Dianeal Low Calcium With Dextrose
a829d4eb-3ec5-453c-b270-793969af15b3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
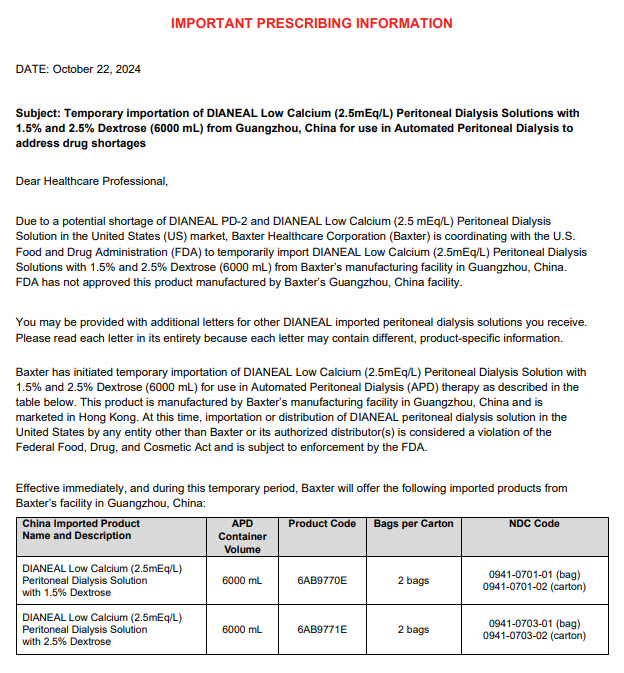
6AB9770E 6000ml (APPROX 225ml EXCESS) Baxter Logo Dianeal ® Low Calcium(2.5mEq/L) Peritoneal Dialysis Solution With 1.5% Dextrose EACH 100ml CONTAINS 1.5 g DEXTROSE HYDROUS USP 538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE 18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM CHLORIDE USP pH 5.2 (4.5 TO 6.5) mEq/L SODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5 CHLORIDE 95 LACTATE 40 OSMOLARITY 344 mOsmol/L(CALC) STERILE NON PYROGENIC POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE DIRECTION OF A PHYSICIAN READ PACKAGE INSERT FOR FULL INFORMATION FOR INTRAPERITONEAL ADMINISTRATION ONLY CAUTIONS SQUEEZE AND INSPECT INNER BAG WHICH MAINTAINS PRODUCT STERILITY DISCARD IF LEAKS ARE FOUND DO NOT USE UNLESS SOLUTION IS CLEAR DISCARD UNUSED PORTION STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM TEMPERATURE (UNDER 25°C) UNTIL READY TO USE AVOID EXCESSIVE HEAT SEE INSERT Ambu-Flex™ III CONTAINER PL-146 ® PLASTIC MANUFACTURED BY BAXTER HEALTHCARE (GUANGZHOU) CO LTD GUANGZHOU CHINA (AN AFFI LIATE OF BAXTER WORLD TRADE INC USA) HK-42530 DIRECTIONS TO BE USED AS DIRECTED BY THE PHYSICIAN Prescription Drug Manufacturer Address: Jiaoyuan Road, Dongji Industrial District, GETDD,Guangzhou, P.R. China Low Calcium 1.5% Dextrose 1.5 LOW CALCIUM WITH 1.5% DEXTROSE 6000mlX2 LOT G00000000 EXP JAN 00 6AB9770E S/N 0000 6AB9771E 6000ml (APPROX 225ml EXCESS) Baxter Logo Dianeal ® Low Calcium(2.5mEq/L) Peritoneal Dialysis Solution With 2.5% Dextrose EACH 100ml CONTAINS 2.5 g DEXTROSE HYDROUS USP 538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE 18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM CHLORIDE USP pH 5.2 (4.5 TO 6.5) mEq/L SODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5 CHLORIDE 95 LACTATE 40 OSMOLARITY 395 mOsmol/L(CALC) STERILE NON PYROGENIC POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE DIRECTION OF A PHYSICIAN READ PACKAGE INSERT FOR FULL INFORMATION FOR INTRAPERITONEAL ADMINISTRATION ONLY CAUTIONS SQUEEZE AND INSPECT INNER BAG WHICH MAINTAINS PRODUCT STERILITY DISCARD IF LEAKS ARE FOUND DO NOT USE UNLESS SOLUTION IS CLEAR DISCARD UNUSED PORTION STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM TEMPERATURE (UNDER 25°C) UNTIL READY TO USE AVOID EXCESSIVE HEAT SEE INSERT Ambu-Flex™ III CONTAINER PL-146 ® PLASTIC MANUFACTURED BY BAXTER HEALTHCARE (GUANGZHOU) CO LTD GUANGZHOU CHINA (AN AFFI LIATE OF BAXTER WORLD TRADE INC USA) HK-42531 DIRECTIONS TO BE USED AS DIRECTED BY THE PHYSICIAN Prescription Drug Manufacturer Address: Jiaoyuan Road, Dongji Industrial District, GETDD,Guangzhou, P.R. China Low Calcium 1.5% Dextrose 2.5 LOW CALCIUM WITH 2.5% DEXTROSE 6000mlX2 LOT G00000000 EXP JAN 00 6AB9771E S/N 0000
Medication Information
Description
6AB9770E 6000ml (APPROX 225ml EXCESS) Baxter Logo Dianeal ® Low Calcium(2.5mEq/L) Peritoneal Dialysis Solution With 1.5% Dextrose EACH 100ml CONTAINS 1.5 g DEXTROSE HYDROUS USP 538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE 18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM CHLORIDE USP pH 5.2 (4.5 TO 6.5) mEq/L SODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5 CHLORIDE 95 LACTATE 40 OSMOLARITY 344 mOsmol/L(CALC) STERILE NON PYROGENIC POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE DIRECTION OF A PHYSICIAN READ PACKAGE INSERT FOR FULL INFORMATION FOR INTRAPERITONEAL ADMINISTRATION ONLY CAUTIONS SQUEEZE AND INSPECT INNER BAG WHICH MAINTAINS PRODUCT STERILITY DISCARD IF LEAKS ARE FOUND DO NOT USE UNLESS SOLUTION IS CLEAR DISCARD UNUSED PORTION STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM TEMPERATURE (UNDER 25°C) UNTIL READY TO USE AVOID EXCESSIVE HEAT SEE INSERT Ambu-Flex™ III CONTAINER PL-146 ® PLASTIC MANUFACTURED BY BAXTER HEALTHCARE (GUANGZHOU) CO LTD GUANGZHOU CHINA (AN AFFI LIATE OF BAXTER WORLD TRADE INC USA) HK-42530 DIRECTIONS TO BE USED AS DIRECTED BY THE PHYSICIAN Prescription Drug Manufacturer Address: Jiaoyuan Road, Dongji Industrial District, GETDD,Guangzhou, P.R. China Low Calcium 1.5% Dextrose 1.5 LOW CALCIUM WITH 1.5% DEXTROSE 6000mlX2 LOT G00000000 EXP JAN 00 6AB9770E S/N 0000 6AB9771E 6000ml (APPROX 225ml EXCESS) Baxter Logo Dianeal ® Low Calcium(2.5mEq/L) Peritoneal Dialysis Solution With 2.5% Dextrose EACH 100ml CONTAINS 2.5 g DEXTROSE HYDROUS USP 538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE 18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM CHLORIDE USP pH 5.2 (4.5 TO 6.5) mEq/L SODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5 CHLORIDE 95 LACTATE 40 OSMOLARITY 395 mOsmol/L(CALC) STERILE NON PYROGENIC POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE DIRECTION OF A PHYSICIAN READ PACKAGE INSERT FOR FULL INFORMATION FOR INTRAPERITONEAL ADMINISTRATION ONLY CAUTIONS SQUEEZE AND INSPECT INNER BAG WHICH MAINTAINS PRODUCT STERILITY DISCARD IF LEAKS ARE FOUND DO NOT USE UNLESS SOLUTION IS CLEAR DISCARD UNUSED PORTION STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM TEMPERATURE (UNDER 25°C) UNTIL READY TO USE AVOID EXCESSIVE HEAT SEE INSERT Ambu-Flex™ III CONTAINER PL-146 ® PLASTIC MANUFACTURED BY BAXTER HEALTHCARE (GUANGZHOU) CO LTD GUANGZHOU CHINA (AN AFFI LIATE OF BAXTER WORLD TRADE INC USA) HK-42531 DIRECTIONS TO BE USED AS DIRECTED BY THE PHYSICIAN Prescription Drug Manufacturer Address: Jiaoyuan Road, Dongji Industrial District, GETDD,Guangzhou, P.R. China Low Calcium 1.5% Dextrose 2.5 LOW CALCIUM WITH 2.5% DEXTROSE 6000mlX2 LOT G00000000 EXP JAN 00 6AB9771E S/N 0000
Health Care Provider Letter
Package/label Principal Display Panel
6AB9770E 6000ml
(APPROX 225ml EXCESS)
BaxterLogo
Dianeal
®Low Calcium(2.5mEq/L)
Peritoneal Dialysis Solution
With 1.5% Dextrose
EACH 100ml CONTAINS1.5 g DEXTROSE HYDROUS USP
538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE
18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM
CHLORIDE USP
pH5.2 (4.5 TO 6.5)
mEq/LSODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5
CHLORIDE 95 LACTATE 40
OSMOLARITY344 mOsmol/L(CALC)
STERILE NON PYROGENIC
POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE
DIRECTION OF A PHYSICIAN
READ PACKAGE INSERT FOR FULL INFORMATION
FOR INTRAPERITONEAL ADMINISTRATION ONLY
CAUTIONSSQUEEZE AND INSPECT INNER BAG WHICH
MAINTAINS PRODUCT STERILITY DISCARD IF
LEAKS ARE FOUND
DO NOT USE UNLESS SOLUTION IS CLEAR
DISCARD UNUSED PORTION
STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM
TEMPERATURE (UNDER 25°C) UNTIL READY TO USE
AVOID EXCESSIVE HEAT SEE INSERT
Ambu-Flex™ IIICONTAINER PL-146 ®PLASTIC
MANUFACTURED BY
BAXTER HEALTHCARE (GUANGZHOU) CO LTD
GUANGZHOU CHINA
(AN AFFI LIATE OF BAXTER WORLD TRADE INC USA)
HK-42530
DIRECTIONSTO BE USED AS DIRECTED BY THE PHYSICIAN
Prescription Drug
Manufacturer Address:
Jiaoyuan Road, Dongji Industrial District,
GETDD,Guangzhou, P.R. China
Low Calcium 1.5% Dextrose
1.5 LOW CALCIUM WITH 1.5% DEXTROSE 6000mlX2
LOT G00000000 EXP JAN 00 6AB9770E S/N 0000
6AB9771E 6000ml
(APPROX 225ml EXCESS)
BaxterLogo
Dianeal
®Low Calcium(2.5mEq/L)
Peritoneal Dialysis Solution
With 2.5% Dextrose
EACH 100ml CONTAINS2.5 g DEXTROSE HYDROUS USP
538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE
18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM
CHLORIDE USP
pH5.2 (4.5 TO 6.5)
mEq/LSODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5
CHLORIDE 95 LACTATE 40
OSMOLARITY395 mOsmol/L(CALC)
STERILE NON PYROGENIC
POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE
DIRECTION OF A PHYSICIAN
READ PACKAGE INSERT FOR FULL INFORMATION
FOR INTRAPERITONEAL ADMINISTRATION ONLY
CAUTIONSSQUEEZE AND INSPECT INNER BAG WHICH
MAINTAINS PRODUCT STERILITY DISCARD IF
LEAKS ARE FOUND
DO NOT USE UNLESS SOLUTION IS CLEAR
DISCARD UNUSED PORTION
STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM
TEMPERATURE (UNDER 25°C) UNTIL READY TO USE
AVOID EXCESSIVE HEAT SEE INSERT
Ambu-Flex™ IIICONTAINER PL-146 ®PLASTIC
MANUFACTURED BY
BAXTER HEALTHCARE (GUANGZHOU) CO LTD
GUANGZHOU CHINA
(AN AFFI LIATE OF BAXTER WORLD TRADE INC USA)
HK-42531
DIRECTIONSTO BE USED AS DIRECTED BY THE PHYSICIAN
Prescription Drug
Manufacturer Address:
Jiaoyuan Road, Dongji Industrial District,
GETDD,Guangzhou, P.R. China
Low Calcium 1.5% Dextrose
2.5 LOW CALCIUM WITH 2.5% DEXTROSE 6000mlX2
LOT G00000000 EXP JAN 00 6AB9771E S/N 0000
Structured Label Content
Health Care Provider Letter
Package/label Principal Display Panel (PACKAGE/LABEL PRINCIPAL DISPLAY PANEL)
6AB9770E 6000ml
(APPROX 225ml EXCESS)
BaxterLogo
Dianeal
®Low Calcium(2.5mEq/L)
Peritoneal Dialysis Solution
With 1.5% Dextrose
EACH 100ml CONTAINS1.5 g DEXTROSE HYDROUS USP
538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE
18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM
CHLORIDE USP
pH5.2 (4.5 TO 6.5)
mEq/LSODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5
CHLORIDE 95 LACTATE 40
OSMOLARITY344 mOsmol/L(CALC)
STERILE NON PYROGENIC
POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE
DIRECTION OF A PHYSICIAN
READ PACKAGE INSERT FOR FULL INFORMATION
FOR INTRAPERITONEAL ADMINISTRATION ONLY
CAUTIONSSQUEEZE AND INSPECT INNER BAG WHICH
MAINTAINS PRODUCT STERILITY DISCARD IF
LEAKS ARE FOUND
DO NOT USE UNLESS SOLUTION IS CLEAR
DISCARD UNUSED PORTION
STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM
TEMPERATURE (UNDER 25°C) UNTIL READY TO USE
AVOID EXCESSIVE HEAT SEE INSERT
Ambu-Flex™ IIICONTAINER PL-146 ®PLASTIC
MANUFACTURED BY
BAXTER HEALTHCARE (GUANGZHOU) CO LTD
GUANGZHOU CHINA
(AN AFFI LIATE OF BAXTER WORLD TRADE INC USA)
HK-42530
DIRECTIONSTO BE USED AS DIRECTED BY THE PHYSICIAN
Prescription Drug
Manufacturer Address:
Jiaoyuan Road, Dongji Industrial District,
GETDD,Guangzhou, P.R. China
Low Calcium 1.5% Dextrose
1.5 LOW CALCIUM WITH 1.5% DEXTROSE 6000mlX2
LOT G00000000 EXP JAN 00 6AB9770E S/N 0000
6AB9771E 6000ml
(APPROX 225ml EXCESS)
BaxterLogo
Dianeal
®Low Calcium(2.5mEq/L)
Peritoneal Dialysis Solution
With 2.5% Dextrose
EACH 100ml CONTAINS2.5 g DEXTROSE HYDROUS USP
538 mg SODIUM CHLORIDE USP 448 mg SODIUM LACTATE
18.3 mg CALCIUM CHLORIDE USP 5.08 mg MAGNESIUM
CHLORIDE USP
pH5.2 (4.5 TO 6.5)
mEq/LSODIUM 132 CALCIUM 2.5 MAGNESIUM 0.5
CHLORIDE 95 LACTATE 40
OSMOLARITY395 mOsmol/L(CALC)
STERILE NON PYROGENIC
POTASSIUM CHLORIDE TO BE ADDED ONLY UNDER THE
DIRECTION OF A PHYSICIAN
READ PACKAGE INSERT FOR FULL INFORMATION
FOR INTRAPERITONEAL ADMINISTRATION ONLY
CAUTIONSSQUEEZE AND INSPECT INNER BAG WHICH
MAINTAINS PRODUCT STERILITY DISCARD IF
LEAKS ARE FOUND
DO NOT USE UNLESS SOLUTION IS CLEAR
DISCARD UNUSED PORTION
STORE UNIT IN MOISTURE BARRIER OVERWRAP AT ROOM
TEMPERATURE (UNDER 25°C) UNTIL READY TO USE
AVOID EXCESSIVE HEAT SEE INSERT
Ambu-Flex™ IIICONTAINER PL-146 ®PLASTIC
MANUFACTURED BY
BAXTER HEALTHCARE (GUANGZHOU) CO LTD
GUANGZHOU CHINA
(AN AFFI LIATE OF BAXTER WORLD TRADE INC USA)
HK-42531
DIRECTIONSTO BE USED AS DIRECTED BY THE PHYSICIAN
Prescription Drug
Manufacturer Address:
Jiaoyuan Road, Dongji Industrial District,
GETDD,Guangzhou, P.R. China
Low Calcium 1.5% Dextrose
2.5 LOW CALCIUM WITH 2.5% DEXTROSE 6000mlX2
LOT G00000000 EXP JAN 00 6AB9771E S/N 0000
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:05.052461 · Updated: 2026-03-14T22:26:18.292818