Emetrol
a8469128-592a-4682-82c7-4d2a6f97a998
34390-5
HUMAN OTC DRUG LABEL
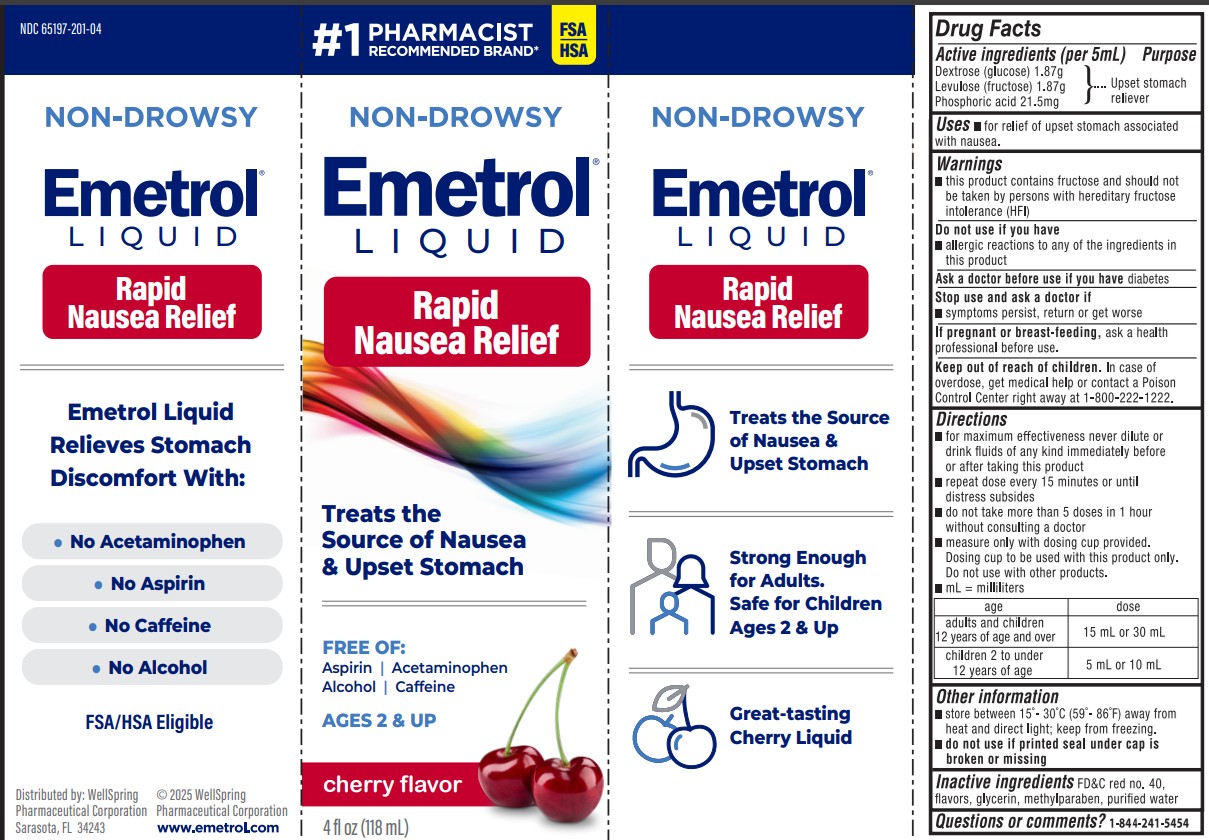
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Phosphorated carbohydrate solution* *each 5 mL contains: 1.87 g Dextrose (glucose) 1.87 g Levulose (fructose) 21.5 mg Phosphoric acid
Purpose
Upset Stomach Reliever
Medication Information
Purpose
Upset Stomach Reliever
Description
Phosphorated carbohydrate solution* *each 5 mL contains: 1.87 g Dextrose (glucose) 1.87 g Levulose (fructose) 21.5 mg Phosphoric acid
Uses
For relief of upset stomach associated with nausea due to overindulgence in food and drink.
Warnings
- This product contains fructose and should not be taken by persons with hereditary fructose intolerance (HFI).
Directions
- for maximum effectiveness never dilute or drink fluids of any kind immediately before or after taking this product
- adults and children 12 years of age and over: one to two tablespoons
- children 2 to under 12: one or two teaspoons
- repeat dose every 15 minutes or until distress subsides
- do not take more than 5 doses in 1 hour without consulting a doctor
- measure only with dosing cup provided. Dosing cup to be used with Emetrol only. Do not use with other products.
Package Label
Emetrol Loquid Rapid Nausea Relief
Treats the source of Nausea & upset stomach
Distributed By
WellSpring Pharmaceutical Corporation
Sarasota, FL 34243 USA
(c) WellSpring 2019
Other Information
- Store between 20-25ºC (68-77ºF) away from heat and direct light; keep from freezing
- Do not use if printed foil seal under bottle cap is broken or missing
Active Ingredients
Phosphorated carbohydrate solution*
*each 5 mL contains:
- 1.87 g Dextrose (glucose)
- 1.87 g Levulose (fructose)
- 21.5 mg Phosphoric acid
Inactive Ingredients
FD&C red no. 40, flavors, glycerin, methylparaben, and purified water.
Do Not Use If You Have
- allergic reactions to any of the ingredients in this product
Questions Or Comments?
call 1-844-241-5454
Stop Use and Ask A Doctor If
- symptoms persist, return or get worse
If Pregnant Or Breast Feeding
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Ask A Doctor Before Use If You Have
- diabetes
Structured Label Content
Uses
For relief of upset stomach associated with nausea due to overindulgence in food and drink.
Purpose
Upset Stomach Reliever
Warnings
- This product contains fructose and should not be taken by persons with hereditary fructose intolerance (HFI).
Directions
- for maximum effectiveness never dilute or drink fluids of any kind immediately before or after taking this product
- adults and children 12 years of age and over: one to two tablespoons
- children 2 to under 12: one or two teaspoons
- repeat dose every 15 minutes or until distress subsides
- do not take more than 5 doses in 1 hour without consulting a doctor
- measure only with dosing cup provided. Dosing cup to be used with Emetrol only. Do not use with other products.
Package Label (PACKAGE LABEL)
Emetrol Loquid Rapid Nausea Relief
Treats the source of Nausea & upset stomach
Distributed By
WellSpring Pharmaceutical Corporation
Sarasota, FL 34243 USA
(c) WellSpring 2019
Other Information (Other information)
- Store between 20-25ºC (68-77ºF) away from heat and direct light; keep from freezing
- Do not use if printed foil seal under bottle cap is broken or missing
Active Ingredients (Active ingredients)
Phosphorated carbohydrate solution*
*each 5 mL contains:
- 1.87 g Dextrose (glucose)
- 1.87 g Levulose (fructose)
- 21.5 mg Phosphoric acid
Inactive Ingredients (Inactive ingredients)
FD&C red no. 40, flavors, glycerin, methylparaben, and purified water.
Do Not Use If You Have (Do not use if you have)
- allergic reactions to any of the ingredients in this product
Questions Or Comments? (Questions or Comments?)
call 1-844-241-5454
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- symptoms persist, return or get worse
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- diabetes
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:17.097699 · Updated: 2026-03-14T23:08:57.795400