Eraxis
a88d9010-55fb-4a02-baff-042cd27688ea
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
ERAXIS is an echinocandin antifungal indicated for the treatment of the following infections: • Candidemia and other forms of Candida infections (intra-abdominal abscess and peritonitis) in adults and pediatric patients (1 month of age and older) ( 1.1 ) • Esophageal candidiasis in adults ( 1.2 ) Limitations of use • ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida or in sufficient numbers of neutropenic patients. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established. ( 1.3 , 5.3 , 8.4 ) • ERAXIS is associated with high relapse rates in esophageal candidiasis. ( 1.3 , 14.2 )
Dosage and Administration
Adults Pediatric Patients 1 Month of Age and Older Candidemia and other forms of Candida infections 200 mg loading dose on Day 1, followed by 100 mg once daily maintenance dose thereafter for at least 14 days after the last positive culture ( 2.1 ) 3 mg/kg (not to exceed 200 mg) loading dose on Day 1, followed by 1.5 mg/kg (not to exceed 100 mg) once daily maintenance dose thereafter for at least 14 days after the last positive culture ( 2.2 ) Esophageal candidiasis 100 mg loading dose on Day 1, followed by 50 mg once daily maintenance dose thereafter for a minimum of 14 days and for at least 7 days following resolution of symptoms ( 2.1 ) Not Approved Rate of Infusion for Adults and Pediatric Patients The rate of infusion should not exceed 1.1 mg/minute [equivalent to 1.4 mL/minute or 84 mL/hour when reconstituted and diluted per instructions] ( 2.3 , 2.4 )
Contraindications
ERAXIS is contraindicated in: • Patients with known hypersensitivity to anidulafungin, any component of ERAXIS, or other echinocandins [see Warnings and Precautions (5.2) ] • Patients with known or suspected Hereditary Fructose Intolerance (HFI) [see Warnings and Precautions (5.4) ]
Warnings and Precautions
• Hepatic Effects : Risk of abnormal liver tests, hepatitis, hepatic failure; monitor hepatic function during therapy. ( 5.1 , 13.2 ) • Hypersensitivity : Anaphylaxis, including shock has been reported. Risk of infusion-related adverse reactions, possibly histamine-mediated, including rash, urticaria, flushing, pruritus, bronchospasm, dyspnea, and hypotension; to reduce occurrence, do not exceed a rate of infusion of 1.1 mg/minute. ( 2.4 , 5.2 ) • Risk of Neonatal Toxicity Associated with Polysorbates : ERAXIS contains polysorbate 80, an inactive ingredient. Thrombocytopenia, renal dysfunction, hepatomegaly, cholestasis, ascites, hypotension and metabolic acidosis haves been reported in low-birth weight infants receiving high doses of polysorbate. ERAXIS is not approved in pediatric patients younger than 1 month of age. ( 5.3 , 8.4 ) • Hereditary Fructose Intolerance (HFI) : ERAXIS contains fructose. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before ERAXIS administration. ( 5.4 , 8.4 )
Adverse Reactions
Laboratory abnormalities in liver tests have been seen in healthy volunteers and pediatric patients treated with ERAXIS. In some patients with serious underlying medical conditions who were receiving multiple concomitant medications along with ERAXIS, clinically significant hepatic abnormalities have occurred. Isolated cases of significant hepatic dysfunction, hepatitis, or hepatic failure have been reported in patients; a causal relationship to ERAXIS has not been established [see Adverse Reactions (6.1) and Nonclinical Toxicology (13.2) ] . Patients who develop abnormal liver tests during ERAXIS therapy should be monitored for evidence of worsening hepatic tests and evaluated for risk/benefit of continuing ERAXIS therapy.
How Supplied
ERAXIS (anidulafungin) for Injection is supplied in a single-dose vial of sterile, lyophilized, preservative-free, white to off-white powder. ERAXIS (anidulafungin) is available in the following packaging configuration: Single-Dose Vial of ERAXIS 50 mg NDC 0049-0114-28 One - 50 mg vial Single-Dose Vial of ERAXIS 100 mg NDC 0049-0116-28 One - 100 mg vial
Description
ERAXIS is an echinocandin antifungal indicated for the treatment of the following infections: • Candidemia and other forms of Candida infections (intra-abdominal abscess and peritonitis) in adults and pediatric patients (1 month of age and older) ( 1.1 ) • Esophageal candidiasis in adults ( 1.2 ) Limitations of use • ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida or in sufficient numbers of neutropenic patients. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established. ( 1.3 , 5.3 , 8.4 ) • ERAXIS is associated with high relapse rates in esophageal candidiasis. ( 1.3 , 14.2 )
Medication Information
Warnings and Precautions
• Hepatic Effects : Risk of abnormal liver tests, hepatitis, hepatic failure; monitor hepatic function during therapy. ( 5.1 , 13.2 ) • Hypersensitivity : Anaphylaxis, including shock has been reported. Risk of infusion-related adverse reactions, possibly histamine-mediated, including rash, urticaria, flushing, pruritus, bronchospasm, dyspnea, and hypotension; to reduce occurrence, do not exceed a rate of infusion of 1.1 mg/minute. ( 2.4 , 5.2 ) • Risk of Neonatal Toxicity Associated with Polysorbates : ERAXIS contains polysorbate 80, an inactive ingredient. Thrombocytopenia, renal dysfunction, hepatomegaly, cholestasis, ascites, hypotension and metabolic acidosis haves been reported in low-birth weight infants receiving high doses of polysorbate. ERAXIS is not approved in pediatric patients younger than 1 month of age. ( 5.3 , 8.4 ) • Hereditary Fructose Intolerance (HFI) : ERAXIS contains fructose. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before ERAXIS administration. ( 5.4 , 8.4 )
Indications and Usage
ERAXIS is an echinocandin antifungal indicated for the treatment of the following infections: • Candidemia and other forms of Candida infections (intra-abdominal abscess and peritonitis) in adults and pediatric patients (1 month of age and older) ( 1.1 ) • Esophageal candidiasis in adults ( 1.2 ) Limitations of use • ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida or in sufficient numbers of neutropenic patients. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established. ( 1.3 , 5.3 , 8.4 ) • ERAXIS is associated with high relapse rates in esophageal candidiasis. ( 1.3 , 14.2 )
Dosage and Administration
Adults Pediatric Patients 1 Month of Age and Older Candidemia and other forms of Candida infections 200 mg loading dose on Day 1, followed by 100 mg once daily maintenance dose thereafter for at least 14 days after the last positive culture ( 2.1 ) 3 mg/kg (not to exceed 200 mg) loading dose on Day 1, followed by 1.5 mg/kg (not to exceed 100 mg) once daily maintenance dose thereafter for at least 14 days after the last positive culture ( 2.2 ) Esophageal candidiasis 100 mg loading dose on Day 1, followed by 50 mg once daily maintenance dose thereafter for a minimum of 14 days and for at least 7 days following resolution of symptoms ( 2.1 ) Not Approved Rate of Infusion for Adults and Pediatric Patients The rate of infusion should not exceed 1.1 mg/minute [equivalent to 1.4 mL/minute or 84 mL/hour when reconstituted and diluted per instructions] ( 2.3 , 2.4 )
Contraindications
ERAXIS is contraindicated in: • Patients with known hypersensitivity to anidulafungin, any component of ERAXIS, or other echinocandins [see Warnings and Precautions (5.2) ] • Patients with known or suspected Hereditary Fructose Intolerance (HFI) [see Warnings and Precautions (5.4) ]
Adverse Reactions
Laboratory abnormalities in liver tests have been seen in healthy volunteers and pediatric patients treated with ERAXIS. In some patients with serious underlying medical conditions who were receiving multiple concomitant medications along with ERAXIS, clinically significant hepatic abnormalities have occurred. Isolated cases of significant hepatic dysfunction, hepatitis, or hepatic failure have been reported in patients; a causal relationship to ERAXIS has not been established [see Adverse Reactions (6.1) and Nonclinical Toxicology (13.2) ] . Patients who develop abnormal liver tests during ERAXIS therapy should be monitored for evidence of worsening hepatic tests and evaluated for risk/benefit of continuing ERAXIS therapy.
How Supplied
ERAXIS (anidulafungin) for Injection is supplied in a single-dose vial of sterile, lyophilized, preservative-free, white to off-white powder. ERAXIS (anidulafungin) is available in the following packaging configuration: Single-Dose Vial of ERAXIS 50 mg NDC 0049-0114-28 One - 50 mg vial Single-Dose Vial of ERAXIS 100 mg NDC 0049-0116-28 One - 100 mg vial
Description
ERAXIS is an echinocandin antifungal indicated for the treatment of the following infections: • Candidemia and other forms of Candida infections (intra-abdominal abscess and peritonitis) in adults and pediatric patients (1 month of age and older) ( 1.1 ) • Esophageal candidiasis in adults ( 1.2 ) Limitations of use • ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida or in sufficient numbers of neutropenic patients. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established. ( 1.3 , 5.3 , 8.4 ) • ERAXIS is associated with high relapse rates in esophageal candidiasis. ( 1.3 , 14.2 )
Section 42229-5
Candidemia and other Candida infections (intra-abdominal abscess and peritonitis)
The recommended dose is a single 200 mg loading dose of ERAXIS on Day 1, followed by a 100 mg once daily maintenance dose thereafter. Duration of treatment should be based on the patient's clinical response. In general, antifungal therapy should continue for at least 14 days after the last positive culture.
1.4 Usage
Specimens for fungal culture and other relevant laboratory studies (including histopathology) should be obtained prior to therapy to isolate and identify causative organism(s). Therapy may be instituted before the results of the cultures and other laboratory studies are known. However, once these results become available, antifungal therapy should be adjusted accordingly.
7.4 Rifampin
Administration of multiple doses of anidulafungin and rifampin to patients resulted in no significant alteration in the steady state pharmacokinetics of anidulafungin. No dosage adjustment of anidulafungin is needed when it is co-administered with rifampin [see Clinical Pharmacology (12.3)].
10 Overdosage
During clinical trials a single 400 mg dose of ERAXIS was inadvertently administered as a loading dose. No clinical adverse events were reported. In a study of 10 healthy subjects administered a loading dose of 260 mg followed by 130 mg daily; 3 of the 10 subjects experienced transient, asymptomatic transaminase elevations (≤3 × ULN) [see Warnings and Precautions (5.1)].
Anidulafungin is not dialyzable.
The maximum non-lethal dose of anidulafungin in rats was 50 mg/kg, a dose which is equivalent to 10 times the recommended daily dose for esophageal candidiasis (50 mg/day) or equivalent to 5 times the recommended daily dose for candidemia and other Candida infections (100 mg/day), based on relative body surface area comparisons.
11 Description
ERAXIS for Injection is a sterile, lyophilized product for intravenous (IV) infusion that contains anidulafungin. ERAXIS (anidulafungin) is a semi-synthetic lipopeptide synthesized from a fermentation product of Aspergillus nidulans. Anidulafungin is an echinocandin, a class of antifungal drugs that inhibits the synthesis of 1,3-β-D-glucan, an essential component of fungal cell walls.
ERAXIS (anidulafungin) is 1-[(4R,5R)-4,5-dihydroxy-N2-[[4"-(pentyloxy)[1,1':4',1"-terphenyl]-4-yl]carbonyl]-L-ornithine]echinocandin B. Anidulafungin is a white to off-white powder that is practically insoluble in water and slightly soluble in ethanol. In addition to the active ingredient, anidulafungin, ERAXIS for Injection contains the following inactive ingredients:
50 mg/vial – fructose (50 mg), mannitol (250 mg), polysorbate 80 (125 mg), tartaric acid (5.6 mg), and sodium hydroxide and/or hydrochloric acid for pH adjustment.
100 mg/vial – fructose (100 mg), mannitol (500 mg), polysorbate 80 (250 mg), tartaric acid (11.2 mg), and sodium hydroxide and/or hydrochloric acid for pH adjustment.
The empirical formula of anidulafungin is C58H73N7O17 and the formula weight is 1140.3.
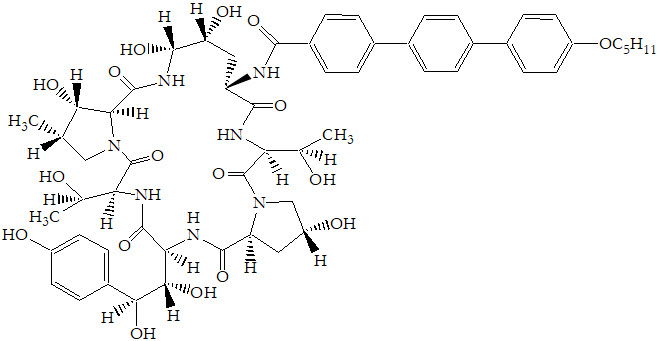
The structural formula is:
Prior to administration, ERAXIS for Injection requires reconstitution with sterile Water for Injection and subsequent dilution with either 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP (normal saline).
7.3 Tacrolimus
Administration of multiple doses of anidulafungin and a single-dose of tacrolimus to healthy subjects resulted in no significant alteration in the steady state pharmacokinetics of either drug. No dosage adjustment of tacrolimus or anidulafungin is needed when the two drugs are co-administered [see Clinical Pharmacology (12.3)].
7.1 Cyclosporine
Administration of multiple doses of anidulafungin and cyclosporine to healthy subjects resulted in no significant alteration in the steady state pharmacokinetics of either drug. No dosage adjustment of cyclosporine or anidulafungin is needed when the two drugs are co-administered [see Clinical Pharmacology (12.3)].
7.2 Voriconazole
Administration of multiple doses of anidulafungin and voriconazole to healthy subjects resulted in no significant alteration in the steady state pharmacokinetics of either drug. No dosage adjustment of voriconazole or anidulafungin is needed when the two drugs are co-administered [see Clinical Pharmacology (12.3)].
16.1 How Supplied
ERAXIS (anidulafungin) for Injection is supplied in a single-dose vial of sterile, lyophilized, preservative-free, white to off-white powder. ERAXIS (anidulafungin) is available in the following packaging configuration:
|
Single-Dose Vial of ERAXIS 50 mg |
|
|
NDC 0049-0114-28 |
One - 50 mg vial |
|
Single-Dose Vial of ERAXIS 100 mg |
|
|
NDC 0049-0116-28 |
One - 100 mg vial |
8.4 Pediatric Use
The safety and effectiveness of ERAXIS for the treatment of candidemia and the following Candida infections: intra-abdominal abscess and peritonitis, have been established in pediatric patients 1 month of age and older. Use of ERAXIS for this indication in this age group is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic, safety data in pediatric patients 1 month of age and older [see Indications and Usage (1), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
The safety and effectiveness of ERAXIS in patients younger than 1 month of age with candidemia/invasive candidiasis has not been established.
Candidemia/invasive candidiasis in pediatric patients younger than 1 month of age has a higher rate of central nervous system (CNS) and multi-organ dissemination than in older patients. In addition, in patients younger than 1 month of age ERAXIS carries a potential risk of life-threatening toxicity associated with high doses of polysorbate 80, an inactive ingredient in ERAXIS [see Warnings and Precautions (5.3)].
The safety and effectiveness of ERAXIS in pediatric patients with esophageal candidiasis has not been established.
ERAXIS is contraindicated in adult and pediatric patients with HFI. Because a diagnosis of HFI may not yet be established in pediatric patients, obtain a careful history of HFI symptoms with fructose/sucrose exposure prior to administration of ERAXIS [see Warnings and Precautions (5.4)].
8.5 Geriatric Use
Of the total number of subjects (N = 197) in the pivotal clinical studies of anidulafungin, 35% were 65 years and over, while 18% were 75 years and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Dosage adjustments are not required for geriatric patients [see Clinical Pharmacology (12.3)].
4 Contraindications
ERAXIS is contraindicated in:
-
•Patients with known hypersensitivity to anidulafungin, any component of ERAXIS, or other echinocandins [see Warnings and Precautions (5.2)]
-
•Patients with known or suspected Hereditary Fructose Intolerance (HFI) [see Warnings and Precautions (5.4)]
6 Adverse Reactions
The following most serious adverse reactions are described elsewhere in other labeling sections:
-
•Hepatic Adverse Reactions [see Warnings and Precautions (5.1)]
-
•Anaphylactic and Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
1.3 Limitations of Use
-
•ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida, and has not been studied in sufficient numbers of neutropenic patients to determine efficacy in this group. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established [see Warning and Precautions (5.3), Use in Specific Populations (8.4)].
-
•ERAXIS is associated with high relapse rates in esophageal candidiasis [see Clinical Studies (14.2)].
1 Indications and Usage
ERAXIS is an echinocandin antifungal indicated for the treatment of the following infections:
-
•Candidemia and other forms of Candida infections (intra-abdominal abscess and peritonitis) in adults and pediatric patients (1 month of age and older) (1.1)
-
•Esophageal candidiasis in adults (1.2)
Limitations of use
-
•ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida or in sufficient numbers of neutropenic patients. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established. (1.3, 5.3, 8.4)
-
•ERAXIS is associated with high relapse rates in esophageal candidiasis. (1.3, 14.2)
8.7 Renal Insufficiency
Dosage adjustments are not required for patients with any degree of renal insufficiency including those on hemodialysis. Anidulafungin has negligible (<1%) renal clearance. In a clinical study of subjects with mild, moderate, severe or end stage (dialysis-dependent) renal insufficiency, anidulafungin pharmacokinetics were similar to those observed in subjects with normal renal function. Anidulafungin is not dialyzable and may be administered without regard to the timing of hemodialysis [see Clinical Pharmacology (12.3)].
12.1 Mechanism of Action
Anidulafungin is an anti-fungal drug [see Microbiology (12.4)].
8.6 Hepatic Insufficiency
No dosing adjustments are required for patients with any degree of hepatic insufficiency. Anidulafungin is not hepatically metabolized. Anidulafungin pharmacokinetics were examined in subjects with Child-Pugh class A, B or C hepatic insufficiency. Anidulafungin concentrations were not increased in subjects with any degree of hepatic insufficiency. Though a slight decrease in AUC was observed in patients with Child-Pugh C hepatic insufficiency, it was within the range of population estimates noted for healthy subjects [see Clinical Pharmacology (12.3)].
1.2 Esophageal Candidiasis
ERAXIS is indicated for the treatment of esophageal candidiasis in adults [see Indications and Usage (1.3), Clinical Studies (14.2)].
5 Warnings and Precautions
-
•Hepatic Effects: Risk of abnormal liver tests, hepatitis, hepatic failure; monitor hepatic function during therapy. (5.1, 13.2)
-
•Hypersensitivity: Anaphylaxis, including shock has been reported. Risk of infusion-related adverse reactions, possibly histamine-mediated, including rash, urticaria, flushing, pruritus, bronchospasm, dyspnea, and hypotension; to reduce occurrence, do not exceed a rate of infusion of 1.1 mg/minute. (2.4, 5.2)
-
•Risk of Neonatal Toxicity Associated with Polysorbates: ERAXIS contains polysorbate 80, an inactive ingredient. Thrombocytopenia, renal dysfunction, hepatomegaly, cholestasis, ascites, hypotension and metabolic acidosis haves been reported in low-birth weight infants receiving high doses of polysorbate. ERAXIS is not approved in pediatric patients younger than 1 month of age. (5.3, 8.4)
-
•Hereditary Fructose Intolerance (HFI): ERAXIS contains fructose. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before ERAXIS administration. (5.4, 8.4)
14.2 Esophageal Candidiasis
ERAXIS was evaluated in a double-blind, double-dummy, randomized Phase 3 study. Three hundred patients received ERAXIS (100 mg loading dose IV on Day 1 followed by 50 mg/day IV) and 301 received oral fluconazole (200 mg loading dose on Day 1 followed by 100 mg/day). Treatment duration was 7 days beyond resolution of symptoms for a minimum of 14 and a maximum of 21 days.
Of the 442 patients with culture confirmed esophageal candidiasis, most patients (91%) had C. albicans isolated at the baseline.
Treatment groups were similar in demographic and other baseline characteristics. In ERAXIS-treated patients, the age range was 16–69 years, the gender distribution was 42% male and 58% female, and the race distribution was 15% White, 49% Black/African American, 15% Asian, 0.3 % Hispanic, 21% other races.
In this study, of 280 patients tested, 237 (84.6%) tested HIV positive. In both groups the median time to resolution of symptoms was 5 days and the median duration of therapy was 14 days.
Efficacy was assessed by endoscopic outcome at end of therapy (EOT). Patients were considered clinically evaluable if they received at least 10 days of therapy, had an EOT assessment with a clinical outcome other than 'indeterminate', had an endoscopy at EOT, and did not have any protocol violations prior to the EOT visit that would affect an assessment of efficacy.
An endoscopic success, defined as cure (endoscopic grade of 0 on a 4-point severity scale) or improvement (decrease of one or more grades from baseline), was seen in 225/231 (97.4%) ERAXIS-treated patients and 233/236 (98.7%) fluconazole-treated patients (Table 13). The majority of these patients were endoscopic cures (grade=0). Two weeks after completing therapy, the ERAXIS group had significantly more endoscopically-documented relapses than the fluconazole group, 120/225 (53.3%) vs. 45/233 (19.3%), respectively (Table 13).
|
Endoscopic Response at End of Therapy |
||||
|
Response |
ERAXIS
|
Fluconazole
|
Treatment Difference Calculated as ERAXIS minus fluconazole
|
95% CI |
|
Endoscopic Success, n (%) |
225 (97.4) |
233 (98.7) |
-1.3% |
-3.8%, 1.2% |
|
Cure |
204 (88.3) |
221 (93.6) |
||
|
Improvement |
21 (9.1) |
12 (5.1) |
||
|
Failure, n (%) |
6 (2.6) |
3 (1.3) |
||
|
|
||||
|
Endoscopic Relapse Rates at Follow-Up, 2 Weeks Post-Treatment |
||||
|
ERAXIS |
Fluconazole |
Treatment Difference |
95% CI |
|
|
Endoscopic Relapse, n/N (%) |
120/225 (53.3%) |
45/233 (19.3%) |
34.0% |
25.8%, 42.3% |
Clinical success (cure or improvement in clinical symptoms including odynophagia/dysphagia and retrosternal pain) occurred in 229/231 (99.1%) of the ERAXIS-treated patients and 235/236 (99.6%) of the fluconazole-treated patients at the end of therapy. For patients with C. albicans, microbiological success occurred in 142/162 (87.7%) of the ERAXIS-treated group and 157/166 (94.6%) of the fluconazole-treated group at the end of therapy. For patients with Candida species other than C. albicans, success occurred in 10/12 (83.3%) of the ERAXIS-treated group and 14/16 (87.5%) of the fluconazole-treated group.
2 Dosage and Administration
|
Adults |
Pediatric Patients 1 Month of Age and Older |
|
Candidemia and other forms of Candida infections |
|
|
200 mg loading dose on Day 1, followed by 100 mg once daily maintenance dose thereafter for at least 14 days after the last positive culture (2.1) |
3 mg/kg (not to exceed 200 mg) loading dose on Day 1, followed by 1.5 mg/kg (not to exceed 100 mg) once daily maintenance dose thereafter for at least 14 days after the last positive culture (2.2) |
|
Esophageal candidiasis |
|
|
100 mg loading dose on Day 1, followed by 50 mg once daily maintenance dose thereafter for a minimum of 14 days and for at least 7 days following resolution of symptoms (2.1) |
Not Approved |
Rate of Infusion for Adults and Pediatric Patients
The rate of infusion should not exceed 1.1 mg/minute [equivalent to 1.4 mL/minute or 84 mL/hour when reconstituted and diluted per instructions] (2.3, 2.4)
3 Dosage Forms and Strengths
For injection: 50 mg, and 100 mg of anidulafungin as a white to off-white sterile lyophilized powder in a single-dose vial for reconstitution.
5.1 Hepatic Adverse Reactions
Laboratory abnormalities in liver tests have been seen in healthy volunteers and pediatric patients treated with ERAXIS. In some patients with serious underlying medical conditions who were receiving multiple concomitant medications along with ERAXIS, clinically significant hepatic abnormalities have occurred. Isolated cases of significant hepatic dysfunction, hepatitis, or hepatic failure have been reported in patients; a causal relationship to ERAXIS has not been established [see Adverse Reactions (6.1) and Nonclinical Toxicology (13.2)]. Patients who develop abnormal liver tests during ERAXIS therapy should be monitored for evidence of worsening hepatic tests and evaluated for risk/benefit of continuing ERAXIS therapy.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post approval use of anidulafungin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune: Anaphylactic shock, anaphylactic reaction, bronchospasm [see Warnings and Precautions (5.2)].
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.3 Preparation for Administration
ERAXIS for Injection must be reconstituted with sterile Water for Injection and subsequently diluted only with 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP (normal saline), prior to administration.
The compatibility of reconstituted ERAXIS with intravenous substances, additives, or medications other than 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP (normal saline) has not been established. Do NOT dilute with other solutions or co-infuse with other medications or electrolytes. The infusion solution must not be frozen.
1.1 Candidemia and Other Forms of Candida
ERAXIS is indicated for the treatment of candidemia and the following Candida infections: intra-abdominal abscess and peritonitis in adults and pediatric patients 1 month of age and older [see Clinical Studies (14.1) and Microbiology (12.4)].
7.5 Amphotericin B Liposome for Injection
Administration of multiple doses of anidulafungin and liposomal amphotericin B to patients resulted in no significant alteration in the steady state pharmacokinetics of anidulafungin. No dosage adjustment of anidulafungin is needed when it is co-administered with liposomal amphotericin B [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology And/or Pharmacology
In 3 month studies, liver toxicity, including single cell hepatocellular necrosis, hepatocellular hypertrophy and increased liver weights were observed in monkeys and rats at doses equivalent to 5–6 times human exposure. For both species, hepatocellular hypertrophy was still noted one month after the end of dosing.
Principal Display Panel 50 Mg Vial Label
1 Sterile Single-dose Vial
Eraxis
®
(anidulafungin)
for Injection
50 mg/vial
For Intravenous Infusion Only
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
Principal Display Panel 100 Mg Vial Label
1 Sterile Single-dose Vial
Eraxis
®
(anidulafungin)
for Injection
100 mg/vial
For Intravenous Infusion Only
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
Principal Display Panel 50 Mg Vial Carton
NDC 0049-0114-28
Unit Pack Contains:
1 Sterile Single-dose Vial
Eraxis
®
50 mg
Eraxis
®
(anidulafungin)
for Injection
50 mg/vial
For Intravenous Infusion Only
Store Vial in Refrigerator
at 2° C - 8° C (36° F - 46° F).
Do Not Freeze.
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
Principal Display Panel 100 Mg Vial Carton
NDC 0049-0116-28
Unit Pack Contains:
1 Sterile Single-dose Vial
Eraxis
®
100 mg
Eraxis
®
(anidulafungin)
for Injection
100 mg/vial
For Intravenous Infusion Only
Store Vial in Refrigerator
at 2° C - 8° C (36° F - 46° F).
Do Not Freeze.
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
2.4 Dilution and Administration of the Infusion
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If particulate matter or discoloration is identified, discard the solution.
5.2 Anaphylactic and Hypersensitivity Reactions
Anaphylactic reactions, including shock were reported with the use of ERAXIS. If these reactions occur, ERAXIS should be discontinued and appropriate treatment administered [see Adverse Reactions (6)].
Infusion-related adverse reactions, possibly histamine-mediated, have been reported with ERAXIS, including rash, urticaria, flushing, pruritus, bronchospasm, dyspnea, and hypotension [see Adverse Reactions (6)]. To reduce occurrence of these reactions, do not exceed a rate of ERAXIS infusion of 1.1 mg/minute [see Dosage and Administration (2.4)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal carcinogenicity studies of anidulafungin have not been conducted.
Anidulafungin was not genotoxic in the following in vitro studies: bacterial reverse mutation assays, a chromosome aberration assay with Chinese hamster ovary cells, and a forward gene mutation assay with mouse lymphoma cells. Anidulafungin was not genotoxic in mice using the in vivo micronucleus assay.
Anidulafungin produced no adverse effects on fertility in male or female rats at intravenous doses of 20 mg/kg/day (equivalent to 2 times the proposed therapeutic maintenance dose of 100 mg/day on the basis of relative body surface area).
5.3 Risk of Neonatal Toxicity Associated With Polysorbates
ERAXIS contains polysorbate 80, an inactive ingredient. Thrombocytopenia, renal dysfunction, hepatomegaly, cholestasis, ascites, hypotension, and metabolic acidosis have been reported in low-birth weight infants receiving high doses of polysorbate. Polysorbate toxicity has not been reported with ERAXIS. ERAXIS is not approved in pediatric patients younger than 1 month of age [see Indications and Usage (1.1, 1.3), Use in Specific Populations (8.4)].
5.4 Risk in Patients With Hereditary Fructose Intolerance (hfi)
ERAXIS contains fructose, an inactive ingredient, and may precipitate a metabolic crisis that may include, but is not limited to life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure in patients with HFI. Obtain careful history of HFI symptoms (nausea, vomiting, abdominal pain) with fructose/sucrose exposure prior to ERAXIS administration because a diagnosis of HFI may not yet be established in pediatric patients [see Contraindications (4) and Use in Specific Populations (8.4)].
Structured Label Content
Section 42229-5 (42229-5)
Candidemia and other Candida infections (intra-abdominal abscess and peritonitis)
The recommended dose is a single 200 mg loading dose of ERAXIS on Day 1, followed by a 100 mg once daily maintenance dose thereafter. Duration of treatment should be based on the patient's clinical response. In general, antifungal therapy should continue for at least 14 days after the last positive culture.
1.4 Usage
Specimens for fungal culture and other relevant laboratory studies (including histopathology) should be obtained prior to therapy to isolate and identify causative organism(s). Therapy may be instituted before the results of the cultures and other laboratory studies are known. However, once these results become available, antifungal therapy should be adjusted accordingly.
7.4 Rifampin
Administration of multiple doses of anidulafungin and rifampin to patients resulted in no significant alteration in the steady state pharmacokinetics of anidulafungin. No dosage adjustment of anidulafungin is needed when it is co-administered with rifampin [see Clinical Pharmacology (12.3)].
10 Overdosage (10 OVERDOSAGE)
During clinical trials a single 400 mg dose of ERAXIS was inadvertently administered as a loading dose. No clinical adverse events were reported. In a study of 10 healthy subjects administered a loading dose of 260 mg followed by 130 mg daily; 3 of the 10 subjects experienced transient, asymptomatic transaminase elevations (≤3 × ULN) [see Warnings and Precautions (5.1)].
Anidulafungin is not dialyzable.
The maximum non-lethal dose of anidulafungin in rats was 50 mg/kg, a dose which is equivalent to 10 times the recommended daily dose for esophageal candidiasis (50 mg/day) or equivalent to 5 times the recommended daily dose for candidemia and other Candida infections (100 mg/day), based on relative body surface area comparisons.
11 Description (11 DESCRIPTION)
ERAXIS for Injection is a sterile, lyophilized product for intravenous (IV) infusion that contains anidulafungin. ERAXIS (anidulafungin) is a semi-synthetic lipopeptide synthesized from a fermentation product of Aspergillus nidulans. Anidulafungin is an echinocandin, a class of antifungal drugs that inhibits the synthesis of 1,3-β-D-glucan, an essential component of fungal cell walls.
ERAXIS (anidulafungin) is 1-[(4R,5R)-4,5-dihydroxy-N2-[[4"-(pentyloxy)[1,1':4',1"-terphenyl]-4-yl]carbonyl]-L-ornithine]echinocandin B. Anidulafungin is a white to off-white powder that is practically insoluble in water and slightly soluble in ethanol. In addition to the active ingredient, anidulafungin, ERAXIS for Injection contains the following inactive ingredients:
50 mg/vial – fructose (50 mg), mannitol (250 mg), polysorbate 80 (125 mg), tartaric acid (5.6 mg), and sodium hydroxide and/or hydrochloric acid for pH adjustment.
100 mg/vial – fructose (100 mg), mannitol (500 mg), polysorbate 80 (250 mg), tartaric acid (11.2 mg), and sodium hydroxide and/or hydrochloric acid for pH adjustment.
The empirical formula of anidulafungin is C58H73N7O17 and the formula weight is 1140.3.
The structural formula is:
Prior to administration, ERAXIS for Injection requires reconstitution with sterile Water for Injection and subsequent dilution with either 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP (normal saline).
7.3 Tacrolimus
Administration of multiple doses of anidulafungin and a single-dose of tacrolimus to healthy subjects resulted in no significant alteration in the steady state pharmacokinetics of either drug. No dosage adjustment of tacrolimus or anidulafungin is needed when the two drugs are co-administered [see Clinical Pharmacology (12.3)].
7.1 Cyclosporine
Administration of multiple doses of anidulafungin and cyclosporine to healthy subjects resulted in no significant alteration in the steady state pharmacokinetics of either drug. No dosage adjustment of cyclosporine or anidulafungin is needed when the two drugs are co-administered [see Clinical Pharmacology (12.3)].
7.2 Voriconazole
Administration of multiple doses of anidulafungin and voriconazole to healthy subjects resulted in no significant alteration in the steady state pharmacokinetics of either drug. No dosage adjustment of voriconazole or anidulafungin is needed when the two drugs are co-administered [see Clinical Pharmacology (12.3)].
16.1 How Supplied
ERAXIS (anidulafungin) for Injection is supplied in a single-dose vial of sterile, lyophilized, preservative-free, white to off-white powder. ERAXIS (anidulafungin) is available in the following packaging configuration:
|
Single-Dose Vial of ERAXIS 50 mg |
|
|
NDC 0049-0114-28 |
One - 50 mg vial |
|
Single-Dose Vial of ERAXIS 100 mg |
|
|
NDC 0049-0116-28 |
One - 100 mg vial |
8.4 Pediatric Use
The safety and effectiveness of ERAXIS for the treatment of candidemia and the following Candida infections: intra-abdominal abscess and peritonitis, have been established in pediatric patients 1 month of age and older. Use of ERAXIS for this indication in this age group is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic, safety data in pediatric patients 1 month of age and older [see Indications and Usage (1), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
The safety and effectiveness of ERAXIS in patients younger than 1 month of age with candidemia/invasive candidiasis has not been established.
Candidemia/invasive candidiasis in pediatric patients younger than 1 month of age has a higher rate of central nervous system (CNS) and multi-organ dissemination than in older patients. In addition, in patients younger than 1 month of age ERAXIS carries a potential risk of life-threatening toxicity associated with high doses of polysorbate 80, an inactive ingredient in ERAXIS [see Warnings and Precautions (5.3)].
The safety and effectiveness of ERAXIS in pediatric patients with esophageal candidiasis has not been established.
ERAXIS is contraindicated in adult and pediatric patients with HFI. Because a diagnosis of HFI may not yet be established in pediatric patients, obtain a careful history of HFI symptoms with fructose/sucrose exposure prior to administration of ERAXIS [see Warnings and Precautions (5.4)].
8.5 Geriatric Use
Of the total number of subjects (N = 197) in the pivotal clinical studies of anidulafungin, 35% were 65 years and over, while 18% were 75 years and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Dosage adjustments are not required for geriatric patients [see Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
ERAXIS is contraindicated in:
-
•Patients with known hypersensitivity to anidulafungin, any component of ERAXIS, or other echinocandins [see Warnings and Precautions (5.2)]
-
•Patients with known or suspected Hereditary Fructose Intolerance (HFI) [see Warnings and Precautions (5.4)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following most serious adverse reactions are described elsewhere in other labeling sections:
-
•Hepatic Adverse Reactions [see Warnings and Precautions (5.1)]
-
•Anaphylactic and Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
1.3 Limitations of Use
-
•ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida, and has not been studied in sufficient numbers of neutropenic patients to determine efficacy in this group. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established [see Warning and Precautions (5.3), Use in Specific Populations (8.4)].
-
•ERAXIS is associated with high relapse rates in esophageal candidiasis [see Clinical Studies (14.2)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ERAXIS is an echinocandin antifungal indicated for the treatment of the following infections:
-
•Candidemia and other forms of Candida infections (intra-abdominal abscess and peritonitis) in adults and pediatric patients (1 month of age and older) (1.1)
-
•Esophageal candidiasis in adults (1.2)
Limitations of use
-
•ERAXIS has not been studied in adult and pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida or in sufficient numbers of neutropenic patients. The dosage of ERAXIS for the treatment of Candida dissemination into the CNS and the eye has not been established. (1.3, 5.3, 8.4)
-
•ERAXIS is associated with high relapse rates in esophageal candidiasis. (1.3, 14.2)
8.7 Renal Insufficiency
Dosage adjustments are not required for patients with any degree of renal insufficiency including those on hemodialysis. Anidulafungin has negligible (<1%) renal clearance. In a clinical study of subjects with mild, moderate, severe or end stage (dialysis-dependent) renal insufficiency, anidulafungin pharmacokinetics were similar to those observed in subjects with normal renal function. Anidulafungin is not dialyzable and may be administered without regard to the timing of hemodialysis [see Clinical Pharmacology (12.3)].
12.1 Mechanism of Action
Anidulafungin is an anti-fungal drug [see Microbiology (12.4)].
8.6 Hepatic Insufficiency
No dosing adjustments are required for patients with any degree of hepatic insufficiency. Anidulafungin is not hepatically metabolized. Anidulafungin pharmacokinetics were examined in subjects with Child-Pugh class A, B or C hepatic insufficiency. Anidulafungin concentrations were not increased in subjects with any degree of hepatic insufficiency. Though a slight decrease in AUC was observed in patients with Child-Pugh C hepatic insufficiency, it was within the range of population estimates noted for healthy subjects [see Clinical Pharmacology (12.3)].
1.2 Esophageal Candidiasis
ERAXIS is indicated for the treatment of esophageal candidiasis in adults [see Indications and Usage (1.3), Clinical Studies (14.2)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Hepatic Effects: Risk of abnormal liver tests, hepatitis, hepatic failure; monitor hepatic function during therapy. (5.1, 13.2)
-
•Hypersensitivity: Anaphylaxis, including shock has been reported. Risk of infusion-related adverse reactions, possibly histamine-mediated, including rash, urticaria, flushing, pruritus, bronchospasm, dyspnea, and hypotension; to reduce occurrence, do not exceed a rate of infusion of 1.1 mg/minute. (2.4, 5.2)
-
•Risk of Neonatal Toxicity Associated with Polysorbates: ERAXIS contains polysorbate 80, an inactive ingredient. Thrombocytopenia, renal dysfunction, hepatomegaly, cholestasis, ascites, hypotension and metabolic acidosis haves been reported in low-birth weight infants receiving high doses of polysorbate. ERAXIS is not approved in pediatric patients younger than 1 month of age. (5.3, 8.4)
-
•Hereditary Fructose Intolerance (HFI): ERAXIS contains fructose. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before ERAXIS administration. (5.4, 8.4)
14.2 Esophageal Candidiasis
ERAXIS was evaluated in a double-blind, double-dummy, randomized Phase 3 study. Three hundred patients received ERAXIS (100 mg loading dose IV on Day 1 followed by 50 mg/day IV) and 301 received oral fluconazole (200 mg loading dose on Day 1 followed by 100 mg/day). Treatment duration was 7 days beyond resolution of symptoms for a minimum of 14 and a maximum of 21 days.
Of the 442 patients with culture confirmed esophageal candidiasis, most patients (91%) had C. albicans isolated at the baseline.
Treatment groups were similar in demographic and other baseline characteristics. In ERAXIS-treated patients, the age range was 16–69 years, the gender distribution was 42% male and 58% female, and the race distribution was 15% White, 49% Black/African American, 15% Asian, 0.3 % Hispanic, 21% other races.
In this study, of 280 patients tested, 237 (84.6%) tested HIV positive. In both groups the median time to resolution of symptoms was 5 days and the median duration of therapy was 14 days.
Efficacy was assessed by endoscopic outcome at end of therapy (EOT). Patients were considered clinically evaluable if they received at least 10 days of therapy, had an EOT assessment with a clinical outcome other than 'indeterminate', had an endoscopy at EOT, and did not have any protocol violations prior to the EOT visit that would affect an assessment of efficacy.
An endoscopic success, defined as cure (endoscopic grade of 0 on a 4-point severity scale) or improvement (decrease of one or more grades from baseline), was seen in 225/231 (97.4%) ERAXIS-treated patients and 233/236 (98.7%) fluconazole-treated patients (Table 13). The majority of these patients were endoscopic cures (grade=0). Two weeks after completing therapy, the ERAXIS group had significantly more endoscopically-documented relapses than the fluconazole group, 120/225 (53.3%) vs. 45/233 (19.3%), respectively (Table 13).
|
Endoscopic Response at End of Therapy |
||||
|
Response |
ERAXIS
|
Fluconazole
|
Treatment Difference Calculated as ERAXIS minus fluconazole
|
95% CI |
|
Endoscopic Success, n (%) |
225 (97.4) |
233 (98.7) |
-1.3% |
-3.8%, 1.2% |
|
Cure |
204 (88.3) |
221 (93.6) |
||
|
Improvement |
21 (9.1) |
12 (5.1) |
||
|
Failure, n (%) |
6 (2.6) |
3 (1.3) |
||
|
|
||||
|
Endoscopic Relapse Rates at Follow-Up, 2 Weeks Post-Treatment |
||||
|
ERAXIS |
Fluconazole |
Treatment Difference |
95% CI |
|
|
Endoscopic Relapse, n/N (%) |
120/225 (53.3%) |
45/233 (19.3%) |
34.0% |
25.8%, 42.3% |
Clinical success (cure or improvement in clinical symptoms including odynophagia/dysphagia and retrosternal pain) occurred in 229/231 (99.1%) of the ERAXIS-treated patients and 235/236 (99.6%) of the fluconazole-treated patients at the end of therapy. For patients with C. albicans, microbiological success occurred in 142/162 (87.7%) of the ERAXIS-treated group and 157/166 (94.6%) of the fluconazole-treated group at the end of therapy. For patients with Candida species other than C. albicans, success occurred in 10/12 (83.3%) of the ERAXIS-treated group and 14/16 (87.5%) of the fluconazole-treated group.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
|
Adults |
Pediatric Patients 1 Month of Age and Older |
|
Candidemia and other forms of Candida infections |
|
|
200 mg loading dose on Day 1, followed by 100 mg once daily maintenance dose thereafter for at least 14 days after the last positive culture (2.1) |
3 mg/kg (not to exceed 200 mg) loading dose on Day 1, followed by 1.5 mg/kg (not to exceed 100 mg) once daily maintenance dose thereafter for at least 14 days after the last positive culture (2.2) |
|
Esophageal candidiasis |
|
|
100 mg loading dose on Day 1, followed by 50 mg once daily maintenance dose thereafter for a minimum of 14 days and for at least 7 days following resolution of symptoms (2.1) |
Not Approved |
Rate of Infusion for Adults and Pediatric Patients
The rate of infusion should not exceed 1.1 mg/minute [equivalent to 1.4 mL/minute or 84 mL/hour when reconstituted and diluted per instructions] (2.3, 2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 50 mg, and 100 mg of anidulafungin as a white to off-white sterile lyophilized powder in a single-dose vial for reconstitution.
5.1 Hepatic Adverse Reactions
Laboratory abnormalities in liver tests have been seen in healthy volunteers and pediatric patients treated with ERAXIS. In some patients with serious underlying medical conditions who were receiving multiple concomitant medications along with ERAXIS, clinically significant hepatic abnormalities have occurred. Isolated cases of significant hepatic dysfunction, hepatitis, or hepatic failure have been reported in patients; a causal relationship to ERAXIS has not been established [see Adverse Reactions (6.1) and Nonclinical Toxicology (13.2)]. Patients who develop abnormal liver tests during ERAXIS therapy should be monitored for evidence of worsening hepatic tests and evaluated for risk/benefit of continuing ERAXIS therapy.
6.2 Post Marketing Experience (6.2 Post-marketing Experience)
The following adverse reactions have been identified during post approval use of anidulafungin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune: Anaphylactic shock, anaphylactic reaction, bronchospasm [see Warnings and Precautions (5.2)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.3 Preparation for Administration
ERAXIS for Injection must be reconstituted with sterile Water for Injection and subsequently diluted only with 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP (normal saline), prior to administration.
The compatibility of reconstituted ERAXIS with intravenous substances, additives, or medications other than 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP (normal saline) has not been established. Do NOT dilute with other solutions or co-infuse with other medications or electrolytes. The infusion solution must not be frozen.
1.1 Candidemia and Other Forms of Candida
ERAXIS is indicated for the treatment of candidemia and the following Candida infections: intra-abdominal abscess and peritonitis in adults and pediatric patients 1 month of age and older [see Clinical Studies (14.1) and Microbiology (12.4)].
7.5 Amphotericin B Liposome for Injection
Administration of multiple doses of anidulafungin and liposomal amphotericin B to patients resulted in no significant alteration in the steady state pharmacokinetics of anidulafungin. No dosage adjustment of anidulafungin is needed when it is co-administered with liposomal amphotericin B [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In 3 month studies, liver toxicity, including single cell hepatocellular necrosis, hepatocellular hypertrophy and increased liver weights were observed in monkeys and rats at doses equivalent to 5–6 times human exposure. For both species, hepatocellular hypertrophy was still noted one month after the end of dosing.
Principal Display Panel 50 Mg Vial Label (PRINCIPAL DISPLAY PANEL - 50 mg Vial Label)
1 Sterile Single-dose Vial
Eraxis
®
(anidulafungin)
for Injection
50 mg/vial
For Intravenous Infusion Only
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
Principal Display Panel 100 Mg Vial Label (PRINCIPAL DISPLAY PANEL - 100 mg Vial Label)
1 Sterile Single-dose Vial
Eraxis
®
(anidulafungin)
for Injection
100 mg/vial
For Intravenous Infusion Only
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
Principal Display Panel 50 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 50 mg Vial Carton)
NDC 0049-0114-28
Unit Pack Contains:
1 Sterile Single-dose Vial
Eraxis
®
50 mg
Eraxis
®
(anidulafungin)
for Injection
50 mg/vial
For Intravenous Infusion Only
Store Vial in Refrigerator
at 2° C - 8° C (36° F - 46° F).
Do Not Freeze.
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
Principal Display Panel 100 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 100 mg Vial Carton)
NDC 0049-0116-28
Unit Pack Contains:
1 Sterile Single-dose Vial
Eraxis
®
100 mg
Eraxis
®
(anidulafungin)
for Injection
100 mg/vial
For Intravenous Infusion Only
Store Vial in Refrigerator
at 2° C - 8° C (36° F - 46° F).
Do Not Freeze.
Pfizer
Hospital
Distributed by
Roerig
Division of Pfizer Inc.
New York, NY 10001
Rx only
2.4 Dilution and Administration of the Infusion
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If particulate matter or discoloration is identified, discard the solution.
5.2 Anaphylactic and Hypersensitivity Reactions
Anaphylactic reactions, including shock were reported with the use of ERAXIS. If these reactions occur, ERAXIS should be discontinued and appropriate treatment administered [see Adverse Reactions (6)].
Infusion-related adverse reactions, possibly histamine-mediated, have been reported with ERAXIS, including rash, urticaria, flushing, pruritus, bronchospasm, dyspnea, and hypotension [see Adverse Reactions (6)]. To reduce occurrence of these reactions, do not exceed a rate of ERAXIS infusion of 1.1 mg/minute [see Dosage and Administration (2.4)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal carcinogenicity studies of anidulafungin have not been conducted.
Anidulafungin was not genotoxic in the following in vitro studies: bacterial reverse mutation assays, a chromosome aberration assay with Chinese hamster ovary cells, and a forward gene mutation assay with mouse lymphoma cells. Anidulafungin was not genotoxic in mice using the in vivo micronucleus assay.
Anidulafungin produced no adverse effects on fertility in male or female rats at intravenous doses of 20 mg/kg/day (equivalent to 2 times the proposed therapeutic maintenance dose of 100 mg/day on the basis of relative body surface area).
5.3 Risk of Neonatal Toxicity Associated With Polysorbates (5.3 Risk of Neonatal Toxicity Associated with Polysorbates)
ERAXIS contains polysorbate 80, an inactive ingredient. Thrombocytopenia, renal dysfunction, hepatomegaly, cholestasis, ascites, hypotension, and metabolic acidosis have been reported in low-birth weight infants receiving high doses of polysorbate. Polysorbate toxicity has not been reported with ERAXIS. ERAXIS is not approved in pediatric patients younger than 1 month of age [see Indications and Usage (1.1, 1.3), Use in Specific Populations (8.4)].
5.4 Risk in Patients With Hereditary Fructose Intolerance (hfi) (5.4 Risk in Patients with Hereditary Fructose Intolerance (HFI))
ERAXIS contains fructose, an inactive ingredient, and may precipitate a metabolic crisis that may include, but is not limited to life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure in patients with HFI. Obtain careful history of HFI symptoms (nausea, vomiting, abdominal pain) with fructose/sucrose exposure prior to ERAXIS administration because a diagnosis of HFI may not yet be established in pediatric patients [see Contraindications (4) and Use in Specific Populations (8.4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:02.356609 · Updated: 2026-03-14T22:31:45.401488