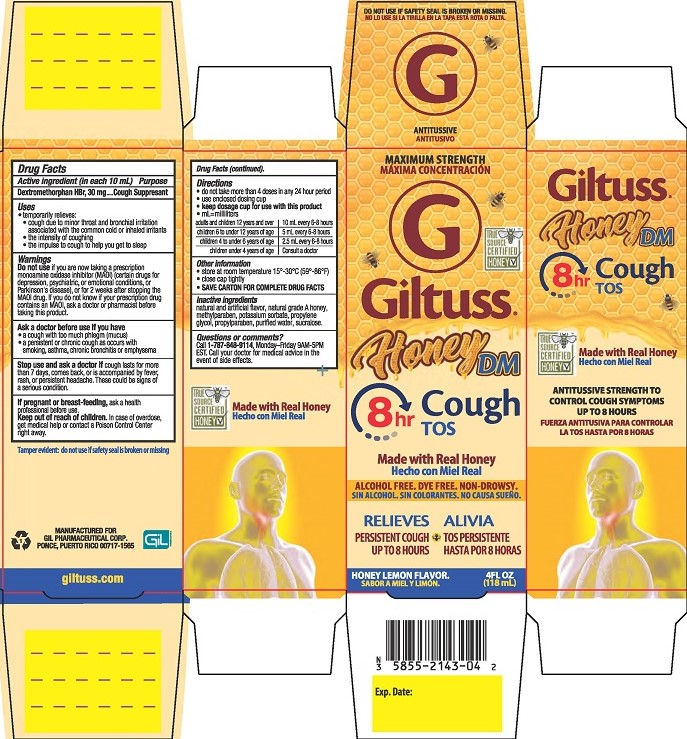
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Warnings and Precautions
Warnings
Indications and Usage
Uses
- temporarily relieves:
- cough due to minor throat and bronchial irritation associated with the common cold or inhaled irritants
- the intensity of coughing
- the impulse to cough to help your child get to sleep
Dosage and Administration
Directions
- do not take more than 4 doses in any 24-hour period
- use enclosed dosing cup
- keep dosage cup for use with this product
- ml = milliliters
adults and children 12 years and over 10 mL every 6-8 hours children 6 to under 12 years of age 5 mL every 6-8 hours children 4 to under 6 years of age 2.5 mL every 6-8 hours children under 4 years of age Consult a doctor
Description
Drug Facts
Section 42229-5
Drug Facts
Section 44425-7
Other information
- store at room temperature 15°-30°C (59°-86°F)
- close cap tightly
- SAVE CARTON FOR COMPLETE DRUG FACTS
Section 50565-1
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor ifcough lasts for more than 7 days, comes back, or is accompanied by fever, rash or persistent headache. These could be signs of a serious condition.
Section 50569-3
Ask a doctor before use if you have
- a cough with too much phlegm (mucus)
- a persistent or chronic cough as occurs with smoking, asthma, chronic bronchitis, or emphysema
Section 50570-1
Do not useif you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Section 51727-6
Inactive ingredients natural and artificial flavor, natural grade A honey, methylparaben, potassium sorbate, propylene glycol, propylparaben, purified water, sucralose.
Section 51945-4
MAXIMUM STRENGTH
Giltuss®
Honey DM Cough
Made with real honey
ALCOHOL FREE. DYE FREE. NON-DROWSY
RELIEVES
PERSISTENT COUGH UP TO 8 HOURS
HONEY LEMON FLAVOR.
4FL OZ (118 mL)
Section 53413-1
Questions or comments? Call 1-787-848-9114, Monday-Friday 9AM - 5PM EST. Call your doctor for medical advice in the event of side effects.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purposes
Cough Suppresant
Section 55106-9
Active ingredient (in each 10 mL)
Dextromethorphan HBr, 30 mg
Structured Label Content
Indications and Usage (34067-9)
Uses
- temporarily relieves:
- cough due to minor throat and bronchial irritation associated with the common cold or inhaled irritants
- the intensity of coughing
- the impulse to cough to help your child get to sleep
Dosage and Administration (34068-7)
Directions
- do not take more than 4 doses in any 24-hour period
- use enclosed dosing cup
- keep dosage cup for use with this product
- ml = milliliters
adults and children 12 years and over 10 mL every 6-8 hours children 6 to under 12 years of age 5 mL every 6-8 hours children 4 to under 6 years of age 2.5 mL every 6-8 hours children under 4 years of age Consult a doctor
Warnings and Precautions (34071-1)
Warnings
Section 42229-5 (42229-5)
Drug Facts
Section 44425-7 (44425-7)
Other information
- store at room temperature 15°-30°C (59°-86°F)
- close cap tightly
- SAVE CARTON FOR COMPLETE DRUG FACTS
Section 50565-1 (50565-1)
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor ifcough lasts for more than 7 days, comes back, or is accompanied by fever, rash or persistent headache. These could be signs of a serious condition.
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- a cough with too much phlegm (mucus)
- a persistent or chronic cough as occurs with smoking, asthma, chronic bronchitis, or emphysema
Section 50570-1 (50570-1)
Do not useif you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Section 51727-6 (51727-6)
Inactive ingredients natural and artificial flavor, natural grade A honey, methylparaben, potassium sorbate, propylene glycol, propylparaben, purified water, sucralose.
Section 51945-4 (51945-4)
MAXIMUM STRENGTH
Giltuss®
Honey DM Cough
Made with real honey
ALCOHOL FREE. DYE FREE. NON-DROWSY
RELIEVES
PERSISTENT COUGH UP TO 8 HOURS
HONEY LEMON FLAVOR.
4FL OZ (118 mL)
Section 53413-1 (53413-1)
Questions or comments? Call 1-787-848-9114, Monday-Friday 9AM - 5PM EST. Call your doctor for medical advice in the event of side effects.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purposes
Cough Suppresant
Section 55106-9 (55106-9)
Active ingredient (in each 10 mL)
Dextromethorphan HBr, 30 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:02.104989 · Updated: 2026-03-14T23:13:00.870746