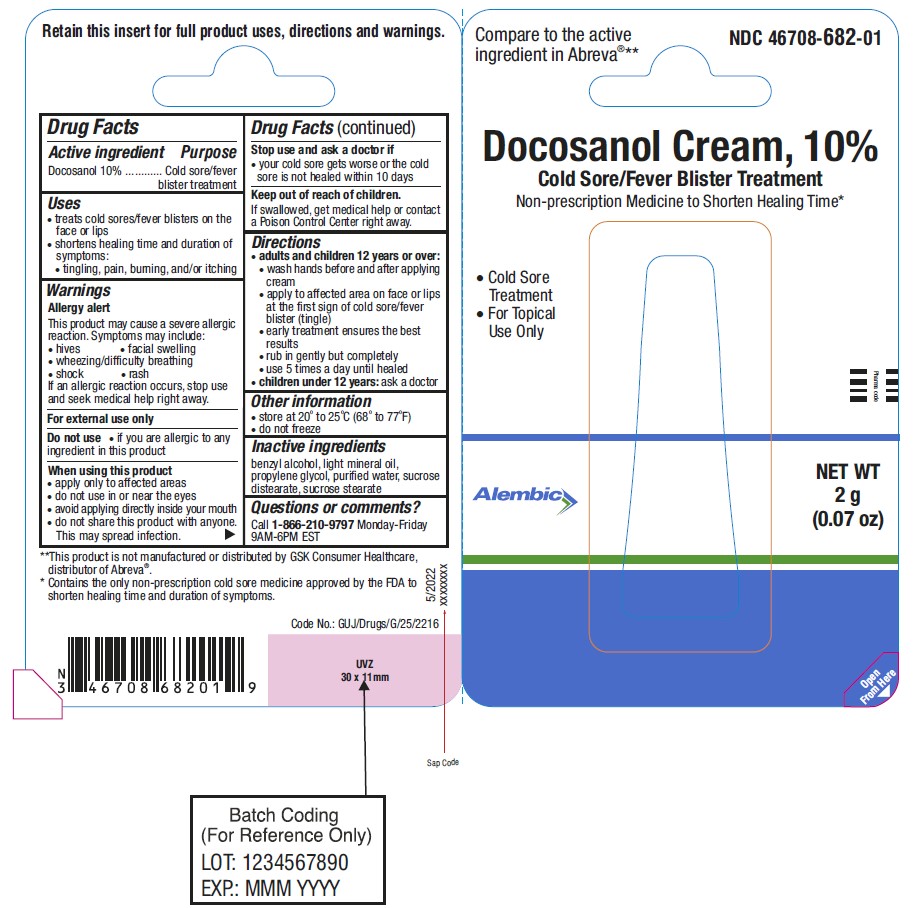
Docosanol Cream, 10%
a87a077d-9b0e-490d-b4ac-26c7045828db
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Docosanol 10%
Purpose
Cold sore/fever blister treatment
Medication Information
Purpose
Cold sore/fever blister treatment
Description
Docosanol 10%
Uses
- treats cold sores/fever blisters on the face or lips
- shortens healing time and duration of symptoms:
• tingling, pain, burning, and/or itching
Warnings
Allergy Alert
This product may cause a severe allergic reaction. Symptoms may include:
- hives
- facial swelling
- wheezing/difficulty breathing
- shock
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
For external use only
Do not use
- if you are allergic to any ingredient in this product
When using this product
- apply only to the affected areas
- do not use in or near the eyes
- avoid applying directly inside your mouth
- do not share this product with anyone. This may spread the infection.
Stop use and ask a doctor if
- your cold sore gets worse or the cold sore is not healed within 10 days
Directions
- adults and children 12 years or over:
• wash hands before and after applying cream
• apply to affected area on face or lips at the first sign of cold sore/fever blister (tingle)
• early treatment ensures the best results
• rub in gently but completely
• use 5 times a day until healed
- children under 12 years:ask a doctor
Active Ingredient
Docosanol 10%
Other Information
- store at 20° to 25°C (68° to 77°F)
- do not freeze
Inactive Ingredients
benzyl alcohol, light mineral oil, propylene glycol, purified water, sucrose distearate, sucrose stearate
Questions Or Comments?
Call 1-866-210-9797 Monday-Friday 9AM-6PM EST
Principal Display Panel
Tube Pack
NDC 46708-682-01
Docosanol Cream, 10%
Compare to the active ingredient in Abreva® **
Cold Sore/Fever Blister Treatment
Non-prescription Medicine to Shorten Healing Time*
Cold Sore Treatment
For Topical Use Only
NET WT 2g (0.07 oz)
**This product is not manufactured or distributed by GSK Consumer Healthcare, distributor of Abreva®.
*Contains the only non-prescription cold sore medicine approved by the FDA to shorten healing time and duration of symptoms.
RETAIN THIS INSERT FOR FULL PRODUCT USES, DIRECTIONS AND WARNINGS
Pump Pack
NDC 46708-682-03
Docosanol Cream, 10%
Compare to the active ingredient in Abreva® **
Cold Sore/Fever Blister Treatment
Non-prescription Medicine to Shorten Healing Time*
Cold Sore Treatment
For Topical Use Only
NET WT 2g (0.07 oz)
**This product is not manufactured or distributed by GSK Consumer Healthcare, distributor of Abreva®.
*Contains the only non-prescription cold sore medicine approved by the FDA to shorten healing time and duration of symptoms.
RETAIN THIS INSERT FOR FULL PRODUCT USES, DIRECTIONS AND WARNINGS
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
- treats cold sores/fever blisters on the face or lips
- shortens healing time and duration of symptoms:
• tingling, pain, burning, and/or itching
Purpose
Cold sore/fever blister treatment
Warnings
Allergy Alert
This product may cause a severe allergic reaction. Symptoms may include:
- hives
- facial swelling
- wheezing/difficulty breathing
- shock
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
For external use only
Do not use
- if you are allergic to any ingredient in this product
When using this product
- apply only to the affected areas
- do not use in or near the eyes
- avoid applying directly inside your mouth
- do not share this product with anyone. This may spread the infection.
Stop use and ask a doctor if
- your cold sore gets worse or the cold sore is not healed within 10 days
Directions
- adults and children 12 years or over:
• wash hands before and after applying cream
• apply to affected area on face or lips at the first sign of cold sore/fever blister (tingle)
• early treatment ensures the best results
• rub in gently but completely
• use 5 times a day until healed
- children under 12 years:ask a doctor
Active Ingredient (Active ingredient)
Docosanol 10%
Other Information (Other information)
- store at 20° to 25°C (68° to 77°F)
- do not freeze
Inactive Ingredients (Inactive ingredients)
benzyl alcohol, light mineral oil, propylene glycol, purified water, sucrose distearate, sucrose stearate
Questions Or Comments? (Questions or comments?)
Call 1-866-210-9797 Monday-Friday 9AM-6PM EST
Principal Display Panel
Tube Pack
NDC 46708-682-01
Docosanol Cream, 10%
Compare to the active ingredient in Abreva® **
Cold Sore/Fever Blister Treatment
Non-prescription Medicine to Shorten Healing Time*
Cold Sore Treatment
For Topical Use Only
NET WT 2g (0.07 oz)
**This product is not manufactured or distributed by GSK Consumer Healthcare, distributor of Abreva®.
*Contains the only non-prescription cold sore medicine approved by the FDA to shorten healing time and duration of symptoms.
RETAIN THIS INSERT FOR FULL PRODUCT USES, DIRECTIONS AND WARNINGS
Pump Pack
NDC 46708-682-03
Docosanol Cream, 10%
Compare to the active ingredient in Abreva® **
Cold Sore/Fever Blister Treatment
Non-prescription Medicine to Shorten Healing Time*
Cold Sore Treatment
For Topical Use Only
NET WT 2g (0.07 oz)
**This product is not manufactured or distributed by GSK Consumer Healthcare, distributor of Abreva®.
*Contains the only non-prescription cold sore medicine approved by the FDA to shorten healing time and duration of symptoms.
RETAIN THIS INSERT FOR FULL PRODUCT USES, DIRECTIONS AND WARNINGS
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:05.486574 · Updated: 2026-03-14T23:08:40.140455