Sei Bella Flawless Liquid Foundation - 380 N
a7d9e03c-a2d0-07eb-e053-2995a90a158f
34390-5
HUMAN OTC DRUG LABEL
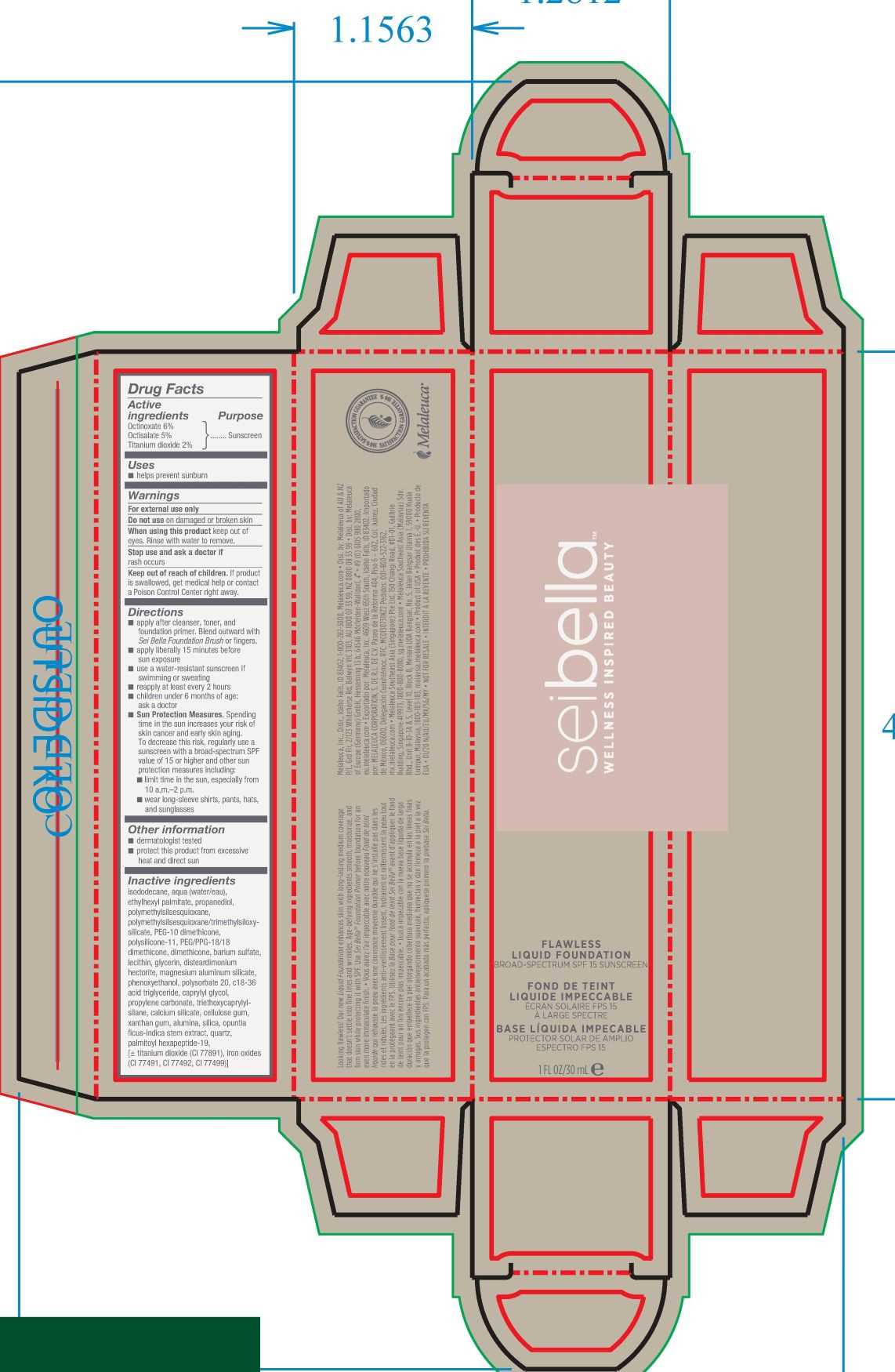
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Octinoxate 7.5%
Purpose
Sunscreen
Medication Information
Purpose
Sunscreen
Description
Octinoxate 7.5%
Use
■ helps prevent sunburn
Section 44425-7
■ protect this product from excesssive heat and direct sun
Section 50566-9
Stop use and ask a doctor if rash occurs
Section 50567-7
When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1
Do not use on damaged or broken skin
Section 51945-4
Warnings
For external use only
Directions
■ apply after cleanser, toner, and foundation primer. Blend outward with Sei Bella foundation brush or fingers.
■ apply liberally 15 minutes before sun exposure
■ use a water-resistant sunscreen if swimming or sweating
■ reapply at least every 2 hours
■ children under 6 months: ask a doctor
■ Sun Protection Measures. Spending time in the sun increases you risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad-spectrum SPF value of 15 or higher and other sun protection measures including:
■ limit time in the sun, especially from 10 a.m.-2 p.m.
■ wear long-sleeve shirts, pants, hats, and sunglasses
Active Ingredient
Octinoxate 7.5%
Other Information
■ dermatologist and allergy tested
Inactive Ingredients
isododecane, water, titanium oxide, dodecane, ethylhexyl palmitate, propanediol, polymethylsilsesquioxane, trimethylsiloxysilicate, peg-10 dimethicone, polysilicone-11, peg/ppg-18/18 dimethicone, dimethicone, barium sulfate, lecithin, glycerin, disteardimonium hectorite, magnesium aluminum silicate, phenoxyethanol, polysorbate 20, c18-36 acid triglyceride, caprylyl glycol, propylene carbonate, triethoxycaprylylsilane, calcium silicate, xanthum gum, cellulose gum, ouptnia ficus-indica stem extract, quartz, palmitoyl hexapeptide-19, may contain: iron oxide CI 77492, iron oxide 77491, iron oxide 77499
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use
■ helps prevent sunburn
Section 44425-7 (44425-7)
■ protect this product from excesssive heat and direct sun
Section 50566-9 (50566-9)
Stop use and ask a doctor if rash occurs
Section 50567-7 (50567-7)
When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1 (50570-1)
Do not use on damaged or broken skin
Section 51945-4 (51945-4)
Purpose
Sunscreen
Warnings
For external use only
Directions
■ apply after cleanser, toner, and foundation primer. Blend outward with Sei Bella foundation brush or fingers.
■ apply liberally 15 minutes before sun exposure
■ use a water-resistant sunscreen if swimming or sweating
■ reapply at least every 2 hours
■ children under 6 months: ask a doctor
■ Sun Protection Measures. Spending time in the sun increases you risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad-spectrum SPF value of 15 or higher and other sun protection measures including:
■ limit time in the sun, especially from 10 a.m.-2 p.m.
■ wear long-sleeve shirts, pants, hats, and sunglasses
Active Ingredient
Octinoxate 7.5%
Other Information (Other information)
■ dermatologist and allergy tested
Inactive Ingredients (Inactive ingredients)
isododecane, water, titanium oxide, dodecane, ethylhexyl palmitate, propanediol, polymethylsilsesquioxane, trimethylsiloxysilicate, peg-10 dimethicone, polysilicone-11, peg/ppg-18/18 dimethicone, dimethicone, barium sulfate, lecithin, glycerin, disteardimonium hectorite, magnesium aluminum silicate, phenoxyethanol, polysorbate 20, c18-36 acid triglyceride, caprylyl glycol, propylene carbonate, triethoxycaprylylsilane, calcium silicate, xanthum gum, cellulose gum, ouptnia ficus-indica stem extract, quartz, palmitoyl hexapeptide-19, may contain: iron oxide CI 77492, iron oxide 77491, iron oxide 77499
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:28.795841 · Updated: 2026-03-14T23:00:56.097890